Journal of Modern Physics
Vol.08 No.04(2017), Article ID:74995,15 pages
10.4236/jmp.2017.84041
Electronic Band Structure of Graphene Based on the Rectangular 4-Atom Unit Cell
Akira Suzuki1, Masashi Tanabe1, Shigeji Fujita2
1Department of Physics, Faculty of Science, Tokyo University of Science, Tokyo, Japan
2Department of Physics, University at Buffalo, State University of New York, Buffalo, NY, USA

Copyright © 2017 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: February 13, 2017; Accepted: March 27, 2017; Published: March 30, 2017
ABSTRACT
The Wigner-Seitz unit cell (rhombus) for a honeycomb lattice fails to establish a 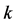 -vector in the 2D space, which is required for the Bloch electron dynamics. Phonon motion cannot be discussed in the triangular coordinates, either. In this paper, we propose a rectangular 4-atom unit cell model, which allows us to discuss the electron and phonon (wave packets) motion in the
-vector in the 2D space, which is required for the Bloch electron dynamics. Phonon motion cannot be discussed in the triangular coordinates, either. In this paper, we propose a rectangular 4-atom unit cell model, which allows us to discuss the electron and phonon (wave packets) motion in the 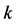 -space. The present paper discusses the band structure of graphene based on the rectangular 4-atom unit cell model to establish an appropriate
-space. The present paper discusses the band structure of graphene based on the rectangular 4-atom unit cell model to establish an appropriate 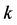 -vector
-vector 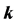 for the Bloch electron dynamics. To obtain the band energy of a Bloch electron in graphene, we extend the tight-binding calculations for the Wigner-Seitz (2- atom unit cell) model of Reich et al. (Physical Review B, 66, Article ID: 035412 (2002)) to the rectangular 4-atom unit cell model. It is shown that the graphene band structure based on the rectangular 4-atom unit cell model reveals the same band structure of the graphene based on the Wigner-Seitz 2-atom unit cell model; the
for the Bloch electron dynamics. To obtain the band energy of a Bloch electron in graphene, we extend the tight-binding calculations for the Wigner-Seitz (2- atom unit cell) model of Reich et al. (Physical Review B, 66, Article ID: 035412 (2002)) to the rectangular 4-atom unit cell model. It is shown that the graphene band structure based on the rectangular 4-atom unit cell model reveals the same band structure of the graphene based on the Wigner-Seitz 2-atom unit cell model; the 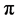 -band energy holds a linear dispersion (
-band energy holds a linear dispersion (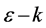 ) relations near the Fermi energy (crossing points of the valence and the conduction bands) in the first Brillouin zone of the rectangular reciprocal lattice. We then confirm the suitability of the proposed rectangular (orthogonal) unit cell model for graphene in order to establish a 2D
) relations near the Fermi energy (crossing points of the valence and the conduction bands) in the first Brillouin zone of the rectangular reciprocal lattice. We then confirm the suitability of the proposed rectangular (orthogonal) unit cell model for graphene in order to establish a 2D 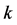 -vector responsible for the Bloch electron (wave packet) dynamics in graphene.
-vector responsible for the Bloch electron (wave packet) dynamics in graphene.
Keywords:
Graphene, Rectangular 4-Atom Unit Cell Model, Primitive Orthogonal Basis Vector, Bloch Electron (Wave Packet) Dynamics, 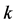 -Vector, Dirac Points, Linear Dispersion Relation
-Vector, Dirac Points, Linear Dispersion Relation

1. Introduction
The electronic band structure of graphene plays an important role for under- standing its unique properties [1] [2] [3] [4] . Graphene is a perfect two- dimensional crystal consisting of a single layer of carbon atoms arranged in a honeycomb lattice. A carbon atom contains four valence electrons, one 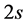 - electron, and three
- electron, and three 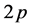 -electrons. They are
-electrons. They are 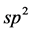 -hybridized, that is, one
-hybridized, that is, one 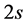 - electron and two
- electron and two 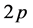 -electrons form strong
-electrons form strong 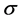 -bonds between carbon atoms leading to the honeycomb structure with the carbon-carbon distance of 0.142 nm. The remaining
-bonds between carbon atoms leading to the honeycomb structure with the carbon-carbon distance of 0.142 nm. The remaining 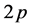 -electron occurs as a
-electron occurs as a 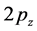 -orbital, which is oriented perpendicularly to the planar structure, and forms a
-orbital, which is oriented perpendicularly to the planar structure, and forms a  -bond with the neigh- bouring carbon atoms. The
-bond with the neigh- bouring carbon atoms. The  -bonds are completely filled and form a deep valence band [2] . The smallest gap between the bonding and the anti-bonding
-bonds are completely filled and form a deep valence band [2] . The smallest gap between the bonding and the anti-bonding 




Indeed, the band structure of graphene can be seen as a triangular lattice with a basis of two atoms per unit cell. This 2-atom unit cell (Wigner-Seitz (WS) cell) model has customarily been used to obtain the graphene band structure for the 


where 



where 


is the electron velocity where 

The potential energy 

where 






Figure 1. (a) Lattice structure of graphene. Carbon atoms at vertices. Each honeycomb lattice consists of equivalent carbon (C+) ions labeled by A and B. The Wigner-Seitz 2-atom unit cell (dotted lines) spanned by the basis (lattice unit) vectors 






does not establish the 

Our purpose of this paper is to explore the suitability of the rectangular (orthogonal) unit cell model for the Bloch electron band structure and to discuss the 

In Section 2, the rectangular 4-atom unit cell of graphene is introduced. Section 3 presents our tight-binding calculations of the energy band of a Bloch electron in graphene, based on the rectangular 4-atom unit cell model described in Section 2. Section 4 illustrates why we have to utilize the rectangular (orthogonal) unit cell rather than the WS unit cell when considering the electron dynamics of graphene. Results and discussion are given in Section 5. Finally conclusions and some remarks are given in Section 6.
2. The Rectangular 4-Atom Unit Cell Model for Graphene
Graphene is made out of honeycomb lattice (carbon atoms arranged in hexagonal structure) and the 2D honeycomb lattice has a reflection symmetry relative to the 


Figure 2. (a) Lattice structure of graphene, made out of the rectangular 4-atom unit cell (a square dotted line) spanned by the basis vectors 






The reason why we choose a rectangular (orthogonal) unit cell rather than a triangular (WS) unit cell for graphene will be explained later.
The lattice basis (unit) vectors 

The lattice constants for the rectangular (orthogonal) unit cell in the Cartesian coordinates are 




The reciprocal-lattice vectors for the rectangular unit cell are given (from Equation (5)) by

The first Brillouin zone is a rectangle as shown in Figure 2(b) with sides of length, 





3. Tight-Binding Approach
The single-particle band structure of graphene can be analytically calculated within the tight-binding approximation assuming that electrons are tightly bound to their C+ ion [8] [9] [10] [11] . In order to obtain the band structure of graphene based on the 4-atom rectangular unit cell model [7] for graphene, we follow the tight-binding approach employed by Reich et al. [8] .
The Schrödinger equation for a single electron in the lattice potential field 

where the Hamiltonian 

and the lattice potential 




where 



where





where the matrices, 





Here 









Since the atomic wave functions are well localized around the carbon atoms, only the nearest-neighbor hopping of Bloch electrons are taken into considera- tion of the following calculations. In other words, as for the electron in atom A orbital, it can hop to a nearest orbital of atom B or atom D. Similarly, the electron in the orbital of atom B can hop to the nearest orbital of atom A or atom C, the electron in the orbital of atom C to the nearest orbital of atom B or atom D, and the electron in the orbital of atom D to the nearest orbital of atom A or atom C.
Let us consider the nearest-neighbor hopping between the orbital of atom A and atom B. The matrix elements 

Here we only take into account the nearest-neighbor hopping between carbon atoms. From the symmetry of the lattice stracture of graphene, the adjacent hoppings are all the same. We introduce the parametric parameter 

Since the nearest-neighbor vector 



Similarly, the nearest-neighbor hopping integrals are given by

We note that the hopping parameter 
The matrix element 

where 



Next we evaluate the matrix elements



Introducing the parametric parameter 

the matrix element 

Similarly we obtain

We note that the matrix element 

In a similar manner, we obtain

We can solve Equation (17) for 



where 

4. Electron Dynamics of Graphene
In semiclassical (wave packet) theory for electron dynamics, it is necessary to introduce a wave vector 


Graphene forms a 2D honeycomb lattice. Let us first consider the Wigner- Seitz (WS) unit cell (rhombus, dotted lines shown in Figure 1(a)). The potential energy 

where the position vector 




In the field theoretical formulation, the field point 

where 

We assume that the wave packet is composed of superposable plane-waves characterized by the 

where 


for all 


The wave function must be Fourier-analyzable. In the rhombic system, however, if we choose the 








Ashcroft and Mermin (AM) [6] introduced a translation operator 



They used

to establish Equation (35). The translation operator 

as seen from

terms of the orthogonal basis vectors 
If 








We assume that the “electron” (“hole”') (wave packet) has the charge 




Thus, “electrons” are the majority carriers in graphene. The thermally activated electron densities are then given by

where 

At finite temperature phonons are present in the system. The excitation of phonons can be discussed based on the same rectangular unit cell introduced for the conduction electrons. We note that phonons can be discussed naturally based on the orthogonal unit cells. It is difficult to describe phonons in the WS cell model.
5. Results and Discussion
Based on the rectangular 4-atom unit cell model, we obtained the band disper- sion of graphene by applying the tight-binding theory of Reich et al. [8] . The obtained (





Figure 4 shows the energy band profile along the high symmetric points (indicated by
Figure 3. (Color online) Band structure of graphene based on the rectangular 4-atom unit cell model. The energy is maximal at the 


Figure 4. (Color online) Band profile along the symmetric points of graphene for the rectangular 4-atom unit cell model. One of the cross in points (so-called Dirac points) is indicated by P. Inset is the 1st Brillouin zone of the rectangular unit cell model. Conduction bands (red and blue) are computed from Equation (29) while valence bands (black and green) are computed from Equation (30). Note that the band profile is plotted within the one quarter of the first Brillouin zone.
space. There are two crossing points P and Q in the first BZ (cp. Figure 3), at which the band energy is crossing (i.e.,
Let us take a close look at the behavior of the band energy close to the crossing points, at which the band energy equals to zero. The conduction and valence bands are degenerate at this point. The dispersion relation for small momenta 



where 

with


The second BZ of the reciprocal lattice for the rectangular 4-atom unit cell is shown by the shaded area in Figure 6. The high symmetric points in the reciprocal lattice of the rectangular 4-atom unit cell are indicated by
Figure 5. (Color online) The linear dispersion near the point P in the first BZ of the rectangular 4-atom unit cell, where the two bands (valence and conduction) cross each other. The energy is maximal at the 



Figure 6. The first Brillouin zone (rectangular) and the second Brillouin (shaded) zone of the rectangular 4-atom unit cell. The region of the first and second BZ’s (i.e., the hexagonal region) of the rectangular 4-atom unit cell is identical to the first BZ of the Wigner-Seitz 2-atom unit cell.
and W, while those in the reciprocal lattice of the 2-atom unit cell are indicated by
The approximate results (






6. Conclusions and Some Remarks
The current authors proposed a rectangular 4-atom unit cell model for graphene [7] . Based on this model, we obtained the band energy by applying the tight- binding approach employed by Reich et al. [8] . We proved the band structure based on the rectangular 4-atom unit cell model for graphene gives the same band structure of graphene based on the prevalent graphene model based on the Wigner-Seitz 2-atom unit cell model.
The rectangular 4-atom unit cell for graphene has the sides perpendicular to each other (see Figure 2). The 



The transport electrons in graphene originating from the 



A material (density) wave such as a phonon wave can be presented by a traveling wave function of the form: 



In the currently prevailing theory [2] [3] [4] [5] [8] [9] [10] [11] [17] the solid state theory dealing with a hexagonal crystal starts with a primitive non- orthogonal unit cell, the 2-atom unit cell. This theoretical model has difficulties in particular for a superconductor. The ground state of a superconductor must be condensed in a single particle-state in accordance with Nernst’s theorem (the third law of thermodynamics). Many fermions have a distribution in energy and hence a many-fermion system cannot be a superconductor. Only many-boson system can be a superconductor.
In the prevailing theory [2] [3] [4] [5] [8] [9] [10] [11] [17] a 2-atom unit cell model is used to set up 




In the present work we have shown that the non-orthogonal 2-atom and orthogonal 4-atom models give the same band energies in the mean field appro- ximation. In separate publication we report the superconductivity in graphite intercalation compounds C8K, C6Ca and in compound MgB2.
Cite this paper
Suzuki, A., Tanabe, M. and Fujita, S. (2017) Electronic Band Structure of Graphene Based on the Rectangular 4-Atom Unit Cell. Journal of Modern Physics, 8, 607-621. https://doi.org/10.4236/jmp.2017.84041
References
- 1. Geim, A.K. and Noboselov, K.S. (2007) Nature Materials, 6, 183-191.
https://doi.org/10.1038/nmat1849 - 2. Neto, A.H.C., Guinea, F., Peres, N.M.R., Novoselov, K.S. and Geim, A.K. (2009) Reviews of Modern Physics, 81, 109.
https://doi.org/10.1103/RevModPhys.81.109 - 3. Sarma, S.D., Adam, S., Hwang, E. and Rossi, E. (2011) Reviews of Modern Physics, 83, 407.
https://doi.org/10.1103/RevModPhys.83.407 - 4. Katsnelson, M.I. (2012) Graphene: Carbon in Two Dimensions. Cambridge University Press, Cambridge.
https://doi.org/10.1017/CBO9781139031080 - 5. Reich, S., Thomsen, C. and Maultzsch, J. (2004) Carbon Nanotubes: Basic Concepts and Physical Properties. Wiley-VCH Verlag GmbH, Berlin.
- 6. Ashcroft, N.W. and Mermin, N.D. (1976) Solid State Physics. Saunders, Philadelphia, 75, 133-135, 137-139, 228-229.
- 7. Fujita, S. and Suzuki, A. (2010) Journal of Applied Physics, 107, Article ID: 013711.
https://doi.org/10.1063/1.3280035 - 8. Reich, S., Maultzsch, J., Thomsen, C. and Ordejön, P. (2002) Physical Review B, 66, Article ID: 035412.
https://doi.org/10.1103/PhysRevB.66.035412 - 9. Wallace, P.R. (1947) Physical Review, 71, 622.
https://doi.org/10.1103/PhysRev.71.622 - 10. McClure, J.W. (1957) Physical Review, 108, 612.
https://doi.org/10.1103/PhysRev.108.612 - 11. Slonczewski, J.S. and Weiss, P.R. (1958) Physical Review, 109, 272.
https://doi.org/10.1103/PhysRev.109.272 - 12. Fujita, S. and Ito, K. (2007) Quantum Theory of Conducting Matter: Newtonian Equations of Motion for a Bloch Electron. Springer, New York, 120-121, 192-193.
https://doi.org/10.1007/978-0-387-74103-1 - 13. Bloch, F. (1929) Zeitschrift für Physik, 52, 555-600.
https://doi.org/10.1007/BF01339455 - 14. Fujita, S., Jovaini, A., Godoy, S. and Suzuki, A. (2012) Physics Letters A, 376, 2808-2811.
- 15. Fujita, S., Takato, Y. and Suzuki, A. (2011) Modern Physics Letters B, 25, 223.
https://doi.org/10.1142/S0217984911025675 - 16. Zhang, Y., Tan, Y.W., Stormer, H.L. and Kim, P. (2005) Nature, 438, 201-204.
https://doi.org/10.1038/nature04235 - 17. Saito, R., Dresselhaus, G. and Dresselhaus, M.S. (2005) Physical Properties of Carbon Nanotubes. Imperial College Press, London.







