Food and Nutrition Sciences
Vol. 3 No. 2 (2012) , Article ID: 17528 , 6 pages DOI:10.4236/fns.2012.32036
Effect of Opium on Lipid Profile and Expression of Liver X Receptor Alpha (LXRα) in Normolipidemic Mouse
![]()
1Department of Biochemistry, Afzalipour School of Medicine, Kerman University of Medical Sciences, Kerman, Iran; 2Physiology Research Centre, Afzalipour School of Medicine, Kerman University of Medical Sciences, Kerman, Iran; 3Department of Laboratory Science, Islamic Azad University, Borūjerd Branch, Borūjerd, Iran; 4Afzalipour School of Medicine, Kerman University of Medical Sciences, Kerman, Iran; 5Department of Basic Science, Tehran Payame Noor University, Tehran, Iran.
Email: *7abbasi@gmail.com
Received November 2nd, 2011; revised December 19th, 2011; accepted December 27th, 2011
Keywords: Opium; Cholesterol; Atherosclerosis; LXR
ABSTRACT
Many people believe that opium has beneficial effects on lipid profile which results in reduced atherosclerosis. Opium contains several alkaloids and biological active components, which some of them are used for atherosclerosis treatment. The liver X receptor α (LXRα) is an important regulator of cholesterol and glucose homeostasis that belongs to the nuclear receptor superfamily. This study aimed to investigate the effects of opium on glucose, lipid profile and LXRα expression. Sixteen N-mary mice randomly were divided into two groups (control and addict), and were studied for one month. Serum lipid profile, Fasting blood sugar (FBS), Aspartate aminotransferase (AST) and Alanine aminotransferase (ALT) were determined. Also LXR mRNA and protein levels were determined by Reverse Transcription PCR and western blotting. This study showed that opium significantly reduced total cholesterol (P < 0.05), While the difference in blood glucose, triglyceride (TG), High-density lipoprotein cholesterol (HDL-c), Low-density lipoprotein cholesterol(LDL-c) and Very low-density lipoprotein cholesterol(VLDL-c), as well as AST and ALT between addict and control groups were not significant. More importantly, LXR protein and mRNA levels significantly increased (P < 0.05) in intestine of addict group in comparison with control, while the change in LXR protein and mRNA in the liver were not significant compared with control. The results of this study showed that opium addiction reduced total cholesterol and increased LXR expression in intestine. Further researches need to determine effective components.
1. Introduction
In some Asian countries, many people believe that opium leads to reduction in lipid profile and blood glucose. Therefore researchers currently have done more and more investigation to prove or deny these effects. Many studs have shown that opium reduces lipid profile and blood glucose in normal condition [1]. On the other hand some researchers have shown that opium has no effect on these profiles in haypercholesterolemic models [1]. Opium contains eighty different alkaloids and biologically active components [2] and morphine is a major component of opium. lso it has been shown that Phosphatidyl inositol kinase and opioid receptors are involved in the useful effects of these drugs [1]. The mechanism of lipid and glucose reduction of opium is unknown. Cholesterol or oxidized cholesterol performs as a ligand for the nuclear liver x receptor (LXR) and regulated expression of genes involved in cholesterol, and lipid as well as glucose metabolism. LXR have two isoforms; LXRα and LXRβ. LXRα predominant expressed in the liver, intestine, adipose and macrophages, whereas LXRβ has a ubiquitous distribution. The ability of LXRs to control genes involved in cholesterol, triglyceride and glucose metabolism make them striking targets for the drug development for treatment of atherosclerosis and diabetes [3,4]. LXRα with the decrease of intestinal cholesterol absorption, and also with regulation of multiple steps in reverse cholesterol transport has an important role in atherosclerotic plaque reduction. Moreover LXRα has key role in cholesterol and triglyceride metabolism in liver [3,5]. This study intended to investigate the effect of opium on glucose, lipid profile, and LXR expression in mice.
2. Materials and Methods
2.1. Experimental Design
A total 16 N-mary mice were used in this study. The mice (28 g - 32 g) were randomly divided into two groups as control and addicted. The animals acclimatized for 2 weeks in animal room conditions and they were maintained under standard laboratory condition at 22˚C ± 1˚C, and normal photo period (12 h dark/12 h light) used for experiment. The experimental protocol has been approved by the Animal Experimentation Ethic Committee of Kerman Medical University.
The addict mice received the opium (Court of Justice, Kerman, Iran). Opium was suspended in 0.4 ml of hot water, and then was cold and administered by gavage with starting dose of 5 mg/kg twice a day and gradually increased to 40 mg/kg for 8 days and remains steady over the month. GC-mass spectrometry analysis of this opium showed more than 30% alkaloids (from which morphine 16%, thebaine 4.4%, papaverine 3.2% codeine 5.5%, were the most abundant of them) and the rest consisted of non-alkaloidal organic and non-organic substances from which 13.5% was water. After 12 h of fasting, mice were anesthetized by injection of sodium thiopental (50 mg/kg, IP) and sacrificed [1,6]. Liver and intestine removed immediately and washed with Phosphate buffered saline (PBS). Also blood was collected from heart and serum was obtained by centrifugation at 3000g for 10 min and stored at –20˚C until assay. High-density lipoprotein cholesterol (HDL-c), total cholesterol (TC), and triglycerides (TG) were measured enzymatically. Fasting blood glucose was measured by a glucometer (Roche Applied Science) [7].
2.2. Semiquantitative RT-PCR
RNA from liver and intestine were extracted with Accuzol Reagent (Bioneer, Korea) according to the manufacturers’ protocol. Briefly, 100 mg of tissue was homogenized in 1 ml reagent, and then 200 µl of chloroform was added. After vigorous shake the mix was incubated on ice for 5 minutes and centrifuged at 12,000 rpm for 15 minutes at 4˚C, an equal amount of isopropyl alcohol was added to aqueous phase and mixed and incubated at –20˚C for 10 minutes. After centrifuged at 12,000 rpm for 10 minutes at 4˚C, 1 ml of 80% ethanol was added to supernatant and mixed. After that tube was then centrifuged and supernatant was cautiously removed. Pellet was dried. RNA was dissolved in RNase-free water, and incubated for 10 minutes at 56˚C and stored at –70˚C till use. Synthesis of cDNA was performed according to the manufacturer’s protocol (Fermentas, Lithuania). Briefly, 2 µg of Template RNA, 1 µl of oligo dT primer and DEPC-treated water (to reach a total volume of 12 ul) were added into a sterile and nuclease-free tube on ice. Then mixed and centrifuged for a moment and incubated 5 minutes at 65˚C. The tubes were chilled on ice, and then 4 µl of 5× reaction buffer, 1 µl of Ribo Lock™ RNase Inhibitor, 2 µl of 10 mM dNTP, 1 µl of RevertAid™ M-MuLV Reverse Transcriptase were added, mixed and centrifuged, and then incubated at 42˚C for 60 minutes. The reaction was ended by heating the tube at 70˚C for 5 min. For semiquantitative RT-PCR reaction 2 µl cDNA, 1 µl forward primer (Pmol/µl), 1 µl reverse primer (Pmol/µl), 13 µl PCR Master Mix (Cinnagen, Iran) and 8 µl deionized water were added into a sterile tube on ice, and centrifuged for a short time. Thirty five cycles of PCR amplification were achieved with denaturation at 95˚C for 30 s, annealing at 63˚C for 30 s, and extension at 72˚C for 30 s using a PCR machine. All reactions were completed with a single additional cycle at 72˚C for 5 minutes. The samples were electrophoresed on a 2.5% agarose gel and visualized by ethidium bromide staining. The following primers were used in this study [8,9] Beta actin Primer; F: 5’-TGG AAT CCT GTG GCA TCC ATG AAA C-3’ and R: 5’-TAA AAC GCA GCT CAG TAA CAG TCC G-3’. LXR alpha primer; F: 5’-GCG TCC ATT CAG AGC AAG TGT-3’ and R: 5’-TCA CTC GTG GAC ATC CCA GAT-3’.
2.3. Western Blotting
About 50 mg of liver and intestine samples were homogenized in 700 µl RIPA buffer containing 1% protein inhibitor cocktail (Santa Cruz, USA) and 1 µM PMSF. The homogenate samples centrifuged at 14,000 rpm at 4˚C for 15 minutes. After measured by Bradford method, 150 μg of proteins were separated onto a 12.5% SDS-PAGE gel and transferred onto a PVDF membrane (Roche Applied Science) [10,11]. Then Membrane was blocked with 3% non-fat dried milk in Tris-buffered saline with Tween 20 (TBS-T) (Roche Applied Science) for 2 hours. After blocking the membrane incubated with primary antibody for 1.5 hours with anti rabbit polyclonal antibody LXR (1: 300 dilutions, Novus Biological) at room temperature. After washing in TBS-T, the membrane incubated with horseradish peroxidase-conjugated secondary antibody (1: 10,000 dilution, Roche Applied Science) at room temperature for 1.5 hours. Immunoreactive bands were visualized using ECL kit (Roche Applied Science). Band densities were determined with Lab Work analyzing software (UVP, UK) and expressed as the ratio of β-Actin (Cell Signaling Technology Inc., Beverly, MA, USA; 1:1000) [9].
2.4. Statistical Analysis
All results are presented as mean ± S.E.M. Statistical analysis of the results were done with Student’s t test. p values of less than 0.05 were considered statistically significant.
3. Results and Discussion
3.1. Lipid Profile and Blood Glucose
Table 1 shows the effect of opium on blood glucose and lipid profile. No significant differences were observed in plasma TG, VLDL-c, HDL-c, AST and ALT as well as blood glucose between the opium and vehicle groups. However, there was a significant decrease (p < 0.05) in total cholesterol in opium group as compared to normal rats (114 mg/dl ± 4.4 mg/dl vs 96 ± 4.3 in opium vs vehicle treated mice respectively, p < 0.05) (Table 1).
In this study total cholesterol levels reduced significantly by 14 percent. Although TG reduced by opium but, were not significant compared to control. Marmor et al. shown that long-term exposure to opiates considerably reduced coronary artery disease and consequently associated with decreased incidence of fatal myocardial infarcttions [1]. Fatemi SS et al. showed that cholesterol significantly reduced in opium addicts compared with normal group, they also reported that cholesterol, triglyceride and LDL-c significantly reduced in opium extracts compared with normal group [11].
It is worthy of note that the difference in the ALT, AST, blood glucose and HDL-c levels between the two groups were not significant in this study. Furthermore many studies have shown that opium reduced these profiles while they were not significant [2,11]. In the clinical setting serum ALT elevations have been used as a substitute biomarker for steatohepatitis. Therefore ALT is an enzyme mostly produced in hepatic cells, we monitored serum ALT as a biomarker to assess the grading of hepatocellular damage [12]. In this study not only ALT activity had not changed, but AST and ALP remained stable.
Mohamadi et al. showed that compared with control, in the hypercholesterolemic and hypercholesterolemic-addicted rabbits, triglycerides, LDL-c and cholesterol, levels were significantly increased while in addicted rabbit (chow diet) compared with control total cholesterol and LDL-c significantly reduced [1].
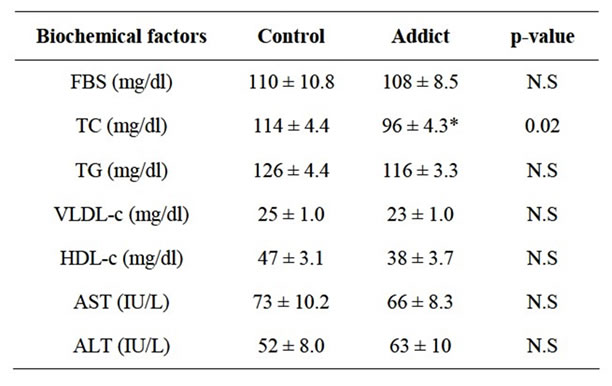
Table 1. Comparison of biochemical factors between addicts and control groups.
3.2. The Effect of Opium on Level of LXRα mRNA and Protein
Intestinal LXRα mRNA significantly increased in addicted mice (opium group) in comparison with control (p < 0.05) (Figure 1), whereas administration of opium had no effect on the mRNA level of LXRα in the liver compared with control animals (Figure 2). Our results showed a significant increase of intestinal LXRα protein in addicted animals with respect to control mice (p < 0.05) (Figure 3), while change in the level of LXRα protein in liver of the addict group in comparison with control mice were not significant (Figure 4).
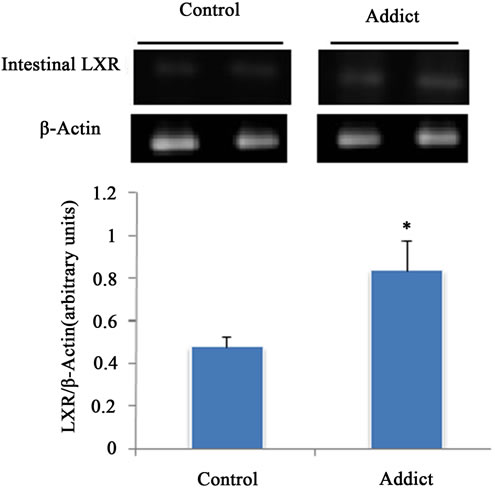
Figure 1. Effect of opium on LXR mRNA expression in male mice intestine (n = 6). LXR mRNA expression significantly increased in opium group. *P < 0.05 considered as signifycant compared with control. Data are presented as means ± SEM.
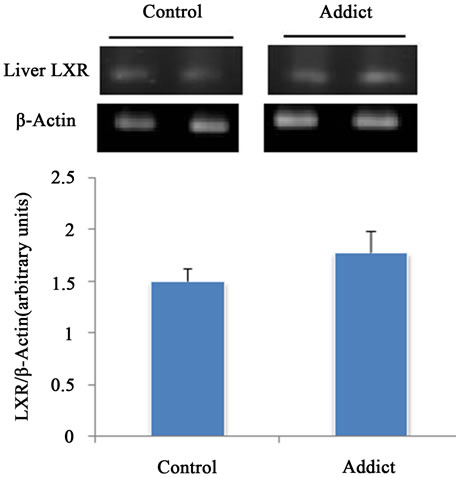
Figure 2. Effect of opium on LXR mRNA expression in male mice liver (n = 6). The different between control and addict groups was not significant. *P < 0.05 considered as significant compared with control. Data are presented as means ± SEM.
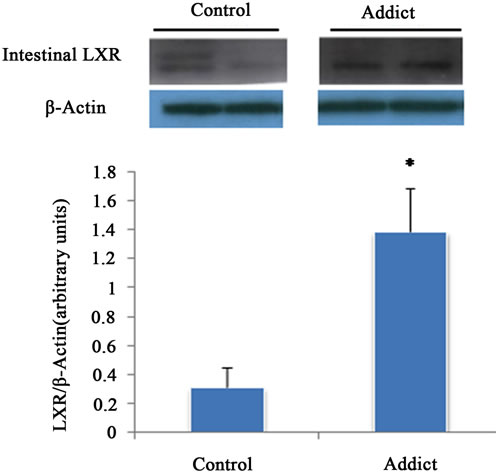
Figure 3. Effect of opium on LXR protein expression in male mice intestine (n = 6). LXR protein expression significantly increased in opium group. *P < 0.05 considered as significant compared with control. Data are presented as means ± SEM.
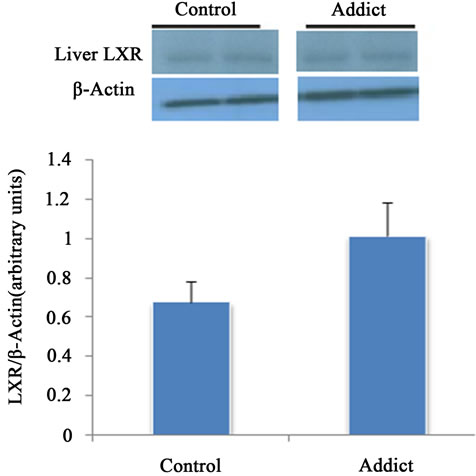
Figure 4. Effect of opium on LXR protein expression in male mice liver (n = 6). The different between control and addict groups was not significant. *P < 0.05 considered as significant compared with control. Data are presented as means ± SEM.
Although cholesterol metabolism regulated in different ways but LXRs have vital role. Brayan LA, et al. showed that the nuclear receptors LXRα and LXRβ played a role in the coordinated regulation of lipid and glucose metabolism. They have shown that activation of LXRs with synthetic agonist leads to the suppression of genes involved in gluconeogenesis and induction of hepatic glucokinase expression and insulin-sensitive glucose transporter 4 (GLUT4) in adipose tissue [13]. In this experiment we showed that opium significantly increased LXRα expression in intestine while there was no change in liver.
Niemann-Pick C1 Like 1 (NPC1L1) is cholesterol transporter which expressed in the brush border membrane of enterocytes in the small intestine. Altmann, SW et al. have shown that cholesterol absorption was decreased by 70% - 90% in knocking out models of NPC1L1wich was likely to reduction when wild type animals were treated with an ezetimibe [14]. Duval C et al. with using a human enterocyte cell line have shown that NPC1L1 expression is down-regulated by LXR in intestine [15]. Furthermore ATP-binding cassette (ABC) transporter proteins, G5 and G8 have a critical role in control of sterol absorption. ABCG5 and ABCG8 are localized at the brush border membrane of enterocytes and act as practical heterodimers [3]. Many studies have shown that over expression of ABCG5 and ABCG8 in mice as well as pharmacological stimulation of their expression did lead to a markedly decreased fractional cholesterol absorption [4], also demonstrated that these transporters played a role in control of cholesterol absorption [3]. It has been shown that LXRα agonists inhibit intestinal cholesterol absorption in mice. Repa JJ et al. initially suggested that this effect may be due to an increase of ABCA1 gene expression [16]. LXR activation is known to decrease cholesterol absorption in vivo via a mechanism involving ABCG5/ABCG8 up-regulations in the small intestine [16]. Furthermore, LXR activation has shown reduced NPC1L1 expression in the duodenum [17]. Treatment by LXR agonists lead to severe hepatic steatosis development in mice. Hepatic steatosis is associated with hepatic insulin resistance and type 2 diabetes [5]. Many studies have shown that this effect is because of up-regulation of lipogenic genes such as fatty acid synthase (FAS), acetyl-coA carboxylase (ACC) and stearoyl-coenzyme A desaturase 1 (SCD1) (a rate limiting enzyme necessary for the biosynthesis of monounsaturated acids) and carbohydrate response element binding protein (ChREBP) (a transcription factor that enhanced conversion of carbohydrates into lipids in liver) [15,5]. Besides of hepatic steatosis, Schultz JR et al. reported that pharmacological LXR activation in mice liver increased plasma triglycerides [18]. On the other hand, Choe SS et al. showed that persistent activation with LXR agonists lead to lipid overloading in pancreatic β-cells and then caused severe β-cell dysfunction and apoptosis, a major process in the development of type 2 diabetes [19]. In this study although opium increased LXR in intestine but did not change it in liver.
4. Conclusion
As indicated above, LXR agonists by down-regulation of NPC1L1, as well as up-regulation of ABCG5, ABCG8 and ABCA1 considerably decrease intestinal cholesterol absorption. But in this study we didn’t measure the levels of these genes, hence further investigations should determine the effect of opium on ABCG5, ABCG8 and ABCA1 as well as NPC1L1 levels in intestine of normal and addicted mice, on the other hand opium contains about eighty different alkaloids which some of them have excellent properties and some other have harmful effects. Therefore, the lipid lowering effect of opium perhaps related to some alkaloids, so more research will be required to determine effective compounds.
5. Acknowledgements
We would like to thank to Mr. Rahim Absalan for preparation of some materials, and we would specially like to thank to Dr. Mohammad Reza Bazrafshan for preparation of antibodies. This study was supported by grants from the Borūjerd Azad University (Borūjerd, Iran).
REFERENCES
- A. Mohammadi, M. Darabi, M. Nasry, M. J. Saabet-Jahromi, R. Malek-Pour-Afshare and H. Sheibani, “Effect of Opium Addiction on Lipid Profile and Atherosclerosis Formation in Hypercholesterolemic Rabbits,” Experimental and Toxicologic Pathology, Vol. 61, No. 2, 2009, pp. 145- 149. doi:10.1016/j.etp.2008.08.001
- S. Asgary, N. Sarrafzadegan, G. A. Naderi and R. Rozbehani, “Effect of Opium Addiction on New and Traditional Cardiovascular Risk Factors: Do Duration of Addiction and Route of Administration Matter?” Lipids in Health and Disease, Vol. 7, No. 42, 2008, pp. 1-5.
- G. A. Graf, W. P. Li, R. D. Gerard, I. Gelissen, A. White, J. C. Cohen and H. H. Hobbs, “Coexpression of ATPBinding Cassette Proteins ABCG5 and ABCG8 Permits Their Transport to the Apical Surface,” Journal of Clinical Investigation, Vol. 110, No. 5, 2002, pp. 659-669.
- J. K. Kruit, A. K. Groen, T. J. Van Berkel, F. Kuipers, “Emerging Roles of the Intestine in Control of Cholesterol Metabolism,” World Journal of Gastroenterology, Vol. 12, No. 40, 2006, pp. 6429-6439.
- D. H. Volle and J. A. Lobaccaro, “Role of the Nuclear Receptors for Oxysterols LXRs in Steroidogenic Tissues: Beyond the “Foie Gras”, the Steroids and Sex,” Molecular and Cellular Endocrinology, Vol. 265-266, 2007, pp. 183-189. doi:10.1016/j.mce.2006.12.018
- H. Najafipoura, J. Joukar, R. Malekpour-Afshar, F. Mirzaeipour and H. R. Nasri, “Passive Opium Smoking Does Not Have Beneficial Effect on Plasma Lipids and Cardiovascular Indices in Hypercholesterolemic Rabbits with Is-Chemic and Non-Ischemic Hearts,” Journal of Ethnopharmacology, Vol. 127, No. 2, 2010, pp. 257-263. doi:10.1016/j.jep.2009.11.011
- M. O. Ebesunun, O. Popoola and E. O. Agbedana, “The Effect of Garlic on Plasma Lipids and Lipoproteins in Rats Fed on High Cholesterol Enriched Diet,” Biokemistri, Vol. 19, No. 2, 2007, pp. 53-58.
- A. C. Provost, M. O. Pequignot, K. M. Sainton, S. Gadin, S. Salle, D. Marchant, D. B. Hales and M. Abitbol, “Expression of SR-BI Receptor and StAR Protein in Rat Ocular Tissues,” Comptes Rendus Biologies, Vol. 326, No. 9, 2003, pp. 841-851. doi:10.1016/j.crvi.2003.09.001
- Z. Hajializadeh, S. Esmaeili-Mahani, V. Sheibani, A. Kaeidi, M. Atapour and M. Abbasnejad. “Changes in the Gene Expression of Specific G-Protein Subunits Correlate with Morphine Insensitivity in Streptozotocin-Induced Diabetic Rats,” Neuropeptides, Vol. 44, No. 4, 2010, pp. 299- 304. doi:10.1016/j.npep.2010.02.004
- S. J. Thornton, E. Wong, D. S. Lee, S. D. Lee and K. M. Wasan, “Effect of Dietary Fat on Hepatic Liver X Receptor Expression in P-Glycoprotein Deficient Mice: Implications for Cholesterol Metabolism,” Lipids in Health and Disease, Vol. 7, No. 21, 2008, pp. 1-7.
- S. S. Fatemi, M. Hasanzadeh, A. Arghami and M. R. Sargolzaee, “Lipid Profile Comparison between Opium Addicts and Non-Addicts,” Journal of Tehran University Heart Center, Vol. 3, No. 3, 2008, pp. 169-172.
- S. Zheng, L. Hoos, J. Cook, G. Tetzloff, G. Davis, M. V. Heek and J. J. Hwa, “Ezetimibe Improves High Fat and Cholesterol Diet-Induced Non-Alcoholic Fatty Liver Disease in Mice,” European Journal of Pharmacology, Vol. 584, No. 1, 2008, pp. 118-124. doi:10.1016/j.ejphar.2008.01.045
- B. A. Laffitte, L. C. Chao, J. Li, R. Walczak, S. Hummasti, S. B. Joseph, A. Castrillo, D. C. Wilpitz, D. J. Mangelsdorf, J. L. Collins, E. Saez and P. Tontonoz, “Activation of Liver X Receptor Improves Glucose Tolerance through Coordinate Regulation of Glucose Metabolism in Liver and Adipose Tissue,” PNAS, Vol. 100, No. 9, 2003, pp. 5419-5424. doi:10.1073/pnas.0830671100
- S. W. Altmann, H. R. Davis, X. Yao, M. Laverty, D. S. Compton, L. Zhu, J.H. Crona, M. A. Caplen, L. M. Hoos, G. Tetzlo, T. Priestley, D. A. Burnett, C. D. Strader and M. P. Graziano, “The Identification of Intestinal Scavenger Receptor Class B, Type I (SR-BI) by Expression Cloning and Its Role in Cholesterol Absorption,” Biochimica et Biophysica Acta, Vol. 1580, No.1, 2002, pp. 77-93. doi:10.1016/S1388-1981(01)00190-1
- C. Fievet and B. Staels, “Liver X Receptor Modulators: Effects on Lipid Metabolism and Potential Use in the Treatment of Atherosclerosis,” Biochemical Pharmacology, Vol. 77, No. 8, 2009, pp. 1316-1327. doi:10.1016/j.bcp.2008.11.026
- J. J. Repa, K. E. Berge, C. Pomajzl, J. A. Richardson, H. Hobbs and D. J. Mangelsdorf, “Regulation of ATP-Binding Cassette Sterol Transporters ABCG5 and ABCG8 by the Liver X Receptors Alpha and Beta,” Journal of Biological Chemistry, Vol. 277, No. 21, 2002, pp. 18793- 18800. doi:10.1074/jbc.M109927200
- S. Colin, E. Bourguignon, A. Boullay, J. Tousaint, S. Huet, F. Caira, B. Staels, B. Lestavel, J. A. Lobaccaro and P. Delerive, “Intestine-Specific Regulation of PPAR-α Gene Transcription by Liver X Receptors,” Endocrinology, Vol. 149, No. 10, 2008, pp. 5128-5135. doi:10.1210/en.2008-0637
- J. R. Schultz, H. Tu, A. Luk, J. J. Repa, J. C. Medina, L. Li, S. Schwendner, S. Wang, M. Thoolen, D. J. Mangelsdorf, K. D. Lustig and B. Shan, “Role of LXRs in Control of Lipogenesis,” Genes and Development, Vol. 14, No. 22, 2000, pp. 2831-2838. doi:10.1101/gad.850400
- S. S. Choe, A.H. Choi, J. W. Lee, K.H. Kim, J. J. Chung, J. Park, K. M. Lee, K. G. Park, I. K. Lee and J. B. Kim, “Chronic Activation of Liver X Receptor Induces BetaCell Apoptosis through Hyperactivation of Lipogenesis: Liver X Receptor-Mediated Lipotoxicity in Pancreatic Beta-Cells, ” Diabetes, Vol. 56, No. 6, 2007, pp. 534-543. doi:10.2337/db06-1059
NOTES
*Corresponding author.

