Journal of Modern Physics
Vol.06 No.14(2015), Article ID:61511,36 pages
10.4236/jmp.2015.614217
Mathematical Physics in Diffusion Problems
Takahisa Okino
Department of Applied Mathematics, Faculty of Engineering, Oita University, Oita City, Japan

Copyright © 2015 by author and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).


Received 14 October 2015; accepted 24 November 2015; published 27 November 2015

ABSTRACT
Using the divergence theorem and the coordinate transformation theory for the general Fickian second law, fundamental diffusion problems are investigated. As a result, the new findings are obtained as follows. The unified diffusion theory is reasonably established, including a self-diffusion theory and an N (N ³ 2) elements system interdiffusion one. The Fickian first law is incomplete without a constant diffusion flux corresponding to the Brown motion in the localized space. The cause of Kirkendall effect and the nonexistence of intrinsic diffusion concept are theoretically revealed. In the parabolic space, an elegant analytical method of the diffusion equation is mathematically established, including a nonlinear diffusion equation. From the Schrödinger equation and the diffusion equation, the universal expression of diffusivity proportional to the Planck constant is reasonably obtained. The material wave equation proposed by de Broglie is also derived in relation to the Brown motion. The fundamental diffusion theories discussed here will be highly useful as a standard theory for the basic study of actual interdiffusion problems such as an alloy, a compound semiconductor, a multilayer thin film, and a microstructure material.
Keywords:
Brown Particle, Boltzmann Factor, Markov Process, Parabolic Law, Error Function

1. Introduction
First of all, we state that the basic diffusion equation of the general nonlinear Fickian second law is discussed in accordance with the fundamental mathematical physics in the present work. The extended diffusion equations in detail are not thus discussed. Nevertheless, the new findings, which are extremely dominant in the diffusion study, are reasonably obtained. In the diffusion history, the problems relevant to the coordinate transformation of diffusion equation had not been discussed in accordance with the Gauss divergence theorem until recently. That is just a reason why the new diffusion theories are discussed in the present study. It will be gradually clarified in the text that the coordinate transformation theory is essentially indispensable for the diffusion study. It is obvious that analyzing the extended diffusion equation must be based on the fundamental diffusion theory. The new fundamental findings different from the existing diffusion theories obtained here will thus exert a great influence on the actual diffusion problems in detail, just because of fundamental ones.
A great many phenomena in various science fields are expressed by using the well-known evolution equations. The diffusion equation is one of them and mathematically corresponds to the Markov process in relation to the normal distribution rule [1] . In other words, the motion of diffusion particles corresponds to the well-known Brown movement satisfying the parabolic law [2] [3] . It is widely accepted that the Brown problem is a general term of investigating subjects in various science fields relevant to the Markov process, such as material science, information science, life science, and social science [4] - [9] .
In physics, we can also understand the diffusion equation in accordance with the Gauss divergence theorem [10] . If we apply the divergence theorem to the diffusion problem for a material under the condition of no sink and source of the material, it is found that the material conservation law is valid for the diffusion particles, regardless of a thermodynamic state of material. The diffusion equation is also called “the continuous equation” and is extremely fundamental one in physics. In history, the heat conduction equation, which is mathematically equivalent to the diffusion equation, was proposed by Fourier, regardless of the Markov process and the divergence theorem [11] .
In accordance with the industrial requirement, the solid materials, such as alloys, semiconductors, and multilayer materials, have been widely fabricated. The heat treatment is indispensable for their fabrication processes then. The migration of particles in a material is caused by the heat treatment. In relation to the migration of their particles, the diffusion problems of various solid materials have been thus widely investigated [12] . Therefore, the diffusion problem is a fundamental study subject in the materials science including the cases of liquid and gas states.
In the present work, the fundamental problems of the general Fickian second law where a driving force affects the diffusion system are discussed in accordance with the mathematical theory. The present analytical method is applicable to interdiffusion problems of an N elements system of every material in an arbitrary thermodynamic state. Although the physical validity of the present method is investigated by using the diffusion data concerning the solid metals, the mathematical generality discussed here is still kept.
The heat conduction equation proposed by Fourier in 1822 has been applied to investigating the temperature distribution in materials [11] . In 1827, the so-called Brown motion was found, where the self-diffusion of water was visualized by pollen micro particle motions [2] [3] . In 1855, Fick applied the heat conduction equation to diffusion phenomena as it had been [13] . Nevertheless, the Brown motion had not been recognized as a diffusion problem until the Einstein theory of Brown motion in 1905, although it was a typical diffusion problem [3] .
Although the concentration of diffusion particles is a real quantity in physics, the temperature is a thermodynamic state quantity. As far as the shape of heat conduction material is unchangeable during a thermal treatment, the coordinate system of heat conduction equation set in a material is a fixed one, since the coordinate system is not influenced by variations of the material internal structure. On the other hand, strictly speaking, the coordinate system of diffusion equation set in the diffusion field (solvent) is a moving one, since it is generally influenced by such variations.
When the Fickian first and second laws (F1 law and F2 law) were proposed, the Gauss divergence theorem had been already reported in 1840 [10] . Nevertheless, the problem of coordinate system of diffusion equation was not mathematically investigated in accordance with the divergence theorem.
In general, it is indispensable for understanding the diffusion problems to discuss their coordinate systems, since it is, strictly speaking, considered that the diffusion particles, solvent particles and also the diffusion region space simultaneously move against the experimentation system in the diffusion region outside.
Although the Fickian laws were still widely applied to various diffusion phenomena as essential equations in physics, the problem between coordinate systems was not discussed. Recently, the diffusion equation was thus mathematically investigated in accordance with the divergence theorem and the coordinate transformation theory [14] - [16] . It is revealed in the text that the diffusion flux should be determined by taking account of the concerned coordinate system of diffusion equation.
Using the corresponding diffusion flux to the coordinate system of diffusion equation for interdiffusion, one way diffusion, impurity diffusion and self-diffusion, they are uniformly discussed in the text. As a result, we found that the foundation of diffusion problems is included in interdiffusion problems. The interdiffusion theory of an
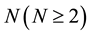 elements system applicable to every material was thus reasonably established [16] . In the analysis of interdiffusion problems, the only difference between a binary system and an N elements system is whether the solvent material is one element or
elements system applicable to every material was thus reasonably established [16] . In the analysis of interdiffusion problems, the only difference between a binary system and an N elements system is whether the solvent material is one element or
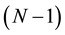 elements.
elements.
The coordinate transformation theory reveals that the corresponding F1 law to the F2 law is incomplete without a constant diffusion flux relevant to the Brown motion in the localized space. Further, it was also found that the Kirkendall effect (K effect) is caused by a shift between the coordinate systems of the diffusion equation like the Doppler effect relevant to a wave equation is caused by a shift between the fixed coordinate system and the moving one for the wave equation [14] - [17] . At the same time, this means that the concept of intrinsic diffusion is unnecessary for understanding the K effect.
As far as the sink and source of a material is nonexistent in the diffusion system, the Gauss divergence theory shows that the general F2 law satisfies the material conservation law even if a driving force affects the diffusion system. The driving force affects entropy and jump frequencies of diffusion particles in the material. The influence of a driving force is thus all incorporated into the diffusivity of diffusion equation then. In the existing diffusion theory, the diffusion fluxes influenced by a driving force are discussed incorporating not influence of both entropy and jump frequencies but only that of jump frequencies on the diffusivity into them, for example such as a drift velocity. However, such concept as a drift velocity is not necessary for analyzing the diffusion equation, since we can solve a nonlinear diffusion equation in accordance with the fundamental theory in the mathematical physics, regardless of the discussion about diffusion fluxes.
The diffusivity is obtained from the Taylor expansion of a probability density function, satisfying the parabolic law [3] . This indicates that the analysis of diffusion equation is essentially possible in the parabolic space. The diffusivity is defined by an interaction between a diffusion particle and the diffusion field near the diffusion particle itself. This indicates that the diffusivity should be essentially investigated in the quantum mechanics, since the behavior of a micro particle should be investigated by analyzing the Schrödinger Equation (S equation) [18] .
Based on the suggestions mentioned above, the elegant analytical method in the parabolic space is reasonably established in the text [19] [20] . As a result, the solutions of the Boltzmann transformation equation for nonlinear interdiffusion phenomena were, for the first time, obtained as analytical expressions in the parabolic space [21] .
From applying the diffusion equation to a problem of diffusion elementary process, we derived the S equation [22] . It is revealed in the text that the diffusivity corresponds to the angular momentum operator in the quantum mechanics. As a result, the universal expression of diffusivity, which is applicable to every material in an arbitrary thermodynamic state, was obtained in proportional to the Planck constant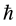 .
.
It was also found that the well-known material wave relation proposed by de Broglie in 1923, which is the most fundamental one in materials science, is obtained from a relation between diffusivity expressions [23] . This gives evidence for the theory discussed in the present study.
In the present work, we review the fundamental diffusion theories relevant to the general F2 law, where they are systematically reframed in points of view different from the previous works, adding some new discussions to them. The new findings obtained here will be widely applicable to fundamental problems as a standard theory in various actual diffusion phenomena.
2. Fundamental Theory of Diffusion Equation
As far as a material is conserved in the given region, the divergence theory shows that the diffusion equation is applicable to diffusion phenomena of every material in an arbitrary thermodynamic state. The diffusion information of a material, such as crystal material or amorphous material, and/or solid, liquid and gas states, is all incorporated into the diffusivity in the given diffusion equation. The diffusion equation with such an arbitrary diffusivity is thus investigated in the following.
In the present study, an abbreviate differential notation for an arbitrary independent variable
 and the well- known Dirac’s bracket notation for an arbitrary vector are used as follows [24] ;
and the well- known Dirac’s bracket notation for an arbitrary vector are used as follows [24] ;
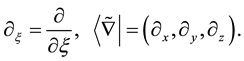
If an operator Q is Hermite one,
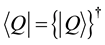 is valid in the Hermite conjugate
is valid in the Hermite conjugate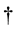 . Here, the notation
. Here, the notation
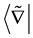 is thus defined as
is thus defined as
 because of
because of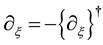 .
.
2.1. Diffusion Equation
The function
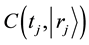 is defined as a normalized concentration where a diffusion particle in the initial state
is defined as a normalized concentration where a diffusion particle in the initial state
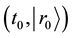 exists in the time and space state
exists in the time and space state
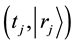 after j times jumps. A diffusion particle moves at random. It is, therefore, considered that the jump frequency
after j times jumps. A diffusion particle moves at random. It is, therefore, considered that the jump frequency
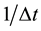 and jump displacement
and jump displacement
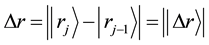 are equivalent in probability to their mean values of all diffusion particles in the collective system.
are equivalent in probability to their mean values of all diffusion particles in the collective system.
Since it is also considered that the probability of diffusion-jump from the state of




is thus valid. The Taylor expansion of both sides of Equation (2-1) yields


The substitution of Equations (2-2) and (2-3) into Equation (2-1) gives

Since the averaged





satisfying the well-known parabolic law [3] . Substituting Equations (2-5) into Equations (2-4), the diffusion equation is obtained as

where

The existence of a driving force affects diffusivity



where

On the other hand, using a drift velocity


in the usual textbooks (see Appendix A) [25] . However, the diffusion flux


In the present study, the discussion about diffusion fluxes is not necessary for analyzing the diffusion equation, since we can solve the nonlinear diffusion equation in accordance with the fundamental theory in the mathematical physics, regardless of diffusion fluxes.
In the following, therefore, Equation (2-6) is directly solved in accordance with the mathematical physics. It is in the nature of things that its analytical method is applicable to the diffusion problems relevant to a driving force then.
For a collective system composed of N




where





in the diffusion field. Defining the outflow flux as a plus value, the diffusion flux is

In mathematics, Equation (2-12) yields


where






2.2. Coordinate System of Diffusion Equation
As discussed later, the unified theory of diffusion problems shows that the foundation of diffusion is in the interdiffusion problems. In the interdiffusion problems, solvent particles as well as diffusion particles move in the diffusion region space. When a micro particle in a material jumps from a site M to an interstice space N, if we call such an interstice space “micro hole” including a vacancy in case of a crystal material, the micro hole of site N is annihilated and a new micro hole is generated at the site M after jumping.
The diffusion region space interacts with the space of diffusion system outside in accordance with the annihilation and/or generation of micro holes like the diffusion system becomes a thermal equilibrium state resulting from an antinomy between the principle of increase of entropy and that of minimum of free energy.
The coordinate system of the basic diffusion equation must be set in the diffusion field, since the diffusivity depends on an interaction between a diffusion particle and the diffusion field near the diffusion particle itself. In general, therefore, the diffusion problem should be investigated among three coordinate systems, where the origins of two coordinate systems are a point P of the coordinate system



In the present work, we investigate an interdiffusion problem applicable to every material in an arbitrary thermodynamic state. It is thus investigated using the original diffusion equation (2-11) of the coordinate system




The origins P







It is generally considered that P










must be physically valid.
Hereafter, we use the suffix P, Q and R for a physical quantity relevant to




When the coordinate origin of






are valid under the condition of



between differential operators are obtained then.
Using Equation (2-16) for Equation (2-11), the diffusion equations of


where



since

In the same manner of the above, the diffusion equation of


Since



where


3. Interdiffusion Problems
The interdiffusion problems of an N elements system in a diffusion couple composed of arbitrary materials A and B are discussed as follows.
In an amorphous material, we then estimate a specimen cross section perpendicular to the x axis where each cross section interval l corresponds to the averaged jump distance in the solid state or to the averaged collision distance in the gas or liquid state. In a crystal material, we estimate the jump distance l between the nearest neighbor crystal cross sections perpendicular to the x axis.
When materials A and B are in the solid state during a thermal diffusion, we conceive that the cross section S of the material A is uniform and equal to that of the material B. If materials A and B are in the fluid state, we conceive that they are in the receptacle corresponding to the case of solid materials. The interface of diffusion couple between materials A and B is smoothly joined. In that case, the origin



The material A is composed of N elements and we define the normalized concentration





The concentration and diffusivity of an element j are





The remained


The cross section of diffusion system is uniform and the shape variation of diffusion couple is usually negligible during the thermal diffusion. In that case, the relation of normalized concentrations yielding

is usually accepted on each cross section between
3.1. Correlation of Diffusion Fluxes with Coordinate Systems
Equations (2-17) and (3-1) yield

because of



using an arbitrary function












in the diffusion region


Using Equation (3-3) for the F1 law, the following relation of

is valid regardless of a coordinate system. The relation of

is also valid as discussed later in the self-diffusion theory.
Using Equations (3-5) and (3-6) for Equation (2-20), the relation of


is obtained. Here,








In the same manner, the following relations


are also obtained from Equations (2-13) and (2-18). Equations (3-7)-(3-9) show that the diffusion flux independent of the space coordinate corresponds to the velocity between the coordinate systems. The relation of

must be physically valid because of

because of Equations (3-7)-(3-10).
In the following,



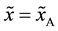



where the suffix









Equation (3-11) yields

Equation (3-12) shows the movement of



because of
3.2. Kirkendall Effect in Interdiffusion Problems
In the metallurgy field, the interface of





regardless of the space coordinate and it satisfies the parabolic law.
As far as the material shape of a diffusion couple is unchangeable, the K effect also reveals that the total quantity of element I diffusing across the K interface is different from that of element II. In other words, for the micro hole concentration








In the binary system interdiffusion problems, the F1 law shows that the relation of

is valid because of



However, the definition of diffusivity shows that the diffusivity is proportional to a statistical jump frequency of a diffusion particle at a point







In the present diffusion theory, the behavior of micro holes is visualized by the movement of an inert marker in the diffusion region because of the inert characteristic. Therefore, the K effect means a shift between the coordinate systems




satisfying the parabolic law.
The concept of intrinsic diffusion is thus unnecessary for understanding the K effect. Further, the K effect occurs in the interdiffusion not only of metal crystal but also of every material, since it is caused by a shift between the coordinate systems.
Equation (3-15) gives evidence that the K effect is caused by a shift between the coordinate systems, since it does not depend on the space coordinate. Substituting


Here, we must notice that




must be concretely investigated so as to be suitable for the concerned interdiffusion problems, since the m value is experimentally obtained.
3.3. Unified Theory of Diffusion Problems
The experimental results show that the relation of


Equation (3-17) of



In the following, various diffusion problems are systematically investigated by applying the diffusion flux of Equation (2-18) to them for

Equation (2-18) is then expressed as

(a) Interdiffusion:
Under the condition of
(i)
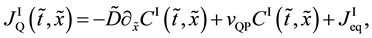
(ii)
(b) One-way diffusion:
Under the condition of
(i)
(ii)
(c) Impurity diffusion:
Under the condition of


(i)

(d) Self-diffusion:
Under the condition of


(i)

If we rewrite





the above theory is still valid in the N elements system. In the diffusion problems, it was thus found that the behavior of diffusion region space plays an important role for understanding diffusion mechanisms.
3.4. Self-Diffusion Theory
The diffusivity expression of Equation (2-5) is valid regardless of pure materials [3] . The Einstein theory of Brown motion shows that the self-diffusion of water is visualized by diffusion phenomena of pollen micro particles. It is thus obvious that the self-diffusion occurs in a pure material. However, even if we apply the F2 law to the self-diffusion problems in a pure material, we cannot understand whether the self-diffusion occurs or not, since the concentration is unchangeable in the self-diffusion system.
Langevin theoretically investigated not the diffusion equation of collective motion but the motion equation of a diffusion particle with a viscosity resistance in a pure liquid material [27] . It was thus revealed that the diffusion particle moves in accordance with the normal distribution rule.
The theories of Langevin as well as Einstein are relevant to diffusion particles with a driving force in a pure material. In other words, the diffusion phenomena of a pure material itself are not directly investigated. However, Equation (2-5) shows that the self-diffusion occurs in a pure material even if there is no driving force in the diffusion system.
The self-diffusion problems have been also experimentally investigated as an impurity diffusion problem by using small quantities of the isotope material. Strictly speaking, however, the self-diffusion problems are obviously different from the impurity ones, as can be seen from the difference of (i) between (c) and (d) in Section 3.3.
As discussed in Section 2.1, the corresponding F 1 law to the F2 law is incomplete without the intrinsic diffusion flux. In the following, therefore, the self-diffusion problems of a pure material are directly investigated in accordance with the intrinsic diffusion flux, regardless of a driving force.
(1) Application of intrinsic diffusion flux to self-diffusion problem
We cannot experimentally confirm




Using the coordinate notation



is obtained. If the concentration difference between




Taking account of


Equation (3-21) shows an instantaneous concentration gradient in the localized time


The behavior of concentration distribution in the local space is thus obtained as

In a pure material or a material in the thermal equilibrium state, Brownian particles move in accordance with Equation (3-22). If


If we apply Equation (3-22) to the interdiffusion problems, the relation of

is valid in accordance with Equation (3-3). Equation (3-23) reveals that Equation (3-6) is valid because of

Using the notation


under the condition of

The diffusion region of self-diffusion problem of a pure material is, for convenience, divided into 2 regions as a binary system interdiffusion problem composed of a pure material I of










Using Equation (3-24) for


are obtained, where







On the other hand, if we pay attention to both sides of







(2) Physical meaning of intrinsic diffusion flux
Here, we estimate the




where





We cannot actually observe


where


Equation (3-25) independent of

From the historical point of view, if the F1 law were investigated at an early stage in accordance with the Gauss divergence theory, we might understand the Brown motion behavior before the Einstein theory [2] [3] [14] .
3.5. Diffusion Equation of Micro Holes
The space coordinate dependence of micro holes has been neglected in the diffusion theory mentioned above. In the metallurgy field, however, the experimental results of multiple markers show that their movements depend on the space coordinate in accordance with a marker position set in the initial diffusion couple [28] - [30] . This indicates that we must investigate the space coordinate dependence of micro holes in the diffusion study in detail, since the behavior of micro holes is visualized by the characteristic of inert markers.
In that case, Equation (3-1) is rewritten as

where




material A and that of



For the thermal equilibrium concentration of micro holes



where


where


The K-effect shows that the concentration of micro holes yields a supersaturated section and an unsaturated one in the diffusion region. Therefore, the averaged value of diffusion flux yielding

is considered to be negligible. We thus assume that the averaged contribution of the generation/annihilation term to the total diffusion flux in the diffusion system is negligible.
Here, if we integrate Equation (3-29) with respect to x, the diffusion flux relation of

is obtained by neglecting the contribution of Equation (3-30). In that case, Equations (3-27), (2-11) and (3-31) yield

under the condition of

Equation (3-32) shows that we can accept a micro hole as a virtual diffusion particle.
The diffusion equation of


using a common interdiffusion coefficient



should be investigated. Equation (3-34) shows that the behavior of multiple markers depends on the time and space, since the obtained

3.6. Application of Present Theory to Actual Diffusion Problems
Hereinbefore, we discussed some fundamental problems in the bulk diffusion of an ideal diffusion couple. The interdiffusion problems for a many elements system, such as an alloy, a compound semiconductor, a multilayer thin film, a microstructure material and so on, have been widely investigated [4] [31] - [34] . It is then fundamentally indispensable for the material science investigation to understand the N (
In the diffusion study history, the concept of intrinsic diffusion has been accepted in order to understand the K effect. In the existing theory of interdiffusion problems, the Darken equation derived from the intrinsic diffusion concept has been widely used for analyzing them [35] . The application of the Darken theory to a many elements system interdiffusion was extremely complicated even if it was a ternary elements system [36] . On the other hand, the simple relation among interdiffusion coefficients obtained here will be extremely useful for analyzing various interdiffusion problems.
As can be seen from the Appendix [B], we revealed that the Darken equation is not mathematically valid. It is also reported that the analytical results obtained by using the Darken equation are not consistent with the experimental results [37] . This fact is consistent with the present theory where there is no such concept of intrinsic diffusion in the diffusion phenomena.
Under the condition of Equation (3-27), therefore, it is necessary for analyzing a many elements system interdiffusion problem to solve the diffusion equations (3-33) and (3-34) of each element in accordance with their initial and boundary conditions. Here, it seems that these diffusion equations are independent of each other since they have a common interdiffusion coefficient. However, it is not independent because of Equation (3-27).
In the parabolic space discussed later, the analytical method of a nonlinear diffusion equation is established. The analytical solutions of Equations (4-48) and (4-49) are thus obtained then. Using them for analyzing each of the above equations, we can fundamentally solve an N (
Since the present analytical theory is fundamental, it will be necessary for actual diffusion problems to modify it partly in accordance with the concerned problems like the Boyle Charles law for an ideal gas state is modified into the van der Waals equation in the actual case by incorporating an interaction between gas particles into it.
4. Analysis of Diffusion Equation in Parabolic Space
Einstein theoretically revealed that the Brownian particles randomly move in accordance with the parabolic law [3] . After that, it was experimentally confirmed by Perrin [38] . The parabolic law is universally shown in phenomena relevant to the normal distribution in a probability problem. The Brown problem is a study subject not only in the material science but also in the complex-system sciences relevant to the Markov process. Therefore, the analytical method of the Brown problem is mathematically common in various science fields.
The diffusion junction depth is directly relevant to the parabolic law. The diffusion problem is justifiably one of the Brown problems. This indicates that the solutions of diffusion problems are possible in the parabolic space. In other words, the analytical method discussed here is widely applicable to various Brown problems.
The F2 law is a continuous equation in the conservation system under the condition of no sink and source of diffusion particles, and it is one of the basic equations in physics. When the existence of a driving force or the sink and/or source of diffusion particles is negligible in a given diffusion system, Equation (2-6) is a parabolic type linear partial differential equation in the evolution equations. A lot of diffusion problems have been investigated by solving the diffusion equation of the time and space

When the diffusivity depends on the concentration, the diffusivity becomes a function of the independent variables


4.1. Definition of Parabolic Space
For convenience, hereafter we use the coordinate notation






and

From the similar calculation, the differential operators in the parabolic space are obtained as

where


4.2. Diffusion Equation in Parabolic Space
If we directly apply Equation (4-1) to homogeneous partial differential Equation (2-6), the ellipse type differential equation is obtained as

where

If



which is the well-known Boltzmann transformation equation [21] . Further, it is rewritten as

The integral calculation of Equation (4-4) yields the integro-differential equation given by

where
Here, if we define

the expression of

is obtained from using Equation (4-6) for Equation (4-5). Boltzmann transformation equation (4-3) is essentially equivalent to Equation (4-7).
We can accept Equation (4-7) as a diffusion flux in the parabolic space. The F1 law in the time and space cannot be used directly for analyzing a diffusion problem. However, the diffusion flux of Equation (4-7) is possible for analyzing a diffusion problem under the initial condition of Equation (4-6).
Further, as described later, the solutions of three dimensions problems in the parabolic space

In the parabolic space

where the component of


using


Equation (4-8) in the parabolic space



is generally valid, if the diffusivity D depends on independent variables. Equation (4-10) is extremely useful for analyzing a nonlinear diffusion equation.
4.3. Analytical Solutions of Linear Diffusion Equation
The utility of the present method is confirmed through solving some typical diffusion problems by concrete calculations. The mathematical method used here is also widely applicable to the Brown problems in various science fields.
(1) One dimension parabolic space
We first solve the linear diffusion equation given by

under the initial and boundary conditions of

The initial condition of Equation (4-12) is rewritten as

in the parabolic space
Substituting




is obtained. Using Equation (4-14) for Equation (4-5), the ordinary differential equation of

is obtained.
From the definition of the well-known error function, the general solution of Equation (4-15) is obtained as

where


In the analytical method in the parabolic space, the solution is thus easily obtained only by the elementary integral. It is, therefore, obvious that the present analytical method is extremely superior in analyzing diffusion equations to the existing methods.
(2) Parabolic space of n dimensions
In case of


Using this equation for

is obtained from Equation (4-8).
The operation of


We solve the ellipse type differential equation (4-19) corresponding to Equation (2-6) under the initial and boundary conditions in the following.
The initial and boundary values of Equation (2-6) given by

correspond to

in the parabolic space
In the analysis of Equation (4-19), it is easily found that the equation of

satisfies Equation (4-19). In other words, Equation (4-22) is the particular solution of Equation (4-19). When the general solution of Laplace equation of

is obtained as

in accordance with the mathematical theory.
Assuming the equation of

and using the variable separation method, the solution of Equation (4-23) is possible. Substituting Equation (4-25) into Equation (4-23), the relation of

is valid. In order to satisfy this equation for an arbitrary

must be valid under the condition of

Equation (4-27) yields



where



In case of



into Equation (4-25), the general solution of Equation (4-23) is obtained as

where


under the condition of


where

The solution of Equation (4-19) is thus obtained as

If we use



The present study reveals that the particular solution is easily possible and the complementary function of Equation (4-19) is a constant value to satisfy the given initial and boundary conditions because of the singular characteristic of initial conditions. In other words, the diffusion behavior is incorporated into the inhomogeneous term of the Poisson equation. As a result, the solution is obtained as a linear combination of the error functions.
Although the diffusion problems have not ever been investigated in the parabolic space, it is obvious that the present solution is thus exceedingly simple and elegant, compared with the usual solution of

for

(3) Analysis of inhomogeneous diffusion equation
The research subjects of linear diffusion phenomena in detail are often expressed by an inhomogeneous partial differential equation. Therefore, the application of the present method to those problems is investigated. Generally, such linear diffusion equations are expressed as

where L is the linear operator of

From the mathematical theory, the solution of inhomogeneous Equation (4-34) is obtained as

where


and


Equation (4-1) shows that the differential operators in the parabolic space are not able to apply to Equation (4-34). However, the linear operator in the parabolic space given by

is possible for analyzing the homogeneous Equation (4-37). In other words, we can obtain the complementary function by the present method. In order to obtain a solution of an inhomogeneous diffusion equation, therefore, we must obtain a particular solution by the existing methods.
4.4. Analytical Solutions of Nonlinear Diffusion Equation
Using the experimental profile



(1) Analytical method in parabolic space
When the diffusivity is affected by a driving force, the mathematical solutions of the nonlinear equations (2-11) and (4-3) or (4-7) are impossible if no other relation between diffusivity and concentration is given. In that case, the diffusivity depends on the independent variables and Equation (4-10) is generally valid in mathematics then. Equations (4-7) and (4-10) are thus simultaneously solved in the following.
The integro-differential equation (4-5) is superior in the approximate calculation to the second order differential equation (4-3). For example, using an effective diffusivity


is valid because of the characteristic of the integral calculation, where D is rewritten as an interdiffusion coefficient
In order to clarify the validity of the present method, we applied the present method to the typical problems of a binary system interdiffusion where the complete solid solution is formed in the interdiffusion region between solid metals. The reason for that is as follows. The Boltzmann Matano method has been widely used for the analysis of various interdiffusion problems. Uncountable papers have been reported and useful findings have been thus accumulated. Even if the present results are thus evaluated by comparing them with the Boltzmann Matano results, the generality of present method is still kept from the viewpoints of mathematical physics.
In the following analysis, the relations of

are adopted as an initial condition in the parabolic space. The coordinate origin




because of


The countless experimental results always reveal that the




The relation of


Using a constant value



where



Substituting Equations (4-5) and (4-41) into Equation (4-10) yields the diffusion flux relation of

For the diffusion flux, the physical speculation produces the relation yielding

where




In relation to a behavior of the error function, Equation (4-44) is divided into following two equations. One is

and the other is

where





There is evidence for the validity of the above division as shown in the following. When the relation of

is valid, Equation (4-46) is approximately rewritten as


under the condition of Equation (4-40). In the typical interdiffusion problems between solid metals, Equation (4-47) has been widely accepted [40] - [43] . The derivation of Equation (4-47) gives evidence for the validity of division of Equation (4-44) into Equations (4-45) and (4-46).
Since it is thus confirmed that the division of Equation (4-44) into Equations (4-45) and (4-46) is reasonable, Equation (4-45) is analyzed in accordance with mathematical theory. As can be seen from the analyzing process in Ref. [19] , the calculations are considerably complex. As a result, the general solution of

was obtained as an analytical expression using initial values, where




Here, the relation of



Subsequently, using the obtained solution of Equation (4-48) for Equation (4-46), Equation (4-46) was analyzed also through considerably complex calculations. The general solution of

was thus obtained also as an analytical expression.
Here, the notations used for Equations (4-48) and (4-40) are as follows:

The validity of the present analytical method is numerically investigated by comparing Equations (4-48) and (4-50) with the results of the Boltzmann Matano method. As a result, we found that the present solutions agree well with those of the Boltzmann Matano method [19] .
When the diffusivity does not depend on independent variables, the relation of





Equations (4-48) and (4-50) agree with

The K effect


by transforming Equation (3-15) into the parabolic space under the condition of
(2) Meaning of general solutions obtained as analytical expressions
Since the general solutions are obtained as Equations (4-48) and (4-50), the interdiffusion problems are fundamentally solved as follows:
We denote an impurity diffusivity


posed of the element I. We first investigate an interdiffusion problem of the diffusion couple between pure materials A and B. It is then well-known that the relations of


are physically valid.
The solution








Substituting these






Subsequently, using






The solutions of an arbitrary binary system interdiffusion problem are thus possible by using impurity diffusivities in the concerned diffusion system. In the N elements system interdiffusion problems, using an impurity diffusivity of an element in the concerned diffusion couple, their solutions are also possible.
In the diffusion history, the nonlinear F2 equation had not been analytically solved until recently, although the numerical solutions were possible. In order to understand the diffusion problems, the corresponding diffusion fluxes to the F2 equation had been thus widely discussed in the existing theories, since it was considered that the mathematical solutions of nonlinear diffusion equation are impossible. For example, such a diffusion flux in the Appendix A has been discussed.
However, the discussion about such a diffusion flux is now unnecessary for understanding the diffusion problems, since the analytical solutions of Equations (4-48) and (4-50) relevant to the nonlinear F2 equation are obtained, regardless of the diffusion fluxes. In other words, as can be seem from Equation (4-49), we can understand the influence of a driving force on the diffusion flux from the obtained solutions.
Based on the above discussions, it is extremely meaningful that the solutions of the nonlinear F2 equation are obtained as analytical expressions.
(3) Nonlinear problems of three dimensions in parabolic space
The analytical solutions of linear diffusion equations were obtained in the parabolic space. Further, we could also obtain the physically meaningful solutions of nonlinear diffusion equation as analytical expressions in the one dimension parabolic space.
In mathematics, it is almost impossible to solve two or three dimensional nonlinear diffusion equation. However, we investigate two or three dimensional problems in the parabolic space in the following.
We rewrite Equation (4-2) into

where

It is impossible to analyze Equation (4-54) exactly since






because of
By reference to Equation (4-19), the solution of Equation (4-56) is obtained as

where

5. Universal Expression of Diffusivity
From applying the diffusion equation to a collision problem between two micro particles in the diffusion elementary process, the S equation is reasonably derived. It is revealed that the diffusivity corresponds to the angular momentum operator in the quantum theory. The universal diffusivity expression, which is applicable to every material in an arbitrary thermodynamic state, is obtained using an essential diffusion constant then. Further, the correlation between the well-known material wave relation and the diffusivity expression is discussed in a minute time and space region.
5.1. Derivation of Schrödinger Equation
If the movement of a material particle is not the Markov process but the continuous process, Equation (2-1) is rewritten as

where
From the Taylor expansion of both sides of Equation (5-1), the well-known wave equation is obtained as

where



where A and





The collective behavior of micro particles relevant to the Markov process is expressed by Equation (2-6) derived from their random movements. However, it is revealed in the quantum mechanics that a micro particle has a wave characteristic. The relation of material wave yielding

was thus proposed by de Broglie in 1923, where




where



where m is the particle mass and

When Equation (5-4) is applied to a micro particle behavior, the left-hand side of Equation (5-4) means the particle motion in accordance with the Markov process. On the other hand, the right-hand side means the continuous process resulting from the wave characteristic of the micro particle during a free motion because of the wave factor
An activation energy caused by a local thermal fluctuation generates micro holes in a material and at the same time micro particles are possible to jump instantaneously to the nearest neighbor micro holes through each energy barrier. The macro behavior of their collective motion is investigated by using the diffusion equation (2-6).
On the other hand, behavior of a micro particle of mass m in the diffusion field is investigated by the well-known Langevin equation of

where





The Langevin equation shows that the behavior of a Brown particle in the minute time and space region is incorporated into the force



caused by a collision between two micro particles in the minute time and space region.
Since the physical essence is still kept even if we consider the simplest collision problem of one dimensional case, we thus investigate a perfect elastic collision problem between micro particles A and B of the same kind. We consider that the particle A moves with a velocity



If we can then clarify the distinction between A and B after the collision, the particle A decelerates from the velocity


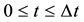
On the other hand, if we cannot clarify the distinction between the particles A and B after the collision, it seems that the particle A decelerates from the velocity




If we accept the above latter case, the relation of






It is considered that the above former case corresponds to the Markov process caused by a collision and the latter case corresponds to a continuous process caused by the impossibility of identification between the micro particles. It is thus mathematically necessary for the transformation from the Markov process into the continuous process to introduce the imaginary time into a collision problem between micro particles.
In physics, it is difficult to accept the imaginary time in accordance with the continuity of time. In mathematics, however, the product of a physical quantity Q and an imaginary time




Rewriting the concentration



Assuming




where


Here, using the real functions




Further, substituting Equation (5-11) into Equation (5-9) and multiplying both sides by

because of
From the partial differential equation of probability, the diffusivity of

is obtained. When a micro particle randomly jumps from a position to another one, the jump orientation becomes the spherical symmetry in probability. Using the relation of





where



The relation of


Substituting Equation (5-13) into Equation (5-12) gives

Here, if we define the imaginary differential operator given by

Equation (5-14) is rewritten as

Further, the substitution of Equation (5-6) into Equation (5-16) yields the S Equation (5-5). The defined Equation (5-15) is one of the essential operators in the quantum mechanics.
It is considered that the relation of



is thus easily obtained because of



At the same time, this gives evidence for the validity of Equation (5-13) derived from the correlation between the diffusion equation and the S equation. It is, therefore, considered that the diffusivity is fundamentally relevant to the Planck constant

5.2. New Universal Diffusivity
The definition of diffusivity shows that the diffusivity should be essentially determined by analyzing the S equation, since the diffusion particle such as an atom and/or a molecule moves as a material wave packet in the minute space. However, the existing diffusivity has been defined by the classical mechanics. In the following, therefore, a new diffusivity is investigated.
The existence probability P of a micro particle in the collective system of a heat quantity Q and an absolute temperature T is given by the well-known Boltzmann factor of

where

The jump frequency of a diffusion particle is relevant to a factor
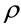



where the molecular or the atomic weight n is expressed as




Equation (3-32) shows that diffusion particles move randomly with a common diffusivity in the concerned diffusion field. On the other hand, Equation (5-18) shows that the diffusivity depends on the mass of diffusion particle. For the diffusion experimentation in a super gravity field, therefore, it is considered that an individuality of a micro particle resulting from the mass effect will be shown in the diffusivity, compared with one in a usual laboratory.
Hereafter, the investigation of

5.3. Influence of a Driving Force on Diffusivity
The new diffusivity of Equation (5-18) is derived regardless of the bulk diffusion problems. However, it is considerably difficult to determine


If we incorporate the potential energy


for a material composed of micro particles in the state of an absolute temperature



Equation (5-18) is thus valid when a driving force is nonexistent in the diffusion system. In other words, the relation of

is considered to be valid between Equations (2-6) and (5-18) for the common


As can be seem from Appendix A in the existing theory, the effect of a driving force on the diffusivity is incorporated into the difference of a jump frequency between nearest neighbor crystal plane 1 and plane 2. In relation to the situation, we discuss the difference between the present diffusivity expression and the existing one as follows.
The existence probability of a micro particle in a material depends on the Boltzmann factor of Equation (5-17). The potential energy



The difference of existence probabilities between a micro particle on the plane 1 and one on plane 2 corresponds to that of the frequencies between them. The effect of a driving force on diffusivity should be thus incorporated into the Boltzmann factor as a potential energy

When the potential energy is not so much large, i.e.,


incorporating a driving force effect into the Boltzmann factor for



is valid. Although the diffusivity gradient of

has the velocity dimension, it is directly relevant to a driving force F because of the definition of mechanics
Substituting Equations (5-22)-(5-24) into the one dimension diffusion equation of the general nonlinear Fickian second law given by

the diffusion equation is obtained as

where


In view of the derivation process, the diffusivity expression of Equation (5-27) is applicable to every material in an arbitrary thermodynamic state.
5.4. Operators in Quantum Mechanics
In the quantum mechanics, energy E, momentum





We cannot observe imaginary physical quantities. Therefore, the eigenvalues of their operators are meaningful in the quantum theory.
In a collision problem mentioned above, the concept of acceleration disappears in the quantum theory because of
It is physically considered that the wave image of a micro particle disappears in the quantum theory when the corresponding wave packet exists in the minute space region



and it converges at zero in a minute time. For the wave image, the relations of






In mathematics, except for physical constant quantities, physical variables with odd time or space dimensions should be thus accepted as physical operators in the minute time and space region.
The kinetic energy








Using relations of





yields the operator




We investigate a characteristic of diffusivity in the minute time and space regions in the following. Equation (2-5) is rewritten as

for a particle image. Equation (5-4) is also rewritten as

because of



is obtained as an operator.
Substituting Equation (5-32) into Equation (2-6), we have

Multiplying both sides of Equation (5-33) by



6. Results
In 1855, Fick applied the heat conduction equation proposed by Fourier in 1822 to diffusion phenomena as it had been. In general, however, the discussion of coordinate transformation of the general nonlinear F2 law is indispensable for understanding the diffusion phenomena. Nevertheless, the coordinate transformation of a diffusion equation had not been discussed for a long time until the recent works [14] - [16] [19] [20] [22] .
Applying the divergence theorem to a diffusion equation, therefore, we investigated the problems of coordinate transformation in the diffusion systems. In relation to the investigation, a new diffusion flux of Equation (2-13) was defined in accordance with the mathematical physics. The new diffusion flux revealed that the F1 law is incomplete without the intrinsic diffusion flux.
The coordinate transformation theory shows also that the K effect is caused by a shift between the coordinate systems like the Doppler effect of the wave equation is caused by a shift between them. The theoretical Equation (3-15) relevant to the K effect was reasonably obtained in accordance with the empirical Equation (3-14) then. At the same time, it was also revealed that the concept of intrinsic diffusion is nonexistent in the diffusion theory, although we had accepted as an indispensable concept for understanding the K effect for a long time until recently.
In relation to the coordinate transformation theory, further, using the new diffusion flux applicable to various diffusion problems, the unified diffusion theory relevant to every material in an arbitrary thermodynamic state was established, including a self-diffusion theory and also an N elements system interdiffusion theory.
The self-diffusion theory has been indirectly discussed in the existing one, where the self-diffusion behavior of a pure material is visualized by any other diffusion particles in the pure material. In the present theory, the self-diffusion mechanism of a pure material itself is discussed using the intrinsic diffusion flux. In an N elements system interdiffusion, it was found that each of diffusion particles moves randomly with a common diffusivity

The essence of diffusion phenomena is shown in the parabolic law. Based on the concept, a new diffusion flux applicable to analyzing diffusion problems was derived as Equation (4-7) and/or (4-8) in the parabolic space. The analytical method in the parabolic space was thus established. It was then revealed that the analysis in the parabolic space is exceedingly superior in calculation to the existing analytical methods. For example, the solutions of nonlinear diffusion equation were, for the first time, obtained as the analytical expressions of Equations (4-48) and (4-50). The effect of a driving force on the diffusivity was then obtained as Equation (4-49) from the solution of Equation (4-48).
The definition of diffusivity indicates that the diffusivity should be essentially investigated as a few many bodies problem in the quantum mechanics. Based on the concept, the S equation was derived from applying the diffusion equation to a diffusion elementary process. In the analytical process, we found that the diffusivity corresponds to an angular momentum operator in the quantum mechanics. The new universal diffusivity expression was thus obtained as Equation (5-27) using an essential diffusion constant of Equation (5-19). It is essentially applicable to every material in an arbitrary thermodynamic state.
It was also found that the material wave equation proposed by de Broglie, which is the most fundamental relation in the materials science, was derived from the relation between diffusivity expressions of Equations (2-5) and (5-13) in the diffusion elementary process. This gives evidence for the validity for the present diffusion theory.
7. Discussion
Even if a driving force exists in the diffusion system under the condition of no sink and source, the divergence theorem shows that a collective behavior of micro particles depends only on a diffusivity of the general nonlinear F2 law. Whenever the diffusivity of a micro particle in a material is given, the collective behavior of micro particles is thus determined only by solving the diffusion equation. The diffusivity depends on a material internal structure composed of micro particles. Therefore, the investigation of diffusivity is the most important subject in the diffusion study. Based on the concept discussed here, the present study was performed.
In relation to the Darken equation, even if a concentration profile is reasonably reproduced by using intrinsic diffusion coefficients as parameters, we cannot directly evaluate its validity of the used diffusivity values, since the diffusivity profile is not obtained by a direct observation. In other words, we can usually perform the numerical simulation to a high precision only by using fitting parameters for the given equation, regardless of a physical validity. As far as the Darken equation is not mathematically correct as shown in the Appendix B, it is thus necessary for us to reexamine a great many results of interdiffusion problems obtained by using the unsuitable Darken equation.
As revealed in the text and Appendix B, we believe that the F1 law and the intrinsic diffusion concept as well as the Darken equation are misunderstanding matters in the diffusion history. Nevertheless, they have been accepted as reasonable ones for such a long time in the concerned field.
If we consider serious influences of their misunderstanding matters on student education and young researcher training in the concerned field, they should be solved as soon as possible, since they have been still described as plausible ones in a lot of existing textbooks. One of the present work aims is thus to make them universally known in the concerned field.
Since the general solutions of nonlinear diffusion equation are analytically obtained in the parabolic space, the diffusion problems under the condition of no sink and source are essentially solved. The influence of a driving force on the diffusivity is thus reasonably obtained as Equation (4-49) from the analytical solutions of Equations (4-48) and (4-50). The discussions about diffusion fluxes are thus now essentially unnecessary for fundamental diffusion problems.
In the actual diffusion problems in detail, further investigation will be required for various difficult problems, for example, such a problem where the sink and source exists in the concerned diffusion system. Even in that case, however, it is no doubt that the fundamental theories discussed here are still useful for their fundamental investigations, just because of fundamental ones.
The investigations relevant to the many elements system interdiffusion problems, such as an alloy, a compound semiconductor, a multilayer thin film, and a microstructure material, are extremely important as an actual problem. Under the condition of Equation (3-27), corresponding Equations (4-48) and (4-50) to solutions of Equation (3-33) and using solutions of Equation (3-34) obtained numerically for a many elements system interdiffusion problems, their solutions are possible.
As mentioned at the beginning of this section, the foundation of diffusion problem is relevant to not the diffusion flux but the diffusion equation for the concerned diffusion system. The influence of a driving force on a diffusion system should be thus incorporated into the diffusivity of the concerned diffusion equation, e.g., Equation (5-25), although the problem relevant to a driving force is discussed in the existing theory using a drift velocity in the diffusion flux representation (see Appendix A).
In the present study, the fundamental diffusion problems are surveyed in accordance with the mathematical physics. As a result, the new useful findings for the diffusion study are reasonably derived from the essential discussions about the general nonlinear Fickian diffusion equation. In particular, the analytical method established in the parabolic space will be applicable not only to diffusion problems but also to Brown problems in various science fields as a useful method.
Finally, we hope that the new findings are useful for the material science researches and also that the present aims are accomplished.
Cite this paper
TakahisaOkino, (2015) Mathematical Physics in Diffusion Problems. Journal of Modern Physics,06,2109-2144. doi: 10.4236/jmp.2015.614217
References
- 1. Markov, A.A. (1960) American Mathematical Society Translations Series, 15, 1-14.
- 2. Brown, R. (1828) Philosophical Magazine N. S., 4, 161-173.
- 3. Einstein, A. (1905) Annals of Physics, 18, 549-560.
http://dx.doi.org/10.1002/andp.19053220806 - 4. Skvortsov, A.A., Orlov, A.M. and Muradov, V.E. (2009) Technical Physics Letters, 35, 606-609.
http://dx.doi.org/10.1134/S1063785009070062 - 5. Ting, P.Y. (1994) IEEE Transactions on Neural Networks, 5, 622-638.
http://dx.doi.org/10.1109/72.298232 - 6. You, Y.L., Xu, W.Y., Tannenbaum, A. and Kaveh, M. (1996) IEEE Transaction on Image Processing, 11, 1539-1553.
- 7. Das, I., Agrawal, N.R., Gupta Sanjeev, K., Gupta Sujeet, K. and Rastogi, R.P. (2009) Journal of Physical Chemistry A, 113, 5296-5301.
http://dx.doi.org/10.1021/jp8064147 - 8. Raji, A., Kuceyeski, A. and Weiner, M. (2012) Neuron, 73, 1204-1215.
http://dx.doi.org/10.1016/j.neuron.2011.12.040 - 9. Zhou, Q. (2015) International Journal of Multimedia and Ubiquitous Engineering, 10, 173-182.
http://dx.doi.org/10.14257/ijmue.2015.10.1.16 - 10. Gauss, C.F. (1840) Res Beob Magn Vereins, 4, 1.
- 11. Fourier, J.B.J. (1822) Theorie analytique de la chaleur. Didot, Paris, 499-508.
- 12. Mehrer, H. (2007) Diffusion in Solid. Springer-Verlag, Berlin Heidelberg.
http://dx.doi.org/10.1007/978-3-540-71488-0 - 13. Fick, A. (1855) Philosophical Magn J Sci, 10, 31-39.
- 14. Okino, T. (2012) Journal of Modern Physics, 3, 1388-1393.
http://dx.doi.org/10.4236/jmp.2012.310175 - 15. Okino, T. (2013) Journal of Modern Physics, 4, 1495-1498.
http://dx.doi.org/10.4236/jmp.2013.411180 - 16. Okino, T. (2014) Applied Physics Research, 6, 1-7.
http://dx.doi.org/10.5539/apr.v6n2p1 - 17. Smigelskas, A.D. and Kirkendall, E.O. (1947) Transactions of the American Institute of Mining, Metallurgical and Petroleum Engineers, 171, 130-142.
- 18. Schrödinger, E. (1926) Annals of Physics, 79, 361-376.
http://dx.doi.org/10.1002/andp.19263840404 - 19. Okino, T. (2011) Materials Transactions, 52, 2220-2227.
http://dx.doi.org/10.2320/matertrans.M2011137 - 20. Okino, T. (2012) Journal of Modern Physics, 3, 255-259.
http://dx.doi.org/10.4236/jmp.2012.33034 - 21. Boltzmann, L. (1894) Annual Review of Physical Chemistry, 53, 959-964.
http://dx.doi.org/10.1002/andp.18942891315 - 22. Okino, T. (2013) Journal of Modern Physics, 4, 612-615.
http://dx.doi.org/10.4236/jmp.2013.45088 - 23. de Broglie, L. (1923) Nature, 112, 540.
http://dx.doi.org/10.1038/112540a0 - 24. Dirac, P.A.M. (1958) Principles of Quantum Mechanics. Clarendon Press, Oxford.
- 25. Manning, J.R. (1972) Theory of Diffusion. In Diffusion American Society for Metals, 1-23.
- 26. Matano, C. (1933) Japanese Journal of Physics, 8, 109-113.
- 27. Langevin, P. (1908) Comptes Rendus de l’Académie des Sciences (Paris), 146, 530-533.
- 28. Cornet, J.F. and Calais, D (1972) Journal of Physics and Chemistry of Solids, 33, 1675-1684.
http://dx.doi.org/10.1016/S0022-3697(72)80463-3 - 29. Cornet, J.F. (1974) Journal of Physics and Chemistry of Solids, 35, 1247-1252.
http://dx.doi.org/10.1016/S0022-3697(74)80148-4 - 30. Iijima, Y., Hirano, K. and Kikuchi, M. (1982) Transactions of the Japan Institutetute of Metals, 23, 19-23.
http://dx.doi.org/10.2320/matertrans1960.23.19 - 31. Alizadeh, R., Mahmudi, R., Ngan, A.H.W. and Langdon, T.G. (2015) Journal of Materials Science, 50, 4940-4951.
http://dx.doi.org/10.1007/s10853-015-9041-x - 32. Saad, A., Fedotov, A., Velichko, O., Pachynin, V. and Davydko, A. (2006) Physica Status Solidi (b), 12, 2665-2671.
http://dx.doi.org/10.1002/pssb.200541272 - 33. Wang, C.W., Yiu, P., Chu, J.P., Shek, C.H. and Hsueh, C.H. (2015) Journal of Materials Science, 50, 2085-2092.
http://dx.doi.org/10.1007/s10853-014-8770-6 - 34. Gong, M.M., Castro, R.H.R. and Liu, F. (2015) Journal of Materials Science, 50, 4610-4621.
http://dx.doi.org/10.1007/s10853-015-9010-4 - 35. Darken, L.S. (1948) Transactions of the American Institute of Mining, Metallurgical and Petroleum Engineers, 175, 184-201.
- 36. Xin, L., Schnell, S.K., Simon, J.M., Krgüer, P., Bedeaux, D., Kjelstrip, S., Bardow, A. and Vlugt, T.J.H. (2013) International Journal of Thermophysics, 34, 1169-1196.
http://dx.doi.org/10.1007/s10765-013-1482-3 - 37. Meyer, A. and Kargl, F. (2013) Int J Microgravity Sci Appl, 30, 30-35.
- 38. Perrin, J. (1909) Annales de Chimie et de Physique, 18, 5-114.
- 39. Shewmon, P.G. (1963) Diffusion in Solids. Series in Materials Science and Engineering. McGraw-Hill, New York.
- 40. Onishi, M., Ikeda, T., Wakamatu, Y. and Shimozaki, T. (1988) Transactions of the Japan Institutetute of Metals, 29, 383-387.
http://dx.doi.org/10.2320/matertrans1960.29.383 - 41. da Silva, L.C.C. and Mehl, R.F. (1951) Transactions of the American Institute of Mining, Metallurgical and Petroleum Engineers, 191, 155-157.
- 42. Hoshino, H., Iijima, Y. and Hirano, K. (1982) Transactions of the Japan Institutetute of Metals, 21, 674-682.
http://dx.doi.org/10.2320/matertrans1960.21.674 - 43. Onishi, M. and Miura, H. (1977) Transactions of the Japan Institutetute of Metals, 18, 107-112.
http://dx.doi.org/10.2320/matertrans1960.18.107 - 44. Boltzmann, L. (1872) Wiener Berichte, 66, 275-370.
- 45. Ohya, M. and Watanabe, N. (2010) Entropy, 12, 1194-1245.
http://dx.doi.org/10.3390/e12051194 - 46. John, L. and Haller, J. (2013) Journal of Modern Physics, 4, 1393-1399.
http://dx.doi.org/10.4236/jmp.2013.410167
Appendix A
In Ref. [25] , the influence caused by a driving force in the planar diffusion phenomena is investigated in relation to jump frequencies of diffusion particles between the nearest neighbor crystal planes.
The net diffusion flux J of diffusing particles across a plane between two nearest neighbor crystal planes 1 and 2 is expressed as

where




The relation between concentration



where a is a lattice constant and

Here, the following relations are considered as;

where
Substituting Equations (A-2), (A-3) into Equation (A-1), the relation of

is obtained. Using the notations given by


the diffusion flux of

is derived. Here,



On the other hand, if we use the Taylor expansion in the above calculations, the relation of

and

are valid. Using Equations (A-7), (A-8) for Equation (A-1), the relation similar to Equation (A-6) is obtained as

where


Equation (A-9) should be essentially equivalent to Equation (2-7) as shown by

However, it seems that Equation (A-11) is not generally valid. The reason is as follows.
When a driving force exists in the diffusion system, it affects entropy and jump frequencies of diffusion particles. As a result, the diffusivity is affected by a driving force, since the diffusivity depends directly on entropy and a frequency. The problem of entropy is extremely important for the diffusion phenomena. However, the effect of entropy is not taken into account in the derivation process of Equation (A-6) or (A-9). Therefore, the diffusion flux discussed here is not generally applicable to the diffusion problems relevant to a driving force.
Equations (4-48) and (4-50) reveal that we can solve a nonlinear diffusion equation even if a driving force exists in the diffusion system. Further, the effect of a driving force on the diffusivity is obtained as Equation (4-49). The diffusion flux discussed here is thus unnecessary for solving a nonlinear diffusion equation.
Further, the divergence theorem shows that the diffusion equation is obtained from the diffusion flux of Equation (A-9) as

As discussed in Section 2.1, even if a driving force exists in the diffusion system, the diffusion equation (2-6) of

must be valid for the diffusion particles, except such an external force as Coulomb force for an ion particle.
Equation (A-12) is obviously inconsistent with Equation (A-13). This is evidence that Equation (A-6) neglecting an influence of entropy caused by a driving force is incorrect. In relation to the Equation (5-24), the diffusivity gradient is directly relevant to a driving force. Although the last term in the right-hand side of Equation (A-12) is the derivation term of a driving force because of Equation (5-24), we cannot understand such an effect of

For example, one can easily notice that a force moment of vector quantity is not relevant to a kinetic energy of scalar quantity, although they have the same dimension of


In general, the diffusion problems should be discussed using not the diffusion flux but the original diffusion equation, since the mathematical theory shows that the diffusion flux should be originally understood after the diffusion equation has been solved.
Appendix B
In the text, the K-effect was reasonably understood as a shift between the coordinate systems. This indicates that the intrinsic diffusion concept is not only needless but also nonexistent in the diffusion phenomena.
Based on the intrinsic diffusion concept, Darken derived a relation between the interdiffusion coefficient and intrinsic diffusion coefficient [35] . However, he repeated misunderstandings inconsistent with the fundamental mathematics theory in the analyzing process. Nevertheless, the so-called Darken Equation (D equation) has been widely applied to interdiffusion problems for such a long time since 1948. In the following, we reveal that the D equation is incorrect in mathematics.
When a driving force exists in the diffusion system, the diffusion phenomena are expressed as

In that case, using the Equation (2-17) transformed from Equation (2-6) for

is valid, where the coordinate notation




Equation (B-1) yields

because of



where


Nevertheless, Darken solved Equation (B-2) as an ordinary differential equation with respect to


is valid. Using an unsuitable initial value


Here, the functional equation (B-5) is not valid, since the left-hand side depends only on



Under the condition of


In any case, if we substitute Equation (B-5) into Equation (B-6), the relation of

is obtained. By comparing Equation (B-7) with the F1 law of

He also failed to notice the mistake relevant to the basic theory of physics, since we cannot directly compare


It seems that






In addition, even if we accept Equation (A-6) in Appendix A, it is not suitable to use it for the movement of K interface in the present case, since the drift velocity

Hereinbefore, it was revealed that the D equation is obviously inconsistent with the theory in mathematical physics. Equation (B-8) is thus entirely meaningless and at the same time the concept of intrinsic diffusion is an illusion in the diffusion history. That the D equation has been highly applied to various interdiffusion problems by a lot of researchers for a long time since 1948 is one of the extremely few cases of misunderstanding problems in the science history.

