Food and Nutrition Sciences
Vol. 2 No. 5 (2011) , Article ID: 5797 , 7 pages DOI:10.4236/fns.2011.25066
Effects of Sodium Lactate on the Survival of Listeria monocytogenes, Escherichia Coli O157:H7, and Salmonella spp. in Cooked Ham at Refrigerated and Abuse Temperatures*
![]()
1SEQ CHAPTER \h \r 1Residue Chemistry and Predictive Microbiology Research Unit, Eastern Regional Research Center, Agricultural Research Service, United States Department of Agriculture, Wyndmoor, USA; 2Food Safety and Intervention Technologies SEQ CHAPTER \h \r 1 Research Unit, Eastern Regional Research Center, Agricultural Research Service, United States Department of Agriculture, Wyndmoor, USA.
Email: #Andy.Hwang@ars.usda.gov, Shiowshuh.Sheen@ars.usda.gov, Vijay.Juneja@ars.usda.gov
Received April 26th, 2011; revised May 11th, 2011; accepted May 18th, 2011.
Keywords: Ham, Lactate, Listeria Monocytogenes, Escherichia Coli O157:H7, Salmonella spp.
ABSTRACT
The objective of this study was to determine the effect of sodium lactate on the survival of Listeria monocytogenes, Escherichia coli O157:H7, and Salmonella spp. in cooked ham during storage at refrigerated and abuse temperatures. Cooked ham was added with 0% - 3% lactate, inoculated with a multiple-strain mixture of L. monocytogenes, E. coli O157:H7, or Salmonella spp. and stored at 4˚C - 15˚C for up to 35 day. The growth of the three pathogens was inhibited in ham containing 3% lactate, and no growth of E. coli O157:H7 and Salmonella spp. occurred at the lowest storage temperatures of 6 and 8˚C, respectively. In ham containing no lactate, the average growth rates were 0.256 - 0.380 log CFU/day for L. monocytogenes at 4˚C - 8˚C, 0.242 - 0.315 log CFU/day for E. coli O157:H7 at 8˚C - 15˚C, and 0.249 - 0.328 log CFU/day for Salmonella spp. at 10˚C - 15˚C. The addition of 1% or 2% lactate significantly (P < 0.05) reduced the growth rates of the three pathogens, and the effect was more profound at lower temperatures. Salmonella spp. were more sensitive to the effect of lactate than L. monocytogenes and E. coli O157:H7. Polynomial models were developed to describe the growth rates of the three pathogens as affected by the lactate concentration and storage temperature. Results from this study demonstrate the effect of lactate on the growth of L. monocytogenes, E. coli O157:H7, and Salmonella spp. in cooked ham and indicate the effective lactate concentrations and storage temperatures that can be used to enhance the microbiological safety of ready-to-eat ham products.
1. Introduction
Refrigerated ready-to-eat (RTE) meat products are processed with a heat treatment to kill vegetative microorganisms; therefore, the products are generally free of vegetative pathogens. However, reports have indicated that the products are susceptible to recontamination with Listeria monocytogenes, Escherichia coli O157:H7, and Salmonella spp. after heat processing. This is evidenced by several outbreaks of foodborne illness that were linked to the consumption of RTE meats [1-4]. L. monocytogenes causes listeriosis with a high mortality rate. E. coli O157:H7 causes serious illnesses that include hemorrhagic colitis and hemolytic uremic syndromes, while Salmonella spp. cause severe gastroenteritis in human [5,6]. Post-thermal processing contamination of RTE meats mainly occurs during product handling, slicing, or packaging in the manufacturing facilities. RTE meats are consumed without prior cooking, therefore controlling the growth of the pathogens that may contaminate RTE meats is particularly important for ensuring the microbial safety of the products. Since L. monocytogenes is capable of growing at refrigerated temperatures, it is a particular concern among pathogens that may contaminate RTE meats. RTE meats contaminated with L. monocytogenes were implicated in several listeriosis outbreaks [1,2]. Although not as prominent as L. monocytogenes in RTE meats, E. coli O157:H7 and Salmonella spp. have been increasingly linked to RTE meats as frequent contaminants in raw meats used as ingredients for RTE meat products. A survey reported that Salmonella spp. were found in 1.9% retail beef samples [7], and, in 2007, 9.9 million kg of fresh and frozen ground beef were recalled due to E. coli O157:H7 contamination [8]. This implicates that both pathogens are likely to be introduced into RTE meat processing environment through the raw meat ingredient.
Salts of lactate have been increasingly used in meat, poultry, and smoked seafood products to control the growth of foodborne pathogens [9,10]. They are used in RTE meats to enhance microbial food safety and shelf life and as flavor agents. Lactate is affirmed as generally recognized as safe (GRAS) and can be used as a direct food ingredient in meat and poultry products at levels not to exceed 4.8% by weight of total formulation [11,12]. The antimicrobial effect of lactate, alone or in combination with other food additives, have been examined and reported. Ukuku et al. [13] used a solution of 1% sodium lactate, 1% hydrogen peroxide, 25 µg/ml nisin, and 0.5% citric acid as a washing solution to treat E. coli O157:H7-inoculated whole cantaloupes and honey dew melons. The levels of the pathogen on the surface of melons were reduced by 3 - 4 log CFU/cm2. RaybaudiMassilia et al. [14] used a dipping solution containing 1% N-acetylL-cysteine, 1% glutathione, and 1% calcium lactate to treat fresh-cut Fuji apple and reported reductions of 2.0 log CFU/g for L. monocytogenes, 1.5 log CFU/g for S. Enteritidis, and 3.0 log CFU/g for E. coli O157:H7 in apple pieces after a 30-d storage at 5˚C. Quilo et al. [15] examined a treatment of 200 ppm peroxyacetic acid followed by 3% lactate for beef trimmings inoculated with E. coli and S. Typhimurium. They reported a significant reduction of S. Typhimurium, but not E. coli, on the trimmings during a 7-d refrigerated storage. Maks et al. [16] reported that lactate and pediocin together rendered L. monocytogenes more sensitive to heat in bologna. Studies have also examined the combination of lactate with UV light (254 nm) in inactivating L. monocytogenes on frankfurters [17], lactate in bioactive alginate coatings to control L. monocytogenes on coldsmoked salmon slices and fillets [18], the combination of lactate and oregano oil in ground beef to control Salmonella spp. [19], and the combination of lactate and lauric arginate to control L. monocytogenes on frankfurters [20,21]. These and many other studies [3,22-25] show that studies examining the antimicrobial effect of lactate in RTE meats have been mainly focused on L. monocytogenes; few have examined the effect of lactate on E. coli O157:H7 and Salmonella spp. The objective of this study was to examine the effects of lactate on L. monocytogenes, Escherichia coli O157:H7, and Salmonella spp. in RTE cooked ham under refrigerated and temperature-abuse conditions. L. monocytogenes was included in this study to serve as a comparison to E. coli O157:H7 and Salmonella spp.
2. Materials and Methods
2.1. L. monocytogenes, E. coli O157:H7, and Salmonella spp.
Five strains of L. monocytogenes, MFS 2 (pork processing plant isolate), MFS 102 (frankfurters isolate), Scott A (clinical isolate), 101M (beef and pork sausage isolate), and F6854 (turkey frankfurters isolate), four strains of E.coli O157:H7, EC505B (beef isolate), EC204P (pork isolate), C7927 (apple cider isolate), and C9490 (hamburger isolate), and four species of Salmonella ssp., S. Typhimurium (beef isolate), S. Kentucky, S. Enteritidis (clinic isolate), and S. Saintpaul (stuffed ham isolate) were used in this study. All bacterial strains were obtained from the culture collection of the Microbial Food Safety Research Unit, Eastern Regional Research Center, Agricultural Research Service, U.S. Department of Agriculture. Each bacterial strain was maintained at –80˚C in Brain Heart Infusion (BHI; Difco Laboratories Inc., Detroit, MI., U.S.A.) broth containing 10% glycerol (Sigma Chemical Co., St. Louis, MO., U.S.A.). A loopful of cell suspension of each strain was subcultured twice in 10 ml BHI broth and incubated at 37˚C for 24 h. Cell suspension from each strain was mixed together, and the mixture was diluted with sterile 0.1% (w/v) peptone water (PW) to achieve a population of approximately 105 CFU/ml for use as inoculum. The cell count of each inoculum was determined by spreadplating 0.1 ml diluted cell mixture onto PALCAM medium base formulated with antimicrobic supplement (Becton, Dickinson and Company, Sparks, MD., U.S.A.) for L. monocytogenes, Sorbitol MacConkey agar (SMAC; Becton, Dickinson and Company) for E. coli O157:H7, and Xylose-Lysine-Tergitol agar (XLT; Becton, Dickinson and Company) for Salmonella spp. Plates were incubated at 37˚C for 24 - 48 h before typical colonies were counted.
2.2. Ham Preparation
A restructured cooked ham product (76% moisture, 2.9% fat, 16.5% protein, 1.9% salt, and <0.1 μg/g nitrite) purchased from a local supplier was used in this study. The ham, in ground form, was added with sodium lactate to ensure that the selected levels of lactate were presented in the ham. To prepare the ham samples, ham was cut into 1 cm × 1 cm × 1 cm pieces, and the ham pieces, in stomacher bags, were heated in a water bath to 63˚C and held for 30 min to inactivate the native microflora. After the heat treatment, the ham pieces were cooled under running water and kept at –20˚C for overnight. The frozen, heat-treated ham pieces were ground in a Waring blender (Waring Commercial, Torrington, CT., U.S.A.). One hundred grams of ground ham were placed into stomacher bags (Spiral Biotech Inc., Norwood, MA., U.S.A.) and added with sodium lactate (65% syrup, Sigma Chemical Co.). The amount of lactate syrup added was adjusted for its concentration to achieve a final lactate concentration of 0, 1%, 2%, or 3% (w/w) in ground ham. The bags were hand-massaged for 30 s and then pummeled for 2 min in a BagMixer 400 Stomacher (Interscience, St. Nom, France). The bags were stored at 4˚C overnight for equilibration. The pH of ham after equilibration was measured using a SevenMulti pH meter fitted with an InLab RoutinPro pH electrode (Mettler-Toledo, Schwerzenbach, Switzerland). The pHs of samples were pH 5.7 - 6.1. After storage, 4 g of ground ham were placed into 100-ml stomacher bags (Spiral Biotech Inc.) for inoculation.
2.3. Sample Inoculation and Storage
One-tenth of one milliliter of the L. monocytogenes, E. coli O157:H7, or Salmonella spp. inoculum was added into bags and mixed thoroughly by hands. The bags were vacuum-sealed using a Multivac A300 vacuum sealer (Multivac Inc., Kansas, MO., U.S.A.). The inoculum level of L. monocytogenes, E. coli O157:H7, or Salmonella spp. in ham was approximately 102 CFU/g. The samples were stored at 4, 8, and 10˚C for L. monocytogenes, 6, 8, 10, 12, and 15˚C for E. coli O157:H7, and 8, 10, 12, and 15˚C for Salmonella spp. for up to 35 d. The temperatures were refrigerated and abuse temperatures or the growth/no growth boundary temperatures for each pathogen.
2.4. Microbial Analysis
During storage, samples were enumerated for counts of L. monocytogenes, E. coli O157:H7, or Salmonella spp. at 6 different times. At each sampling time, two samples were added with 8 ml PW, and the mixture was pummeled for 2 min. Additional dilutions were prepared in serial PW, and duplicated 0.1 ml of appropriate dilution were surface-plated on PALCAM, SMAC, and XLT to enumerate cell counts of L. monocytogenes, E. coli O157:H7, and Salmonella spp., respectively. All plates were incubated at 35˚C for 24 - 48 h before typical colonies were counted. The experiment was performed twice.
2.5. Average Growth Rate and Effect Analysis
Means of cell counts (log CFU/g) of each pathogen in ham during storage were plotted against sampling times to obtain growth curves. From the curves the average growth rates (AGR, log CFU/d) for each pathogen were estimated by dividing the numbers of net population increase (log CFU/g) by the length of time of storage (d). Since most of the treatments resulted in growth curves without lag phases, the AGR were a good indication of the effect of lactate and storage temperature on the pathogens. To analyze the individual and combined effect of lactate and storage temperature, the AGR were transformed into square root The GR were transformed into square root to stabilize the variance [26] and analyzed by the General Linear Model (GLM) of Statistical Analysis System 9.1 (SAS Institute Inc., Cary, NC., U.S.A.) as a function of lactate concentration, storage temperature, and their interaction with the following quadratic equation:
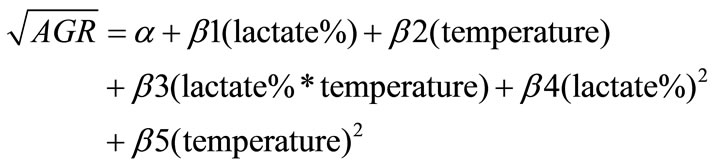
where α is the intercept, and β1- β5 are estimated coefficients.
3. Results and Discussion
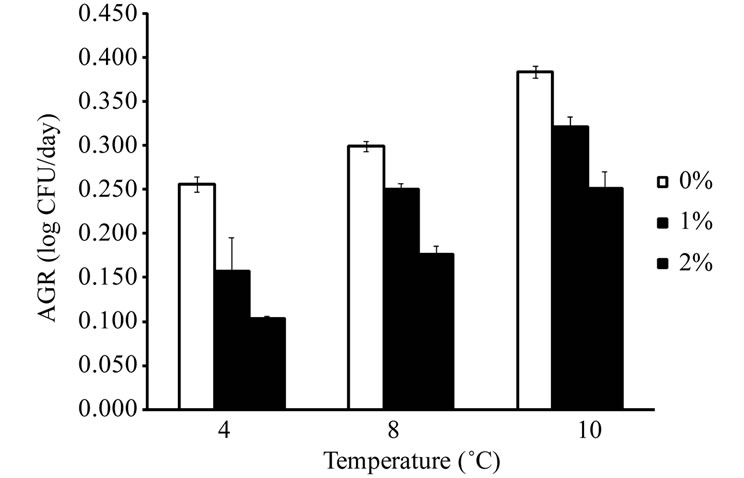
E. coli O157:H7 and Salmonella spp. were not able to grow in ham samples stored at 6˚C and 8˚C, respectively, regardless of the concentration of lactate in ham. The temperatures observed in this study were similar to the minimal growth temperatures (approximately 7˚C - 8˚C) reported for pathogenic E. coli and Salmonella spp. [19,27]. L. monocytogenes, E. coli O157:H7, and Salmonella spp. did not grow (less than 0.5 log CFU/g increase during storage) in ham containing 3% lactate at all storage temperatures. The addition of lactate in ham at 1% or 2% reduced the growth rates of L. monocytogenes, E. coli O157:H7, and Salmonella spp. when compared to ham containing no lactate. The results indicate that lactate at 3% or higher concentrations may prevent the growth of these pathogens in ham at refrigerated and mild abuse temperatures for up to 35 d, while lactate at lower levels slow the growth of these pathogens. The AGR of L. monocytogenes, E. coli O157:H7, and Salmonella spp. in ham containing 0% - 2% lactate are shown in Figure 1. In ham containing no lactate, the AGR were 0.256 - 0.380 log CFU/d for L. monocytogenes at 4˚C - 10˚C, 0.242 - 0.315 log CFU/d for E. coli O157:H7 at 8˚C - 15˚C, and 0.249 - 0.328 log CFU/d for Salmonella spp. at 10˚C - 15˚C. The addition of 1% or 2% lactate in ham significantly (P < 0.05) reduced the AGR of the
 (a)
(a)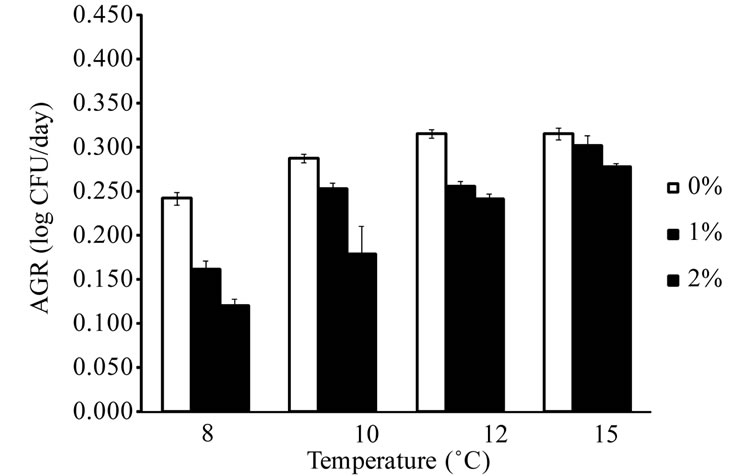 (b)
(b)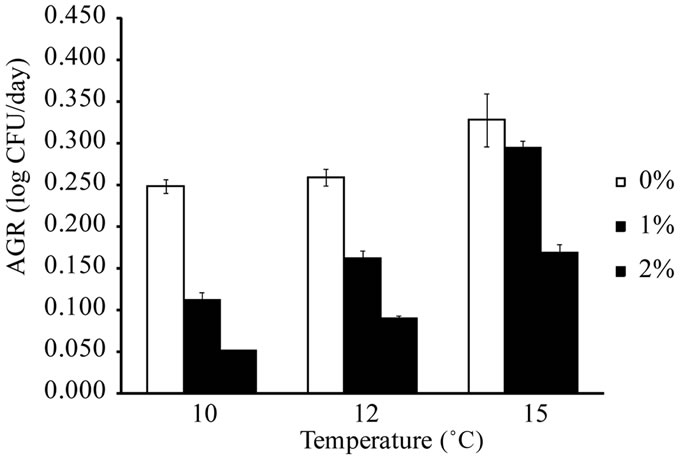 (c)
(c)
Figure 1. Average growth rates of L. monocytogenes (a), E. coli O157:H7 (b), and Salmonella spp. (c) in cooked ham formulated with 0% - 2% lactate and stored at refrigerated and abuse temperatures (n = 4).
three pathogens, and the effect was more profound at lower storage temperatures. The AGR of L. monocytogenes in ham containing no lactate at 4˚C was 0.256 log CFU/d, and it was reduced to 0.158 and 0.104 log CFU/d with the addition of 1% and 2% lactate, respecttively. Lactate at 1% and 2% in ham reduced the AGR of L. monocytogenes by 35% and 60% at 4˚C, respectively, whereas the reductions were 16% and 41% at 8˚C, and 16% and 34% at 10˚C (Figure 1(a)). At the lowest storage temperatures that supported the growth of E. coli O157:H7 and Salmonella spp., the AGR reductions caused by 1% and 2% lactate for E. coli O157:H7 were 33% and 50% at 8˚C (Figure 1(b)), and were 55% and 79% for Salmonella spp. at 10˚C (Figure 1(c)). The lactate-induced AGR reduction decreased as the storage temperature for E. coli O157:H7 and Salmonella spp. increased. The reductions were 4% - 19% and 12% - 38% for E. coli O157:H7 at 12 and 15˚C, and were 10% - 37% and 49% - 65% for Salmonella spp. at 12 and 15˚C, respectively. The results indicated that Salmonella spp. were more sensitive to the growth-rate reducing effect of lactate than L. monocyto-genes and E. coli O157:H7.
The findings regarding the antimicrobial effect of lactate on L. monocytogenes, E. coli O157:H7, and Salmonella spp. in RTE ham is in agreement with results reported by other studies using different foods or growth media. Thompson et al. [23] reported that L. monocytogenes levels in turkey roll containing 2% lactate increased only 2 log CFU/cm2, which was significant lower than those in samples containing no lactate, after a 6-week refrigerated storage. The growth-delaying effect of lactate on L. monocytogenes in food products or microbiological growth media were also reported by Glass et al. [24], Stekelenburg [28], Legan et al. [29], and Abou-Zeid et al. [30]. McWilliam Leitch and Steward [31, 32] reported a reduced viability of E. coli O157:H7 and non-O157 E. coli isolates in a lactate solution (100 mM) at 37˚C. Neetoo et al. [18] reported that lactate (1.2 or 2.4%) suppressed the growth of spoilage aerobic and anaerobic microorganisms at 4˚C. In this study, Salmonella spp. were observed to grow in ham containing 1 and 2% lactate stored at 10˚C - 15˚C, albeit the growth was slower comparing to those in samples containing no lactate. Similar observations have also been reported. Jung et al. [33] examined the growth kinetics of lactic acid-stressed and unstressed S. Typhimurium in broth and chicken patties containing a mixture of 2.2% potassium lactate and 0.16% sodium diacetate and reported that the growth of lactic acid-stressed S. Typhimurium was inhibited by the mixture at 10˚C, whereas the growth of unstressed S. Typhimurium was not inhibited. Juneja et al. [5] also reported that Salmonella spp. was not able to grow at 15˚C in heat-treated ground beef containing 1.5% lactate.
The individual and combined effects of lactate and storage temperature on the 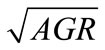 of the three pathogens were analyzed using a quadratic regression. The estimated coefficients for each parameter and their significance levels are shown in Table 1. The resulted
of the three pathogens were analyzed using a quadratic regression. The estimated coefficients for each parameter and their significance levels are shown in Table 1. The resulted
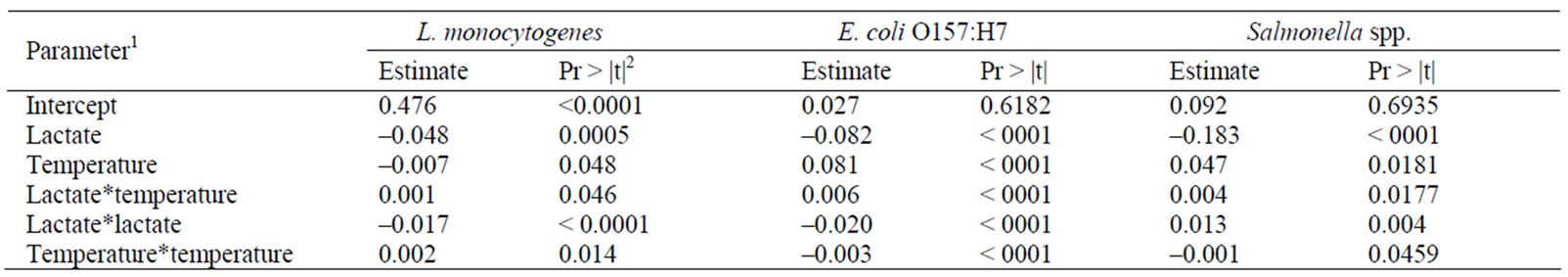
Table 1. Coefficients and significance of fitted parameters.
regression equations that describe the 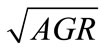 for each of the three pathogens in ham containing 0% - 2% lactate and at storage temperatures that supported their growth are:
for each of the three pathogens in ham containing 0% - 2% lactate and at storage temperatures that supported their growth are:
L. monocytogenes (4˚C - 10˚C)

E. coli O157:H7 (8˚C - 15˚C)

Salmonella spp. (10˚C - 15˚C)

In general lactate concentration, storage temperature, and their interaction significantly (P < 0.05) affect the  of L. monocytogenes, E. coli O157:H7, and Salmonella spp. To determine the goodness-of-fit of the equations, the predicted
of L. monocytogenes, E. coli O157:H7, and Salmonella spp. To determine the goodness-of-fit of the equations, the predicted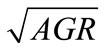 , obtained from the equations, were plotted against the observed
, obtained from the equations, were plotted against the observed 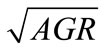 (Figure 2). The linear relationship between the predicted and observed
(Figure 2). The linear relationship between the predicted and observed 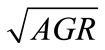 is indicated by the correlation coefficients (r2) of 0.98 for L. monocytogenes, 0.96 for E. coli O157:H7, and 0.98 for Salmonella spp., indicating a consistent closeness of the predicted and observed values within the ranges of lactate concentration and temperature examined. The predicted
is indicated by the correlation coefficients (r2) of 0.98 for L. monocytogenes, 0.96 for E. coli O157:H7, and 0.98 for Salmonella spp., indicating a consistent closeness of the predicted and observed values within the ranges of lactate concentration and temperature examined. The predicted 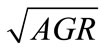 are mostly within ±8% of the observed for L. monocytogenes and E. coli
are mostly within ±8% of the observed for L. monocytogenes and E. coli
 (a)
(a)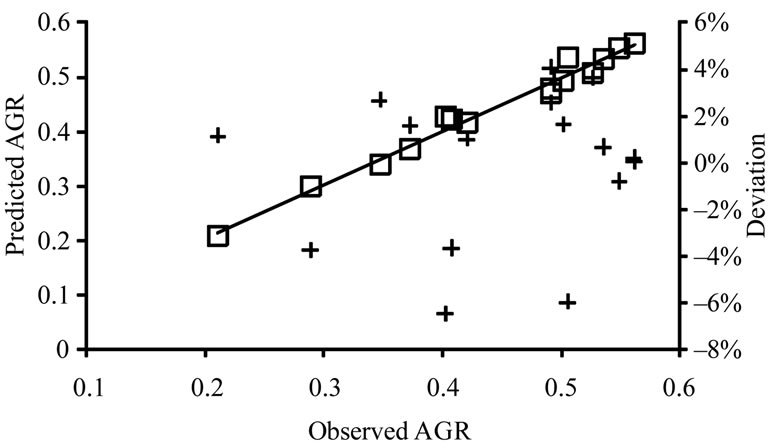 (b)
(b)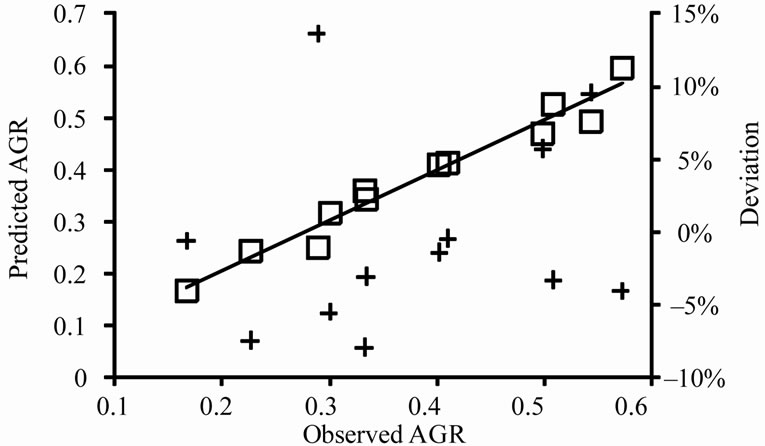 (c)
(c)
Figure 2. Predicted versus observed  (□) for L. monocytogenes (a), E. coli O157:H7 (b), and Salmonella spp. (c) and the deviations of predicted values to the observed values (+).
(□) for L. monocytogenes (a), E. coli O157:H7 (b), and Salmonella spp. (c) and the deviations of predicted values to the observed values (+).
O157:H7, and are within ±10% of the observed for Salmonella spp., indicating that the equations closely describe the observed 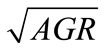 of the three pathogens in ham containing 0% - 2% lactate at the storage temperatures that supported their growth.
of the three pathogens in ham containing 0% - 2% lactate at the storage temperatures that supported their growth.
4. Conclusions
The present study showed that lactate had a broad antimicrobial effect in ham as it delayed the growth of L. monocytogenes as well as E. coli O157:H7 and Salmonella spp. at refrigerated and abuse temperatures. The results indicated the individual and combined effect of lactate and storage temperature on delaying the growth of L. monocytogenes, E. coli O157:H7, and Salmonella spp in ham. The mathematical models that describe the average growth rates of the three pathogens in ham will be beneficial to the manufacturers of RTE meats in formulating lactate concentrations in ham for food-safety and sensory quality purposes.
5. Acknowledgments
The authors would like to thank Ms. Stacy Raleigh for technical assistance with performing the laboratory component of this study.
REFERENCES
- Centers for Disease Control and Prevention, “Multistate Outbreak of Listeriosis—United States, 1998,” Morbidity and Mortality Weekly Report, Vol. 49, 1998, pp. 1085- 1086.
- Centers for Disease Control and Prevention, “Public Health Dispatch: Outbreak of Listeriosis—Northeastern United States,” Morbidity and Mortality Weekly Report, Vol. 51, 2001, pp. 950-951.
- Centers for Disease Control and Prevention, “Investigation Update: Multistate Outbreak of Human Salmonella Montevideo Infections, 2010. http://www.cdc.gov/salmonella/montevideo/index.html.
- S. Ojha and M. Kostrzynska, “Approaches for Reducing Salmonella in Pork Production,” Journal of Food Protection, Vol. 70, No. 11, 2007, pp. 2676-2694.
- V. Juneja, C.-A. Hwang and M. Friedman, “Thermal Inactivation and Postthermal Treatment Growth during Storage of Multiple Salmonella Serotype in Ground Beef as afFected by Sodium Lactate and Oregano Oil,” Journal of Food Protection, Vol. 75, No. 1, 2010, pp. M1-M6.
- T. M. Osaili, C. L. Griffis, E. M. Martin, B. L. Beard, A. E. Keener and J. A. Marcy, “Thermal Inactivation of Escherichia coli O157:H7, Salmonella, and Listeria Monocytogenes in Breaded Pork Patties,” Journal of Food Science, Vol. 72, No. 2, 2007, pp. M56-M61.
- C. Zhao, B. Ge, J. De Villena, R. Sudler, E. Yeh, S. Zhao, D. G. White, D. Wagner and J. Meng, “Prevalence of Campylobacter spp., Escherichia coli, and Salmonella Serovars in Retail Chicken, Turkey, Pork and Beef from the Greater Washington D.C. Area,” Applied and Environmental Microbiology, Vol. 67, No. 12, 2001, pp. 5431-5436. doi:10.1128/AEM.67.12.5431-5436.2001
- The Food Safety and Inspection Service, US Department of Agriculture, “Updated: New Jersey firm Expands Recall of Ground Beef Products Due to Possible E. coli O157:H7 Contamination,” 2007. http://www.fsis.usda.gov/PDF/Recall_040_2007_Exp_Update.pdf.
- A. Lianou, I. Geornaras, P. A. Kendall, J. A. Scanga and J. N. Sofos, “Behavior of Listeria Monocytogenes at 7˚C in Commercial Turkey Breast, with or without Antimicrobials, after Simulated Contamination for Manufacturing, Retail and Consumer Settings,” Food Microbiology, Vol. 24, No. 5, 2007, pp. 433-443. doi:10.1016/j.fm.2006.11.002
- R. L. Thompson, C. E. Carpenter, S. Martini and J. R. Broadbent, “Control of Listeria Monocytogenes in Ready-to-Eat Meats Containing Sodium Levulinate, Sodium Lactate, or a Combination of Sodium Lactate and Sodium Diacetate,” Journal of Food Science, Vol. 78, No. 5, 2008, pp. M239-M244.
- Code of Federal Regulations, “Title 21-Food and Drugs. Part 184, Direct Food Substances Affirmed as Generally Recognized as Safe,” The Office of the Federal Register, the US National Archives and Records Administration, College Park, MD.
- Code of Federal Regulations, “Title 9-Animals and Animal Products. Part 424, Preparation and Processing Operations,” The Office of the Federal Register, the US National Archives and Records Administration, College Park, MD, 2009.
- D. O. Ukuku, M. L. Bari, S. Kawamoto and K. Isshiki, “Use of Hydrogen Peroxide in Combination with Nisin, Sodium Lactate and Citric Acid for Reducing Transfer of Bacterial Pathogens from Whole Melon Surfaces to Fresh-Cut Pieces,” International Journal Food Microbiology, Vol. 104, No. 2, 2005, pp. 225-233. doi:10.1016/j.ijfoodmicro.2005.01.016
- R. M. Raybaudi-Massilia, J. Mosqueda-Melgar, A. Sobrino-Lopez, R. Soliva-Fortuny and O. Martin-Belloso, “Use of Malic Acid and Other Quality Stabilizing Compounds to Assure The Safety of Fresh-Cut ‘Fuji’ Apples by Inactivation of Listeria Monocytogenes, Salmonella Enteritidis and Escherichia Coli O157:H7,” Journal of Food Safety, Vol. 29, No. 2, 2009, pp. 236-252. doi:10.1111/j.1745-4565.2009.00153.x
- S. A. Quilo, F. W. Pohlman, P. N. Dias-Morse, A. H. Brown, P. G. Crandall and R. P. Story, “Microbial, Instrumental Color and Sensory Characteristics of Inoculated Ground Beef Produced Using Potassium Lactate, Sodium Metasilicate or Peroxyacetic Acid as Multiple Antimicrobial Interventions,” Meat Science, Vol. 84, 2010, pp. 470-476. doi:10.1016/j.meatsci.2009.09.018
- N. Maks, L. Zhu, V. Juneja and S. Ravishankar, “Sodium Lactate, Sodium Diacetate and Pediocin: Effect and Interactions on the Thermal Inactivation of Listeria Monocytogenes on Bologna,” Food Microbiology, Vol. 27, No. 1, 2010, pp. 64-69. doi:10.1016/j.fm.2009.08.004
- C. H. Sommers, P. H. Cooke, X. Fan and J. E. Sites, “Ultraviolet Light (254 nm) Inactivation of Listeria Monocytogenes on Frankfurters That Contain Potassium Lactate and Sodium Lactate,” Journal of Food Science, Vol. 74, No. 3, 2009, pp. M114-M119.
- H. Neetoo, M. Ye and H. Chen, “Bioactive Alginate Coatings to Control Listeria Monocytogenes on ColdSmoked Salmon Slices and Fillets,” International Journal of Food Microbiology, Vol. 136, No. 3, 2010, pp. 326- 331. doi:10.1016/j.ijfoodmicro.2009.10.003
- The International Commission on Microbiological Specifications for Foods, “Intestinally Pathogenic Escherichia Coli,” In: T. A. Robert, A. C. Baird-Parker and R. B. Tompkin, Eds., Microorganisms in Foods 5: Characteristics of Microbial Pathogens, Blackie Academic and Professional, London, 1996, p. 131.
- E. M. Martin, C. L. Griffis, K. L. S. Vaughn, C. A. O’Bryan, E. C. Friendly, J. A. Marcy, S. C. Ricke, P. G. Crandall and R. Y. Lary, “Control of Listeria Monocytogenes by Lauric Arginate on Frankfurters Formulated with or with Lactate/Diacetate,” Journal of Food Science, Vol. 74, No. 6, pp. M237-M241.
- A. C. S. Porto-Fett, S. G. Campano, J. L. Smith, A. Oser, B. Shoyer, J. E. Call and J. B. Luchansky, “Control of Listeria Monocytogenes on Commercially-Produced Frankfurters Prepared with and without Potassium Lactate and Sodium Diacetate and Surface Treated with Lauric Arginate Using the Sprayed Lethality in Container (SLIC®) Delivery Method,” Meat Science, Vol. 85, No. 2, 2010, pp. 312-318.doi:10.1016/j.meatsci.2010.01.020
- I. M. Barmpalia, K. P. Koutsoumanis, I. Geornaras, K. E. Belk, J. A. Scanga, P. A. Kendall, G. C. Smith and J. N. Sofos, “Effect of Antimicrobials as Ingredients of Pork Bologna for Listeria Monocytogenes Control during Storage at 4 or 10˚C,” Food Microbiology, Vol. 22, 2005, pp. 205-211. doi:10.1016/j.fm.2004.08.003
- G. K. Bedie, J. Samelis, J. N. Sofos, K. E. Belk, J. A. Scanga and G. C. Smith, “Antimicrobials in the Formulation to Control Listeria Monocytogenes Postprocessing Contamination on Frankfurters Stored at 4˚C in Vacuum Packages,” Journal of Food Protection, Vol. 64, No. 12, 2001, pp. 1949-1955.
- K. A. Glass, D. A. Granberg, A. L. Smith, A. M. McNamara, M. Hardin, J. Mattias, K. Ladwig and E. A. Johnsoni, “Inhibition of Listeria Monocytogenes by Sodium Diacetate and Sodium Lactate on Wieners and Cooked Bratwurst,” Journal of Food Protection, Vol. 65, No. 1,
- 2002, pp. 116-123.
- C.-A. Hwang and M. L. Tamplin, “Modeling the Lag Phase and Growth Rate of Listeria Monocytogenes in Ground Ham Containing Sodium Lactate and Sodium Diacetate at Various Storage Temperatures,” Journal of Food Science, Vol. 72, 2007, pp. M246-M253.
- F. Devlieghere, A. H. Geeraerd, K. J. Versyck, B. Vandewaetere, J. Van Impe and J. Debevere, “Growth of Listeria Monocytogenes in Modified Atmosphere Packed Cooked Meat Products: A Predictive Model,” Food Microbiology, Vol. 18, No. 1, 2001, pp. 53-66. doi:10.1006/fmic.2000.0378
- The International Commission on Microbiological Specifications for Foods, “Salmonellae,” In: T. A. Robert, A. C. Baird-Parker and R. B. Tompkin, Eds., Microorganisms in Foods 5: Characteristics of Microbial Pathogens, Blackie Academic and Professional, London, 1996, p. 225.
- F. K. Stekelenburg, “Enhanced Inhibition of Listeria Monocytogenes in Frankfurter Sausage by the Addition of Potassium Lactate and Sodium Diacetate Mixtures,” Food Microbiology, Vol. 20, No. 1, 2003, pp. 133-137. doi:10.1016/S0740-0020(02)00098-9
- J. D. Legan, D. L. Seman, A. L. Milkowski, J. A. Hirschey and M. H. Vandeven, “Modeling the Growth Boundary of Listeria Monocytogenes in Ready-to-Eat Cooked Meat Products as a Function of the Product Salt, Moisture, Potassium Lactate, and Sodium Diacetate Concentrations,” Journal of Food Protection, Vol. 67, No. 10, 2004, pp. 2195-2204.
- K. A. Abou-Zeid, K. S. Yoon, T. P. Oscar, J. G. Schwarz, F. M. Hashem, F. M. and R. C. Whiting, “Survival and Growth of Listeria Monocytogenes in Broth as a Function of Temperature, pH, and Potassium Lactate and Sodium Diacetate Concentrations,” Journal of Food Protection, Vol. 70, No. 11, 2007, pp. 2620-2625.
- E. C. McWilliam Leitch and C. S. Stewart, “Escherichia Coli O157 and Non-O157 Isolates Are More Susceptible to L-Lactate than to D-Lactate,” Applied and Environmental Microbiology, Vol. 68, No. 9, 2002, pp. 4676- 4678. doi:10.1128/AEM.68.9.4676-4678.2002
- E. C. McWilliam Leitch and C. S. Stewart, “Susceptibility of Escherichia Coli O157 and Non-O157 Isolates to Lactate,” Letters in Applied Microbiology, Vol. 35, 2002, pp. 176-180. doi:10.1046/j.1472-765X.2002.01164.x
- Y. J. Jung, K. J. Min and Y. S. Yoon, “Responses of Acid-Stressed Salmonella Typhimurium in Broth and Chicken Patties to Subsequent Antimicrobial Stress with ε-Polylysine and Combined Potassium Lactate and Sodium Diacetate,” Food Microbiology, Vol. 26, No. 5, 2009, pp. 467-474. doi:10.1016/j.fm.2009.02.007
NOTES
*Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.

