 International Journal of Organic Chemistry, 2011, 1, 224-232 doi:10.4236/ijoc.2011.14033 Published Online December 2011 (http://www.SciRP.org/journal/ijoc) Copyright © 2011 SciRes. IJOC Synthesis of Chlorinated Bicyclic Adduct as Biocids for Sulfate-Reducing Bacteria Mona A. Youssif, Nahla A. Mansour, Azza M. Mazrouaa, Mohamed A. Shenashen Egyptian Petroleum Research Institute, Ca iro , Egypt E-mail: azza_mazroua2005@yahoo.com Received June 21, 2011; revised August 28, 2011; accepted September 12, 2011 Abstract Synthesis of bicyclic systems containing chlorine atoms, and/or ether groups in aromatic rings can be con- sidered as an important method for building bicyclic system and production of new adducts. One of the most important types in the cycloaddition reaction is the Diels-Alder reaction (1,4 cycloaddition). In the present investigation a new ether of allylic type (dienophile) p-allyl bromo phenol was prepared and its structure was confirmed by molecular weight determination, refractive index, infrared spectra, and density. A new adduct was obtained by means of 1,4 cycloaddition reaction of hexachlorocyclopentadiene (HCP) and the new pre- pared dienophile. The reaction takes place without using solvent, catalysts, or elimination of any compound. The effect of variations in temperature, initial molar ratio and reaction duration were studied to determine the optimum conditions of the reaction. The optimum conditions reached were reaction temperature recorded 140˚C, initial molar ratio diene: dienophile was 3:1 and the reaction duration time reached 6 h. Under these optimum conditions the maximum yield was 78%. The new adduct revealed very high biological effect as sulfate-reducing bacteria (SRB). Keywords: 1,4 Cycloaddition, Hexachlorocyclopentadiene, Adduct and Sulfate-Reducing Bacteria (SRB) 1. Introduction The Diels-Alder reaction is an organic chemical reaction (specially,a cyclo-addition) between a conjugated diene and substituted alkene, commonly termed (the dienophile) to form a substituted cyclohexene system [1,2]. The Di- els-Alder reaction is generally considered the “Monalisa” of reaction in organic chemistry since it requires very lit- tle energy to create the very useful cyclohexene ring [3- 5]. Due to the high degree of regio-and stero selectivity (due to the concerted mechanism), the Diels-Alder reac- tion is a very powerful reaction and widely used in syn- thetic organic chemistry [5,6]. The reaction is usually thermodynamically favorable due to the conversion of 2π bonds into new stronger (б) bonds. Diels-Alder reaction is favored by electron withdrawing groups on the electro- philic dienophile and by electron donating groups on the nuclophilic diene. The diene components in Diels-Alder reaction can be an open-chain or cyclic type, it can have many different kinds of substituents. It must be able to exsit in the cis conformation [2,5]. Cyclic dienes that are premantly in the s-cis conformation are exceptionally reactive in Diels-Alder reactions. Cyclic dienes that are permanently in the s-trans conformation will not undergo the Diels-Alder reaction at all [2,5]. Dienophiles in D-A reaction do not react with equal case. The reactivities of the dienophiles depend on the structure, i.e the kind and the position of the substituents of the dienophiles mole- cules. The greater number of the electron attracting sub- stituents on the double or triple bonds, the more abil- ity of the olefins or acetylenes to be react as dienophiles [2,7,8]. The D-A reaction is reversible one, and many ad- ducts dissociate into their components at quite low tem- perature. In this case better yield are obtained by using an excess of one of the reactants, or solvent from which the adduct separates readily [2]. Hexachlorocyclopenta- diene (HCP) and/or its derivatives have been used in diene synthesis since 1954. It has been found that they could be considered as a good dienes which may be used in the diene synthesis due to the importance of its ad- ducts with various type of dienophiles, as they have very important applications and great role in the industial fields [9,10]. (HCP) can be adding to certain dienophiles under certain conditions. Several studies are carried out to compare the reactivity of the dienophiles of allylic type in D-A reaction 1,4 cyclo-addition with (HCP) [9,11-15].  225 M. A. YOUSSIF ET AL. Sulfide production by sulfate-reducing bacteria (SRB) is a major concern for the petroleum industry since it is toxic and corrosive and causes plugging due to the for- mation of insoluble iron sulfides [16-18]. A number of methods for controlling sulfide production in different oil production facilities had been proposed in order to redu- ce activity of SRB [19-21]. However, most of methods are usually inefficient because of microbial resistance or they might be a risk to human health and environment. The sulfate reducing bacteria (SRB) are the most destruct- tive microorganisms in anaerobic minimum concentra- tion inhibition MIC. These bacteria having the ability to oxidize sulfur compounds, it reduce sulphate to sulphide and promote formation of sulphide film, i.e., their char- acteristic form of respiration uses sulphate and results in sulphide formation [22]. According to Iverson, apart from H2S, SRB also produces a highly corrosive pho- sphides containing metabolite at pH = 3 that enhances the dissolution of the metal under anaerobic conditions [23]. Many chemical compounds have the ability to inhibit the growth and metabolism of microorganisms or to kill them [24]. The aim of the work is synthesis of chlorin- ated bicyclic adduct which may be used as biocides for Sulfate-Reducing Bacteria. 2. Experiment 2.1. Materials Para bromophenol: (C6H5BrO) M.Wt. 173.01 g·mol−1, Density 1.84 g·mL−1, M.p. 64˚C - 68˚C, B.p. 235˚C - 236˚C. Allyl bromide: (C3H5Br) M.Wt. 120.99 g·mol−1, Density 1.398 g·mL−1, M.p. –119˚C, B.p. 71˚C. Anhydrous (K2CO3): M.Wt. 183.205 g·mol−1, Density 2.29 g·mL−1, M.p. 891˚C, B.p. decomposes. Acetone: M.Wt. 58.08 g·mol−1, Density 0.7925 g·mL−1, M.p. 95˚C, B.p. 56˚C - 57˚C. NaOH: M.Wt. 39.997 g·mol−1, Density 2.13 g·mL−1, M.p. 318˚C, B.p. 1388˚C. Anhydrous (MgSO4): M.Wt. 120.366 g/mol, Density 2.66 g/cm3, M.p. 1124˚C. Hexach loroc yclop entad ien e (HCP): (diene) B.p. 239˚C/ 753 mm, density at 25˚C, 1.7179 g/mol, M.wt. 272.7, viscosity at 35˚C, 5.04 Cp and 1.5626. 25 D n 2.2. Methods 1) Infrared spectroscopy (IR) The IR analysis was carried out using a FTIR spec- trometer at wavelengths from 500 - 4000 cm–1 and transmittance % from 30 - 100. 2) Nuclear Magnetic Resonance (NMR) Proton NMR spectra in deuterated CHCL3 containing tetramethyl silane as an internal standard were recorded in an A varian instrument division EM-390 90M HZ NMR spectrometer. 3) Density The density was measured at the Egyptian Petroleum Research Institute (EPRI) using Instrument: 6890 plus G. C. Preparation of Allyl P-bromophenylether: One mole of para bromophenol, one mole of allyl bro- mide, one mole of anhydrous K2CO3 and 150 gm of ace- tone were mixed together in a round flasket. The reaction mixture was refluxed for 12 - 14 hours with stirring. The mixture kept to be cool. Potassium bromide was separa- ted by filtration. Cold water and ether were added to the reaction liquid and the organic layer was separated [9, 15,25]. The resulting organic layer was washed with brine (NaOH 10%), and then dried over anhydrous MgSO4. After removing the drying agent by filtration all the un- desirable materials were distilled off. The new allyl ether was collected under subatmospheric pressure of (20 - 23) mmHg, at 152˚C, in a yield of 75% wt. The new ether has the following properties: average mo- lecular weight: 213.4 g, colour: pale oily yellow liquid, solubility: soluble in CCl4, xylene, = 1.4508 and d25 = 0.9432. The infrared spectrum is given in Figure 1. which indicates that: the transmission absorbance could be due to: C-Br at 660 cm–1, -CH bending aromatic at 813 - 997 cm–1, -CO ether at 1235 cm–1 , C=C aromatic at 1421, CH2 bending at 1484 cm–1 , C=C aliphatic at 1692 cm–1, para aromatic subs. At 1742 - 1866 cm–1, CH2 stretching sym. At 2857 cm–1, CH asym. at 2919, and CH-Aromatic sym. at 3078. 25 D n Synthesis of 1,2,3,4,7,7, Hexachloro 5 parabromoph- enoxymethyl bicyclo 2,2,1heptene-2: In a round bottomed 50 ml flask, 0.075 mole (13.6 gm) of HCP was mixed with 0.025 mole of allyl p-bromo- phenylether. The flask was stoppered carefully and pla- ced in an oil bath for a constant reaction time of 6 hours at each of evaluated temperature (90˚C - 160˚C). The condensation reaction was carried out without using sol- vents or catalysts. The flask was removed from the oil bath, cooled, and opened. The unreacted diene, dieno- phile as well as the adduct were separated from the reac- tion mixture by careful fraction distillation. This was carried out under subatmospheric pressure of (20 - 23) mmHg [9,13-25]. After three days the adduct was obtained in its past form, then recrystallization took place using (1:3) mix- ture hexanemethylene chloride. The new adduct was pre- pared in its pure form as white yellowish needles, soluble in benzene, DMF and chloroform. Average molecular weight (486.7 gm), m.p. 120˚C - 122˚C, the infrared spectra-I.R. Figure 2 which indicat that the transmission Copyright © 2011 SciRes. IJOC 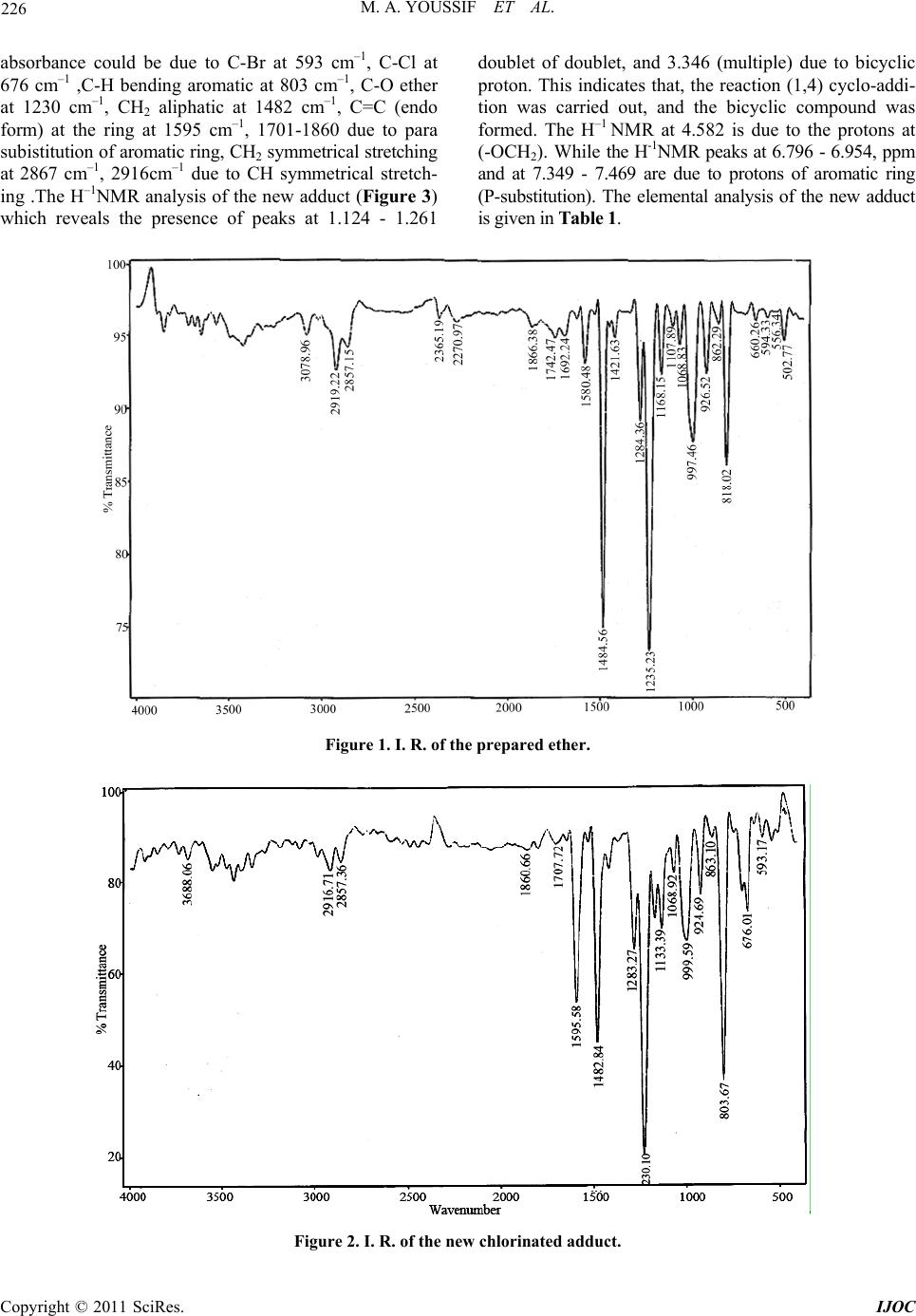 M. A. YOUSSIF ET AL. Copyright © 2011 SciRes. IJOC 226 absorbance could be due to C-Br at 593 cm–1, C-Cl at 676 cm–1 ,C-H bending aromatic at 803 cm–1, C-O ether at 1230 cm–1, CH2 aliphatic at 1482 cm–1, C=C (endo form) at the ring at 1595 cm–1, 1701-1860 due to para subistitution of aromatic ring, CH2 symmetrical stretching at 2867 cm–1, 2916cm–1 due to CH symmetrical stretch- ing .The H–1NMR analysis of the new adduct (Figure 3) which reveals the presence of peaks at 1.124 - 1.261 doublet of doublet, and 3.346 (multiple) due to bicyclic proton. This indicates that, the reaction (1,4) cyclo-addi- tion was carried out, and the bicyclic compound was formed. The H–1 NMR at 4.582 is due to the protons at (-OCH2). While the H-1NMR peaks at 6.796 - 6.954, ppm and at 7.349 - 7.469 are due to protons of aromatic ring (P-substitution). The elemental analysis of the new adduct is given in Table 1. Figure 1. I. R. of the prepared ether. Figure 2. I. R. of the new chlorinated adduct. 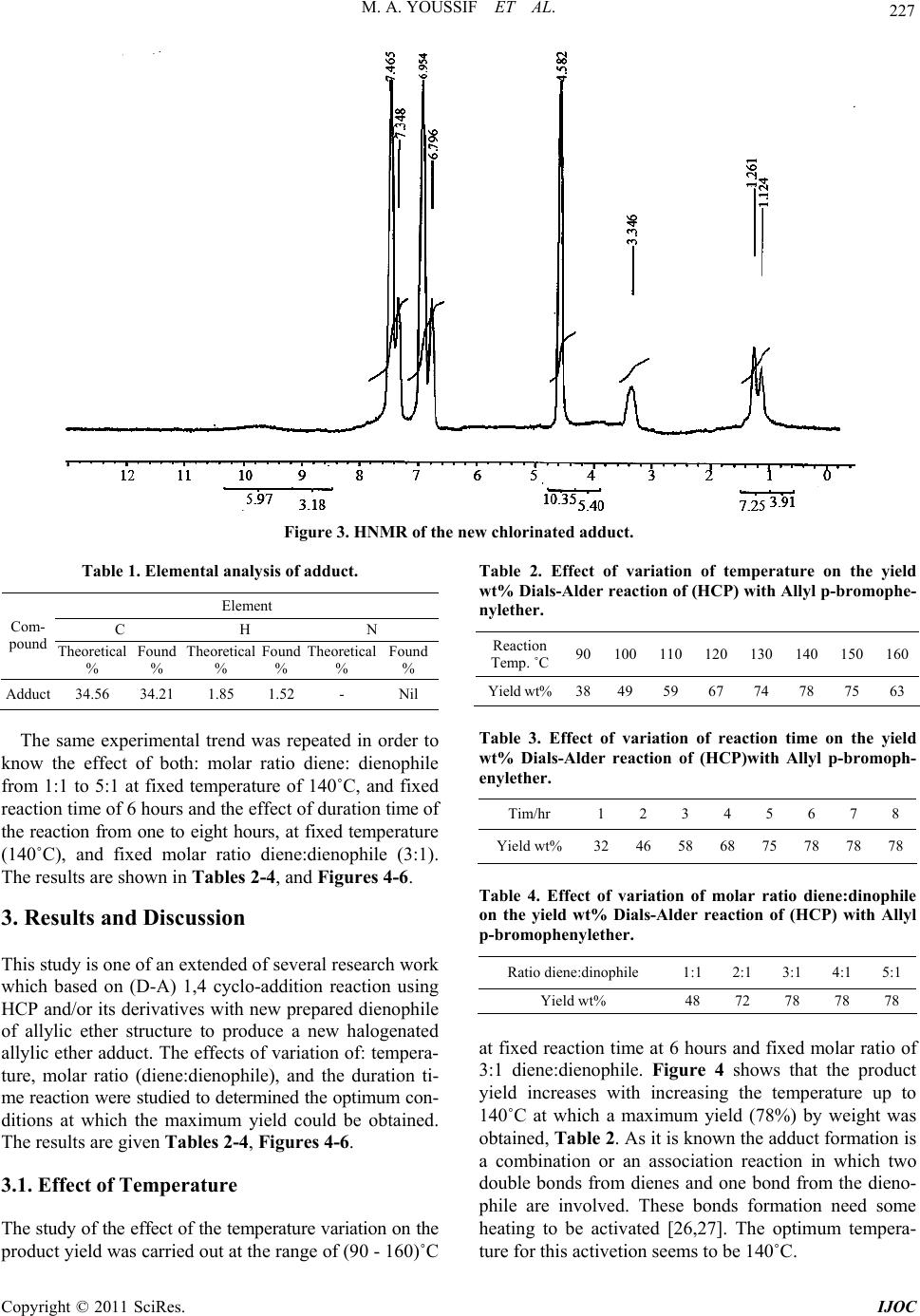 227 M. A. YOUSSIF ET AL. Figure 3. HNMR of the new chlorinated adduct. Table 1. Elemental analysis of adduct. Element C H N Com- pound Theoretical % Found % Theoretical % Found % Theoretical % Found % Adduct 34.56 34.21 1.85 1.52 - Nil The same experimental trend was repeated in order to know the effect of both: molar ratio diene: dienophile from 1:1 to 5:1 at fixed temperature of 140˚C, and fixed reaction time of 6 hours and the effect of duration time of the reaction from one to eight hours, at fixed temperature (140˚C), and fixed molar ratio diene:dienophile (3:1). The results are shown in Tables 2-4, and Figures 4-6. 3. Results and Discussion This study is one of an extended of several research work which based on (D-A) 1,4 cyclo-addition reaction using HCP and/or its derivatives with new prepared dienophile of allylic ether structure to produce a new halogenated allylic ether adduct. The effects of variation of: tempera- ture, molar ratio (diene:dienophile), and the duration ti- me reaction were studied to determined the optimum con- ditions at which the maximum yield could be obtained. The results are given Tables 2-4, Figures 4-6. 3.1. Effect of Temperature The study of the effect of the temperature variation on the product yield was carried out at the range of (90 - 160)˚C Table 2. Effect of variation of temperature on the yield wt% Dials-Alder reaction of (HCP) with Allyl p-bromophe- nylether. Reaction Temp. ˚C 90 100 110 120 130 140 150 160 Yield wt%3849 59 67 74 78 75 63 Table 3. Effect of variation of reaction time on the yield wt% Dials-Alder reaction of (HCP)with Allyl p-bromoph- enylether. Tim/hr 1 2 3 4 5 6 7 8 Yield wt% 32 46 58 68 75 78 78 78 Table 4. Effect of variation of molar ratio diene:dinophile on the yield wt% Dials-Alder reaction of (HCP) with Allyl p-bromophenylether . Ratio diene:dinophile 1:1 2:1 3:1 4:1 5:1 Yield wt% 48 72 78 78 78 at fixed reaction time at 6 hours and fixed molar ratio of 3:1 diene:dienophile. Figure 4 shows that the product yield increases with increasing the temperature up to 140˚C at which a maximum yield (78%) by weight was obtained, Tab le 2. As it is known the adduct formation is a combination or an association reaction in which two double bonds from dienes and one bond from the dieno- phile are involved. These bonds formation need some heating to be activated [26,27]. The optimum tempera- ture for this activetion seems to be 140˚C. Copyright © 2011 SciRes. IJOC 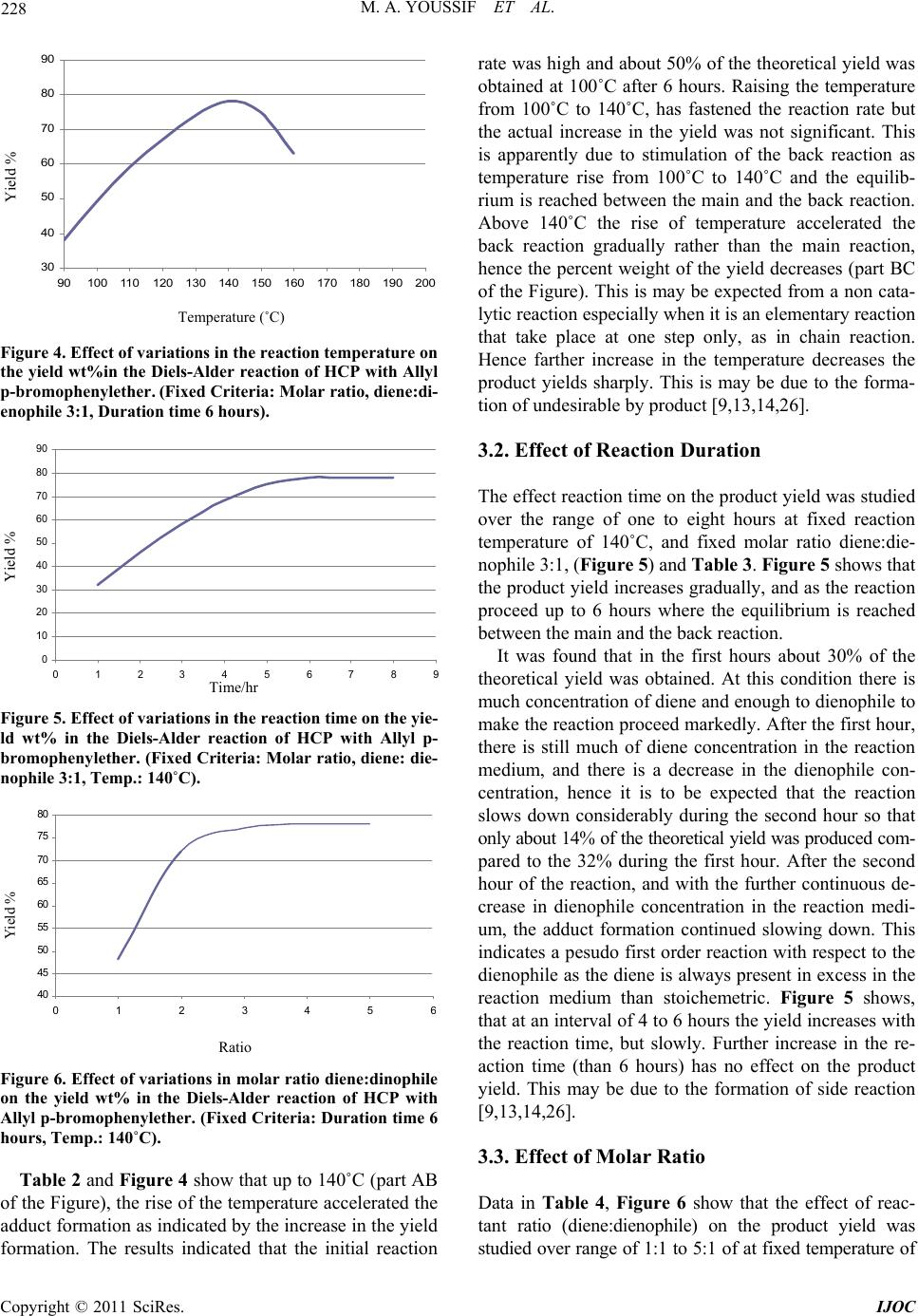 M. A. YOUSSIF ET AL. 228 Figure 4. Effect of variations in the reaction temperature on the yield wt%in the Diels-Alder reaction of HCP with Allyl p-bromophenylether . (Fixed Criteria: Molar ratio, diene:di- enophile 3:1, Duration time 6 hours). Figure 5. Effect of variations in the reaction time on the yie- ld wt% in the Diels-Alder reaction of HCP with Allyl p- bromophenylether. (Fixed Criteria: Molar ratio, diene: die- nophile 3:1, Temp.: 140˚C). Figure 6. Effect of variations in molar ratio diene:dinophile on the yield wt% in the Diels-Alder reaction of HCP with Allyl p-bromophenylether. (Fixed Criteria: Duration time 6 hours, Temp.: 140˚C). Table 2 and Figure 4 show that up to 140˚C (part AB of the Figure), the rise of the temperature accelerated the adduct formation as indicated by the increase in the yield formation. The results indicated that the initial reaction rate was high and about 50% of the theoretical yield was obtained at 100˚C after 6 hours. Raising the temperature from 100˚C to 140˚C, has fastened the reaction rate but the actual increase in the yield was not significant. This is apparently due to stimulation of the back reaction as temperature rise from 100˚C to 140˚C and the equilib- rium is reached between the main and the back reaction. Above 140˚C the rise of temperature accelerated the back reaction gradually rather than the main reaction, hence the percent weight of the yield decreases (part BC of the Figure). This is may be expected from a non cata- lytic reaction especially when it is an elementary reaction that take place at one step only, as in chain reaction. Hence farther increase in the temperature decreases the product yields sharply. This is may be due to the forma- tion of undesirable by product [9,13,14,26]. 30 40 50 60 70 80 90 90100 110120 130 140 150 160 170 180 190 200 Tem Yi eld Yield % eratu re °C Temperature (˚C) 3.2. Effect of Reaction Duration 0 10 20 30 40 50 60 70 80 90 0123456789 The effect reaction time on the product yield was studied over the range of one to eight hours at fixed reaction temperature of 140˚C, and fixed molar ratio diene:die- nophile 3:1, (Figure 5) and Table 3. Figure 5 shows that the product yield increases gradually, and as the reaction proceed up to 6 hours where the equilibrium is reached between the main and the back reaction. Yield% Yield % It was found that in the first hours about 30% of the theoretical yield was obtained. At this condition there is much concentration of diene and enough to dienophile to make the reaction proceed markedly. After the first hour, there is still much of diene concentration in the reaction medium, and there is a decrease in the dienophile con- centration, hence it is to be expected that the reaction slows down considerably during the second hour so that only about 14% of the theoretical yield was produced com- pared to the 32% during the first hour. After the second hour of the reaction, and with the further continuous de- crease in dienophile concentration in the reaction medi- um, the adduct formation continued slowing down. This indicates a pesudo first order reaction with respect to the dienophile as the diene is always present in excess in the reaction medium than stoichemetric. Figure 5 shows, that at an interval of 4 to 6 hours the yield increases with the reaction time, but slowly. Further increase in the re- action time (than 6 hours) has no effect on the product yield. This may be due to the formation of side reaction [9,13,14,26]. Tim/hr Time/hr 40 45 50 55 60 65 70 75 80 012345 Rati Yield % Ratio 3.3. Effect of Molar Ratio Data in Table 4, Figure 6 show that the effect of reac- tant ratio (diene:dienophile) on the product yield was studied over range of 1:1 to 5:1 of at fixed temperature of Copyright © 2011 SciRes. IJOC  229 M. A. YOUSSIF ET AL. 140˚C, and fixed reaction time of 6 hours. It was found that at equimolar ratio of reactants about (50%) of the theoretical yield was obtained. When was doubling the initial ratio of the diene under the same conditions about (70%) of the theoretical yield was obtained. At (3:1) mole ratio of diene:dienophile 78% of the product yield was obtained. Further increase in the initial molar ratio of the diene has no effect on the reaction rate. This is indicated that, this increase of the ratio of the diene does not affect the rate of the adduct formation, although it may be promote polymerization reaction leading to the produc- tion of resin. This is in concordance with [9,13,14,26]. It is known that the adduct formation reaction takes place between one molecule of diene and one molecule of dienophile. Data in Table 4 show that an initial equi- molar ratio of reactance seems to be the best ratio for such reaction to be proceeding with a good rate. How- ever, the presence of electron withdrawing chlorine atom in the diene molecule decreases the electron density at the dou- ble bonds, leading to a decrease in the reactivity of diene. So it must be need, for a much of diene in the reaction me- dium, in the form of a higher initial molar ratio of diene. The D-A reaction of HCP and p-bromo allyl ether is an exothermic reaction which takes place under autoge- netic pressure (closed system) to facilitate the reaction rate in absence of solvent or catalyst. The better yield is obtained by using an excess of one of component (di- ene). This is compatible with (Carrutuners). The new ether (dienophile) is prepared and characterized by av- erage molecular weight (213.4 g), density (0.9432) and infrared spectra Figure 1. The new adduct was prepared in its pure form as white yellowish needles and its structure was confirmed by molecular weight, IR Fig- ure 2 and H1 NMR spectra Figure 3 and elemental analysis Table 1. To compare the effect of kind of the substituent in the dienophilic molecule of allylic structure on (D-A) 1,4 cycloaddition with (HCP). The following explanation is carried out. The maximum product yield in this present work is relatively higher than that obtained in the previ- ous work [26], although the two dienophiles are the same allylic structure. This implies that, the reason is related to kind of the substituent in the dienophile molecule has the major effect on the electron density of the terminal dou- ble bond (alpha position) of the side chain, and conse- quently on the product yield. Our explanation is that the presence of methyl group in ref [25] which has an electron donating activator group (has +M, +I effect) which pushes the electron by means of mesomeric effect to the benzylic ring, and consequen- tly by resonance to the terminal alkenyl double bond. Consequently the reactivity of the dienophilic olefin to react in the (D-A) 1,4 cyclo-addition relatively decreas- ing. Whereas in the present work the bromine atom in the dienophilic molecules has (–I, +M) effect due to the fact that when there is a conflict between the mesomeric ef- fect (M) and inductive effect (I), the conflict between the mesomeric effect predominates in most cases, except in case of halogens, the (–I) effect is more powerful than (+M) effect. So, the bromine atom pulls the electrons from the adjacent benzylic carbon, then by resonance it pulls the electron from the alkenyl double bond. Thus the electron density on the terminal double bond of the al- kynl chain decreases. So it increase the reactivity of the olefinic molecule (dienophile) to be reactive and the (D-A) 1,4 cyclo addition is more predominates than the previous work [25]. On the other hand study of (D-A) 1,4 cycloaddition was carried out using dimethoxy tetrachloro cyclopeta- diene (DMTCP), which is a drevative of HCP, with p- chloro-allylether [9]. It was found that, the maximum yield is (84.6) at optimum conditions (120˚C, 5 hours and 2:1 molar ratio diene:dienophile). The maximum yield in the previous study is higher than the present one. Although the two dienophiles in the studies share the same allylic structures, their substituent’s in the two di- enophiles are approximately of equal activity, and their positions in the dienophilic molecules are in the same (P- positions). So the difference in the product yields is re- lated to the nature of the diene molecule: the presence of two electron donating groups (methoxy groups) in the study (10) increase the electron density on the double bond of the diene molecule, and thus it increases its reac- tivity. Whereas the presence of six chlorine atoms in HCP diene molecule decreases the electron density on its double bond, and hence its reactivity decreases compare- ing to the other diene (DMTCP). This conclusion is in concordance with that mentioned in ref [9,13]. 3.4. Antibacterial Activity As a result of the numerous problems caused by sulfate reducing bacteria in oil and gas production operations, aggressive measures have been taken to monitor and control bacterial populations. Measurements of bacterial growth are not usually considered until after corrosion failures point due to microbial induced corrosion. By the time microbial induced corrosion is discovered, exten- sive and costly damage to the operating systems has of- ten already occurred [28]. The antibacterial activities of the synthesized compound was tested against sulfur re- ducing bacteria Desulfomonas p igra. The biocidal activi- ties of the synthesized compound against SRB strain were investigated at different concentrations using the serial dilution method, Table 5. Both the minimum in- hibitory concentrations (MIC) and the lethal concentra- Copyright © 2011 SciRes. IJOC  M. A. YOUSSIF ET AL. 230 Table 5. The antibacterial activities of the synthesized com- pound. Concentration SRB Count (colony/ml sample) 10–1 00.00 10–2 00.00 10–3 00.00 10–4 268 10–5 4.98 × 103 Control 7.72 × 103 tions (LC) was detected, the results are shown in Table 5. The minimum inhibitory concentration (MIC) is the lowest concentration of the compound that didn’t show microbial growth [29]. This means that, the lower the MIC value of a compound, the higher is its activity. The adduct is efficient compound since its MIC value is (0.00073), and has three lethal concentration val- ues (10–1 and 10–2, 10–3). King [30] has showing greater impacts of 2,4-DBP on aerobic bacteria, and the behavior of these compounds as potent uncouplers of oxidative phosphorylation [31,32]. Antibiotic activity towards microbes and meiofauna [30, 33-37] and nephrotoxic and hepatotoxic activity in higher organisms [38,39] have previously been documented for bromophenols and other brorninated metabolites. The in- hibition pattern for sulfate reduction suggests that sul- fate-reducing bacteria are sensitive bromo phenols, though not to the extent noted for aerobes. The effect of bromo- phenols on sulfate reduction appears to be tempered by the rapid dehalogenation bromophenols under anoxic condi- tions [30,40]. The interaction between adduct molecules and cellular membrane causes, a strong damage of the selective permeability of these membranes which disturbs the metabolic pathway within the cytoplasm. 4. Conclusions • A new dienophile had been allylic type (Allyl P- bromophenylether) was prepared and its structure had been confirmed. • The Diels-A1, 4-addition reaction has been studied using a diene, H.Cl.C.D., and dienophile, allyl P- bromophenylether. • The condensation reaction took place under pressure without catalyst or solvent. • Reaction conditions were studied including, tem- perature 90˚C - 160˚C, reactant molar ratio 1:1 to 5:1, and reaction time 1to 8 hours. • The structure of the new adduct was confirmed by; molecular weight, H1 NMR and IR spectra. • The optimum conditions for maximum product yield were found to be as following; temperature = 140˚C, reactant molar ratio = 3:1 with a reaction time of six hours. • Maximum adduct yield was = 78%. • The new adduct revealed very high effect as sul- fate-reducing bacteria (SRB). 5. References [1] T. J. Brocksom, Joanita, M. L. Ferreira and U. Brocksom, “The Diels-Alder Reaction: An Update,” Journal of the Brazilian Chemical Society, Vol. 12, No. 5, 2001, pp. 597-662. [2] W. Carruthers, “Cycloaddition Reaction in Organic Syn- thesis,” Pergamon, Oxford, 1990. [3] H. B. Kagan and O. Riant, “Chtalytic Asymmetric Diels- Alder Reaction,” Chemical Reviews, Vol. 92, 1992, pp. 1007-1019. doi:10.1021/cr00013a013 [4] K. C. Nicolaou, S. A. Snyder, T. Montagnon and G. Vas- silikogiannakis, “The Diels-Alder Reaction in Total Syn- thesis,” Angewandte Chemie International Edition, Vol. 41, No. 10, 2002, pp. 1668-1698. doi:10.1002/1521-3773(20020517)41:10<1668::AID-AN IE1668>3.0.CO;2-Z [5] S. A. Kozmin, S. He and V. H. Rawal, “Organic Synthe- ses,” Coll , Vol. 10, 2004, p. 442. [6] V. L. Júnior and S. M. G. Valdo, “Niobium Pentachloride Activation of Enone Derivatives: Diels-Alder and Con- jugate Addition Products Mauricio Gomes Constantino,” Molecules, Vol. 7, 2002, pp. 456-465. [7] J. March, “Advanced Organic Chemistry: Reactions, Mechanisms, and Structure,” 2nd Edition, John Wiley & Sons, New York, 1977, pp. 5-51. [8] T. L. Finar, “Organic Chemistry,” 6th Edition, ELBS, Singapore, 1973. [9] M. A. Youssif, “Condensation of 5,5-Dimethoxy Tetra- chlorocyclopentadiene with Dienophiles of Allylic Types,” Ph.D. Thesis, Pub Azu Pet Institution, Buku, 1983. [10] M. A. Hassan and M. A. Youssif, “Proportion of Some Fire-Resistant Coating Containing Diglycreal-Borat and Hexachlorocyclopentadiene,” Egyptian Journal of Petro- leum, Vol. 1, 1992, pp. 35-40. [11] A. L. Saad, M. A. Hassan, M. A. Youssif and M. G. Ah- med, “Studies of Electrical Properties of Some Fire Re- tarding Poly (Vinyl Chloride) Compositions,” Journal of Applied Polymer Science, Vol. 65, No. 1, 1997, pp. 27-35. doi:10.1002/(SICI)1097-4628(19970705)65:1<27::AID- APP4>3.0.CO;2-S [12] M. A. Youssif, A. M. Badawi, E. Noaman, A. M. Maz- roua, S. M. Madboly and N. A. Mansour, “Synthesis of a Novel Hexachloro Amino Ester Adduct as Biologically Active Agents,” Egyptian Journal of Petroleum, Vol. 16, No. 2, 2007, pp. 1-14. [13] M. A. Youssif, A. M. Badawi, E. Noaman, A. M. Maz- roua, S. M. Madboly and N. A. Mansour, “Synthesis and Optimization of New Chlorinated Antitumor Mono Ep- oxy Adduct,” Egyptian Journal of Petroleum, Vol. 16, No. 2, 2007, pp. 15-28. [14] A. M. Mazroua, A. M. Hassan, and M. A. Youssif, Copyright © 2011 SciRes. IJOC  231 M. A. YOUSSIF ET AL. “Study on 1,4 Cycloaddition Reaction. Fire Retardant Halogenated Organic Adducts and Poly (Vinyl Chloride) Textile Compositions,” Egyptian Journal of Petroleum, Vol. 13, No. 2, 2004, pp. 23-29. [15] F. F. Mogazulenusku, T. A. Omadov and A. A. Hussie- nov, “Condensation of Hexachlorocyclopentadiene with Orthochlorohpenyl Allyl Ether,” Journal of Agricultural and Food Chemistry, Vol. 4, 1980, pp. 51-54. [16] R. Cord-Ruwish, R. W. Kleinitz and F. Widdel, “Sul- fate-Reducing Bacteria and Their Activities in Oil Pro- duction,” Journal of Petroleum Technology, Vol. 39, 1987, pp. 97-106. [17] C. Hubert, M. Nemati, G. Jenneman and G. Voordouw, “Corrosion Risk Associated with Microbial Souring Con- trol Using Nitrate or Nitrite,” Applied Microbiology and Biotechnology, Vol. 68, No. 2, 2005, pp. 272-282. doi:10.1007/s00253-005-1897-2 [18] M. Nemati, G. E. Jenneman and G. Voordouw, “Mecha- nistic Study of Microbial Control of Hydrogen Sulfide Production in Oil Reservoirs,” Biotechnology and Bioen- gineering, Vol. 74, No. 5, 2001, pp. 424-434. doi:10.1002/bit.1133 [19] F. B. Cooling, C. L. Maloney, E. Negel, J. Tabinowski and J. M. Odom, “Inhibition of Sulfate Respiration by 1,8-Dihydroxyanthraquinone and Other Anthraquinone Derivatives,” Applied Microbiology and Biotechnology, Vol. 62, 1996, pp. 2999-3004. [20] L. R. Gardner and P. S. Stewart, “Action of Glutaralde- hyde and Nitrite against Sulfate-Reducing Bacterial Biofilms,” Journal of Industrial Microbiology & Bio- technology, Vol. 29, No. 6, 2002, pp. 354-360. doi:10.1038/sj.jim.7000284 [21] J. Larsen, “Downhole Nitrate Applications to Control Sulfate Reducing Bacteria Activity and Reservoir Sour- ing,” Corrosion, Vol. 25, 2002, pp. 1-10. [22] F. Sanogu, R. Javaherdashti and N. Aksiiz, “Corrosion of a Drilling Pipe Steel in an Environment Containing Sul- phate-Reducing Bacteria,” International Journal of Pres- sure Vessels and Piping, Vol. 73, No. 2, 1997, pp. 127- 131. doi:10.1016/S0308-0161(97)00022-7 [23] E. I. Sungur and A. Cotuk, “Microbial Corrosion of Gal- vanized Steel by a Fresh Water Strain of Sulphate Re- ducing Bacteria (Desulfovibrio sp.),” Corrosion Science, Vol. 49, No. 3, 2007, pp. 1097-1109. doi:10.1016/j.corsci.2006.05.050 [24] Z. Yuxia, Z. Tao, M. Wanshan, Z. Haibin and C. Suifeng, “Synthesis, Structural Determination and Antibacterial Activity of Compounds Derived from Vanillin and 4- Aminoantipyrine,” Hauxue Shiji, Vol. 24, 2002, p. 117. [25] M. A. Youssif, “The Condensation Reaction of Hexa- chlorocyclopentadiene with Allyl P-Tolylether,” Faculty of Science Zagazig University, 1991, pp. 98-109. [26] A. A. Hussienov, “Condensation of hexachlorocyclopen- tadiene with Dienophiles of Allylic Types,” Ph.D. Thesis, Pub Azu Pet Institution, Buku, 1982. [27] W. Carrutners, “Modern Methods of Organic Chemistry Synthesis,” 3rd Edition, Cambridge University Press, Cam- bridge, 1996, p.183. [28] H. H. Downs, “Behavior of Ni-Base Alloy 625 in Metha- nol Supercritical Water Systems,” 15th International Corrosion Congress, Granada, September 2002. [29] I. Argyropoulou, A. Geronikaki, V. Paola and F. Zani, “Arkivoc,” Vol. vi, 2009, pp. 89-102. [30] G. M. King, “Dehalogenation in Marine Sediments Con- taining Natural Sources of Halophenols,” Applied and Environmental Microbiology, Vol. 54, No. 12, 1988, pp. 3079-3085. [31] M. Stockdale and M. J. Selwyn, “Influence of Ring Sub- stituents on the Action of Phenols on Some Dehydro- genases, Phosphokinases and the Soluble ATPase from Mtochondria,” European Journal of Biochemistry, Vol. 21, No. 3, 1971, pp. 416-423. doi:10.1111/j.1432-1033.1971.tb01486.x [32] M. Stockdale and M. J. Selwyn, “lnfluence of Ring Sub- stituents on the Activity of Phenols as Inhibitors and Un- couplers of Mitochondrial Respiration,” European Jour- nal of Biochemistry, Vol. 21, No. 4, 1971, pp. 565-574. doi:10.1111/j.1432-1033.1971.tb01502.x [33] J. Azariah, V. N. R. Rao and V. T. Sridharan, “Effect of the Epidermal Secretions of Hemichordate. Ptychodera Flava on Growrn of Amphora Coffeaeformis and Cycio- reiia Meneghiniana (Diatoms),” Proceedings of the In- dian Academy of Sciences, Vol. 87B, 1978, pp. 145-149. [34] G. M. King, “Inhibition of Microbial Activity in Marine Sediments by a Bromophenol from a Hemichordate,” Nature, Vol. 323, 1986, pp. 257-259. doi:10.1038/323257a0 [35] S. J. Weiss, S. T. Test, C. M. Eckmann, D. Roos and S. Regiani, “Brominating Oxidants Generated by Human Eosinophils,” Science, Vol. 234, No. 4773, 1986, pp. 200- 203. doi:10.1126/science.3018933 [36] P. Jensen, R. Emnch and K. Weber, “Brominated Me- tabolltes and Reduced Numbers of Meiofauna Organisms in the Burrow Wall Lining of the Deep-Sea Enteropneust Stereobalanus Canadensis,” Deep Sea Research, Vol. 39, 1992, pp. 2147-2253. doi:10.1016/0198-0149(92)90067-4 [37] R. Teeyapant, H. J. Woerdenbag, P. Kreis, J. Hacker, et al., “Antibiotic and Cytotoxlc Activity of Brominated Compounds from the Marine Sponge Verongia Aero- phoba,” Journal of Biosciences, Vol. 48, No. 11-12, 1993, pp. 939-945. [38] B. D. Kerger, S. M. Roberts and R. C. James, “Compari- son of Human and Mouse Liver Microsomal Metabolism of Bromobenzene and Chlorobenzene to 2- and 4-Halo- phenols,” Drug Metabolism and Disposition, Vol. 16, No. 5, 1988, pp. 672-677. [39] E. Caslllas and M. S. Myers, “Effect of Bromobenzene and Obromophenol on Kidney and Hver of English Sole (Parophrys Vetulus),” Comparative Biochemistry and Physiology, Vol. 93, No. 1, 1989, pp. 43-48. doi:10.1016/0742-8413(89)90008-X [40] C. C. Steward, T. C. Dixon, Y. P. Chen and C. R. Lovell, “Enrichment and Isolation of a Reductively Debroml- Copyright © 2011 SciRes. IJOC  M. A. YOUSSIF ET AL. Copyright © 2011 SciRes. IJOC 232 natlng Bactenum from the Burrow of a Bromometabo- lite-Producing Marine Hemichordate,” Canadian Journal of Microbiology, Vol. 41, No. 7, 1995, pp. 637-642. doi:10.1139/m95-086
|