Food and Nutrition Sciences
Vol. 3 No. 2 (2012) , Article ID: 17524 , 7 pages DOI:10.4236/fns.2012.32034
Mixes of Carrot Juice and Some Fermented Dairy Products: Potentiality as Novel Functional Beverages
![]()
Food Science and Technology Department, Faculty of Agriculture, Alexandria University, Alexandria, Egypt.
Email: *m_m_youssef@yahoo.com
Received August 16th, 2011; revised October 2nd, 2011; accepted October 10th, 2011
Keywords: Yoghurt; Rayeb; Probiotics; Anthocyanins; β-Carotene; Antioxidants
ABSTRACT
The present study aimed to formulate and evaluate yoghurt and Rayeb (Traditional Egyptian natural fermented milk) mixes with red and yellow carrot juices. Out of 16 different mixing ratios (Eight for yoghurt and eight for Rayeb), data indicated that the most significantly acceptable mixes as judged by panelists were: yoghurt-red carrot juice (3:1 v/v), yoghurt-yellow carrot juice (2:1 v/v), Rayeb-red carrot juice (2:1 v/v) and Rayeb-yellow carrot juice (4:1 v/v). The aforementioned mixes were further investigated versus their counterpart controls (yoghurt and Rayeb). It was obvious that the formulated mixes contained considerably higher concentrations of the bioactive components mainly, ascorbic acid and anthocyanins (red-carrot mixes) and β-carotene (yellow-carrot mixes) along with antioxidant activity (DMPD Radical Scavenging Activity) and higher counts of probiotic LAB, as compared to the controls.
1. Introduction
Although there is no consensus on the exact definition of the “Functional Food” term, according to the American Dietetic Association, functional food is defined as: Any modified food or food ingredient that may provide a health benefit beyond the traditional nutrients that contains [1]. Functional food development has enjoyed heightened interest by commercial, academic and governmental sectors over the past decade [2].
The interest in the role of phytochemicals especially as dietary antioxidants in human health has prompted research in the field of food science. Fruits and vegetables are good sources of antioxidants. Consequently, there are a number of commercial polyphenol rich beverages, which base their marketing strategies on antioxidant potency [3].
Carrot (Daucus carota L.) is an inexpensive and highly nutritious vegetable, since it contains appreciable amounts of vitamins B1, B2 and B6 along with carotenes. Dietary carotenes are associated with lowering risk of many cancers. Meanwhile, vitamin A is an antioxidant which plays a key role in growth and repair of tissues in addition to help the body to fight with infections, keep eyes healthy, nourish epithelial tissues in the lungs and skin as well [4].
Extensively reviewed data about Leuconostoc, relative to their habitat, taxonomy, metabolism and genetics, their implications in health and safety, and their present and potential use in dairy technology and functional foods was done[5]. It is worth to mention that the development of probiotics in the last two decades has signaled an important advance in food industry. The number of scientific publications on probiotics has increased a lot stimulated by factors as exciting scientific and clinical findings using well-documented probiotic organisms [6].
The aim of the present work was to formulate mixes of carrot juice (yellow and red varieties) with two fermented dairy products namely, yoghurt and Rayeb, in order to design new functional beverages. Meanwhile, such beverages were evaluated from sensorial, physicochemical and microbiological points of view.
2. Materials and Methods
2.1. Materials
Fresh yellow and red carrots (Daucus carota) were purchased from local farm and immediately transferred to cooling room (0˚C and 90% RH) until used.
Yoghurt and Rayeb milk (Traditional Egyptian natural fermented milk) were obtained from pilot plant belonging to Department of Dairy Science and Technology, Faculty of Agriculture, Alexandria University, Egypt.
2.2. Preparation of Carrot Juice
Both yellow and red carrots were “topped” and “tailed” using a sharp knife, thoroughly washed with tap water, trimmed and peeled using a hand peeler (Approximately 1.00 mm of the periderm was removed by peeling and 2 cm of both tip and top were also removed by trimming), and were cut into sticks by sharp knife (1 × 3 × 1 cm). The sticks were then blanched for 15 min in boiling water with a ratio of 1:1 w/v carrot: water. The sticks along with blanching water were blended in a household blendor at a maximum velocity for 6 min to get fine paste. Juices were kept in polyethylene bags and frozen at –18˚C until used.
2.3. Preparation of Yoghurt-Carrot Juice Mixes
One hundered ml of pasteurized milk were tempered to 40˚C then inoculated with 2 g of commercial yoghurt starter culture (Streptococcus thermophilus and Lactobacillus delbrueckii ssp. Bulgaricus) obtained from Dairy Pilot Plant, Faculty of Agriculture, Alexandria University. Incubation was conducted at 45˚C for 8 hr, then cooled to 4˚C. Mixes of milk, starter culture and yellow or red carrot juice were prepared by the same method using ratios at 1:0, 1:1, 2:1, 3:1 and 4:1 of milk to carrot juice, respectively.
2.4. Preparation of Rayeb-Carrot Juice Mixes
One hundred ml of non-pasteurized milk were tempered to 40˚C and fermented with natural flora for 24 hr. Mixes of milk and yellow or red carrot juice were prepared by the same method using ratios at 1:0, 1:1, 2:1, 3:1 and 4:1 of milk to carrot juice, respectively. Fermentation was stopped by lowering the temperature to 4˚C and samples were held overnight at this degree.
2.5. Sensory Evaluation
Samples were subjected to sensory evaluation along with their controls. Ten trained panelists were asked to evaluate the samples according to the method described [7] on hedonic scale consisting of 9 points from 1 (Extremely dislike) to 9 (Extremely like). Colour, taste, consistency and overall acceptability were evaluated.
2.6. Microbiological Analysis
Food samples were aseptically removed from the bottles and 10 g from each sample were homogenized in 90 ml of sterile distilled water. Serial dilutions were prepared and 1 ml aliquots were plated in each specific medium and incubated at different temperatures, times and growth media as shown in Table 1.
2.7. Gross Chemical Composition
Moisture content was determined by drying the sample in vacuum oven at 70˚C to a constant weight [10], method No. 945.43. Ash was determined by incineration the sample at 550˚C in an electrical Muffle furnace [10], method No. 923.03.
Crude protein (N × 6.25) was determined according to AOAC method [10], method No. 2001.11. Fat content was determined according to Folch et al. method [12], using a mixture of chloroform and methanol (2:1 v/v). Carbohydrate content, was calculated by difference.
2.8. Determination of β-Caroten
Carotenoids were extracted from samples with 80% acetone (1:50 w/v). Absorbance of the extract was measured at 480 nm using Speckol Spectrocoluorimeter (Spekol 11, Carl Zeiss Jena, Germany). The β-carotene concentration was figured out by using the extinction coefficient ( ) of 2273 [13,14].
) of 2273 [13,14].
2.9. Determination of Anthocyanins
Anthocyanins were extracted with 1% conc. HCl in 95% methanol (v/v). The ratio between sample and solvent was 1:50 (w/v). The micromolar concentration of anthocyanins in the extracts was obtained by multiplying the absorbance at 530 nm by 33.3 based on the molar extinction coefficient (1.0 cm light path) of cyanidine chloride being 30,000 [15]. Anthocyanin content was calculated as mg cyanidine chloride/g sample.
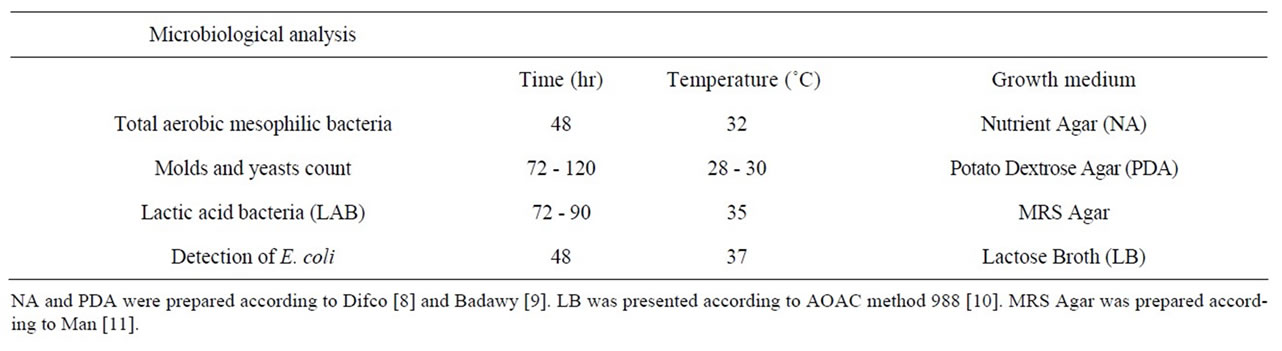
Table 1. Media and incubation conditions used for microbiological analysis.
2.10. DMPD Radical Scavenging Activity
Antioxidant activity was measured using N,N-Dimethylp-phenylenediamine dihydrocholoride (DMPD). A dose response curve was derived for ascorbic acid by plotting the absorbance at 505 nm as percentage of the uninhibited radical cation solution.
Antioxidant activity was expressed as ascorbic acid equivalent antioxidant capacity using the calibration curve plotted with different amounts of ascorbic acid [16].
2.11. Statistical Analysis
Data were expressed as mean of triplicates ± SD. Data of sensory evaluation were subjected to analysis of variance (ANOVA) and Duncan’s multiple range test to separate the treatment means [17]. The analysis was computed using SAS program.
3. Results and Discussion
3.1. Sensory Properties of Yoghurt-Carrot Juice Mixes
Data presented in Table 2 reveal that the colours of yoghurt-red carrot juice mixes at different ratios were less scored than the control, with ratio of 3:1 being the only exception. In accordance, the taste of the control was higher scored than all mixes. In contrast, scores given for consistency of mixes were significantly comparable to the control. As for the overall acceptability, significant differences could be traced between the control and mixes. Since the mix at ratio 3:1 possessed the highest score for the overall acceptability, it was chosen to be investigated in details.
Table 2 shows that yoghurt-yellow carrot juice (YYC) mix at ratio of 2:1 exhibited the highest score given by the panelists for the overall acceptability as compared to the control and the other mixes. Accordingly, the aforementioned mix was chosen for the further investigation.
3.2. Chemical Composition of Yoghurt-Carrot Juice Mixes
Data given in Table 3 indicate that the moisture content ranged between 86.04% and 89.30% for the control and mixes. The yoghurt red carrot juice (YRC) mix at 3:1 v/v had the highest crude protein content (25.70%) and the lowest fat content (29.06%) as compared to the control and yoghurt-yellow carrot juice (YYC) mix.
The control exhibited the highest ash content (5.31%), while YYC had the least ash content being 4.16%. Carbohydrate contents (Table 3) can be ascendingly ordered as followes: YYC (40.84%), YRC (40.56%) and the yoghurt control (31.05%).
The YRC exhibited the highest iron content being 11.83 mg/100g followed by the control (10.34 mg/100g), while YYC was tailed behind (9.01 mg/100g). The same trend was also observed for phosphorus content (Table 3).
Increment of iron and phosphrous in YRC mix can be explained on the basis that the resultant lactic acid due to fermentation improves the mineral availability by degradation of some components in the raw material, decrease in pH and growth of lactic acid bacteria is coupled to production of different organic acid [18]. It has been shown that metal chelates originating from vegetables, such as phytate, tannins and oxalate, may be degraded during fermentation as a result of microbial and/or plant enzymatic activities. In contrast, decline of iron and

Table 2. Sensory evaluation of the control yoghurt and its mixes with red and yellow carrot juices.
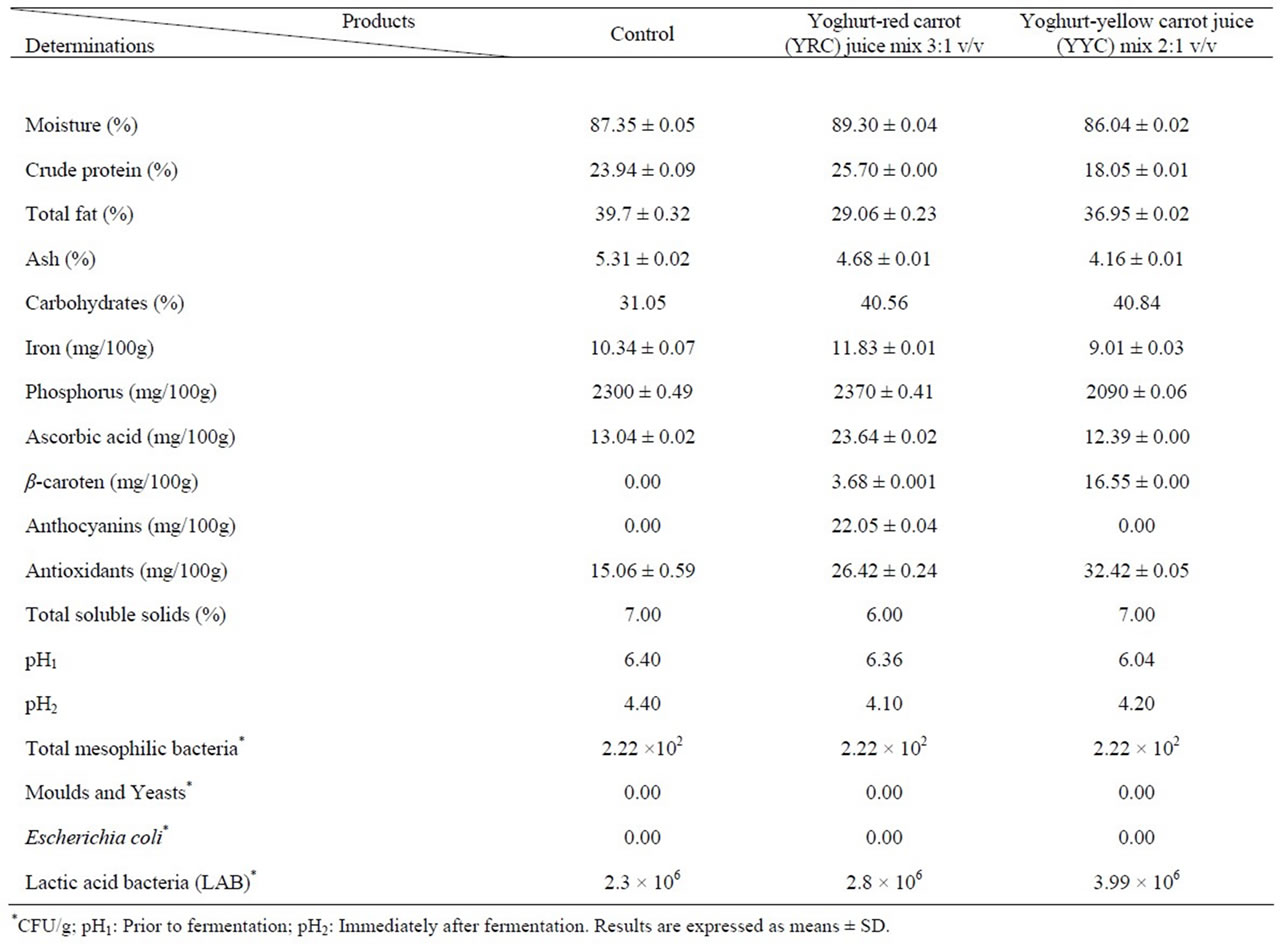
Table 3. Chemical composition (on dry weight basis) and microbial counts of yoghurt and its mixes with red and yellow carrot juices.
phosphrous in YYC mix may be due to the translocation of minerals from the solution into lactic acid bacteria cells for various functions in the bacteria [18].
It is worth to mention that YRC mix had almost double vitamin C content as compared to each of the control and YYC mix (Table 3). Notwithstanding, the YYC mix exhibited almost 5 folds of β-carotene as compared to YRC mix, while β-carotene could not be traced in the control (Table 3).
No anthocyanins could be detected in any of the control or YYC mix, on contrary to YRC mix which possessed considerable amount of anthocyanins being 22.05 mg/100g. Table 3 indicates that YYC mix had more than double of antioxidants as the control had (32.42 versus 15.06 mg/100g). Meanwhile, YRC mix contained higher content (26.42 mg/100g), than its counterpart of the control (15.06 mg/100g).
Total soluble solids were 7.0, 6.0 and 7.0 for the control, YRC and YYC, respectively (Table 3). The pH was measured twice (prior and immediately after fermentation). It was obvious that the pH values declined considerably form 6.40 to 4.40 (control), from 6.36 to 4.10 (YRC mix) and from 6.04 to 4.20 (YY mix) as shown in Table 3. Such declines in pH values are suspected due to formation of lactic acid during fermentation.
3.3. Microbial Counts of Yoghurt-Carrot Juice Mixes
Counts of total mesophilic bacteria were quite identical (2.22 × 102 CFU/g) for the yoghurt control, YRC mix and YYC mix. On the other hand, neither moulds and yeasts nor Escherichia coli could be detected neither in the control nor in the two mixes under investigation (Table 3).
The point of interest is that the count of lactic acid bacteria (LAB) increased considerably from 2.3 × 106 CFU/g (control) to 2.8 × 106 CFU/g (YRC mix) and 3.99 × 106 CFU (YYC), as it is shown in Table 3. Such increment of LAB counts can be attributed to the presence of carrot juice as a source of nutrients for bacteria. In accordance in other studies synbiotic potential of carrot juice supplemented with Lactobacillus spp. was discussed [19]. Their data revealed that both bacterial strains namely lactobacillus rhamnosus and Lactobacillus bulgaricus were capable of growing in carrot juice, reaching nearly 5 × 109 CFU/g after a 48 h fermentation and the pH was reduced to 3.5 - 3.7 or below [19]. Meanwhile, some biochemical characteristics of the fermented juice, such as β-carotene content and antioxidant activity, were also preserved, indicating that the metabolism of the Lactobacillus spp. did not degrade these components after 4 weeks of storage at 4˚C.
3.4. Sensory Properties of Rayeb-Carrot Juice Mixes
Data presented in Table 4 indicate that colours of Rayeb red-carrot juice (RRC) mixes were significantly comparable to the control at 1:1 and 3:1 ratios (v/v). No significant differences in taste scores could be figured out regarding RRC mixes. In contrast, these mixes were significantly different from the control in terms of their consistencies. As for overall acceptability, all mixes with an exception of 1:1 ratio mix were significantly comparable to the control. The 2:1 ratio mix was chosen for the further investigation, since it gained the highest score given for overall acceptability among the four RRC mixes.
Rayeb-yellow carrot juice (RYC) mix at 4:1 ratio was significantly similar to the control in terms of its colour and taste. Meanwhile, RYC mix at 3:1 ratio was significantly similar to the control in terms of consistency. The RYC mix at 4:1 ratio was chosen for further investigation because it had the highest score given by panelists for the overall acceptability as compared to the other RYC mixes.
3.5. Chemical Composition of Rayeb-Carrot Juice Mixes
Table 5 shows the chemical composition (on dry weight basis) of Rayeb (Natural fermented milk) and its mixes with red (RRC) and yellow (RYC) carrot juices. The RRC mix (2:1 v/v) possessed the highest moisture content being 89.46%, and the RYC mix (4:1 v/v) had the lowest moisture content (85.35%), while the control exhibited moisture content of 87.81%. Notwithstanding, crude protein content ranged between 29.75% (RYC) and 35.10% (Control). Accordingly, mixing of Rayeb milk with carrot juice resulted in decline of crude protein content. The RYC mix exhibited the highest total fat content (34.28%) as compared to 30.70% (RRC mix) and 33.80% (Control). Ash content varied from 4.30% (RYC mix) to 5.89% (RRC mix). As for carbohydrate contents, a range from 25.69% (Control) to 32.18% (RRC mix) was figured out.
Iron content varied from 13.22 mg/100g (RYC mix) to 17.14 mg/100g (RRC). Increment and decrease of iron content due to fermentation can be explained on the basis discussed previously for yoghurt-carrot juice mix. On the other hand, phosphorus content decreased in the mix as compared to its counterpart in the control (Table 5).
The RRC mix explored the highest ascorbic acid content (32.84 mg/100g) as compared to the value of 17.47 mg/100g (RYC) and 20.99 mg/100 g (Control). Meanwhile, it was obvious that RYC mix had almost twice content of β-carotene as compared to RRC mix versus nil for control (Table 5).
The RRC mix was the only product which contains appreciable amount (37.16 mg/100 g) of anthocyanins
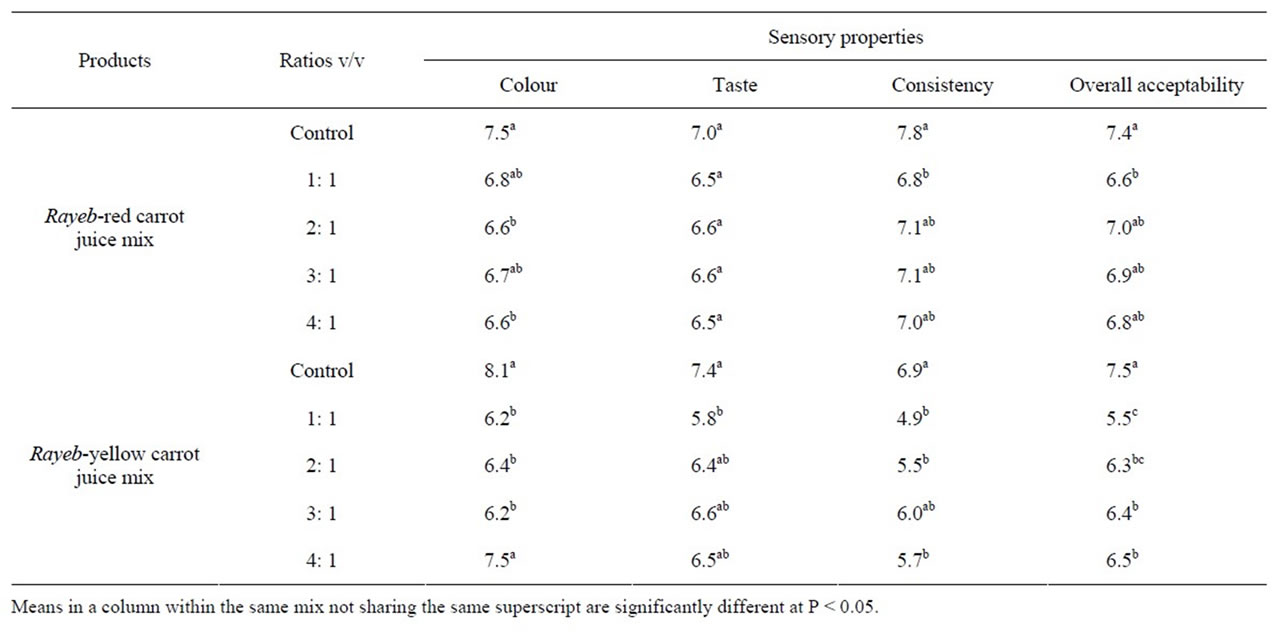
Table 4. Sensory evaluation of the control Rayeb and its mixes with red and yellow carrot juices.
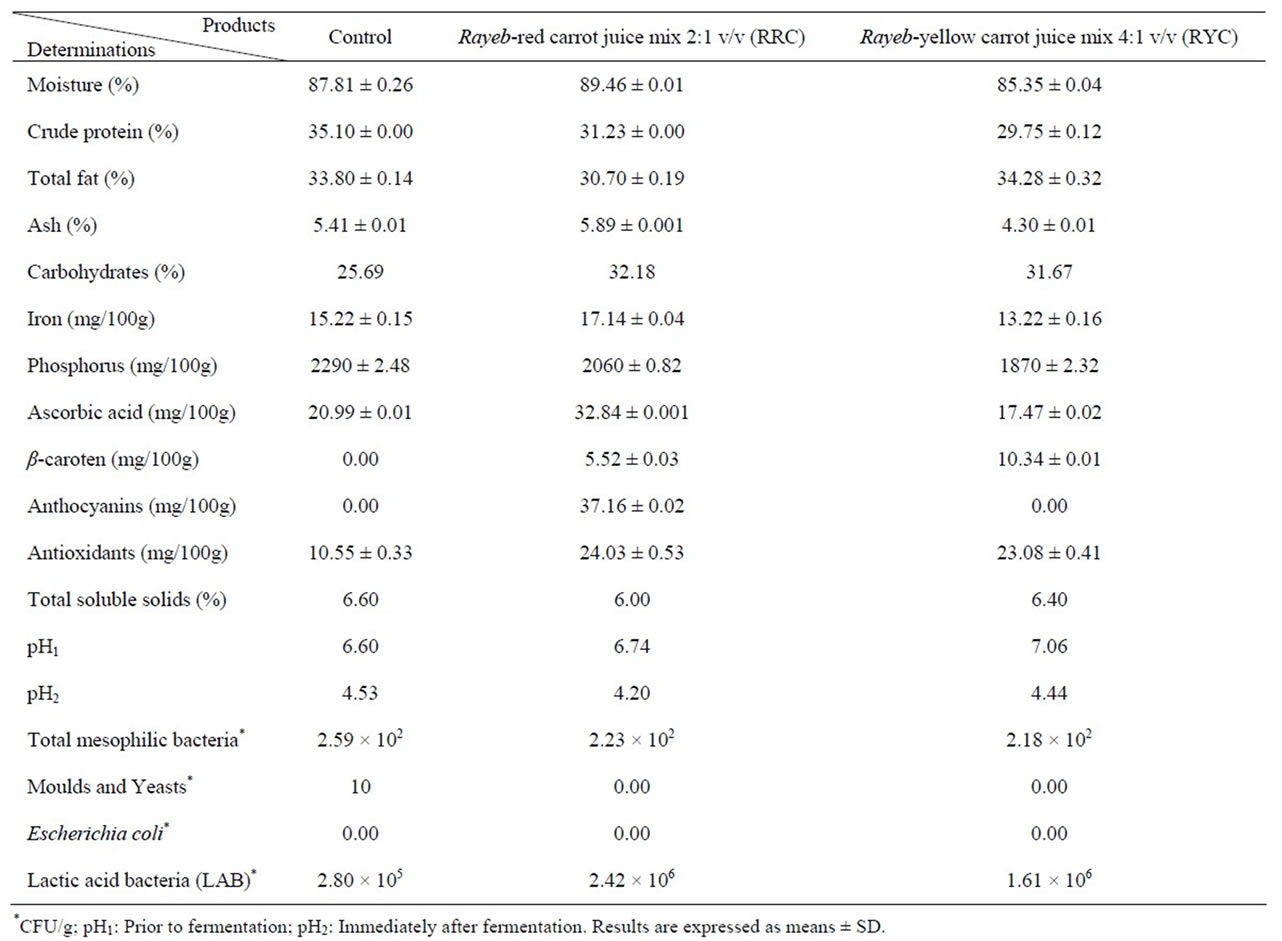
Table 5. Chemical composition (on dry weight basis) and microbial counts of Rayeb and its mixes with red and yellow carrot juices.
versus nil for each of RYC mix and the control (Table 5). Antioxidants (mg/100g) increased considerably on mixing Rayeb with carrot juices. So, the RRC mix had 24.03 while RYC mix exhibited a value of 23.08 versus 10.55 for the control (Table 5).
Total soluble solids were 6.6%, 6.0% and 6.4% for the control, RRC mix and RYC mix, respectively (Table 5). Notwithstanding, the pH was measured prior to fermentation and immediately after fermentation. Dramatic declines in pH were figured out. Such declines were as follows: from 6.60 to 4.53 (Control), from 6.74 to 4.20 (RRC mix) and from 7.00 to 4.44 (RYC mix). Such declines in pH can be attributed to producing organic acids (mainly lactic acid) as a result of fermentation by lactic acid bacteria present in Rayeb.
3.6. Microbial Counts of Rayeb-Carrot Juice Mixes
Counts of total mesophilic bacteria were found to decline as a result of mixing Rayeb with carrot juices. In contrast, RRC and RYC mixes exhibited higher counts (2.42 × 106 CFU/g) and (1.61 × 106 CFU/g) of lactic acid bacteria than the Rayeb control (2.80 × 105 CFU/g). Such an elevation of lactic acid bacteria can be explained on the basis that presence of carrot juices in the medium of bacteria represents pivotal source of nutrients and thereby increase the growth rate of bacteria.
4. Conclusion
High quality and functional beverage mixes were able to be produced by mixing each of yoghurt and Rayeb (Traditional Egyptian natural fermented milk) with red carrot juice and yellow carrot juice. Such mixes can act as one of the most effective means for overcoming vitamin A deficiency. Moreover, these mixes contain high concentrations of natural antioxidants (vitamin C, β-carotene and anthocyanins) along with lactic acid bacteria (LAB) which act as a very good probiotics. All these components possess health benefits, as it has been extensively reported in numerous literatures. Epidmiological studies have demonstrated or at least suggested numerous health effects related to probiotics, prebiotics and natural antioxidants [20].
REFERENCES
- A. Bloch and C. A. Thomson, “Position of the American Dietetic Association: Phytochemicals and Functional Foods,” Journal of American Dietetic Association, Vol. 95, No. 4, 1995, pp. 493-496. doi:10.1016/S0002-8223(95)00130-1
- P. J. Jones and S. Jew, “Functional Food Development: Concept to Reality,” Trends in Food Science & Technology, Vol. 18, No. 7, 2007, pp. 387-390. doi:10.1016/j.tifs.2007.03.008
- E. Genzālez-Molina, D. A. Moreno and Garcia-Viguera. A New Drink Rich in Healthy Bioactive Combining Lemon and Pomegranate Juices,” Food Chemistry, Vol. 115, No. 4, 2009, pp. 1364-1372. doi:10.1016/j.foodchem.2009.01.056
- B. Singh, P. S. Panesar and V. Nanda, “Utilization of Carrot Pomace for the Preparation of a Value Added Product,” World Journal of Dairy & Food Sciences, Vol. 1, No. 1, 2006, pp. 22-27.
- D. Hemme and C. Foucaud-Scheunemonn, “Leuconostoc, Characteristics, Use in Dairy Technology and Prospects in Functional Foods,” International Dairy Journal, Vol. 14, No. 6, 2004, pp. 467-494. doi:10.1016/j.idairyj.2003.10.005
- F. C. Prado, J. L. Parada, A. Pandey and C. R. Soccoi. Review: Trends in Non-Dairy Probiotic Beverages,” Food Research International, Vol. 41, No. 2, 2008, pp. 111-123. doi:10.1016/j.foodres.2007.10.010
- S. Hooda and S. Jood, “Organoleptic and Nutritional Evaluation of Wheat Biscuits Supplemented with Untreated and Treated Fenugreek Flour,” Food Chemistry, Vol. 90, No. 3, 2005, pp. 427-435. doi:10.1016/j.foodchem.2004.05.006
- DIFCO, Difco Manual of Dehydrated Culture Media and Reagents for Microbiological and Clinical Laboratory Procedures,” 8th Edition, Difco Laboratories, Detroit, 1948.
- D. M. Badawy, “Chemical, Technological and Microbiological Studies on Karkade (Hibiscus abdariffa) Petals Extract,” M.Sc Thesis, Cairo University, Cairo, 2007.
- AOAC, Official Methods of Analysis of AOAC International, 17th Edition, Association of Official Analytical Chemists, Maryloand, 2000.
- J. C. Man, M. Rogosa and M. E. Sharpe, “A Medium for the Cultivation of Lactobacilli,” Journal of Applied Bacterial, Vol. 23, No. 1, 1960, pp. 130-135. doi:10.1111/j.1365-2672.1960.tb00188.x
- J. Folch, M. Less and S. Stanley, “A Simple Method for the Isolation and Purification of Total Lipids from Animal Tissues,” Journal Bio-Chemistry, Vol. 226, No. 1, 1957, pp. 497-509.
- A. Jensen, “Chlorophylls and Carotenoids,” In: J. A. Hellebuts and J. S. Carigie, Eds., Hand Book of Physiological and Biochemical Methods, Cambridge University Press, London, 1978, pp. 59-70.
- A. Ben-Amotz and M. Avronm, “The Factors Which Determines Massive -Carotene Accumulation in the Halotolerant Alga Dunaliella bardawil,” Plant Physiology, Vol. 72, No. 3, 1983, pp. 593-597. doi:10.1104/pp.72.3.593
- F. T. Halaweish and D. K. Dougall, “Sinapolyl Glucose Synthesis by Extracts of Wild Carrot Cell Cultures,” Plant Science, Vol. 71, No. 2, 1990, pp. 179-184. doi:10.1016/0168-9452(90)90007-B
- V. Fogliano, V. Verde, G. Randazzo and A. Ritieni, “Method for Measuring Antioxidant Activity and Its Application to Monitoring the Antioxidant Capacity of Wines,” Journal of Agricultural and Food Chemistry, Vol. 47, No. 3, 1999, pp. 1035-1040. doi:10.1021/jf980496s
- R. B. D. Steel and T. H. Torrie, “Principles and Procedures of Statistics,” McGraw Hill Co., Boston, 1980.
- S.W. Bergqvist, A.S. Sandberg, N. G. Carlsson and T. Andlid, “Improved Iron Solubility in Carrot Juice Fermented by Homoand Hetero-Fermentative Lactic Acid Bacteria,” Food Microbiology, Vol. 22, No. 1, 2005, pp. 53-61. doi:10.1016/j.fm.2004.04.006
- F. Nazzaro, F. Fratianni, A. Sada and P. Orlando, “Synbiotic Potential of Carrot Juice Supplemented with Lactobacillus spp. and Inulin or Fructooligosaccharides,” Journal of the Science of Food and Agriculture, Vol. 88, No. 13, 2008, pp. 2271-2276. doi:10.1002/jsfa.3343
- W. Grajek, A. Olejnik and A. Sip, “Probiotics, Prebiotics and Antioxidants as Functional Foods: A Review,” Acta Biochimica Polonica, Vol. 52, No. 3, 2005, pp. 665-671.
NOTES
*Corresponding author.

