Advances in Microbiology
Vol.3 No.1(2013), Article ID:29236,7 pages DOI:10.4236/aim.2013.31017
Microbial Adhesion on Orthodontic Ligating Materials: An in Vitro Assessment
1Teeth’N’Jaws Center, Chennai, India
2Microbiology Division, Central Leather Research Institute, (Council of Scientific and Industrial Research) Adyar, Chennai, India
Email: *gnanamani3@gmail.com
Received November 2, 2012; revised December 5, 2012; accepted January 6, 2013
Keywords: Adhesion; Growth; Orthodontic Ligatures; Growth OD; CFU; Metabolic Activity; XTT
ABSTRACT
Orthodontic fixed appliance therapy is the commonest mode of treatment for most types of malocclusions (teeth irregularities). However, these materials are liable for microbial adhesion, which predisposes the wearer to increased microbial burden. The present study aims to evaluate, microbial adhesion and growth on commonly used orthodontic ligating materials (Teflon coated wire, stainless steel wire, elastic rings) under in vitro condition. Furthermore, the role of saliva on adhesion and microbial colonization on said materials was also assessed. Experiments were conducted with three different orthodontic ligating materials each in 6 numbers. Growth OD, metabolic activity and cell viability were the experimental variables in addition to SEM (Scanning Electron Microscopy) analysis performed. Results revealed irespective of the nature of the ligating materials, microbial adhesion and growth were observed in all the materials and suggested that the chosen materials promotes microbial adhesion. Nevertheless, stainless steel ligatures were less prone to adhesion compared to Teflon coated and elastic ligatures. Presence of saliva accelerates adhesion and growth.
1. Introduction
Orthodontic fixed appliance therapy is the commonest mode of treatment for most types of malocclusions and the most commonly used orthodontic materials are brackets, tubes, band material, ligating materials and archwires. However, these materials are liable for microbial adhesion, greatly inhibit oral hygiene and create new retentive areas for plaque and debris, which in turn predisposes the wearer to increased microbial burden and possibility of subsequent infection. Literatures on microbial adhesion to various types of brackets are already available, however no reports are on ligating materials and band materials. As reported by Magno et al. [1] fixed appliances promote continuous accumulation and retention of microbial growth. Available reports suggests, it is difficult to remove the microbial growth [2,3] or clean the orthodontic appliances fixed at the critical sites and it finally leads to enamel decalcification and white spot lesion formation around the orthodontic appliances [4]. More than 50% of the patients encountered enamel demineralization after the removal of fixed orthodontic appliances [2,4,5]. Transformations of oral microbial biota by the presence of orthodontic fixed appliances pose significant impact on the patient’s oral and general health [5-7]. It is a common belief that plaque formation during treatment with fixed appliances is mainly attributed to the complexity of the bracket design [8-10] and ligating methods. Kitada et al. [11] proposed an alarming report on steady increase in occurrence of infections during the orthodontic usage. As reported by Atack et al. [12] frequently encountered (50% - 60%) opportunistic pathogens in humans are from the oral cavities of young adults.
Cannon et al. [13] reported that oral cavity serve as niche for colonization by yeast cells. Białasiewicz et al. [14] reported higher percentage of yeast cells in the oral cavity of children compared to adult control. According to Jewtuchowicz et al. [15] use of dental devices significantly increased the prevalence of yeasts in periodontal pockets in patients having complaints with gingivitis. A yearlong case study conducted by Arslan et al. [16] emphasizes, influence of saliva on yeast growth on orthodontic appliances and in addition demonstrated, an increased risk of infection overtime in fixed orthodontic appliance increases with poor oral hygiene and is evidenced by the elevated level of yeast cells in saliva.
Furthermore, Budtz-Jorgensen [17] reported high oral colonization of yeast cells in individuals wearing full or partial removable dentures. Hibino et al. [18] reported adherence of yeast cells on orthodontic appliances and transformation of healthy individuals to yeast cell carriers. However, no reports are available on the direct microbial adhesion and growth on orthodontic ligating materials. Thus, the present study aimed to assess microbial adhesion and growth on various orthodontic ligating materials under in vitro conditions using yeast cells as test organism.
With regard to saliva, different opinions are on whether saliva promotes microbial growth or prohibits the growth. In the present study, since the ligating materials are to be in the oral environment for six - eight weeks, it is necessary to study the role of saliva in adhesion of microbes on orthodontic ligating materials. Hence, experiments on impact of saliva on microbial adhesion on chosen orthodontic ligating material was studied under in vitro conditions.
2. Materials and Methods
2.1. Organisms
A clinical strain of yeast cells (Candida albicans (CLCA 0520)) obtained from patient suffering from oral thrush was further identified (CHROM agar, Germ tube) by standard protocols and subcultured in Sabouraud dextrose (Hi Media, Mumbai, India) agar slants and used for in vitro study.
2.2. Medium and Growth Conditions
Sabouraud Dextrose (SD) broth (HiMedia, India) contains Dextrose (20 g/L) and Peptone (10 g/L) was used for the sub-culturing of yeast cells. Yeast Nitrogen Base (YNB) broth with 500 mM dextrose used for growth.
2.3. Materials
Orthodontic ligating materials: 1) Stainless steel ligatures (diameter 0.010 inches), manufactured by TP orthodontics, USA; 2) Teflon coated stainless steel ligatures (diameter 0.012 inches); and 3) Elastic rings (Clear) (Outer Diameter 3.10 mm (transparent rings), manufactured by Ortho-Organizer, USA; each in six numbers were used in the present study.
2.4. Preparation of Saliva Pre-Coated Orthodontic Ligatures
Whole un-stimulated saliva was collected from six healthy adult volunteers (refrained from eating for a minimum of 2 hours prior to collection). The donors were asked to rinse their mouth gently with water and then to chew de-sugared chewing gum for 1 hour to stimulate salivary glands. The saliva was collected, pooled and centrifuged at 1000 rpm for 15 min at 4˚C and the supernatant used immediately for coating [19,20].
2.5. Assessment of Microbial Adhesion and Growth
In order to assess the optimum period required for maximum adhesion of yeast cells on ligating materials, cells were grown in YNB medium for 48 hours at 37˚C and subjected to centrifugation at 12,000 g, washed the deposited cells twice with PBS (Phosphate Buffered Saline; pH 7.2) and suspended in the same buffer and the cell suspension with optical density (600 nm) of 0.5 OD (5 × 107 cells/ml) was used as the optimum cell concentration. Orthodontic ligatures (all three types) (n = 6) immersed in cell suspension individually for the scheduled period of 30, 45, 60, 90 and 120 minutes at 37˚C with mild shaking (50 RPM; Remi Shaker, India). After scheduled time points, ligatures were carefully removed and gently washed with PBS.
The ligatures with adhered cells were then allowed to grow in 6-well plates containing sabouraud dextrose broth with additional 50 mM dextrose for 48 hours at 37˚C. Cell growth, cell viability and metabolic activity assays were performed according to the procedures summarized below.
2.6. Role of Saliva on Adhesion
To assess the role of saliva, the ligatures were pre-coated with saliva and the yeast cells adhesion and growth was tested as summarized above. In brief, ligatures coated with saliva was exposed to yeast cells for the period of 1 hour for initial adherence and then subjected to gentle wash to remove the non-adhered cells and then incubated in SD broth for the period of 48 hours at 37˚C. Cell growth, cell viability and metabolic activity assays were performed according to the procedures summarized below.
2.7. Analysis of Growth OD, Fresh and Dry Weight Measurements
Ligatures obtained from the above said experiments were kept under sonication with 0.5 ml of 1.5 M PBS (pH 7.2) and then with additional volume of 0.5 ml and the cell suspension was pooled. The optical density of the pooled cell suspension was measured at 600 nm using UV-Visible spectrophotometer (Schimadzu, Japan). The final cell suspension was centrifuged at 10,000 × g at 4˚C and the weight of the pellet measured was taken as fresh weight and the weight of the pellet free from moisture (dehydrated using 90% ethanol) was considered as dry weight.
2.8. Assay of Cell Viability—Colony Forming Unit (CFU)
For cell viability assessment, cell suspension obtained from the above step was serially diluted and the dilution of 10−7 and 10−6 were spread plated in 20 ml of Sabouraud dextrose agar plate, incubated for 48 hours at 37˚C and counted the colony forming units.
2.9. Metabolic Activity Assay—XTT Reduction
Metabolic activity of viable cells was assessed according to Ramage et al. [19] using XTT (2,3-bis [2-methoxy-4- nitro-5-sulfophenyl]-5-[(phenylamino) carbonyl]-2H-tetrazolium hydroxide) reduction assay. Briefly, XTT (Sigma, USA) dissolved in PBS (pH 7.2) at a final concentration of 1 mg/ml filter sterilized using a 0.22 μm-pore-size filter and stored at –70˚C until required. Ligatures containing adhered cells were transferred to a new 6-well plate containing 3 ml of PBS, 50 µl of XTT solution (1 mg/ml in PBS; Sigma, USA) and 4 µl of menadione solution (0.4 mM of menadione in 1 mM in acetone; Sigma, USA), incubated for 4 hours at 37˚C. The solution containing reduced XTT formazan products were made cells free (centrifuged at 6000 g, 5 min) and read at 492 nm.
2.10. Scanning Electron Microscopy
Microbial adhesion on ligatures (with and without saliva pre-coating) was processed for scanning electron microscopy, according to the procedure summarized by Bozzola and Russel [21] using JEOL JSN 6360 (Japan)] in high vacuum 20 kV. Images processed using soft imaging viewer software and Adobe Photoshop CS 2 (Adobe Systems, Inc., San Jose, CA, USA).
2.11. Statistics
All the experiments carried out for each six pieces (n = 6) of ligatures on different days. Raw data were statistically analyzed and results with P < 0.05 considered as statistically significant.
3. Results
Figure 1 depicts the time period required for the maximum adhesion of yeast cells on the chosen orthodontic ligating materials and suggests, irrespective of the nature of ligating materials, the maximum colony forming units was observed after 60 minutes of exposure to cell suspension. No significant increase in CFU was observed for the additional incubation period.
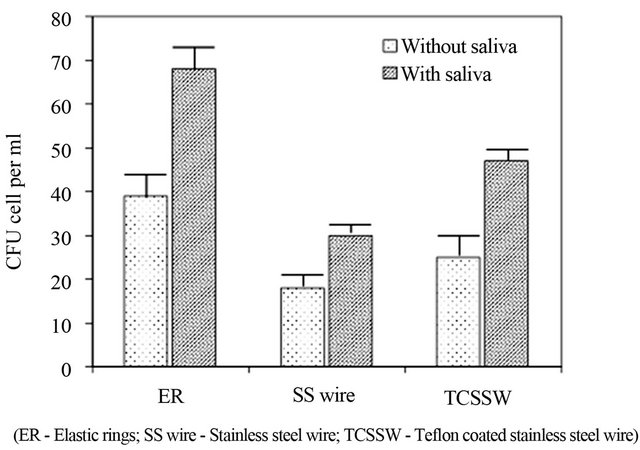
Upon adherence, the growth of yeast cells on ligating materials was assessed from the measurements on CFU and metabolic activity analyses after 48 hours. Results displayed a significant difference amongst the ligatures and the maximum bioburden was encountered with Elastic rings (ER) followed by Teflon coated stainless steel wire (TCSSW) and stainless steel (SS) (Figure 2(a)). Statistical analysis demonstrated the significance at the level of P < 0.001 between ER vs TCSSW, ER vs SS, SS vs TCSSW (Table 1).
With regard to role of saliva on microbial growth on ligatures, Figure 2(b) displayed the level of bioburden assessed and suggests, saliva coating significantly accelerates the adhesion and growth in all the three different
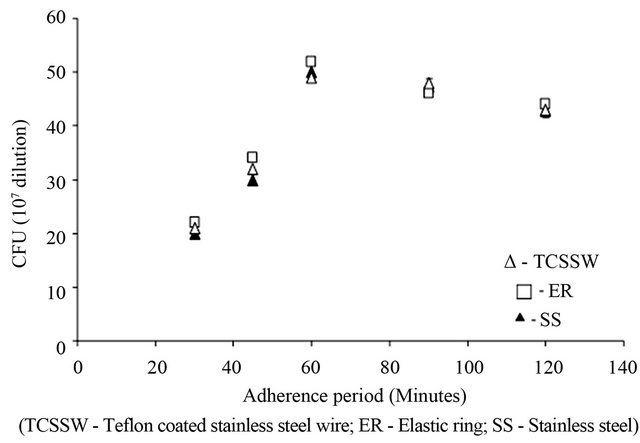
Figure 1. Optimization of adherence period of yeast cells on orthodontic ligatures.
 (a)
(a)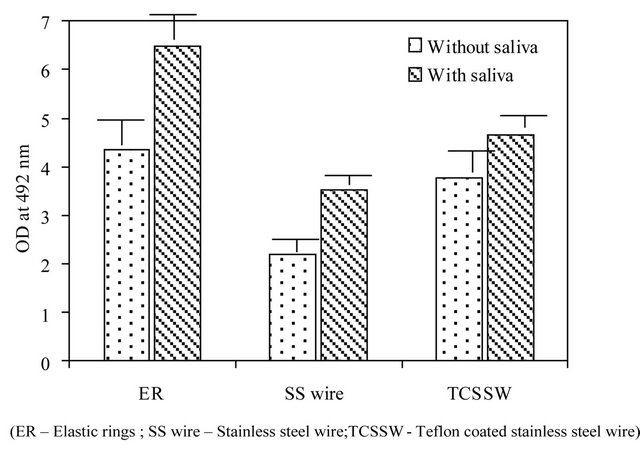 (b)
(b)
Figure 2. (a) Growth (CFU) and (b) Metabolic activity of yeast cells grown on SS wire (stainless steel wire); TCSSW (Teflon coated stainless steel wire) and ER (Elastic rings) Orthodontic ligatures with and without saliva pre-coating.
Table 1. Statistical analysis of yeast cells bioburden estimated in three different orthodontic ligatures (ER: Elastic rings; TCSSW: Teflon coated stainless steel; SS: Stainless steel) for experiments with and without saliva coating (n = 6).
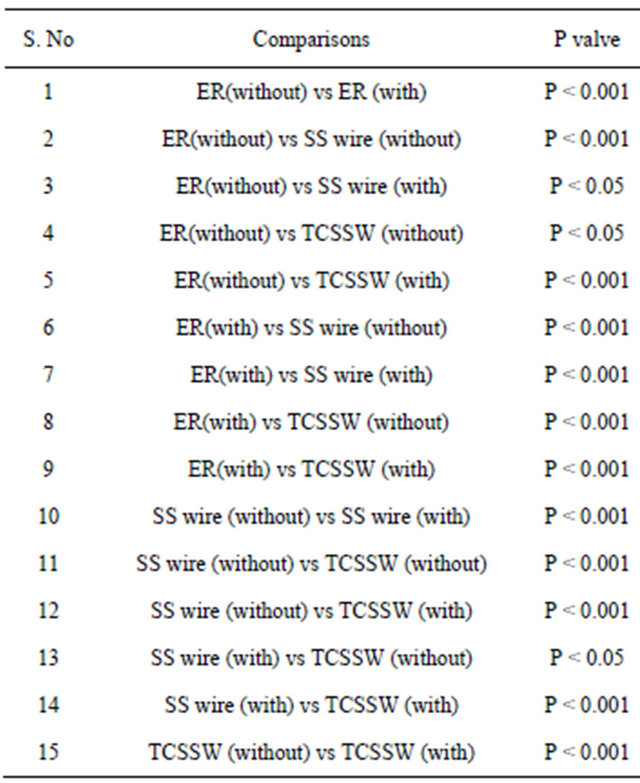
ligatures chosen. About 44%, 37% and 50% increase in bioburden was observed respectively with saliva coated elastic rings, stainless steel and Teflon coated stainless steel compared to plain ligatures. Table 1 demonstrates the results on statistical analysis followed and suggests the significant difference in yeast cells bioburden between with and without saliva coated ligatures. Significance at P < 0.05 was observed for the experiments on ER (without saliva coating) vs TCSSW (without coating) and ER (without coating) vs SS wire (with coating) and SS wire (with) vs TCSSW (without).
With regard to fresh and dry weight measurements on the yeast cells grown on ligatures, Table 2 demonstrated differences in fresh and dry weight of cells obtained from ER, TCSSW and SS ligatures of both with and without saliva coating. Similar to the observations on CFU, both fresh and dry weights (μg) demonstrated maximum bioburden with elastic rings followed by Teflon coated stainless steel and then by stainless steel.
In order to ascertain, whether, the adhered and grown cells were completely removed during the process of rigorous washings, crystal violet staining of ligatures was made that ensures the all the adhered cells were gone to the solution. With regard to metabolic activity (Figure 2(b)), similar to cell viability measurements, elastic rings exhibited higher metabolic activity followed by Teflon coated stainless steel and then by stainless steel. In addition, the saliva coated ligatures demonstrate higher metabolic activity than plain ligatures.
Scanning electron microscopy analysis (Figures 3(a) and (b)) of ligating materials made after 24 hours of incubation with yeast cells demonstrated, adherence of cells in all the ligatures. Images, with out saliva coating illustrate, growth of cells and hyphal structures, however, an increase in growth in the form of increased hyphal structures. Similar to the observations on CFU and metabolic activity, SEM analysis also reflected high bioburden with ER and TCSSW compared to SS.
4. Discussion
In general, orthodontic fixed appliances remain in the mouth for a period of 2 - 3 years, and a study on assessing the microbial adhesion in one of the accessories (ligating materials) is thus significant. In the present study evaluation of microbial adhesion on three different commonly used orthodontic ligating materials was carried out yeast cells as test organism. The reason for the selection of yeast cells was based on their presence in the most frequent microbial infections of oral buccal mucosa [22, 23]. Reports imply, C. albicans colonizes on cementum, enamel and dentin, which serve as a reservoir for the spread of infection [24]. Tronchin et al. [25] demonstrates, yeasts adhere directly to the plastic surface and form a fine layer or biofilm on the surface of the synthetic device.
Since adhesion of cells on the surface is more vital for the growth and development, it is necessary to assess the optimal time taken for the maximum number of cells to adhere on the surface of ligating materials. Experimental results from the optimization study revealed maximum adhesion was observed within 60 minutes of exposure. Once cells adhered, cell elongates with the formation of pseudohyphae and the formation of hyphae was considered as one of the virulence factors associated with greater invasive capacity, tissue invasion, which further provide great resistance to phagocytes [26,27]. These cells are more resistant to antifungal drugs and serve as foci of infection. Adhesion of microbes further depends on the surface properties of the materials and the high surface roughness leads to increased cell adhesion [28]. In addition, surface energy, composition, surface hydrophobicity and zeta potential of the materials also influences adhesion of cells [29]. Further, possibility of alterations in the surface property due to oral environment, food colorants and food habits further accelerates the affinity of microbes [30]. In the present study, all the three ligating materials have a microheterogeneity surface, which promotes the yeast cells adhesion at considerable level.
Table 2. Fresh and dry weight measurements of yeast cells bioburden measured for the experiments on three difference orthodontic ligatures for both with and without saliva coating (ER: Elastic rings; TCSSW: Teflon coated stainless steel; SS: Stainless steel) (n = 6).
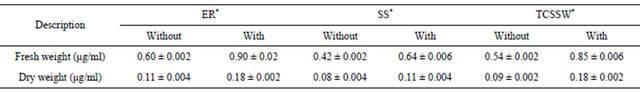
*Values are the mean ± SD of six samples.

Figure 3. Scanning Electron micrograph of yeast cells grown on ligatures of (a) With out saliva coated—Elastic rings, stainless steel wires, Teflon coated stainless steel wires; (b)With saliva pre-coated—Elastic rings, stainless steel wires, Teflon coated stainless steel wires.
Followed by adhesion, the growth of cells for the period of 48 hours and the assessment on CFU, cell viability and metabolic activity reveal, the maximum CFU, viable cells and higher metabolic activity was realized with Elastic rings compared to Teflon coated wire and Stainless steel wires. This could be reasoned to 1) more surface area; and 2) reduced surface energy of elastic rings (data not shown). Further, we also expected, the reduced surface energy of Teflon coating could not allow the cells to adhere and grow, but in contrast, we found significant growth OD and metabolic activity for the cells obtained from Teflon coated materials. Nevertheless, when compared to Stainless steel ligatures no significant difference in growth OD and metabolic activity was observed in Teflon coated ligatures and suggested other than surface energy; some other factors influences the adhesion and growth. This kind of observations needs further exploration with other Teflon coated materials used for clinical purposes.
Further, we observed saliva also influences the adhesion of yeast cells on the chosen ligatures. Despite, presence of antimicrobial proteins such as IgA, histatin 5, lysozyme, lactoferrin, [31] growth OD and the metabolic activity of yeast cells showed significant increase in the samples obtained from saliva pre-coated ligatures compared to ligatures without any saliva pre-coating. The enhanced cell adhesion and growth in saliva pre-coated material might be due to the presence of proteins like human fibronectin and few basic proline-rich protein families, which are acting as receptors for free-living planktonic yeast cells [32-36]. Further, Lee et al. [10] studied the composition of salivary pellicle on the surfaces of orthodontic materials by providing saliva coating on the material to mimic the oral conditions and found orthodontic materials have unique surface properties that differs from other restorative and prosthetic materials, as well as from human tooth enamel and salivary coating tremendously affects the surface characteristics of the materials.
Currently, for time-efficient orthodontics, Elastic rings are used as a material of choice to ligate orthodontic arch wires to brackets, instead of stainless steel ligatures. However, the results of our study suggests, Elastic rings promotes adhesion of yeast cells and growth and lead to predeposition for microbial population on the teeth surface locally. Similar to our findings, Forsberg et al. [37] also observed greater microbial colonization on teeth ligated to archwire with elastic rings compared with teeth ligated with steel wires and recommended the use of elastic rings may be avoided in patients with inadequate oral hygiene. In contrary, Sukontapatipark et al. [5] found no significant differences between brackets ligated with Elastic and Stainless steel ligature materials in regard to microbial contamination. The results of our study emphasize the presence of microbial adhesion on commonly used ligating materials irrespective of their nature and structure. Among the three ligating materials, higher bioburden was observed with Elastic ring compared to other two materials. In addition, saliva accelerated the growth and colonization of microbes in all the evaluated ligating materials as evidenced through the growth OD measurements and metabolic activity assay. As use of orthodontic fixed appliances is popular, problems associated with these materials needs attention. Magno et al. [1] suggests microbial adhesion analysis of orthodontic materials is essential before going for clinical applications. Based on the observation made in the present study, we also recommended the necessity of pre-microbial adhesion analysis to avoid orthodontic ligating materials as potential foci of infection. Davies and Begola [4] pointed out that orthodontists have to use protection barriers and sterilize instruments, hand pieces and pliers. The American Dental association and Center for Disease control and Prevention have confirmed these recommendations because this is the only way to guarantee a safe environment for patients, professionals and coworkers. This will have implications in disposal of used orthodontic ligating materials as they harbour active microorganisms on their surface as shown in our results.
In conclusion, the results of the study demonstrates microbial adhesion and growth on orthodontic ligating materials under in vitro conditions. Maximum microbial adhesion and growth were observed on Elastic ligatures, the most commonly used ligating materials and a least on Stainless steel ligatures, a less commonly used. Presence of saliva accelerates the microbial adhesion and growth in orthodontic ligatures.
5. Acknowledgements
The first author Dr. P. Harikrishnan, acknowledges Director, CLRI, for the permission to undertake the study.
REFERENCES
- A. F. Magno, C. Enoki, I. Y. Ito, M. A. Matsumoto, G. Faria and P. Nelson-Filho, “In Vivo Evaluation of the Contamination of Super Slick Elastomeric Rings by Streptococci mutans in Orthodontic Patients,” American Journal of Orthodontics & Dentofacial Orthopedics, Vol. 133, No. 4, 2008, pp. S104-S109. doi:10.1016/j.ajodo.2006.04.054
- G. S. Baillie and L. J. Douglas, “Effect of Growth Rate on Resistance of Candida albicans Biofilms to Antifungal Agents,” Antimicrobial Agents and Chemotherapy, Vol. 42, No. 8, 1998, pp. 1900-1905.
- M. A. Al-Fattani and L. J. Douglas, “Penetration of Candida Biofilms by Antifungal Agents,” Antimicrobial Agents and Chemotherapy, Vol. 48, No. 9, 2004, pp. 3291-3297. doi:10.1128/AAC.48.9.3291-3297.2004
- D. Davies and A. Begole, “Compliance with Infection Control Procedures among Illinois Orthodontists,” American Journal of Orthodontics and Dentofacial Orthopedics, Vol. 113, No. 6, 1998, pp. 647-654.
- W. Sukontapatipark, M. A. El-Agroudi, N. J. Selliseth, K. Thunold and K. A. Selvig, “Bacterial Colonization Associated with Fixed Orthodontic Appliances-A SEM Study,” European Journal of Orthodontics, Vol. 23, No. 5, 1998, pp. 475-484. doi:10.1093/ejo/23.5.475
- M. G. Azmi and N. Al-Jasser, “The Effect of Fixed Orthodontic Appliance Therapy on Oral Candida Carriage,” Saudi Dental Journal, Vol. 15, No. 3, 2003, pp. 141-144.
- U. Hagg, P. Kaveewatcharanont, Y. H. Samaranayake and L. P. Smaranayake, “The Effect of Fixed Orthodontic Appliances on the Oral Carriage of Candida Species and Enterobacteriaceae,” European Journal of Orthodontics, Vol. 26, No. 6, 2004, pp. 623-629. doi:10.1093/ejo/26.6.623
- R. J. Gibbon, “Bacterial Adhesion to Oral Tissues: A Model for Infectious Diseases,” Journal of Dental Research, Vol. 68, No. 5, 1989, pp. 750-760. doi:10.1177/00220345890680050101
- L. A. Tabak and W. H. Bowen, “Roles of Saliva (Pellicle), Diet and Nutrition on Plaque Formation,” Journal of Dental Research, Vol. 68, 1989, pp. 1560-1566.
- S. J. Lee, H. S. Kho, S. W. Lee and W. S. Yang, “Experimental Salivary Pellicles on the Surface of Orthodontic Materials,” American Journal of Orthodontics & Dentofacial Orthopedics, Vol. 119, No. 1, 2001, pp. 59-66. doi:10.1067/mod.2001.110583
- K. Kitada, A. De Toledo and T. Oho, “Increase in Detectable Opportunistic Bacteria in the Oral Cavity of Orthodontic Patients,” International Journal of Dental Hygiene, Vol. 7, No. 2, 2009, pp. 121-125. doi:10.1111/j.1601-5037.2008.00333.x
- N. E. Atack, J. R. Sandy and M. Addy, “Periodontal and Microbiological Changes Associated with the Placement of Orthodontic Appliances—A Review,” Journal of Periodontology, Vol. 67, No. 2, 1996, pp. 78-85. doi:10.1902/jop.1996.67.2.78
- R. D. Cannon and W. L. Chaffin, “Oral Colonization by Candida albicans,” Critical Reviews in Oral Biology and Medicine, Vol. 10, No. 3, 1999, pp. 359-383. doi:10.1177/10454411990100030701
- D. Bialaisiewicz, A. Kurnatowska and G. Smiech-Slomkowska, “Characteristics of Fungi and Attempts of Their Elimination from the Oral Cavity in Children Treated with Orthodontic Appliances,” Medycyna Doswiadczalna i Mikrobiologia, Vol. 45, No. 3, 1993, pp. 389-392.
- V. M. Jewtuchowics, M. I. Brusca, M. T. Mujica, L. A. Gliosca, J. L. Finquelievich, C. A. Lovannitti and A. C. Rosa, “Subgingival Distribution of Yeast and Their Antifungal Susceptibility in Immunocompetent Subjects with and without Dental Devices,” Acta Odontologica Latinoamericana, Vol. 20, No. 1, 2007, pp. 17-22.
- S. G. Arslan, N. Akpolat, J. D. Kama, T. Ozer and O. Hamamci, “One Year Follow Up of the Effect of Fixed Orthodontic Treatment on Colonization by Oral Candida,” Journal of Oral Pathology & Medicine, Vol. 37, No. 1, 2008, pp. 26-29. doi:10.1111/j.1600-0714.2007.00574.x
- E. Budtz-Jorgensen, “Candida Associated Denture Stomatitis and Angular Cheilitis,” In: L. P. Samaranayake and T. W. Macfarlane, Eds., Oral Candidiasis, Wright, London, 1990, pp. 156-183.
- K. Hibino, R. W. Wong, U. Hagg and L. P. Samaranayake, “The Effects of Orthodontic Appliances on Candida in the Human Mouth,” International Journal of Paediatric Dentistry, Vol. 19, No. 5, 2009, pp. 301-308. doi:10.1111/j.1365-263X.2009.00988.x
- G. Ramage, S. P. Saville, D. P. Thomas, J. L. LopezRibot, “Candida Biofilms: An Update,” Eukaryot Cell, Vol. 4, No. 4, 2005, pp. 633-638. doi:10.1128/EC.4.4.633-638.2005
- Y. Jin, L. P. Samaranayake, Y. Samaranayake and H. K. Yip, “Biofilm Formation of Candida albicans Is Variably Affected by Saliva and Dietary Sugars,” Archives of Oral Biology, Vol. 49, No. 10, 2004, pp. 789-798. doi:10.1016/j.archoralbio.2004.04.011
- J. J. Bozzola and L. D. Russel, “Electron Microscopy: Principles and Techniques for Biologists,” Jones & Bartlett Learning, Burlington, 1999.
- R. L. Sandin, A. L. Rogers, M. I. Fernandezs and E. S. Benekev, “Variations in Affinity to Candida albicans in Vitro among Human Buccal Epithelial Cells,” Journal of Medical Microbiology, Vol. 24, No. 2, 1987, pp. 151-155. doi:10.1099/00222615-24-2-151
- M. I. Brusca, O. Chara, L. Sterin-Borda and A. C. Rosa, “Influence of Different Orthodontic Brackets on Adherence of Microorganisms in Vitro,” The Angle Orthodontist, Vol. 77, No. 2, 2007, pp. 331-336. doi:10.2319/0003-3219(2007)077[0331:IODOBO]2.0.CO;2
- M. Addy, W. C. Shaw, P. Handsford and M. Hopkins, “The Effect of Orthodontic Appliances on the Distribution of Candida and Plaque in Adolescents,” British Journal of Orthodology, Vol. 9, No. 3, 1982, pp. 158-163.
- G. Tronchin, J. P. Bouchara, R. Robert and J. M. Senet, “Adherence of Candida albicans Germ Tubes to Plastic: Ultrastructural and Molecular Studies of Fibrillar Adhesions,” Infection and Immunity, Vol. 56, No. 8, 1988, pp. 1987-1993.
- J. E. Cutler, “Putative Virulence Factor of C. albicans,” Annual Review of Microbiology, Vol. 45, 1991, pp. 187- 218. doi:10.1146/annurev.mi.45.100191.001155
- J. W. Rippon, “Candidiasis and the Pathogenic Yeasts,” In: J. W. Rippon, Ed., Medical Mycology, The Pathogenic Fungi and the Pathogenic Actinomycetes, W B Saunders, Philadelphia, 1982, pp. 484-531.
- J. Bagg and R. W. Silverwood, “Coagglutination Reactions between Candida albicans and Oral Bacteria,” Journal of Medical Microbiology, Vol. 22, No. 2, 1986, pp. 165-169. doi:10.1099/00222615-22-2-165
- R. L. Price, K. Ellison, K. M. Haberstroh and T. J. Webster, “Nanometer Surface Roughness Increases Select Osteoblast Adhesion on Carbon Nanofiber Compacts,” Journal of Biomedical Materials Research Part A, Vol. 70A, No. 1, 2004, pp. 129-138. doi:10.1002/jbm.a.30073
- A. Faltermeier, R. Bürgers and M. Rosentritt, “Bacterial Adhesion of Streptococcus mutans to Esthetic Bracket Materials,” American Journal of Orthodontics and Dentofacial Orthopedics, Vol. 133, No. 4, 2008, pp. S99- S103. doi:10.1016/j.ajodo.2007.03.024
- M. W. J. Dodds, D. A. Johnson and C. Yeh, “Health Benefits of Saliva: A Review,” Journal of Dentistry, Vol. 33, No. 3, 2005, pp. 223-233. doi:10.1016/j.jdent.2004.10.009
- K. G. Skerl, E. Segal, T. Sreevalsan and W. M. Scheld, “In Vitro Binding of Candida albicans Yeast Cells to Human Fibronectin,” Canadian Journal of Microbiology, Vol. 30, No. 2, 1984, pp. 221-227. doi:10.1139/m84-033
- J. M. O’Sullivan, R. D. Cannon, P. A. Sullivan and H. F. Jenkinson, “Identification of Salivary Basic Proline-Rich Proteins as Receptors for Candida albicans Adhesion,” Microbiol, Vol. 143, No. 2, 1997, pp. 341-348. doi:10.1099/00221287-143-2-341
- C. Gyurko, U. Lendenmann, R. F. Troxler and F. G. Oppenheim, “Candida albicans Mutants Deficient in Respiration Are Resistant to the Small Cationic Salivary Antimicrobial Peptide Histatin 5,” Antimicrobiol Agents Chemother, Vol. 44, No. 2, 2000, pp. 348-354. doi:10.1128/AAC.44.2.348-354.2000
- R. S. Millan, N. Elguezabal, P. Regulez, M. D. Moragues, G. Quindos and J. Ponton, “Effect of Salivary Secretary IgA on the Adhesion of Candida albicans to Polystyrene,” Microbiology, Vol. 146, 2000, pp. 2105-2112.
- Y. H. Samaranayake, L. P. Samaranayake, E. H. Pow, V. T. Beena and K. W. Yeung, “Antifungal Effects of Lysozyme and Lactoferrin against Genetically Similar, Sequential Candida albicans Isolates from a Human Immunodeficiency Virus-Infected Southern Chinese Cohort,” Journal of Clinical Microbiology, Vol. 39, No. 9, 2001, pp. 3296-3302. doi:10.1128/JCM.39.9.3296-3302.2001
- C. M. Forsberg, V. Brattstrom, E. Malmberg and C. E. Nord, “Ligature Wires and Elastomeric Rings: Two Methods of Ligation and Their Association with Microbial Colonization of Streptococci mutans and Lactobacilli,” European Journal of Orthodontics, Vol. 13, No. 5, 1991, pp. 416- 420. doi:10.1093/ejo/13.5.416
NOTES
*Corresponding author.

