Journal of Diabetes Mellitus
Vol.07 No.03(2017), Article ID:78812,10 pages
10.4236/jdm.2017.73017
Relationship between C-Reactive Protein, White Blood Cell Count and Metabolic Syndrome in Nigerians with Type 2 Diabetes Mellitus
D. O. Soyoye1*, R. T. Ikem1, B. A. Kolawole1, R. A. Bolarinwa2, O. O. Amjo3, O. T. Yusuff3, F. A. Owolabi3, O. O. Ezekpo3
1Department of Medicine, Obafemi Awolowo University, Ile-Ife, Nigeria
2Department of Haematology and Immunology, Obafemi Awolowo Umiversity, Ile-Ife, Nigeria
3Department of Medicine, Obafemi Awolowo University Teaching Hospital, Ile-Ife, Nigeria

Copyright © 2017 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: July 28, 2017; Accepted: August 27, 2017; Published: August 30, 2017
ABSTRACT
Background: Presence of metabolic syndrome (MS) in people with diabetes confers increased cardiovascular and diabetes-specific micro- and macrovascular complications. The pathogenic pathways for metabolic syndrome are still issues for discussion especially in some special groups like those with type 2 diabetes mellitus (T2DM). Recent evidences suggest that inflammation may play a key role in MS. This study assessed the relationship between MS (and its component risks) and markers of inflammation (high-sensitivity C-reactive protein {hs-CRP} and white blood cells {WBC}). Methods: A cross-sectional study involving 108 patients with T2DM. Anthropometric measurements and clinical examination were conducted. Blood sample was collected for hs-CRP, WBC, glycated haemoglobin etc. Metabolic syndrome was defined using the International Diabetes Federation criteria. Ethical approval was granted and informed consent was obtained from participants. Results: Mean age of male and female participants were 58.00 ± 7.01 years and 55.48 ± 8.35 years respectively (p = 0.092). Eighty-two (75.9%) participants had metabolic syndrome. Median values of hs-CRP and total WBC were 0.89mg/L and 5.73 x103/mm3 respectively. On correlation, hs-CRP showed statistically significant association with waist circumference (r = 0.194; p = 0.044), fasting plasma glucose (r = 0.191; p = 0.048) and serum triglycerides (p = 0.226; r = 0.019). There was no statistically significant association between WBC and the metabolic components. Conclusion: Prevalence of metabolic syndrome is high, and C-reactive protein was associated with waist circumference, fasting plasma glucose and serum triglycerides.
Keywords:
C-Reactive Protein, Inflammatory Markers, Metabolic Syndrome, Type 2 Diabetes Mellitus

1. Introduction
Insulin resistance syndrome, Reaven’s syndrome, deadly quartet, metabolic cardiovascular syndrome etc., are some of the names that have been used to describe the metabolic syndrome (MS) [1] [2] [3] . It denotes a constellation of risk factors whose presence confers increased atherosclerotic cardiovascular and metabolic risks that is more than the sum of the risks associated with individual abnormalities [1] [2] [4] . The traditional component risks of the syndrome include raised fasting plasma glucose, blood pressure, triglycerides, reduced high-density lipoprotein cholesterol (HDL) and central obesity [4] [5] , but it has been suggested that other non-traditional risks such as insulin resistance, inflammation, thrombosis etc., are possible components [4] .
The clinical and epidemiologic implications of this syndrome are reflected by increased incidence of type 2 diabetes (T2DM), cardiovascular disease (CVD) and mortality [6] [7] . The association of MS and T2DM appears to be stronger compared with its association with cerebrovascular disease, coronary heart disease or CVD [3] [8] . Metabolic syndrome not only results in increased occurrence of T2DM, but also in the development of its microvascular and macrovascular complications [9] [10] .
The main pathophysiological abnormality of this syndrome remains open for discourse. Environmental and genetic influences are suggested as contributors to its occurrence [4] . Insulin resistance is generally regarded as the principal underlying abnormality of MS, however, other authors have postulated that this syndrome may be caused by some of its component risks including obesity, dyslipidaemia, abnormalities in the adipose tissue and chronic inflammation [11] [12] .
The relationship between metabolic syndrome and inflammation may still remain unclear, but available evidence suggests that inflammation may play a major role in mediating both insulin resistance in vascular endothelium, and may contribute to the link between conditions such as metabolic syndrome, type 2 diabetes and CVD [13] [14] . Inflammatory markers that may be increased in response to increase in adipose tissue include tumour necrosis factor α (TNF-α), interleukin 6 (IL-6) and C-reactive protein (CRP).
Inflammatory markers in association with metabolic syndrome have been mainly studied among Caucasians, but most studies on metabolic syndrome in Africans have been limited to prevalence studies. Studies on the possible association of inflammation and/or inflammatory markers in people with metabolic syndrome in Africans are sparse. Also, studies of metabolic syndrome among people with diabetes seem surprisingly fewer than expected. This study evaluated the relationship between metabolic syndrome and markers of inflammation (high-sensitivity C-reactive protein {hs-CRP} and total white blood cell {WBC}) among Nigerians.
2. Patient and Methods
This cross-sectional study involved 108 consecutive adult Nigerians with type 2 diabetes attending the diabetes out-patient clinic of a tertiary hospital in South-West Nigeria. Ethical approval was sought and granted by the hospital’s Ethics and Research Committee. Written informed consent was obtained from participants. Participants with features of infection within a month prior to recruitment were excluded. Patients presenting with diabetic emergencies and other acute clinical conditions e.g. myocardial infarction, cerebrovascular disease were similarly excluded. Clinical history and relevant clinical examination were performed and documented. Weight, height and waist circumference (WC) were measured using standard protocols [15] , and body mass index (BMI) was calculated [16] .
2.1. Laboratory Investigation
Blood samples were collected after eight hours overnight fast for determination of serum levels of total cholesterol, low-density lipoprotein (LDL), high-density lipoprotein (HDL), triglycerides, fasting plasma glucose (FPG). Glycated haemoglobin was measured using the Bio-Rad in2it System (Deeside, UK). Sysmex KX-21N Haematology Analyzer (Illnois, USA) was used for full blood count estimation (including white cell counts). High-sensitivity C-reactive protein was measured using a solid phase enzyme-linked immunosorbent assay (Diagnostic Automation Inc., California, USA).
2.2. Definition of Metabolic Syndrome
Metabolic syndrome (MS) was diagnosed using the International Diabetes Federation (IDF) criteria [4] [17] . Since all the participants were patients with type 2 diabetes, waist circumference ≥94 cm in males or ≥80 cm in females, and any one of the following was used as the criteria to diagnose people with metabolic syndrome: systolic BP ≥ 130 or diastolic BP ≥ 85 mm Hg or treatment of previously diagnosed hypertension; serum triglycerides ≥150 mg/dL (1.7 mmol/L) or specific treatment for this lipid abnormality; <40 mg/dL (1.03 mmol/L) in males or <50 mg/dL (1.29 mmol/L) in females or specific treatment for this lipid abnormality [4] [17] .
2.3. Data Analysis
Data was analyzed using the Statistical Package of Social Sciences (SPSS) version 20. Categorical variables were expressed using frequency and percentages. Continuous variables were expressed in means, and differences in means between a continuous variable and dichotomous variable was done using the Student’s t test for normally distributed variables, and using Mann Whitney U test for variables that were not distributed normally. Relationship between hs-CRP and WBC and the components risks of MS was evaluated using the Spearman’s correlation. Statistical significance was set at p < 0.05.
3. Results
This study involved 46 males and 62 females, with mean age of 58.00 ± 7.01 years and 55.48 ± 8.35 years respectively (p = 0.092), and have been diagnosed with type 2 diabetes between one to twenty-five years. Most of the participants were either traders, civil servants or retiree with rates of 41.3%, 25.3% and 21.3% respectively. Seventy (64.8%) participants were hypertensive [male = 27 (25.0%) vs. female 43 (39.8%); p = 0.251]. Females had significantly higher mean BMI and serum LDL level; the mean waist-to-hip ratio (WHR) was significantly higher among males. There was no statistical difference in the mean values of the other clinical or laboratory parameters considered among male and female participants. Table 1 shows mean anthropometric, clinical and laboratory variables of the participants.
Metabolic syndrome defined by the IDF criteria occurred among 82 (75.9%) of the study participants; 25 (54.3%) of the male participants and 57 (91.9%) of the female participants (p < 0.001). Reduced HDL, hypertension, central obesity, and elevated triglycerides occurred among 81.5%, 79.6%, 76.9% and 30.6% of the participants respectively. Three (2.8%), 11 (10.2%), 26 (24.1%), 44 (40.7%) and 24 (22.2%) of the participants had one, two, three, four and five of the components of metabolic syndrome respectively. Median values of hs-CRP and total WBC were 0.89 mg/L and 5.73 × 103/mm3 respectively. Figure 1 and Figure 2
Table 1. Mean anthropometric, clinical and laboratory variables among participants.
FPG = Fasting plasma glucose; LDL = low-density lipoprotein; HDL = high-density lipoprotein; hs-CRP = high sensitivity C-reactive protein; WBC = white blood cells.
Figure 1. Distribution of hs-CRP and component risks of metabolic syndrome among participants.
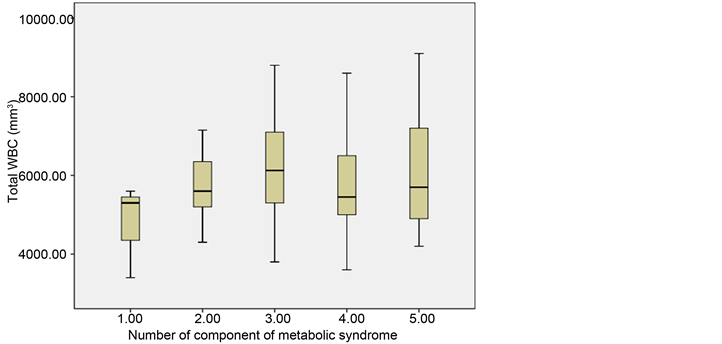
Figure 2. Distribution of total white blood cell and component risks of metabolic syndrome among participants.
shows box plot distribution of number of metabolic syndrome components among participants with serum hs-CRP and total WBC respectively.
Median values of hs-CRP in participants with one, two, three, four and five metabolic risks were 0.47 mg/L (interquartile range, 0.26 - 0.74 mg/L), 1.20 mg/L (interquartile range, 0.72 - 2.77 mg/L), 0.90 mg/L (interquartile range, 0.24 - 2.08 cmg/L), 0.65mg/L (interquartile range, 0.27 - 1.61 mg/L) and 1.04 mg/L (interquartile range, 0.59 - 3.03 mg/L) respectively.
Median values of total WBC in participants with one, two, three, four and five MS components were 5.30 × 103/mm3 (interquartile range, 4.35 - 5.45 × 103/ mm3), 5.80 × 103/mm3 (interquartile range, 5.30 - 6.45 × 103/mm3), 6.15 × 103/mm3 (interquartile range, 5.30 - 7.30 × 103/mm3), 5.60 × 103/mm3 (interquartile range, 5.20 - 6.75 × 103/mm3) and 5.60 × 103/mm3 (interquartile range, 5.00 - 7.15 × 103/mm3) respectively.
Using the Mann Whitney U test, mean hs-CRP was higher in participants with MS (1.52 ± 1.69 mg/L) compared with participants without it (1.39 ± 1.48 mg/L), this did not however attained statistical significance (p = 0.825). Mean total WBC was similar in patients with and without MS (p = 0.233). A consideration of the relationship of hs-CRP with the component risks of MS showed a statistically significant positive correlation between hs-CRP and waist circumference (r = 0.194; p = 0.044), fasting plasma glucose (r = 0.191; p = 0.048) and serum triglycerides (p = 0.226; r = 0.019). There was no statistical significant correlation between total WBC and any of the metabolic risks. Table 2 is a Spearman’s correlation analysis showing the relationship between hs-CRP and total WBC with component risks of metabolic syndrome.
4. Discussion
The high prevalence of MS among Nigerians with T2DM has been stated earlier [18] [19] [20] , consistent with our findings in this study. Metabolic syndrome in people with diabetes presents an innocuous association, as it does not only increase the occurrence of other cardiovascular complications, but may be involved in the pathogenesis of specific diabetic microvascular and macrovascular complications [10] . Inflammation is a non-traditional risk in explaining the occurrence of MS and its component risks. In this study, we evaluated the association of MS and its component risks with hs-CRP and total white blood cells.
This study further emphasized the high occurrence of MS in T2DM. Prevalence of MS may be affected by the population studied and the diagnostic criteria used. In this study, using the IDF criteria, a prevalence rate of 75.9% was found among participants with a significantly higher occurrence among females. Puepet et al. [19] reported a lower prevalence of 63.3% among people with T2DM using IDF criteria in North Central Nigeria, and a male preponderance. The
Table 2. Relationship between high-sensitivity CRP and total WBC and component risks of metabolic syndrome.
hs-CRP = high sensitivity C-reactive protein; WBC = white blood cells; BP = blood pressure.
higher prevalence of MS in this study compared to Puepet et al. may be due to different prevailing occupations in the study participants. Most of the participants in this study were traders who usually sell wares in shops, and hence may be mainly sedentary. Most of the other studies [18] [20] [21] [22] in Nigeria used other diagnostic criteria apart from the IDF criteria. Out of these studies, those [18] [21] [22] done in South-West Nigeria, reported higher prevalence in females as obtained in this study.
C-reactive protein is an acute phase reactant of the pentagastrin family of proteins, and it increases with injury, tissue death or inflammation [23] . Elevated CRP has been suggested as part of the ‘platinum standard’ definition of additional metabolic measurements which may be associated with metabolic syndrome [4] . Studies have confirmed this possible association of CRP and MS. In a study involving 190 obese individuals, Florez et al. [24] reported a higher CRP level among obese patients with MS, and it correlated with waist circumference and BMI among the study participants. In a study by Ye et al. [25] involving 3289 aged 50 to 70 years where MS was diagnosed using the National Cholesterol Education Program-Adult Treatment Panel III (NCEP-ATP III), CRP medians increased with number of components of MS, and CRP was also observed to be correlated with BMI and WC. In this study, higher hs-CRP was observed in diabetic patients with MS compared with diabetics without MS. Median values of hs-CRP increased with numbers of components of MS, though non-linearly. This is similar to findings by Ye et al. and Florez et al. [24] [25] .
There have been some variations in relationship of CRP and component risks of MS. In our study, hs-CRP correlated positively with waist circumference, fasting plasma glucose and serum triglycerides level respectively. Association of hs-CRP with waist circumference has also been reported in other studies [24] [25] [26] [27] . As reported in our study, hs-CRP was associated with waist circumference and triglycerides levels in a study in Netherlands involving 1165 people with central obesity [28] . High-sensitivity CRP was also found to be particularly associated with FPG in a report by Mirhafez et al. [29] as observed in our study.
White cell count is a readily available measure of inflammation in this environment and presumably in most developing nations. Assessment of the association between MS and its components and total peripheral WBC count showed that mean total WBC was not significantly different among participants with MS and those without it. Total WBC increased with numbers of MS, this trend was not however perfectly linear. There was no significant correlation between components of MS and total WBC. Our findings contrast with those reported by Park et al. [30] , where 104 nondiabetic patients undergoing peritoneal dialysis were assessed and MS was defined using the NCEP-ATPIII criteria. They found higher WBC in the participants with MS. Also WBC increased significantly with number of MS, and was positively correlated with BMI, homeostatic model assessment of insulin resistance (HOMA-IR), and triglycerides and negatively correlated with HDL.
Obesity, a major component of the MS is characterized by chronic inflammation and is accompanied by elaboration of cytokines such TNF-α, and IL-6, which is a major stimulus for hepatic CRP secretion [31] [32] . It has been postulated that obesity, by secreting these cytokines and perpetuating a state of chronic low-grade inflammatory, may result in insulin resistance, endothelial and other cardiovascular dysfunctions [33] [34] .
5. Limitations
The cross-sectional method adopted in this study may be a limitation as the association from correlation study may not denote causal link, hence causal association of inflammation with metabolic syndrome may need to be further evaluated in prospective studies. Also the relatively small size of the study may result in non-significant associations where larger studies would have shown significance. This may explain lack of statistical significance obtained between total WBC and MS.
Despite these limitations, we believe, to the best of our knowledge, that this would be the first study that aimed to evaluate the association of inflammatory markers with MS in a cohort of type 2 diabetics in this environment. It should therefore provide a good stimulus for further studies among our diabetic patients with metabolic syndrome.
6. Conclusion
Prevalence of MS is high in our setting, and CRP was associated with the MS in type 2 diabetics. C-reactive protein has a positive correlation with WC, FPG and triglycerides. Diagnosis and treatment of these component risks may reduce the chronic inflammation that characterizes the syndrome.
Cite this paper
Soyoye, D.O., Ikem, R.T., Kolawole, B.A., Bolarinwa, R.A., Amjo, O.O., Yusuff, O.T., Owolabi, F.A. and Ezekpo, O.O. (2017) Relationship between C-Reactive Protein, White Blood Cell Count and Metabolic Syndrome in Nigerians with Type 2 Diabetes Mellitus. Journal of Diabetes Mellitus, 7, 212-221. https://doi.org/10.4236/jdm.2017.73017
References
- 1. Eckel, R.H., Grundy, S.M. and Zimmet, P.Z. (2005) The Metabolic Syndrome. Lancet, 365, 1415-1428. https://doi.org/10.1016/S0140-6736(05)66378-7
- 2. Hjermann, I. (1992) The Metabolic Cardiovascular Syndrome: Syndrome X, Reaven’s Syndrome, Insulin Resistance Syndrome, Atherothrombogenic Syndrome. Journal of Cardiovascular Pharmacology, 20, S5-S10. https://doi.org/10.1097/00005344-199220008-00002
- 3. Li, C. and Ford, E.S. (2006) Definition of the Metabolic Syndrome: What’s New and What Predicts Risk? Metabolic Syndrome and Related Disorders, 4, 237-251. https://doi.org/10.1089/met.2006.4.237
- 4. The IDF Consensus Worldwide Definition of the Metabolic Syndrome (2006) https://www.idf.org/component/attachments/attachments.html?id=705&task=download
- 5. Alberti, K.G.M.M., Eckel, R.H., Grundy, S.M., Zimmet, P.Z., Cleeman, J.I., Donato, K.A., et al. (2009) Harmonizing the Metabolic Syndrome: A Joint Interim Statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation, 120, 1640-1645. https://doi.org/10.1161/CIRCULATIONAHA.109.192644
- 6. Lakka, H.M., Laaksonen, D.E., Lakka, T.A., Niskanen, L.K., Kumpusalo, E., Tuomilehto, J., et al. (2002) The Metabolic Syndrome and Total and Cardiovascular Disease Mortality in Middle-Aged Men. Journal of the American Medical Association, 288, 2709-2716. https://doi.org/10.1001/jama.288.21.2709
- 7. Lorenzo, C., Okoloise, M., Williams, K., Stern, M.P., Haffner, S.M. and San Anthonio Heart Study (2003) The Metabolic Syndrome as Predictor of Type 2 Diabetes: The San Antonio Heart Study. Diabetes Care, 26, 3153-3156. https://doi.org/10.2337/diacare.26.11.3153
- 8. Ford, E.S. (2005) Risks for All-Cause Mortality, Cardiovascular Disease, and Diabetes Associated with the Metabolic Syndrome: A Summary of the Evidence. Diabetes Care, 28, 1769-1778. https://doi.org/10.2337/diacare.28.7.1769
- 9. Moehlecke, M., Leitao, C.B., Kramer, C.K., Rodrigues, T.C., Nickel, C., Silveiro, S.P., et al. (2010) Effect of Metabolic Syndrome and of Its Individual Components on Renal Function of Patients with Type 2 Diabetes Mellitus. Brazilian Journal of Medical and Biological Research, 43, 687-693. https://doi.org/10.1590/S0100-879X2010007500054
- 10. Metascreen Writing Committee, Bonadonna, R., Cucinotta, D., Fedele, D., et al. (2006) The Metabolic Syndrome Is a Risk Indicator of Microvascular and Macrovascular Complications in Diabetes: Results from Metascreen, a Multicenter Diabetes Clinic-Based Survey. Diabetes Care, 29, 2701-2707. https://doi.org/10.2337/dc06-0942
- 11. Reaven, G.M. (1988) Role of Insulin Resistance in Human Disease. Diabetes, 37, 1595-1607. https://doi.org/10.2337/diab.37.12.1595
- 12. Laclaustra, M., Corella, D. and Ordovas, J.M. (2007) Metabolic Syndrome Pathophysiology: The Role of Adipose Tissue. Nutrition, Metabolism & Cardiovascular Diseases, 17, 125-139.
- 13. Sutherland, J.P., Mckinley, B. and Eckel, R.H. (2004) The Metabolic Syndrome and Inflammation. Metabolic Syndrome and Related Disorders, 2, 82-104. https://doi.org/10.1089/met.2004.2.82
- 14. Dokken, B.B. (2008) The Pathophysiology of Cardiovascular Disease and Diabetes: Beyond Blood Pressure and Lipids. Diabetes Spectrum, 21, 160-165. https://doi.org/10.2337/diaspect.21.3.160
- 15. World Health Organization (2008) Waist Circumference and Waist-Hip Ratio Report of a WHO Expert Consultation. WHO, Geneva.
- 16. World Health Organization (1995) World Health Organisation. Physical Status: The Use and Interpretation of Anthropometry. Report of a WHO Expert Committee. WHO Technical Report Series No 854.
- 17. Alberti, K.G., Zimmet, P. and Shaw, J. (2006) Metabolic Syndrome—A New World-Wide Definition. A Consensus Statement from the International Diabetes Federation. Diabetic Medicine, 23, 469-480. https://doi.org/10.1111/j.1464-5491.2006.01858.x
- 18. Ogbera, A.O. (2010) Prevalence and Gender Distribution of the Metabolic Syndrome. Diabetology & Metabolic Syndrome, 2, 1. https://doi.org/10.1186/1758-5996-2-1
- 19. Puepet, F.H., Uloko, A., Akogu, I.Y. and Aniekwensi, E. (2009) Prevalence of the Metabolic Syndrome among Patients with Type 2 Diabetes Mellitus in Urban North-Central Nigeria. African Journal of Endocrinology and Metabolism, 8, 12-14.
- 20. Isezuo, S.A. and Ezunu, E. (2005) Demographic and Clinical Correlates of Metabolic Syndrome in Native African Type-2 Diabetic Patients. Journal of the National Medical Association, 97, 557-563.
- 21. Adediran, O.S., Edo, A.E., Jimoh, A.K. and Ohwovoriole, A.E. (2007) Prevalence of the Metabolic Syndrome among Nigerians with Type 2 Diabetes. Diabetes International, 15, 13-14.
- 22. Alebiosu, C.O. and Odusan, B.O. (2004) Metabolic Syndrome in Subjects with Type-2 Diabetes Mellitus. Journal of the National Medical Association, 96, 817-821.
- 23. Chandrashekara, S. (2014) C-Reactive Protein: An Inflammatory Marker with Specific Role in Physiology, Pathology, and Diagnosis. Internet Journal of Rheumatology and Clinical Immunology, 2, SR3.
- 24. Florez, H., Castillo-Florez, S., Mendez, A., Casanova-Romero, P., Larreal-Urdaneta, C., Lee, D., et al. (2006) C-Reactive Protein Is Elevated in Obese Patients with the Metabolic Syndrome. Diabetes Research and Clinical Practice, 71, 92-100.
- 25. Ye, X., Yu, Z., Li, H., Franco, O.H., Liu, Y. and Lin, X. (2007) Distributions of C-Reactive Protein and Its Associations with Metabolic Syndrome in Middle-Aged and Older Chinese People. Journal of the American College of Cardiology, 49, 1798-1805.
- 26. Zuliani, G., Volpato, S., Galvani, M., Ble, A., Corsi, A.M., et al. (2009) Elevated C-Reactive Protein Levels and Metabolic Syndrome in the Elderly: The Role of Central Obesity Data from the in Chianti Study. Atherosclerosis, 203, 626-632.
- 27. Huffman, F.G., Gomez, G.P. and Zarini, G.G. (2009) Metabolic Syndrome and High-Sensitivity C-Reactive Protein in Cubans. Ethinicity and Disease, 19, 115-120.
- 28. Den Engelsen, C., Koekkoek, P.S., Gorter, K.J., van der Donk, M., Salome, P.L. and Rutten, G.E. (2012) High-Sensitivity C-Reactive Protein to Detect Metabolic Syndrome in a Centrally Obese Population: A Cross-Sectional Analysis. Cardiovascular Diabetology, 11, 25. https://doi.org/10.1186/1475-2840-11-25
- 29. Mirhafez, S.R., Ebrahim, M., Saberi Karimian, M., Avan, A., Tayefi, M., Heidari-Bakavoli, A., et al. (2016) Serum High-Sensitivity C-Reactive Protein as a Biomaker in Patients with Metabolic Syndrome: Evidence-Based Study with 7284 Subjects. European Journal of Clinical Nutrition, 70, 1298-1304. https://doi.org/10.1038/ejcn.2016.111
- 30. Park, J.T., Chang, T.I., Kim, D.K., Choi, H.Y., Lee, J.E., Kim, H.W., et al. (2009) Association of White Blood Cell Count with Metabolic Syndrome in Patients Undergoing Peritoneal Dialysis. Metabolism, 58, 1379-1385.
- 31. Hotamisligil, G.S., Shargill, N.S. and Spiegelman, B.M. (1993) Adipose Expression of Tumor Necrosis Factor-α: Direct Role in Obesity Linked Insulin Resistance. Science, 259, 87-91. https://doi.org/10.1126/science.7678183
- 32. Crichton, M.B., Nichols, J.E., Zhao, Y., Bulun, S.E. and Simpson, E.R. (1996) Expression of Transcripts of Interleukin-6 and Related Cytokines by Human Breast Tumors, Breast Cancer Cells, and Adipose Stromal Cells. Molecular and Cellular Endocrinology, 118, 215-220.
- 33. Monteiro, R. and Azevedo, I. (2010) Chronic Inflammation in Obesity and the Metabolic Syndrome. Mediators of Inflammation, 2010, Article ID: 289645. https://doi.org/10.1155/2010/289645
- 34. Dandona, P., Aljada, A. and Bandyopadhyay, A. (2004) Inflammation: The Link between Insulin Resistance, Obesity and Diabetes. Trends in Immunology, 25, 4-7.