American Journal of Plant Sciences
Vol.4 No.7(2013), Article ID:34731,5 pages DOI:10.4236/ajps.2013.47176
Influence of VAM Fungi, Azotobacter sp. and PSB on Soil Phosphatase Activity and Nutrients (N, P, K, Cu, Zn, Fe and Mn) Status in the Rhizosphere of Stevia rebaudiana (Bert.) Plants
![]()
1Department of Biotechnology, Sri Venkateswara University, Tirupati, India; 2Department of Botany, Sri Venkateswara University, Tirupati, India.
Email: *agni2013@gmail.com
Copyright © 2013 G. Ramakrishnaiah, T. Vijaya. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received April 26th, 2013; revised May 27th, 2013; accepted June 15th, 2013
Keywords: Microbial Inoculants; Macronutrients; Micronutrients
ABSTRACT
The pot culture experiment was carried out to evaluate the influence of different combinations of microbial inoculants (VAM + AZO + PSB) on soil phosphatase activity andnutrients (N, P, K, Cu, Zn, Fe, Cu & Mn) of Steviarebaudia plant rhizosphere on 20th, 40th and 60th day. The study revealed that the combined application of microbial inoculants onsoil Alkaline and Acid phosphatase activity has been found maximum at 60th day in triple inoculated plants than dual inoculated and control. The results also showed that the soil macronutrients (N, P & K) and micronutrients (Zn, Fe, Cu & Mn) have been found to be maximum at 40th day and 60th day respectively in triple inoculated plants than dual inoculated and control.
1. Introduction
Vesicular arbuscular mycorrhizae, Azotobacter and phosphate solubilizing bacteria are the most widely used bio fertilizers, significantly increasing the soil N, P, K, Zn, Fe, Cu & Mn and make them available to the plants. The interaction of the major soil components like minerals, organic matter and microorganisms, shows a profound influence on the biological processes of the soils [1]. On root colonization VAM fungi produce two specialized structures called vesicles and arbuscules in the cortex region of the root. Phosphatases play a major role in the mineralization processes of organic phosphorous and transports of phosphorous which are present in the vacuoles of VAM fungi. Azotobacter involve in the increasing the soil nitrogen by fixing the atmospheric nitrogen. Phosphate solubilizing bacteria (PSB), acting in union with plant roots, are responsible for solubilizing phosphate minerals. Although there are several reports on the microbial inoculants influence on the soil fertility and their support to the plants growth, the synergetic influence of VAM fungi, Azotobacter and PSB on the soil phosphatases activity and nutrients status of the rhizosphere of Stevia rebaudiana plant are limited, hence the present study was undertaken to evaluate the soil phosphatases activity and soil nutrients status under the influence of different combinations of bio-inoculants.
2. Materials and Methods
The pot culture experiment was carried out under green house conditions to know the influence of Glomus mossae, Azotobacter chrocooccum and Pseudomonas striata on soil nutrients status and phosphatases activity of Stevia plant rhizosphere. For the experiment purpose brown colour soil (pH: 6.91) was collected from a cultivation land. The soil was sterilized before treated with the bio-inoculants.
2.1. VAM Inoculum Preparation
Sand: soil (1:1) mixture containing spores and infectedroot segments of Maize (Sorghum vulgare), infected with Glomus mosseae (Nicolson and Gerdeman) and grown for 90 days, then collected the roots that contained VAM fungi. This served asthe mycorrhizal inoculum. The stem cuttings of the tested plant were inoculated with 500 mg of mycorrhizal inoculum (approximately 250 spores), by placing 2 cm below the soil surface where stem cuttings were planted.
2.2. PSB Inoculum Preparation
Culture of Pseudomonas striata was obtained from Regional Biofertilizers Development Centre Bangalore Division, India. P. straiata was inoculated in 500 ml sterilized Pikovskaia’s broth and incubated at 30˚C for 3 days in a BOD chamber. After obtaining the desirable growth (107 - 108 cells/ml), the broth was mixed with wood charcoal by maintaining moisture content and pH 7.0. The slurry thus prepared was mixed with soil at the time of planting.
2.3. Azotobacter sp. Inoculums Preparation
Culture of Azotobacter chrocooccum was obtained from RBDC, Bangalore, India. From this mother culture carrier based inoculums was developed by growing it in nitrogen free nutrient broth as described by Ashby [2] for 3 days at 28˚C temperature. Then the culture was mixed with lignite powder and this carrier based inoculum was mixed with soil at the time of planting the test plants.
Stem cuttings of S. rebaudiana plant were collected from local nursery and were used as test plants. The fol lowing treatments were established and the plants were grown for 60 days with periodic watering and weeding.
T1: Control (No inoculation)T2: Inoculation with VAM fungi (Glomus mosseae)T3: Inoculation with Azotobacter chrocooccumT4: Inoculation with P. striataT5: Inoculation with VAM fungi and A. chrocooccumT6: Inoculation with VAM fungi and P. striataT7: Inoculation VAM fungi, A. chrocoooccum and P. striata.
Twenty grams of inoculums of Glomus mosseae, A. chrocoocuum and P. striata were mixed with sterile soil in the ratio of 1:1 as per the above single or mixed treatments. Stem cuttings of uniform size were transferred to the plastic pots containing the above mixture. Eight replications were maintained for each treatment and the pots were arranged in a completely randomized design. The plants were grown under natural photoperiods (23.5/18˚C day/night, 4000 - 6000 Lux light intensity) for two months during which only deionized water was supplied. The soil (plant rhizospher) nutrients of both macro (N, P, K) and micro nutrients (Zn, Fe, Cu & Mn) and soil phosphatase (alkaline and acid) were measured on every 20th, 40th and 60th day of the plant growth in all the treatments. The soil content of Nitrogen [3], Phosphorus [4], Potassium [5], Copper, Iron, Zinc and Manganese [6] were estimated (Tables 1 and 2). The activities of both the soil Alkaline and Acid phosphatases were also estimated [7] (Table 3).
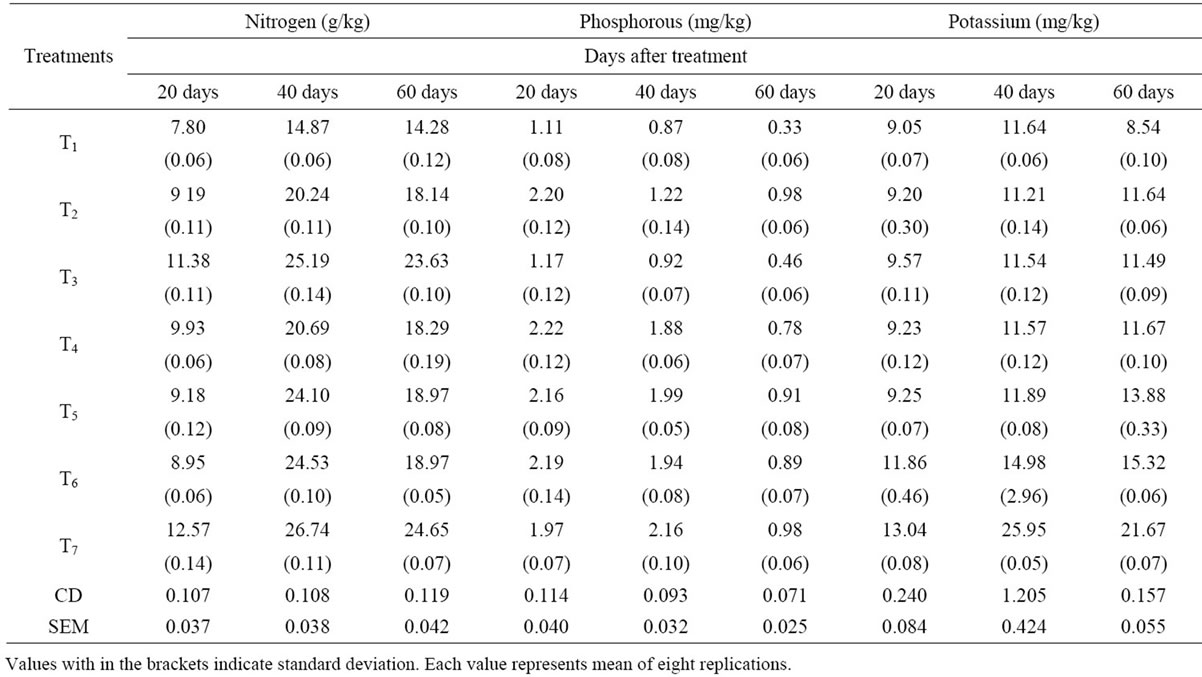
Table 1. Effect of VAM fungi, Azotobacter sp. and PSB on nitrogen, phosphorous and potassium contents of soil.
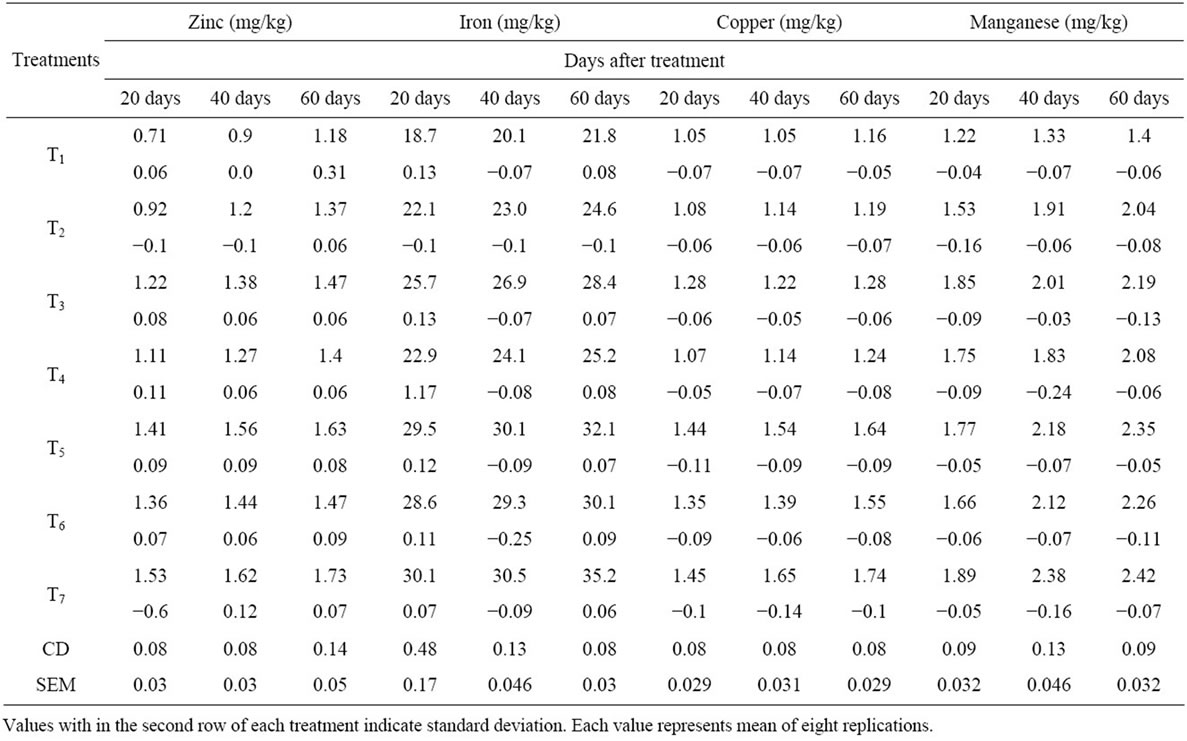
Table 2. Effect of VAM fungi, Azotobacter sp. and PSB on zinc, iron, copper and manganese contents of soil.
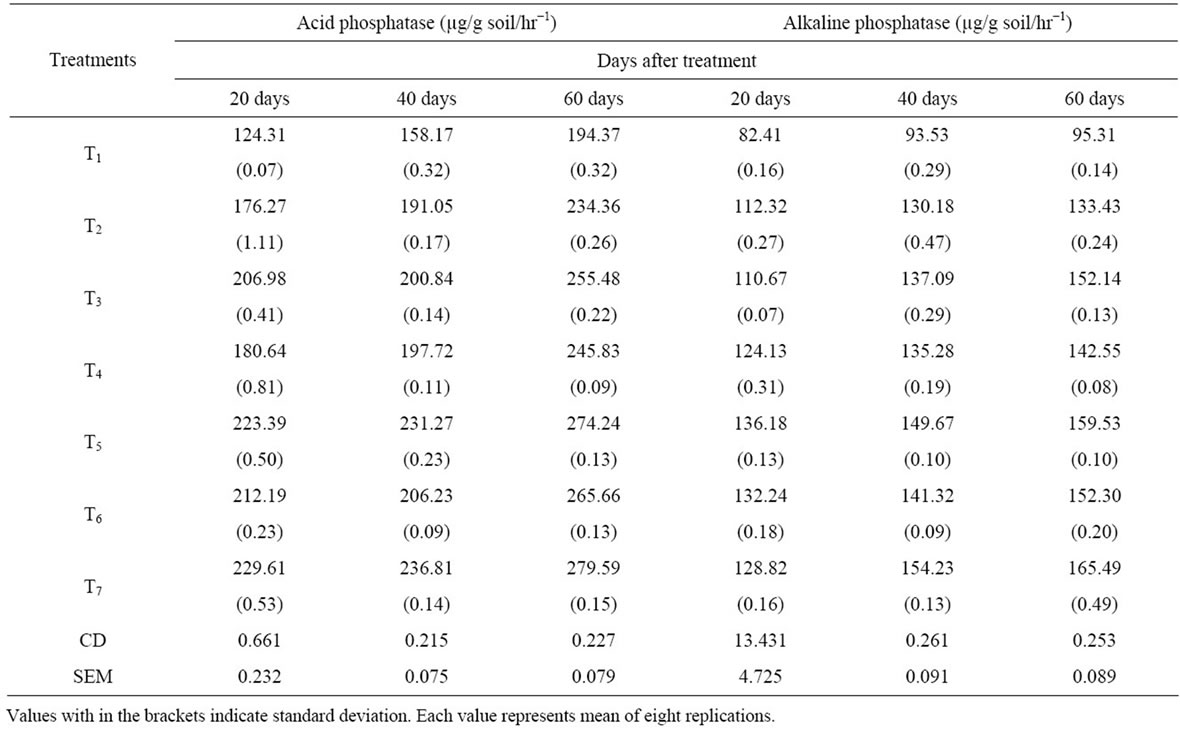
Table 3. Acid and Alkaline Phosphatases activity.
3. Results and Discussion
It was observed that all the treated soils exhibited increased soil nutrient contents compared to the control soils. The amount of soil nitrogen in T7 treatment soil was found to be the highest on 40th day and then decreases to the 60th day. The amount of soil phosphorus was found maximum at 60th day in T7 treated soils. Similarly the amount of soil potassium was found maximum at 60th day in T7 treated soils. Then it is followed by dual inoculated soils (T6, T5, T4, T3, T2 & T1). The amounts of soil micronutrients Zinc, Iron, Copper and Manganese were found maximum at 60th day in all the treated soils than control. It was found that the micronutrients contents were maximum in triple inoculated (VAM fungi + AZO+ PSB) soils than dual inoculated, single inoculated and control.
Both the Acid and Alkaline phosphatases of inoculated soils were found to be significantly influenced by the VAM fungi, Azotobacter and PSB. Maximum acid and alkaline phosphatases were recorded in T7 treated soils followed by T6, T5, T4, T3 and T2 and least amount in control soil. VAM inoculation either individually (T2) or in combination with Azotobacter, PSB (T5 and T6) and both showed significantly higher root colonization. Maximum colonization was found in plants with mixed inoculums (T7) followed by dual inoculum (T5 > T6) and minimum in VAM alone treated soils.
The results of present study on soil nitrogen indicated that there was a significant increase of soil N content in treatments than control. Similar results were also reported [8]. Azatobacter involved in the fixation of atmospheric nitrogen to the nitrates. Mycorrhizal fungi may also contribute to an increased nitrogen status in the mycorrhizosphere by decomposing organic matter. So the combine effect of VAM fungi and Azotobacter increased the N content in the treated soils. In the present study the soil nitrogen content at 40 days was higher than 60 days soils; this is because of the increased absorption of nitrogen by plants during their growth. The content of soil phosphorous in all the treated soils had been increased; this is due to the application of VAM fungi, Azotobacter and PSB, which enhances the phosphorous content. Several scientists have reported increase in the availability and uptake of native soil P in different plants by converting insoluble phosphates in the soil to soluble forms by producing various organic acids [9-11]. Similarly the potassium content in the soil was significantly increased in all the treatments when compared to control. Bio-inoculants are usually considered very effective in increasing the K content in the soil [12]. Sharma [13] reported an increase in K content in the rhizosphere soil of mycorrhizal treated rice and wheat plants. The micronutrients contents (Zn, Fe, Cu and Mn) were increased in the treated soils than control. The results prove that triple and dual treatments increase the Zinc content of the soil. Similar results were reported by Mohammad et al., [14]) and Bi et al., [15]. Mukerji et al., (1996) observed an increase in soil Fe, Mn and Cu in root zone soil of A. catechu, L. leucocephala and P. julifera inoculated with G. fasciculatum. In the present study the increased organic content of the soil in treated plants could be the reason for more concentration of these micro nutrients in addition formation of organic chealates of higher stability with organic compounds preventing the micronutrients from precipitation, fixation, oxidation and leaching. Reports were their on the correlation between soil organic matter and contents of micronutrients [16].
Alkaline and acid phosphatases were considered to be the PO4 metabolism of the fungus. Increase in alkaline and acid phosphatases activities were recorded in all inoculated soils over control. Study in Neem (Azardicta indica) plants noticed an increase in the phosphatase activity in the root zone soil because of VAM inoculation [17]. Similar results were also reported [18,19]. The high alkaline and acid phosphatase activity in triple and dual inoculated soils attributes to the production of more enzymes by the synergetic interaction of VAM fungi, Azotobacter and Phosphate solubilizing bacteria.
REFERENCES
- P. M. Huang, “Foreseeable Impacts of Soil Mineral-Organic Component-Microorganism Interactions on Society Ecosystem Health,” Elsevier, Boston, 2002.
- N. S. Subba Rao, “Advances in Agricultural Microbiology,” Oxford & IBH Publishing Co. Pvt. Ltd., New Delhi, 1982.
- B. V. Subbiah and G. L. Asija, “A Rapid Method for Estimation of Available Nitrogen in Soil,” Current Science, Vol. 25, 1956, pp. 258-260.
- S. R. Olsen and L. E. Sommes, “Phosphorous in Methods of Soil Analysis. Part 2,” 2nd Edition, American Society of Agronomy and Soil Science, Madison, 1982.
- M. L. Jackson, “Soil Chemical Analysis,” Prentice Hall of India Private Limited, New Delhi, 1967.
- W. L. Lindsay and W. A. Norvell, “Development of a DTPA Soil Test for Zinc, Iron, Manganese, and Copper,” Soil Science Society of America Journal, Vol. 42, No. 3, 1978, pp. 421-428. doi:10.2136/sssaj1978.03615995004200030009x
- F. Eivazi and M. A. Tabatabai, “Phosphatases in Soils,” Soil Biology and Biochemistry, Vol. 9, No. 3, 1977, pp. 167-172. doi:10.1016/0038-0717(77)90070-0
- M. Patidar and A. L. Mali, “Residual Effect of Farmyard Manure, Fertilizer and Biofertilizer on Succeeding Wheat (Triticum aestivum),” Indian Journal of Agronomy, Vol. 47, No. 1, 2002, pp. 26-32.
- A. C. Gour and K. P. Ostwal, “Influence of Phosphorous Dissolving Bacilli on the Yield and Phosphorous Uptake of Crop,” Indian Journal of Experimental Biology, Vol. 19, 1972, p. 393.
- S. Banik and B. K. Dey, “Available Phosphate Content of Alluvial Soil as Influenced by Inoculation of Some Isolated Phosphate Solubilizing Microorganisms,” Plant and Soil, Vol. 69, No. 3, 1992, pp. 353-364. doi:10.1007/BF02372456
- A. Raced, “Phosphorous Use Efficiency in Soils of Islamabad,” Proceedings of 4th National Congress of Soil Science, Islamabad, 1994, pp. 115-117.
- G. Bohrer, et al., “Effects of Different Kalahari-Desert VA Mycorrhizal Communities on Mineral Acquisition and Depletion from the Soil by Host Plants,” Journal of Arid Environments, Vol. 55, No. 2, 2003, pp. 193-208. doi:10.1016/S0140-1963(03)00047-8
- S. N. Sharma, “Effect of Phosphate Solubilizing Bacteria on Efficiency of Mussorie Rockphosphate in Rice, Wheat Cropping System,” Indian Journal of Agricultural Sciences, Vol. 73, No. 9, 2003, pp. 478-481.
- M. Mohammad, W. Pan and A. Kennedy, “Wheat Response to Vesicular Arbuscular Mycorrhizal Fungal Inoculation of Soils from Eroded Top Sequence,” Soil Science Society of America Journal, Vol. 59, No. 4, 1995, pp. 1086-1090. doi:10.2136/sssaj1995.03615995005900040020x
- Y. Bi, et al., “Influence of Early Stages of Arbuscular Mycorrhiza on Uptake of Zinc and Phosphorous by Red Clover from Low Phosphorous Soil Amended with Zinc and Phosphorous,” Chemosphere, Vol. 50, No. 6, 2003, pp. 831-837. doi:10.1016/S0045-6535(02)00227-8
- C. Calvaruso, M. P. Turpault and P. Frey-Klett, “Root Associated Bacteria Contributed to Mineral Weathering and to Mineral Nutrition in Trees Soil,” Applied and Environmental Microbiology, Vol. 72, No. 2, 2006. pp. 1258-1266. doi:10.1128/AEM.72.2.1258-1266.2006
- D. A. Sumana, “Influence of VA Mycorrhizal Fungi and Nitrogen Fixing and Mycorrhization Helper Bacteria on Growth of Neem (Azardichta indica A. Juss.),” Ph.D Thesis, University of Agricultural Sciences, Bangalore, 1998.
- M. L. Izaguirre-Mayorala, et al., “Effects of Arbuscular Mycorrhizal Inoculation on Growth, Yield, Nitrogen and Phosphorous Nutrition of Nodulating Bean Varieties in Two Soil Substrates of Contrasting Fertility,” Journal of Plant Nutrition, Vol. 23, No. 8, 2000, pp. 1117-1133. doi:10.1080/01904160009382086
- R. Lakshmipathy, B. Gowda, K. Chandrika and D. J. Bagyaraj, “Symbiotic Response of Garcinia Indica to VA Mycorrhizal Inoculation,” Indian Journal of Forestry, Vol. 26, No. 2, 2003, pp. 143-146.
NOTES
*Corresponding author.

