International Journal of Clinical Medicine
Vol.3 No.3(2012), Article ID:19411,5 pages DOI:10.4236/ijcm.2012.33046
Hemophilic Pseudotumor: A Case Report and Review of Literature
![]()
Department of Internal Medicine, University of Oklahoma Health Science Center, Oklahoma City, USA.
Email: Aneesh-Pakala@ouhsc.edu
Received February 16th, 2012; revised March 19th, 2012; accepted April 6th, 2012
Keywords: Hemophilia; Pseudotumor
ABSTRACT
A hemophilic pseudotumor is one of the rarer complications of hemophilia that results from repetitive bleeding resulting in an encapsulated mass of clotted blood and necrosed tissue. These have become rarer over the years with better treatment modalities for bleeding disorders like factor replacement. In this case report, we describe the natural history and clinical course of a patient with hemophilia who presented with a massive pseudotumor. A review of literature follows which explores all the relevant data to date on the presentation, complications, diagnostic modalities and therapies that have been implemented in the management of this rare complication of a common bleeding disorder.
1. Introduction
Hemophilia A and B are X-linked clotting disorders caused by the deficiency of factors VIII and IX respectively. Clinical presentation varies with the severity of the disease. Hemophilia manifests with hemorrhage; spontaneous bleeding into joints and muscles is a feature of severe hemophilia [1,2]. A hemophilic pseudotumor is the result of repeated episodes of bleeding at a bone fracture site or as a result of sub-periosteal hemorrhage or bleeding into a soft tissue. Inadequate resorption of extravasated blood results in an encapsulated area of clotted blood and necrosed tissue. With successive hemorrhagic episodes, these lesions expand over time, eventually causing symptoms by mass effect.
The majority of all reported hemophilic pseudotumors involve the musculoskeletal system. A few intra-abdominal pseudotumors have been reported but these were in fact tumors of the pelvis that had extended into the abdomen [3,4]. Very few retroperitoneal pseudotumors have been reported in literature. In this case report, we describe a patient with a massive retroperitoneal pseudotumor and review the literature on hemophilic pseudotumors.
2. Case Report
Our patient is a 54 y/o caucasian gentleman with Factor IX deficiency (Christmas disease). He presented to our institution with malnutrition and early satiety due to a large intra-abdominal mass. The patient was born with a glial hematoma and rectal bleeding and was subsequently diagnosed with Factor IX deficiency at the age of 2. Throughout his childhood he had several instances of hemarthrosis and eventually developed ankle fibrosis. At some time during his clinical course, patient was started on Factor IX replacements.
In his early adulthood, he noticed paraesthesias in the right upper thigh that were brought on by prolonged weight bearing and relieved by rest. He did not seek medical attention for a substantial period of time. The pain however worsened over time and began to radiate down to his knee. Patient sought emergent care for the pain but all initial evaluations were inconclusive. After multiple visits to the ER for similar complaints, a CT scan of the pelvis was obtained which revealed a retroperitoneal pseudotumor, arising from the right pelvis and incidentally compressing the bladder. The patient continued to receive factor replacements.
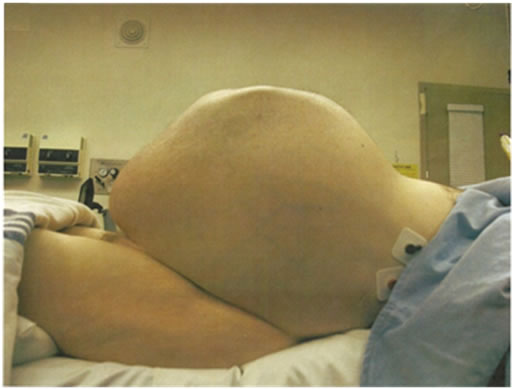
His symptomatology changed over the next decade. The pain shifted to the abdomen and also started to abate for prolonged periods of time. His abdominal girth visibly increased, and went from a 32” waist in his late twenties to a 40” waist in his forties. During this period, he underwent follow up CT scans and MRIs. The pseudotumor showed a progressive increase in size and worsening compression of the surrounding structures. The risk of surgery was considered too high due to the retroperitoneal location of the mass and the pseudotumor was left unresected.
In his fifth decade, the patient’s waist circumference was 50” and he started noticing early satiety, worsening nausea and vomiting. Imaging studies now documented compression of the stomach by the pseudotumor. His nutrition status deteriorated to the point that he was hospitalized and underwent CT guided aspiration of strategically located daughter cysts [branches of the main pseudotumor] with subsequent decompression of the stomach and temporary relief of symptoms. At this point, patient was offered surgical resection of the pseudotumor in the event that his symptoms recurred and were deemed to be life threatening. An MRI obtained at this stage is shown in Figure 1. He was readmitted to the surgery service at our institute with intractable nausea, vomiting and failure to thrive and underwent successful surgical debulking of the pseudotumor. At the time of surgery, his tumor measured 34 × 21 × 22 cm (Figure 2). The resected mass was massive and had a volume of approximately 20 liters. The patient developed post operative enterocutaneous fistulae and died within 4 months of surgery due to infectious complications.
 (a)
(a)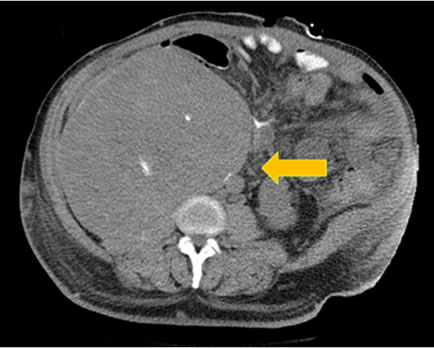 (b)
(b)
Figure 1. MRI images showing vertical and transverse sections of the abdomen.
 (a)
(a)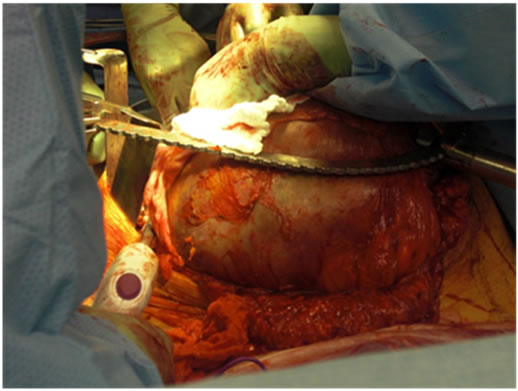 (b)
(b)
Figure 2. The tumor mass before surgery and at the time of resection.
3. Discussion
The hemophilic pseudotumor was first described by Starker [5] who described the entity in the femur of a 14 year old boy. Pseudotumors or “blood cysts” are a rare complication of this hemorrhagic syndrome, having been observed in only 1% - 2% of patients with severe disease. [6] Affected patients are usually diagnosed hemophiliacs who have experienced multiple hemorrhagic episodes from various sites over several years [3,4,7].
As hemorrhagic episodes are now well controlled with factor substitution therapy, the incidence of pseudotumors may have decreased [9,10]. However, factor substitution therapy is ineffective in patients with autoantibodies against factor VIII or factor IX. Among these patients, bleeding also tends to be more severe and varied in anatomical location [11].
Pseudotumors consist of extravasated clotted blood surrounded by a fibrous capsule. They commonly affect bones [5,12-14] but have also been reported to occur in the lung [15], abdomen [3] and intramurally in the wall of the stomach [7,16,17]. The bones most commonly affected are the ones most susceptible to trauma i.e. femur, pelvis, tibia and small bones of the hand in decreasing order of frequency [13,14].
Clinical Presentation
Patients present with painless palpable masses or with painful crises due to episodic acute bleeding into the tumor [4]. Most of the morbidity from pseudotumors is due to their compressive effect on surrounding structures including bone destruction [4,5,13,18], muscle necrosis and skin necrosis [18,19], Depending on the area involved, the symptoms include; palpable masses, numbness, weakness, and neuralgia [4,6].
4. Complications
Complications occur due to the extension of their compressive effect and include perforation through the skin or into adjacent organs, abscess and fistula formation [19, 20], pathologic fractures due to bone destruction and resorption [6] and compartment syndromes due to vascular compromise and joint contractures [21]. In rare cases, rupture of a pseudotumor can cause life threatening exsanguinations. [6,21,22]. Erosion into an artery may also be responsible for massive bleeding. Also, a pseudotumor distorts natural anatomy causing major displacements of bowels, ureters, nerves and blood vessels thus increasing the likelihood of injury during surgery. [4].
5. Pathology
On histological examination, hemophilic pseudotumors resemble hematomas with a dense fibrous capsule. The cyst wall is formed by collagenous connective tissue and the cavity contains a variable amount of organized fibrous tissue, thick “toothpaste-like” debris and liquefied clots. Other features that have been observed within the cyst cavity include bone fragments, foci of hemosiderin loaded histiocytes, vascular neoformation, osteoid neoformation, giant multinucleated cells and foreign body type cells [23].
6. Imaging
6.1. X-Rays
On radiologic imaging, hemosiderin deposits within the cyst result in a dense appearance, mimicking other benign or malignant tumors or infectious processes. The differential diagnosis includes Ewing’s sarcoma, osteomyelitis, primary osteosarcomas, metastatic neoplasms, tuberculosis abscesses, giant cell tumors, plasmacytomas and chondrosarcomas [24]. However, with Hemophilia A or B and a history of recurrent bleeding episodes, hemophilic pseudotumor ought to be one of the top differentials.
6.2. Computed Tomography (CT)
CT scans define the attenuation value of the tumor and thus help to differentiate it from other masses. It clearly delineates the size and extent of the tumor and compression of the adjacent skeleton, tissues and organs. Of note, early in the course of a lesion, CT may prove useful in determining the origin of the pseudotumor and help follow its course. In cases that are conservatively managed, consecutive CTs may help decide whether or not surgery is indicated. CT scans have been proven to be particularly efficient in the detection of daughter cysts, deep seated cysts and tumor extension into adjacent tissues, information that is useful in planning surgery [8,25-27].
6.3. Ultrasonography
Ultrasonography is ineffective for the detection of bony changes. It can be used after surgery to monitor for recurrence of pseudotumors in soft tissues. It has also been suggested as a low cost alternative to monitor the progress of an existing lesion before obtaining a CT scan prior to repeated surgery [27].
6.4. Magnetic Resonance Imaging (MRI)
MRI has been shown to be superior to other modalities for the imaging of soft tissues [28]. MRI is sensitive for the detection of pseudotumors and can provide useful information for decisions regarding therapy and may be used to follow the response of a tumor to treatment [21]. The MRI appearance of pseudotumors shows a heterogenous signal with a dense fibrous or hemosiderin laden capsule that shows low intensity on T1 and T2 weighted images. A unique finding in intramuscular pseudotumors is the presence of mural nodules [21].
The appearance on MRI seems non-specific with a similar appearance in tumors and abscesses, but invariably there are heterogeneous lowand high-signal intensity areas on both pulse sequences, reflecting the presence of blood products in various stages of evolution.
In summary, either CT or MRI is indicated as an imaging modality. Imaging characteristics of pseudotumors are rather non-specific, but given a history of hemophilia, radiologists must be aware of these to avoid misinterpretation as biopsy of a pseudotumor is absolutely contraindicated.
7. Therapy
Given the rarity of hemophilic pseudotumors, there is no consensus among authors about specific management strategies. In view of the differing approaches in various case reports, it would be best to individualize therapy.
Conservation: A conservative approach includes factor replacement and immobilization of the affected area. With long term replacement, the prognosis is favorable [29] and this method has been suggested as the preferred management of pseudotumors caused by recent hemorrhage [30].
Aspiration: Aspiration of the fluid within the cysts for either diagnostic or therapeutic purposes is absolutely contraindicated. First, the cystic contents are too thick to permit successful aspiration. Secondly, there is a substantial risk of relapse, sepsis or the development of a persistent fistula [4,23,31].
Surgery: Surgery has been recommended early in the management of pseudotumors, particularly those of the ilium and femur in order to prevent erosion of bone, ulceration of overlying skin and infection [32]. Indications for surgery include clinical or radiographic evidence of progressive enlargement of the hematoma, hemodynamic deterioration or occurrence of a complication. Surgical extirpation of pseudotumors is complicated by their size, distortion of normal anatomy, involvement of adjacent neurovascular structures and the presence of daughter cysts which must be completely removed to prevent recurrence. The main concern associated with surgery is the risk of uncontrollable hemorrhage. The availability of Factor VIII, IX and activated prothrombin complex concentrates have now made surgery feasible. Recurrence after surgery occurs in the surrounding tissues left behind after removal of the tumor [24]. Surgical management has been associated with the best outcomes especially when selected as the primary line of treatment. Studies also showed that patients in whom surgery was performed after the failure of replacement therapy had poorer outcomes than those in whom it had been the primary modality [23]. Surgery has also been suggested as the preferable treatment for pseudotumors that have been present for years [30].
Radiotherapy: Radiotherapy is a viable alternative where surgery is not feasible and conservative management is not effective. Several mechanisms have been suggested to explain the response of pseudotumors to radiation therapy. These include radiation induced injury to blood vessels [14,37] and fibrosis of the cellular component of the pseudotumor [38]. Radiotherapy has been shown to be a good alternative in the management of recurrent pseudotumors of the long bones [36].
8. Conclusion
Hemophilic pseudotumors are a well documented complication of bleeding disorders that clinicians should be aware of while caring for such patients. Signs and symptoms of compression should be evaluated early on with CT and then MRI. Early surgical consultation along with factor replacements are essential steps after diagnosis is made. Patients with advanced disease and complications might benefit with a conservative approach but further decision making would need an interdisciplinary approach.
REFERENCES
- C. T. A. Brower, “Hemophilia A,” Gene Reviews, 1993.
- C. T. A. Brower, “Hemophilia B,” Gene Reviews, 1993.
- R. Garcia-Perez, G. Torres-Salmeron, F. Sanchez-Bueno, A. Garcia-Lopez and P. Parrilla-Paricio, “Intraabdominal He- mophilic Pseudotumor: Case Report,” Revista Española de Enfermedades Digestivas, Vol. 102, No. 4, 2010, pp. 275-280. doi:10.4321/S1130-01082010000400009
- W. W. Coon and J. A. Penner, “Management of Abdominal Hemophilic Pseudotumor,” Surgery, Vol. 90, No. 4, 1981, pp. 735-40.
- L. Starker, “Knochenusur Durch ein Hamophiles Subperiostales Hamatom,” Mitteilungen aus den Grenzgebieten der Medizin und Chirurgie, Vol. 31, 1918, pp. 381-415.
- T. R. Lewis, H. R. Webb, J. P. Bell, D. P. Beall and J. R. Fish, “Hemophilic Pseudotumor,” The Journal of the Oklahoma State Medical Association, Vol. 98, No. 10, 2005, pp. 485-487.
- F. W. Wright, J. M. Matthews, “Hemophilic Pseudotumor of the Stomach,” Radiology, Vol. 98, No. 3, 1971, pp. 547-549.
- H. Pettersson and A. Ahlberg, “Computed Tomography in Hemophilic Pseudotumor,” Acta Radiologica: Diagnosis (Stockh), Vol. 23, No. 5, 1982, pp. 453-457.
- M. S. Gilbert, “Characterizing the Hemophilic Pseudotumor,” Annals of the New York Academy of Sciences, Vol. 240, 1975, pp. 311-315. doi:10.1111/j.1749-6632.1975.tb53365.x
- H. Pettersson, I. M. Nilsson, U. Hedner, K. Norehn and A. Ahlberg, “Radiologic Evaluation of Prophylaxis in Severe Haemophilia,” Acta Paediatrica, Vol. 70, No. 4, 1981, pp. 565-570. doi:10.1111/j.1651-2227.1981.tb05741.x
- C. A. Lee, E. E. Berntorp and W. K. Hoots, Eds., “Textbook of Hemophilia,” 2nd Edition, Wiley-Blackwell, Hoboken, 2010.
- S. Castro-Boix, J. Pradell-Teigell, R. Boque-Genovard, V. Zanon-Navarro, A. Nadal-Guinard, C. Altisent-Roca, et al., “Hemophilic Pelvic Pseudotumor,” Cirugía Española, Vol. 81, No. 2, 2007, pp. 102-104. doi:10.1016/S0009-739X(07)71273-3
- J. S. Park and K. N. Ryu, “Hemophilic Pseudotumor Involving the Musculoskeletal System: Spectrum of Radiologic Findings,” American Journal of Roentgenology, Vol. 183, No. 1, 2004, pp. 55-61.
- E. E. Brant and H. H. Jordan, “Radiologic Aspects of Hemophilic Pseudotumors in Bone,” American Journal of Roentgenology, Vol. 115, No. 3, 1972, pp. 525-539.
- S. Bicknell and A. Mason, “Hemophilic Pseudotumor of the Chest,” Journal of Thoracic Imaging, Vol. 16, No. 3, 2001, pp. 188-190. doi:10.1097/00005382-200107000-00010
- H. Grossman, W. E. Berdon and D. H. Baker, “Reversible Gastrointestinal Signs of Hemorrhage and Edema in the Pediatric Age Group,” Radiology, Vol. 84, No. 1, 1965, pp. 33-39.
- J. D. Rothstein, W. R. Sandusky and T. E. Keats, “Hematoma as a Cause of Radiographic Deformity of Stomach,” Radiology, Vol. 90, No. 1, 1968, pp. 116-117.
- W. M. Steel, R. B. Duthie and B. T. O’Connor, “Haemophilic Cysts. Report of Five Cases,” Journal of Bone & Joint Surgery, Vol. 51, No. 4, 1969, pp. 614-626.
- I. M. Nilsson, U. Hedner, A. Ahlberg, S. A. Larsson and S. E. Bergentz, “Surgery of Hemophiliacs—20 Years’ Experience,” World Journal of Surgery, Vol. 1, No. 1, 1977, pp. 55-66. doi:10.1007/BF01654733
- Y. A. Kilic, S. V. Dundar, D. Onat and O. Akhan, “Iliopsoas Hemophilic Pseudotumor with Bowel Fistulization,” Bratisl Lek Listy, Vol. 110, No. 11, 2009, pp. 729-731.
- S. Jaovisidha, K. N. Ryu, J. Hodler, M. E. Schweitzer, D. J. Sartoris and D. Resnick, “Hemophilic Pseudotumor: Spectrum of MR Findings,” Skeletal Radiology, Vol. 26, No. 8, 1997, pp. 468-474. doi:10.1007/s002560050268
- M. S. Gilbert, Ed., “Haemophilic Pseudotumor,” Excerpta Medica, Amsterdam, 1975.
- M. Magallon, J. Monteagudo, C. Altisent, A. Ibanez, A. Rodriguez-Perez, J. Riba, et al., “Hemophilic Pseudotumor: Multicenter Experience over a 25-Year Period,” The American Journal of Hematology, Vol. 45, No. 2, 1994, pp. 103-108. doi:10.1002/ajh.2830450202
- R. Hoffman, Ed., “Hematology: Basic Principles and Practice,” 3rd Edition, Churchill Livingstone, New York, 2003.
- M. Sundaram, M. K. Wolverson, J. H. Joist, M. A. Riaz and B. J. Rao, “Case Report 133. Hemophilic Pseudotumor of Ilium and Soft Tissues,” Skeletal Radiology, Vol. 6, No. 1, 1981, pp. 54-57. doi:10.1007/BF00347349
- W. B. Guilford, P. D. Mintz, P. M. Blatt and E.V. Staab, “CT of Hemophilic Pseudotumors of the Pelvis,” American Journal of Roentgenology, Vol. 135, No. 1, 1980, pp. 167-169.
- G. Hermann, H. C. Yeh and M. S. Gilbert, “Computed Tomography and Ultrasonography of the Hemophilic Pseudotumor and Their Use in Surgical Planning,” Skeletal Radiology, Vol. 15, No. 2, 1986, pp. 123-128. doi:10.1007/BF00350205
- J. M. Stafford, T. T. James, A. M. Allen and L. R. Dixon, “Hemophilic Pseudotumor: Radiologic-Pathologic Correlation,” Radiographics, Vol. 23, No. 4, 2003, pp. 852-856. doi:10.1148/rg.234025154
- T. Morissy and S. L. Weinstein, Eds., “Pediatric Orthopedics,” 6th Edition, Lippincott Williams & Wilkins, Philadelphia, 2006.
- E. Gaary, J. B. Gorlin and D. Jaramillo, “Pseudotumor and Arthropathy in the Knees of a Hemophiliac,” Skeletal Radiology, Vol. 25, No. 1, 1996, pp. 85-87. doi:10.1007/s002560050040
- A. K. Ahlberg, “On the Natural History of Hemophilic Pseudotumor,” Journal of Bone and Joint Surgery, Vol. 57, No. 8, 1975, pp. 1133-6.
- R. L. Rosenthal, J. J. Graham and E. Selirio, “Excision of Pseudotumor with Repair by Bone Graft of Pathological Fracture of Femur in Hemophilia,” Journal of Bone and Joint Surgery, Vol. 55, No. 4, 1973, pp. 827-832.
- J. M. Pisco, V. L. Garcia, J. M. Martins and A. M. Mascarenhas, “Hemophilic Pseudotumor Treated with Transcatheter Arterial Embolization: Case Report,” Angiology, Vol. 41, No. 12, 1990, pp. 1070-1074. doi:10.1177/000331979004101208
- F. Alaniz Camino and E. Lozano, “Hemophilic Pseudotumor Treated with Radiation,” Revista de Investigación Clínica, Vol. 33, No. 3, 1981, pp. 281-283.
- M. W. Hilgartner and W. D. Arnold, “Hemophilic Pseudotumor Treated with Replacement Therapy and Radiation. Report of a Case,” Journal of Bone and Joint Surgery, Vol. 57, No. 8, 1975, pp. 1145-1146.
- M. Issaivanan, M. P. Shrikande, M. Mahapatra and V. P. Choudhry, “Management of Hemophilic Pseudotumor of Thumb in a Child,” Journal of Pediatric Hematology/ Oncology, Vol. 26, No. 2, 2004, pp. 128-132. doi:10.1097/00043426-200402000-00016
- H. S. Reinhold, “The Influence of Radiation on Blood Vessels and Circulation. Chapter IV. Structural Changes in Blood Vessels,” Current Topics in Radiation Research Quarterly, Vol. 10, No. 1, 1974, pp. 58-74.
- L. Meyers and N. Hakami, “Pseudotumor of Hemophilia in the Orbit: The Role of Radiotherapy in Management,” The American Journal of Hematology, Vol. 19, No. 1, 1985, pp. 99-104. doi:10.1002/ajh.2830190113

