Journal of Cancer Therapy
Vol.3 No.5(2012), Article ID:23450,7 pages DOI:10.4236/jct.2012.35073
Older Men with Intermediate to High Risk Prostate Cancer-Patterns of Care and Outcomes of Treatment
![]()
1Department of Medicine, UT Health Science Center San Antonio, San Antonio, USA; 2Departments of Radiation Oncology and Urology, UT Health Science Center San Antonio, San Antonio, USA; 3Department of Biostatistics, UT Health Science Center San Antonio, San Antonio, USA; 4Department of Urology, UT Health Science Center San Antonio, San Antonio, USA.
Email: swansong@uthscsa.edu
Received April 18th, 2012; revised May 26th, 2012; accepted June 7th, 2012
Keywords: Prostate Cancer; Elderly; Age Effect
ABSTRACT
There is significant controversy on how aggressively to treat older men with prostate cancer. We identified 1082 patients diagnosed with prostate cancer from 1998-2008 with Gleason score ≥ 7 on biopsy or prostatectomy pathology in the South Texas Veteran’s Healthcare System. Prostate specific antigen (PSA) values, pathology, treatment and response to treatment were analyzed. Mean follow up was 4.99 years. Patients > 74 years had significantly higher pretreatment PSA, higher grade disease, and were received hormone therapy more often. Unadjusted hazard ratios for metastasis and cancer related death were 2.15 (95% CI 1.02, 4.52; p = 0.04) and 2.66 (95% CI 1.18, 6; p = 0.02), respectively. However, after controlling for treatment, Gleason score and pre-treatment PSA, there was no significant difference in cancer specific survival (CSS) by age group. In the patients > 74 years, there was also no significant difference in overall survival (OS) or CSS among patients treated with surgery, radiation or hormones after controlling for Gleason score and pre-treatment PSA. Our oldest patients have worse cancer presumably to later diagnosis, but they do just as well as younger patients with any given treatment modality. Most importantly, they have similar cancer specific survival with hormone therapy as they do with radiation or surgery.
1. Introduction
Prostate cancer is still the most common cancer, excluding cutaneous malignancies, diagnosed in men. In 2010, there were 217,713 new cases of prostate cancer diagnosed in the United States [1]. Although the widespread use of prostate specific antigen (PSA) screening has allowed for both younger age and earlier stage of diagnosis, the vast majority of cases are still diagnosed in men older than 65 years, and approximately 25% of cases are diagnosed in men older than 75 years [2]. The US Preventative Services Task Force (USPSTF) has recently recommended against screening for prostate cancer in men older than 75, based on the observation that the benefits from screening and treatment for improved survival are only likely to be manifest in men that will live more than 10 years [3,4]. Still, given the higher risk of cancer morbidity and mortality in patients with high risk prostate cancer, the National Comprehensive Cancer Network (NCCN) makes no age distinction, but recommends basing initial treatment for Gleason 7 prostate cancer not on age, but on life expectancy [5]. If the patient is expected to live 10 years or more, the recommendation is to treat these high risk tumors aggressively with radical prostatectomy (RP) or radiation therapy (RT) ± short term hormone therapy (HT) ± brachytherapy. If the patient is expected to live less than 10 years, active surveillance (AS) or RT are recommended [5]. For patients with clinically localized Gleason 8 - 10 prostate cancer, treatment with either RT + long term HT, RT + brachytherapy ± short term HT, or RP is indicated regardless of age or life expectancy [5]. Therefore, we have two discordant recommendations; one is not to screen men under the age of 75 and the other is to treat men older than 75 with higher risk cancer aggressively if they are in good health. The concern is that some older men with high risk cancer will suffer adversely from not receiving definitive treatment.
There is little published data specifically on outcomes for older patients with high grade prostate cancer treated with the different treatment modalities and how they compare to younger patients. Only with specific information will we be able to know how to approach these patients.
We analyzed our experience with patients diagnosed with Gleason 7 - 10 prostate cancer. Our aim is to examine patterns of initial treatment chosen for patients younger than 64 years, between 64 and 74 years, and 75 years or older. We then will evaluate whether there is a difference in disease characteristics and outcomes in older patients compared to younger patients.
2. Methods
With institutional review board approval, we reviewed all patients in the South Texas Veteran’s Healthcare System Tumor Registry diagnosed with prostate cancer between January 1, 1998 and December 31, 2008 capturing those with Gleason score greater than or equal to 7 on biopsy or on radical prostatectomy pathology who received treatment. The mean follow up time was 4.99 years. 123 surgical patients had a biopsy Gleason score < 7, but were found to have Gleason 7 - 10 disease on RP pathology. Statistical analysis was performed both with and without the inclusion of these patients, and their inclusion did not lead to significant differences in outcome measure comparison so they are included in our analysis.
Details thought to affect survival were recorded from the patient’s medical record and included age at diagnosis, date of diagnosis, pre-treatment PSA values, date and type of initial treatment, any neoadjuvant or adjuvant treatments, response to treatment including all PSA values, date of biochemical failure, metastasis and death where applicable. Treatment of biochemical failure or metastasis was also recorded. Patients with metastatic disease at the time of diagnosis were excluded as were patients with inadequate follow up.
As per national guidelines, biochemical failure was defined differently depending on the choice of primary treatment. For patients undergoing RP, biochemical failure was defined by the American Urological Association Prostate Cancer Guideline Panel’s definition of a PSA of 0.2 ng/mL or greater followed by a confirmatory PSA value 0.2 ng/mL or greater [6]. For patients undergoing RT, biochemical failure was defined by the Phoenix definition of a PSA rise 2.0 ng/mL above the nadir [7]. Patients receiving either RP or RT were also considered to have failed on the date any salvage treatment was initiated. For patients receiving HT, failure was defined by the development of metastasis, biochemical failure of two consecutive rises (dated to the first) after the initiation of both gonadotropin releasing hormone agonist and anti-androgen therapy or the initiation of another treatment modality. We considered this necessary as a significant response to an antiandrogen was not uncommon. Unfortunately, the antiandrogen treatment wasn’t always started immediately (if at all), making the biochemical endpoint for the androgen ablation patients somewhat in-exact.
Patients occasionally elected observation. Some later would elect more definitive therapy and are classified as to that treatment. Those that received no treatment (65 patients) before the development of metastasis or death were recorded as observation and excluded from evaluation of treatment outcomes. Patients were then stratified by age at the time of diagnosis and by primary treatment: RP, RT, or HT. Age stratification was treated as a categorical value with levels of age as younger than 64 years, 64 to 74 years, and older than 74 years.
Statistical Analysis: Chi square and Kruskal-Wallis tests were used to assess statistically significant difference in selection of primary treatment selection based on age. Multivariate competing risks regression analysis was performed, and hazard ratios were calculated with their 95% confidence intervals (CI). BFS, OS and CSS were determined and displayed using Kaplan-Meier curves. For tests of significance, p-values < 0.05 were considered to be statistically significant, except when determining interaction between variables, when <0.15 was considered statistically significant. Given the different sensitivities in defining failure based on PSA, especially in regards to androgen ablation where there is no concrete definition of becoming androgen insensitive, we did not report the statistical differences between the cohorts.
3. Results
We identified 1082 patients who received treatment for non-metastatic, Gleason 7 - 10 prostate cancer on either biopsy or surgical pathology. Primary treatment was RP in 585, RT in 253, and HT in 244 for a total of 1082 patients included for analysis. We identified 444 patients younger than 64 years, 369 64 - 74 years and 269 over 74 years of age.
Patients in the <64, 64 - 74 and >74 year age groups differed significantly in their pretreatment characteristic. Patients < 64 years had a significantly lower median PSA at diagnosis at 5.4 ng/mL compared with 6.2 ng/mL and 9.8 ng/mL for patients aged 64 - 74 and >74 years, respectively. Also, older patients had a higher incidence of Gleason 8 - 10 cancer (Table 1). There was no significant difference between age groups on the presence of bilateral disease at diagnosis, but patients > 74 years had higher mean percentage of positive biopsy cores, suggesting bulkier disease. Selection of RP as primary treatment decreased dramatically as age increased with 78.8%, 53.7% and 13.8% of patients < 64, 64 - 74, and >74 years receiving surgery, respectively. The use of HT as a primary treatment increased significantly as age increased with only 4.1% of patients younger than 64 years receiving HT compared to 17.6% and 59.9% of patients 64 - 74 and >74 years, respectively (Table 1).
For the entire population, age has a significant effect on overall (p < 0.001) and cancer specific mortality (p =
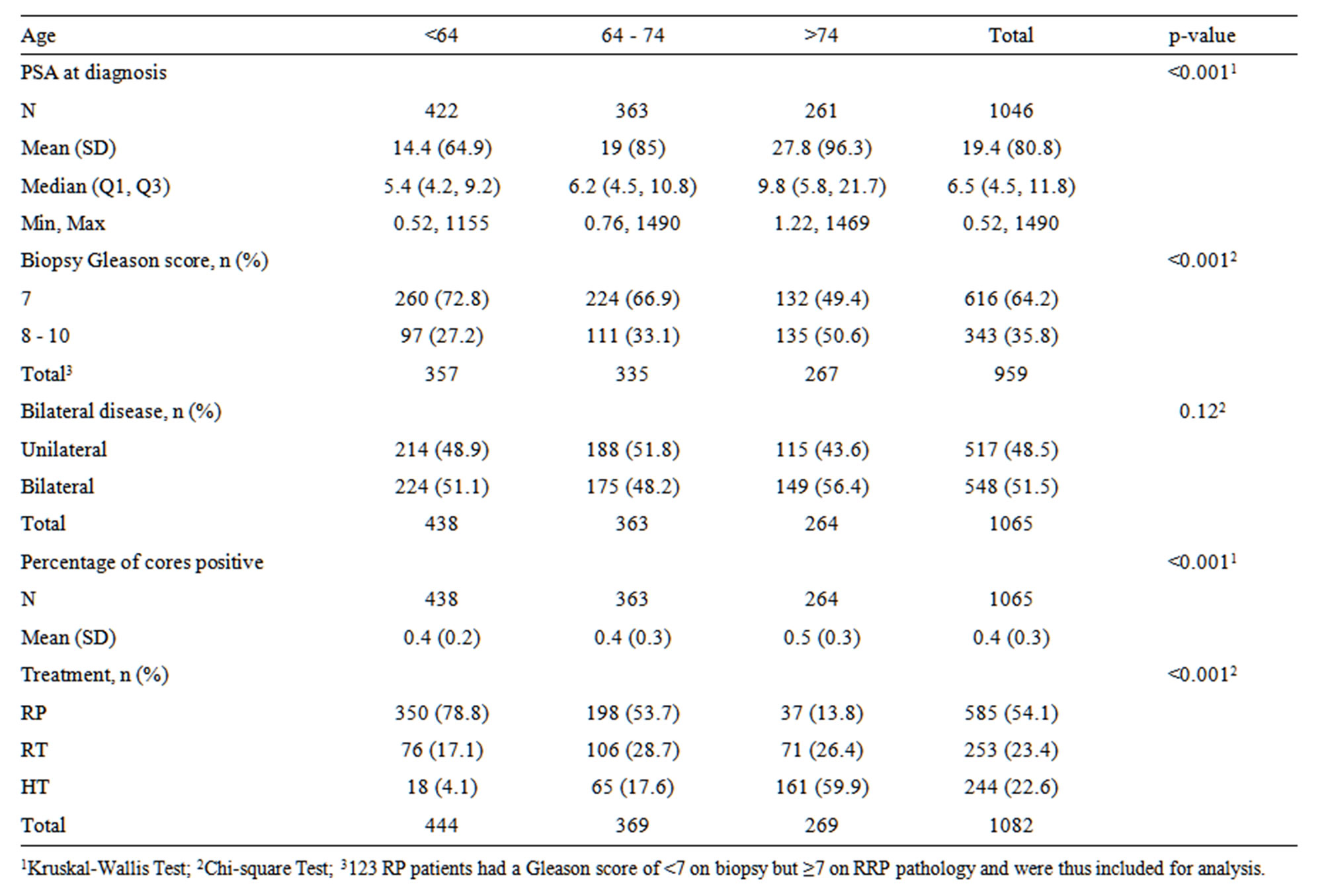
Table 1. Comparison of pretreatment features by age group.
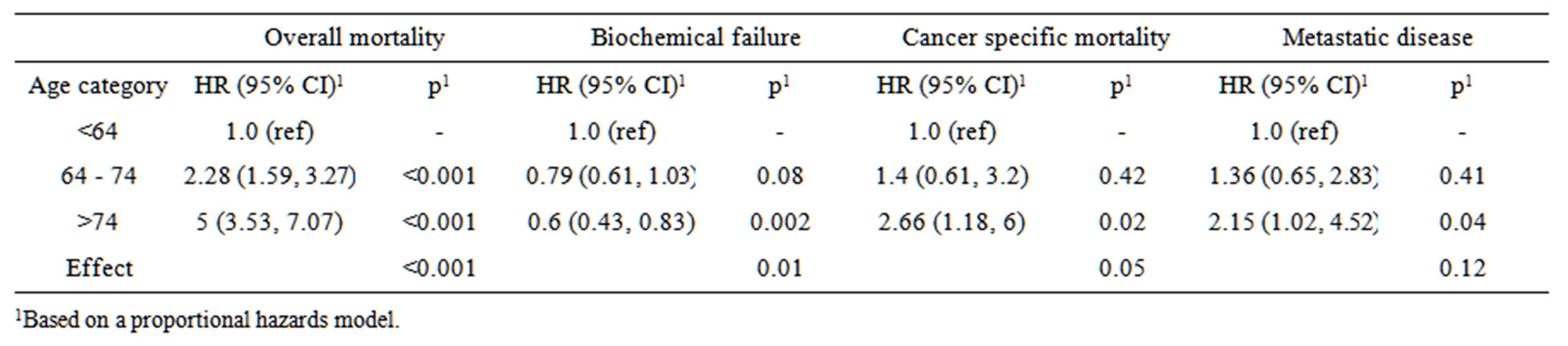
Table 2. Unadjusted effects of age category on overall mortality, biochemical failure, cancer specific mortality, and metastatic disease.
0.05) (Table 2). When compared to patients younger than 64 years, men older than 74 years had an overall mortality 5 times greater and cancer related mortality over 2 times greater. However, upon multivariate analysis after controlling for treatment, Gleason score and pre-treatment PSA, age was no longer associated with a significant difference in CSS.
In evaluation of the older patients alone (>74 years) (Table 3), Gleason score significantly affected 5 year BFFS; 90.9%, 89.7% and 66.2% with Gleason score of 7, 8 and 9 - 10, respectively (p < 0.001). Similarly, Gleason score significantly impacted 5 year cancer specific survival (CSS); 99.2%, 94.8% and 92.2% with Gleason score of 7, 8 and 9 - 10, respectively (p = 0.03). At this point, this did not, however, translate to a significant difference in OS. Also, 5 year BFFS, OS and CSS decreased significantly with increase pre-treatment PSA (p = 0.01, 0.001 and <0.001, respectively). Specifically, for PSA > 20 ng/mL, 5 year OS, BFFS and CSS were 50.7%, 72.5% and 85.5%, respectively. Although bilateral disease was not associated with a significant difference in outcome measures, 5 year BFFS decreased significantly when positive cores were ≥ 50% (p = 0.005).
Also, for men over age 74 years, there was no significant difference in OS or CSS between the primary treatment groups after controlling for Gleason score and

Table 3. Effect of pretreatment characteristics on BFFS, OS and CSS in patients > 74 years.
pre-treatment PSA. Specifically, older men over age 74 years didn’t have significant benefit from surgery. This is different for the men younger than 64 years; those who underwent RP had significantly increased 5 year OS and CSS compared to the HT patients.
For each individual age group, there was no difference in CSS between the three modalities (Table 4). For example, in the <64 years age group, the 5 yr CSS for RP, RT, and HT was 98.9%, 100% and 94.4% (p. 0.98), respectively. For patients 64 - 74 years and >74 years, there was a significant (p < 0.001) decline in OS between those selected for surgery versus radiation or hormone therapy. For any given treatment, there was no difference in CSS or OS between the age groups. For example, for those receiving RP, the 5 yr CSS for those < 64 was 98.9% versus 98.5% for the 64 - 74 year olds and 100% for those older than 74 years.
4. Discussion
We evaluated our experience for the effect of age on prostate cancer characteristics and outcomes for patients with intermediate to high risk non-metastatic prostate cancer and can make several observations.
Even within our high risk cohort, the older (>74 years old) patients were more likely to have the highest grade prostate cancer. In our series, 50.6% of men older than 74 years had Gleason 8 - 10 prostate cancer, compared with 33.1% of men aged 64 - 74 years and 27.2% of men younger than 64 years. This is not the first observation that older men are likely to have higher grade cancer. Autopsy studies show older men are significantly more likely to have higher Gleason scores and more advanced stage cancer than younger men [8]. This has been confirmed by biopsy studies [9]. The reasons for this are uncertain. While it is possible that older patients develop worse disease, the most likely explanation is the delay in diagnosis due to the reluctance to perform what may be unnecessary biopsies in an older patient. This is encouraged by the recommendations that routine screening should not be done in patients over 75 [10]. The result is that physicians delay intervention and only pursue a diagnosis in patients with a higher PSA, thereby detecting the worst cancers. This is supported by our finding of a higher average PSA, which in conjunction with more cores positive, would indicate bulkier disease in our older patients.
In addition, older patients are less likely to be treated with “curative” therapies like surgery and radiation as opposed to androgen ablation or simply following the patient. That was confirmed in our series. Although there were no set criteria, obviously treatment decisions were made based on patient age and perceived comorbidities. Interestingly, between the ages < 64 and >74, the number of patients receiving radiation treatment stayed the same,
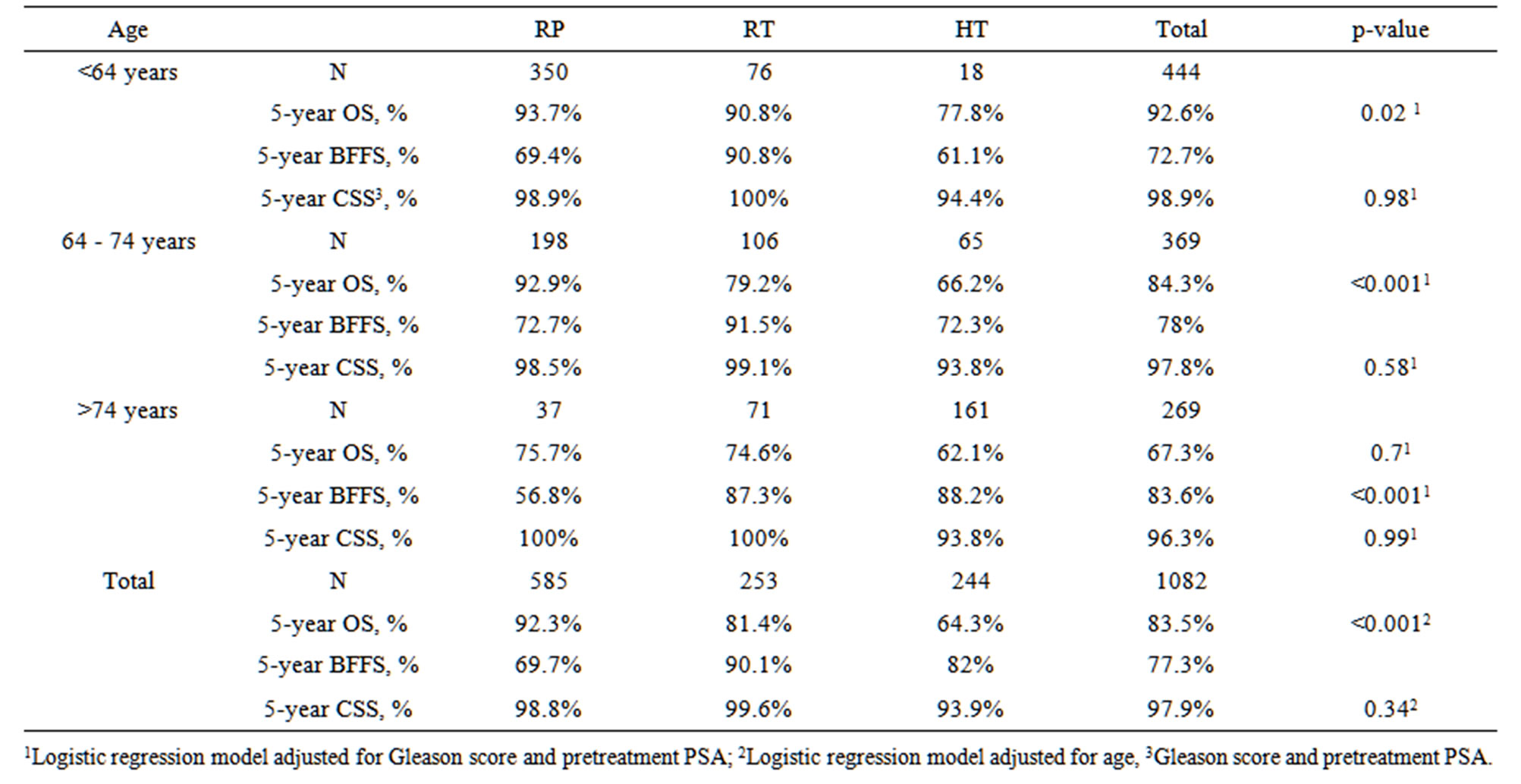
Table 4. Effect of primary treatment on OS, BFFS and CSS by age category.
but the number of patients undergoing surgery versus androgen ablation reversed. That treatment selection is based on perceived longevity is confirmed by our observation that cause specific survival is no different, but the overall survival was significantly different.
Some studies show a detriment to survival in high risk patients that receive non-aggressive treatment. Albertson et al. [11] examined patients treated with immediate or delayed HT with follow up for more than 20 years after diagnosis. Overall they report that men with high grade cancer frequently die of prostate cancer and perhaps require more aggressive treatment. The study included a small number of men aged 70 - 74 years with Gleason 7 (64 patients) and Gleason 8 - 10 (33 patients). The 5 year prostate cancer mortality for the Gleason 7 patients was 17% (from the graphs; 10 year 30%) and for the Gleason 8 - 10 patients was 29% (49% at 10 years). This was clearly worse than our experience. For our >74 year old Gleason 7 - 10 patients the 5 year prostate cancer mortality rate for observed/androgen ablated patients was 6.2%. This was not significantly different from those treated with radiation therapy or surgery. There are several possible reasons for this large discrepancy. We had a larger number of patients. Also, they were diagnosed in the “PSA era” meaning the cancers were probably less bulky than those pre 1984 patients in the Albertson study. Although treatment selection for our patients was empiric, it appears we were able to separate our patients for treatment based on life expectancy. Even though the cancer mortality was not significantly different between our groups, the overall mortality was, with 92.3% 5 year survival for surgery patients, 81.4 % for radiation patients and 64.3% for observation/androgen ablation patients. We were somewhat able to select out patients that would live long enough to potentially benefit from the more aggressive therapies.
The question then arises whether men over the age of 75 years should be treated aggressively. While commonly used in younger patients, the role of RP men older than 74 years is much more controversial. One reason for this is increased risk for perioperative morbidity and mortality in the elderly. Begg et al. [12] analyzed the Surveillance, Epidemiology and End Results (SEER) database and found a doubling in the perioperative mortality rates from 0.4% in men 65 - 69 years old to 0.9% in men older than 74 years. The same study showed a 5-fold increase in perioperative mortality in patients with significant comorbidity (as defined by Charlson score > 2) compared to those without comorbidity. Also, it appears that only younger patients benefit from an aggressive approach. The Scandinavian Prostate Cancer Group 4 randomized controlled trial evaluated cancerspecific mortality in patients treated with RP compared with watchful waiting [13]. In men younger than 65 years, patients randomized to receive RP had a 10% - 18% absolute risk reduction in cancer-specific mortality compared to those randomized to watchful waiting/ delayed androgen ablation. However, in men older than 65, there was no statistically significant risk reduction even when adjusted for PSA level, Gleason score and tumor stage.
In our cohort, patients under 64 were significantly more likely to undergo RP than those 64 - 74 and those older than 74 years. When adjusted for covariates, for our patients older than 74 years, RP offered no benefits over radiation or androgen ablation. This would support the observations cited above that older patients do just as well with non-invasive therapy.
The selection factors for radiation therapy are not as robust as those for surgery. As a result, RT is the most common treatment chosen for men older than age 70 with intermediate to high grade prostate cancer [14]. As long as the dosage is greater than 72 Gray (Gy), the biochemical progression free survival (BPFS) appears equivalent to that of RP or permanent seed implant [15]. We treated 230 patients with external beam radiotherapy (median dose 76 Gy) and/or brachytherapy. Twenty three percent also received adjuvant androgen ablation. While the radiation treated patients did well with a five year cause specific survival approaching 100% (Table 4), they really did no better than the androgen ablative patients regarding OS or CSS.
Given the uncertainties with treatment selection, there have been attempts to more formally apply comorbidity criteria in determining who should be treated aggressively. Several comorbidity indexes appear predictive and are similar in their accuracy, but they have not been widely adapted in decision making [16]. For now, it appears, that treatment decisions will continue to be made empirically, with the lament that some older men may be denied curative therapy that they need [17].
From our data, most noteworthy is that at 5 years there is not a dramatic benefit to more aggressive therapy in our older patients. Specifically, there was no difference in CSS between any of the three treatment modalities. While it can be argued that our follow up was just too short to detect the difference, in the older population length of follow becomes relative. Undoubtedly, if they live long differences may appear, but the biochemical failure rate at this point is only 16.4%, so we would not expect a big upsurge in life threatening metastatic disease in the near future. This would lend some support to the notion that these patients do not need the most aggressive treatment even with high risk disease to live out their lives cancer morbidity and mortality free. A note of caution is the observation that pretreatment PSA > 20 ng/mL in our older patients resulted in a higher risk (p < 0.001) of dying from prostate cancer1 (Table 3).
Study Strengths and Limitations: The principle strength of this study is the large number of patients, including those in the elderly age groups, from a single institution diagnosed and treated in the modern era. Study limitations include those inherent to any retrospective analysis. Selection of treatment was at the discretion of the original treating physician and factors such as age, comorbidities, prostate size and patient preference affected candidacy for surgery, radiation or hormone therapy. Certainly increased overall survival in patients < 64 and 64 - 67 years receiving RP may be due, in part, to the favorable disease characteristics, lack of medical comorbidities and other risk factors that made them suitable surgical candidates in the first place. Additionally, a mean follow up of 4.99 years is not long enough to capture all deaths from prostate cancer or development of metastatic disease. However, evaluation of a more modern cohort evaluating current surgical, radiation and hormone therapy techniques necessitates a shorter follow-up period. Additionally, the relatively short life expectancy of our elderly patients makes 5 years of follow-up more reasonable. Though all our patients were diagnosed and followed within the Veterans Health Care System, a central pathologic review was not performed, and Gleason scores as well as other pathologic features were recorded as interpreted by the original attending pathologists. Finally, differences in definitions of biochemical failure for patients undergoing RP, RT and HT make comparing BFFS between patients in the different initial treatment groups are inherently problematic. Though surgical patients appear to have the lowest BFFS, this may be due to the fact that the PSA threshold for failure is lowest for failure after prostatectomy.
5. Conclusion
Several observations can be made from our cohort. First, due to an apparent delayed diagnosis, even in our high risk population, the older patients have worse cancer. Second, in spite of their more advanced age, they do just as well as the younger patients with any of the three major treatment modalities. Finally, it appears that the older patients do just as well with androgen ablation as with the more aggressive radiation and surgical treatments. It would appear that the recommendation that aggressive treatment be reserved for men that are projected to live longer than 10 years is reasonable and well founded and that these men may do well with androgen ablation alone at some point in the disease process.
REFERENCES
- A. Jemal, R. Siegel, J. Xu, et al., “Cancer Statistics,” CA: A Cancer Journal for Clinicians, Vol. 60, No. 5, 2010, pp. 277-300. doi:10.3322/caac.20073
- J. M. Fitzpatrick, “Management of Localized Prostate Cancer in Senior Adults: The Crucial Role of Comorbidity,” BJU International, Vol. 101, Suppl. 2, 2008, pp. 16- 22. doi:10.1111/j.1464-410X.2007.07487.x
- D. Ilic, D. O’Connor, S. Green, et al., “Screening for Prostate Cancer: An Updated Cochrane Systematic Review,” BJU International, Vol. 107, No. 6, 2011, pp. 882-891. doi:10.1111/j.1464-410X.2010.10032.x
- US Preventive Services Task Force, “Screening for Prostate Cancer: US Preventive Services Task Force Recommendation Statement,” Annals of Internal Medicine, Vol. 122, No. 1, 2008, pp. 185-191. doi:10.1542/peds.2007-2210
- J. L. Mohler, R. R. Bahnson, B. Boston, et al., “NCCN Clinical Practice Guidelines in Oncology: Prostate Cancer,” Journal of the National Comprehensive Cancer Network, Vol. 10, No. 9, 2010, pp. 162-200.
- M. S. Cookson, G. Aus, A. L. Burnett, et al., “Variation in the Definition of Biochemical Recurrence in Patients Treated for Localized Prostate Cancer: The American Urological Association Prostate Guidelines for Localized Prostate Cancer Update Panel Report and Recommendations for a Standard in the Reporting of Surgical Outcomes,” The Journal of Urology, Vol. 177, No. 2, 2007, pp. 540-545. doi:10.1016/j.juro.2006.10.097
- M. Roach, G. Hanks, H. Thames, et al., “Defining Biochemical Failure Following Radiotherapy with or without Hormonal Therapy in Men with Clinically Localized Prostate Cancer: Recommendations of the RTOG-ASTRO Phoenix Consensus Conference,” International Journal of Radiation Oncology, Biology, Physics, Vol. 65, No. 4, 2006, pp. 965-974. doi:10.1016/j.ijrobp.2006.04.029
- N. B. Delongchamps, C. Y. Wang, V. Chandan, et al., “Pathological Characteristics of Prostate Cancer in Elderly Men,” The Journal of Urology, Vol. 182, No. 3, 2009, pp. 927-930. doi:10.1016/j.juro.2009.05.018
- J. C. Sung, J. N. Kabalin and M. K. Terris, “Prostate Cancer Detection, Characterization, and Clinical Outcomes in Men Aged 70 Years and Older Referred for Transrectal Ultrasound and Prostate Biopsies,” Urology, Vol. 56, No. 2, 2000, pp. 295-301. doi:10.1016/S0090-4295(00)00611-7
- K. Lin, R. Lipsitz, T. Miller, et al., “Benefits and Harms of Prostate-Specific Antigen Screening for Prostate Cancer: An Evidence Update for the US Preventive Services Task Force,” Annals of Internal Medicine, Vol. 149, No. 63, 2008, pp. 192-199.
- P. C. Albertsen, J. A. Hanley and J. Fine, “20-Year Outcomes Following Conservative Management of Clinically Localized Prostate Cancer,” The Journal of the American Medical, Vol. 293, No. 17, 2005, pp. 2095-2101. doi:10.1001/jama.293.17.2095
- C. B. Begg, E. R. Riedel, P. B. Bach, et al., “Variations in Morbidity after Radical Prostatectomy,” The New England Journal of Medicine, Vol. 346, 2002, pp. 1138-1144. doi:10.1056/NEJMsa011788
- A. Bill-Axelson, L. Holmberg, F. Filen, et al., “Radical Prostatectomy versus Watchful Waiting in Localized Prostate Cancer: The Scandinavian Prostate Cancer Group- 4 Randomized Trial,” Journal of the National Cancer Institute, Vol. 100, No. 16, 2008, pp. 1144-1154. doi:10.1093/jnci/djn255
- T. Bubolz, J. H. Wasson, L.-Y. Grace, et al., “Treatments for Prostate Cancer in Older Men: 1984-1997,” Urology, Vol. 58, No. 6, 2001, pp. 977-982. doi:10.1016/S0090-4295(01)01434-0
- P. A. Kupelian, L. Potters, D. Khuntia, et al., “Radical Prostatectomy, External Beam Radiotherapy < 72 Gy, External Beam Radiotherapy > or =72 Gy, Permanent Seed Implantation, or Combined Seeds/External Beam Radiotherapy for Stage T1-T2 Prostate Cancer,” International Journal of Radiation Oncology, Biology, Physics, Vol. 58, No. 1, 2004, pp. 25-33. doi:10.1016/S0360-3016(03)00784-3
- S. M. Alibhai, M. Leach, G. A. Tomlinson, et al., “Is There an Optimal Comorbidity Index for Prostate Cancer?” Cancer, Vol. 112, No. 5, 2008, pp. 1043-1050. doi:10.1002/cncr.23269
- K. L. Schwartz, S. M. Alibhai, G. Tomlinson, et al., “Continued Undertreatment of Older Men with Localized Prostate Cancer,” Urology, Vol. 62, No. 5, 2003, pp. 860- 865. doi:10.1016/S0090-4295(03)00690-3
NOTES
1Prostate cancer in the elderly.

