International Journal of Clinical Medicine
Vol. 3 No. 7 (2012) , Article ID: 26178 , 7 pages DOI:10.4236/ijcm.2012.37114
Wrongly Prescribed Half Tablets in a Swiss University Hospital
![]()
Pharmaceutical Care Research Group, Pharmaceutical Sciences, University of Basel, Basel, Switzerland.
Email: isabelle.arnet@unibas.ch
Received September 18th, 2012; revised October 24th, 2012; accepted November 25th, 2012
Keywords: Tablet Splitting; Scored Tablets; Score Lines; Subdivision of Tablets; Prescription Error; Medication Error
ABSTRACT
Background: Prescription of 1/2 tablets is a widespread practice, mainly to achieve dose flexibility and to facilitate swallowing. However, tablet splitting includes several disadvantages, like destruction of galenic formulation, stability problems, and unequal amount of active ingredient that may reduce effectiveness or result in a greater risk of toxicity. Objective: To assess the rate of wrongly prescribed 1/2 tablets in discharge prescriptions at the University Hospital in Basel (UHBS, 600 beds) and to evaluate its consequences for community pharmacists. Setting: Discharge prescriptions written between January 1st and December 31st 2011 and containing the term “1/2” were extracted from the electronic patients’ data management system of the UHBS. Presence of a score line and suitability for splitting were retrieved from two official sources of drug information. Main Outcome Measure: Wrong prescription was assigned for tablets with no score line or not suitable for dose splitting. Results: Of the 36,751 discharge prescriptions that were recorded in 2011 at the UHBS, 3724 (10.1%) contained at least one prescription item with the term “1/2”. The recipient patients were on average 72.9 ± 14.8 years old (median 76 years), 50.9% were women. Of the 4517 analysed items, 49% had a corresponding lower dosage strength available on the market, making splitting unnecessary. Rate of wrongly prescribed 1/2 tablets reached 16.4% (2.8% of all prescriptions) and concerned predominantly unscored tablets. When the lack of information on splitting suitability (5.6%) and on score lines (0.5%) was taken into account, the rate reached 22.4%. Half of all wrong prescriptions could be assigned to 14 different products that were prescribed with an overall rate between 3.1 and 0.2%. Quetiapine (Seroquel®) at all strengths was the most often wrongly prescribed tablet to split (3.1%; no score line), followed by atorvastatin (Sortis®) at all strengths (1.3%; no score line) and oxazepam (Seresta®) 15 mg (1.2%; with decorative score line). Conclusion: Prescribing of 1/2 tablets is common and concerns every 10th discharge prescriptions. It represents a pharmaceutical care issue, since in almost every second case, an identical drug with half the dosage strength is commercially available and a substitution could be offered by the community pharmacist. Further, one out of 5 prescribed 1/2 tablets is wrong or untraceable in the official sources of drug information and represents a safety issue. In all cases, time consuming and costly clarifications must be undertaken, ultimately the physician must be consulted, in order to modify the prescription or to dispense the prescribed 1/2 tablets as off-label use. If splitting is allowed, the patient’s cognitive and physical capacities have to be clarified and appropriate aids have to be offered, e.g. a pill splitter, in order to insure the safe use of the drug.
1. Introduction
Prescribing a drug requires from the physician a cognitive part with decision making (e.g., knowledge of diagnosis, interactions, contraindications), and a technical part with communication of essential information (e.g., drug name, form of administration, dose) [1]. Ordering to split a tablet in half belongs to the technical part and is a widespread practice. According to previously published studies, every fourth tablet is split before taken in ambulatory patients [2,3]. The main reasons for tablet splitting are dose adjustment, ease of swallowing and cost reduction [4-6]. However, tablet splitting also includes several drawbacks such as difficulty of breaking, breaking into unequal parts, and mass loss [6] which seems to be more pronounced with small round tablets [7]. Changing the way in which a dosage form is presented can further alter its characteristics. The degradation of the active substance at the fractured surface (inactivation by water, oxygen or light from the air; by acid from the stomach) may lead to loss of active substance before and after ingestion and consequently to altered absorption characteristics and failure to reach the site of action, which can be clinically relevant e.g., for medications with narrow therapeutic index [8]. Stability problems (friability, adsorption of water) and identification problems may complicate the keeping of the halves. Further, some dosage formulations are unsuitable for splitting, such as controlled release tablets, whose destruction can impair the liberation kinetics of the substance (dose dumping) and lead to dose-dependant side effects. One case has been reported in which a crushed extended-release nifedipine tablet had a fatal outcome [9]. Finally, irritant property or toxicity of the active substance, especially of CMR-substances (carcinogen, mutagen or toxic for reproduction) should be taken into account in order to protect the patient, his family members or healthcare workers and carers. Splitting of such tablets should be executed only with protective measures such as the wearing of gloves and masks [10].
Errors at the stage of ordering are the most common errors encountered in hospitals [11] and belong to medication errors [12]. Such errors affect 50% of hospital admissions and 7% of medication orders at any stage of the treatment process [13]. Although the literature lacks a universally accepted definition of a prescribing error [1,12,14], its most commonly reported type is error in dosage [13], predominantly by ordering the wrong dose [11]. Splitting a tablet that is not intended to be broken is not mentioned in different lists of prescribing errors [1,13,15], except in a recently published French classification of drug-related events (“Choice of an inadequate formulation: form not breakable or not crushable”) [16]. In any case, prescribing a 1/2 tablet that is not allowed to be split may result in reduced drug effectiveness and greater risk of toxicity, and represents a safety issue. Thus, we considered it as a prescribing error according to the most accepted definition of a medication error [1].
The University Hospital in Basel (UHBS) is a 600-bed teaching hospital in the northwest of Switzerland without an e-prescribing system, and serves the region’s 1 million residents.
The objectives of this retrospective data analysis was to assess the rate of prescribed half tablets by physicians at the UHBS at discharge, and to evaluate its consequences for community pharmacists, in view of optimizing seamless care.
2. Methods
Data were extracted from the ISMED database, the electronic patients’ data management system of the University Hospital Basel. Only discharge prescriptions with the term “1/2” and written between January 1st and December 31st 2011 were included. Patient characteristics (age, sex, and ward) stored in the system were also retrieved. This was a retrospective data-based study.
Unnecessary splitting was defined if a commercially available tablet with identical active ingredient had the required strength and pharmaceutical form, or if the prescribed strength could be obtained with a multiple of the tablet with a lower strength.
Of the retrieved drugs, presence of a score line and suitability for splitting were extracted from two sources of drug information, i.e. the Swiss Online Compendium that compiles the Summary of Products Characteristics (SPC) [17] and the internal pharmacy hospital list of the UHBS on splitting and crushing drugs [18]. Tablets were classified as with a score line or not, and suitable for dose splitting or not. If a generic drug name was equivocal, the tablet was classified as having a score line when at least one generic brand with a score line was commercially available. A generic tablet was classified as suitable for dose splitting if dose splitting was allowed for at least one generic brand. Splitting suitability was deduced if a 1/2 tablet was a recommended dosage. Wrong prescription was allocated to drugs that were not suitable for dose splitting, i.e. if a tablet was unscored [19] or if a corresponding ban was found in the SPC (“tablets must not be split” or “tablets are not suitable for splitting” or “the score line is only to facilitate breaking for ease of swallowing and not to divide into equal doses”).
Descriptive statistics were performed, with results given as crude value, mean ± standard deviation or percentage.
3. Results
Of the 36,751 electronic discharge prescriptions that were recorded 2011 at the University Hospital Basel, 3724 (10.1%) contained at least one prescription item with the term “1/2”. They were predominantly delivered by the Internal Medicine (3.2%) and the Emergency wards (1.9%) and represented 34,307 items (average of 9.2 items per prescription). The recipient patients were on average 72.9 ± 14.8 years old (median 76 years), 50.9% were women.
Small typing errors were corrected when the identification of the product was unequivocal (Lamotrin for Lamotrigin; Cosaar 50 mh for Cosaar 50 mg; Beoc zok for Beloc Zok). Minor errors were corrected when the intention regarding the final product was unequivocal e.g., when the strength was incomplete (Co-Diovan 160 mg for Co-Diovan 160/12.5 mg) or wrong (Dipiperon 30 mg tablets for Dipiperon 40 mg tablets); when the galenic form was missing (Madopar 125 mg for Madopar 125 mg tablets) or wrong (Dipiperon 40 mg capsules for Dipiperon 40 mg tablets).
Of the 4888 retrieved single items with “1/2”, 371 were excluded because the prescription was incomplete (Table 1). The analysis of the divisibility included 4517 tablets. For 2215 of them (49%), the prescribed splitting

Table 1. Reasons for exclusion of the retrieved 4888 single items containing “1/2” (n = 371; 7.6%).
was unnecessary, since the prescribed dose was available as a marketed tablet with half strength. The majority of the prescribed 1/2 tablets were scored (87%) and were suitable for dose splitting (77.6%). Wrong prescription of 1/2 tablets reached 16.4% and was due to inexistent score lines (12.6%) or the presence of a clear ban in the sources of drug information (3.8%) (Figure 1). No information was found on splitting suitability and on score lines for 5.6% and 0.5% of the prescribed 1/2 tablets, respectively, increasing the rate of wrong splitting prescription to 22.4%.
Half of all wrong prescriptions (506/1014; 49.9%) concerned 14 different products that were prescribed with an overall rate between 3.1% - 0.2%. The most frequent wrong prescriptions concerned quetiapine at all strengths (3.1%; Seroquel® 25, 50, 100, 200, 300 and 400 mg), an antipsychotic drug as coated tablet with no score line; atorvastatin at all strengths (1.3%; Sortis® 10, 20, 40 and 80 mg), a lipid altering agent as coated tablet with no score line; and oxazepam (1.2%; Seresta® 15 mg), a tablet with a decorative score line.
4. Discussion
In our study, approximately 10% of all discharged prescriptions contained the order to split a tablet, which is close to the 12.5% of drugs prescribed in split form in a German 1680-bed university hospital [20]. Several studies investigated prescribing errors in hospitals and reported various rates from 0.4 [21] to 34 per 100 orders [22] somewhere in the medication process. However, since the definition of what constitutes an error varies widely between studies, interpretation of data is difficult. Errors in discharge prescriptions are frequently described in literature, unfortunately often with few specification of the type of errors [23]. Incomplete prescriptions accounted for 6% in discharged elderly patients in Spain [24], while wrong dosage schedules and missing information were the most common types of the 5.8% prescribing errors observed for discharged patients in a US university teaching hospital [25]. Other authors found 5% omissions or inaccurate information, mostly incorrect dosing forms e.g., nonexistent tablet strengths, in discharge prescription of a US children hospital [26]. These results are in line with our findings of missing strengths (2.6%) and nonexistent strengths (1.4%) as the most frequently encountered missing information on discharge prescriptions.
In our study, prescribing to split a tablet with no score line was the most frequently observed error, and concerned every eighth prescription of a 1/2 tablet (12.6%). There is almost no literature available about the rate of wrong tablet splitting within the medication process in hospitals. A study in Pakistan assessed the prescribing practice of physicians in two teaching hospitals [27]. Of 1218 medication orders, 6.8% required to split a tablet that concerned a modified release form in more than half of the cases. The authors concluded on a prescription rate of wrong 1/2 tablets of 3.5%, which however may be underestimated since the offending errors may have involved other situations than the splitting of modified release form.
Errors in discharge prescriptions are mostly detected by community pharmacists whose core competency is to screen the prescription orders and to correct potential medication errors [28]. Wrong prescription of 1/2 tablets may not cause significant patient harm, since for many drugs, especially those with a wide therapeutic range and a long half-life, dose fluctuations are unlikely to be clinically significant. However, the rate of 2.8% errors of all discharge prescriptions observed in our study represents a major issue, since it may result in much unnecessary work, as pharmacists need to resolve the uncertainty about the prescription. As an trivial example of our study, 1/2 trazodone 150 mg tablet (Trittico®, extended released) was prescribed, however the oval tablets have 2 score lines and can be divided into 3 parts of 50 mg each, leaving the pharmacist probably perplexed. Health professionals need precise information about the divisibility or further characteristics of the drug before prescribing and dispensing the medication. Our study showed that this information was missing in the SPCs, the legal prescribing information for health professionals, for 6.1% of all prescribed 1/2 tablets, leaving the pharmacists with a
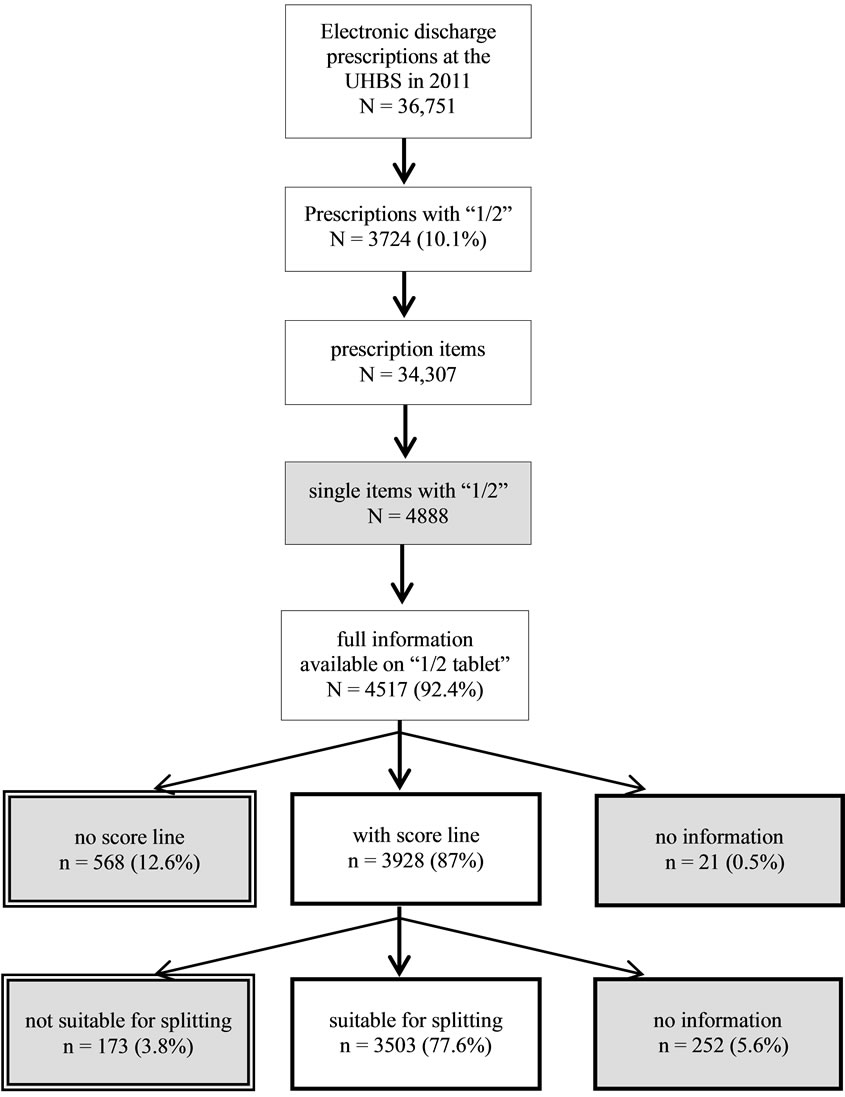
Figure 1. Flow chart of the electronic discharge prescriptions recorded between January 1st and December 31st 2011 at the University Hospital Basel (UHBS) and containing the term “1/2 tablet”. Wrongly prescribed 1/2 tablets concerned tablets without score lines and tablets with a clear splitting ban in the sources of drug information (grey boxes with double line border). If the absence of information on score lines and on splitting suitability in the sources of drug information was taken into account (grey boxes with simple line border), wrongly prescribed splitting of tablets reached 22.4%.
crucial lack of information [29]. Ultimately, pharmacists may need to clarify the prescriber’s intention by contacting him, usually by phone. Each pharmacist’s action may delay drug delivery and hence drug administration. Further, effective instruction of the patients by the pharmacy is a prerequisite to minimise confusion and intake errors, especially when patients received information at the time of discharge that diverges from the finally dispensed medication. All processes may influence drug safety, take time and generate costs that may exceed the potential cost savings that physicians had advocated for splitting tablets [30]. For countries like Switzerland, where medication is dispensed in the original treatment pack, maximum savings from tablet splitting could theoretically be obtained for products that have similar prices for each of the dosages. However, only few drugs have this so called “flat pricing” structure e.g., generics of atorvastatin or simvastatin. The potential of pill splitting to achieve cost savings was evaluated in a retrospective analysis of US pharmacy claims [31]. Among the 11 medications identified by the authors for which pill splitting was cost saving and clinically appropriate, three matched our results (atorvastatin, lisinopril, olanzapine). Unfortunately, all three products are formulated as unscored tablets in Switzerland.
Uneven breaking of a tablet is most probable with unscored tablets, and may result in fluctuations in the administered dose which can be harmful to the patient, especially for medications with a narrow therapeutic index e.g., carbamazepine, digoxin, phenytoin, sodium valproate, theophylline [32]. In our study, two products may put the patient at a higher risk of side effects when halved. Firstly, Digoxin® 0.25 mg (0.22% of the cases), a substance with a narrow therapeutic index, formulated as a round and small tablet, scored on one side only (what contributes to increased inaccuracy of tablet splitting), and without indication on secability in the SPC. We asked the manufacturer about the divisibility of Digoxin®. The tablets might be split to facilitate intake, but not to administrate a half dose, which can be achieved with the lower existing strength with 0.125 mg. Secondly, MST® Continus (0.11% of the cases), that contains morphine in an extended release preparation (dispersion of the substance within a soluble matrix that is slowly eroding), is formulated as tablets with no score line and has a clear ban of splitting in the SPC. For both situations, we suppose oversight or ignorance of the prescriber.
We found quetiapine to be the drug most often wrongly prescribed as half tablet. This atypical antipsychotic agent is approved for treating schizophrenia and bipolar disorders. Quetiapine is also commonly used for off-label conditions such as psychosis, agitation, dementia, anxiety, obsessive-compulsive disorders [33] and delirium [34], however with limited evidence. A recent meta-analysis justified the additional use of quetiapine as evidence-based only in the treatment of generalized anxiety disorder [33]. Most of the small clinical studies used increasing doses, starting at 25 mg/day [35-37]. We could only find one Italian study with quetiapine 12.5 mg to initiate treatment in 41 patients with dementia and concomitant psychotic disorders [38]. According to a clinical pharmacist at the UHBS, quetiapine is used offlabel in case of agitation in geriatric patients, in flexible dosing schedules starting at 5 mg/day, with specially produced capsules of 5 mg strength [personal communication, Hospital Pharmacy 2012-10-8]. Further, a dose of 12.5 mg is administered at the UHBS in the prevention of delirium and is given as 1/2 tablet of 25 mg. However, it is evaluated as inappropriate prescription in the discharge medications by the clinical pharmacists and stopped when intercepted. One may argue, since quetiapine has a broad therapeutic range, and the formulation is without functional coating or modified release, that breaking the tablet into uneven parts is unlikely to lead to toxicity. Nevertheless, the potential problems inherent to splitting a tablet (good dexterity and eyesight, conservation and confusion of the halves, wastage, adherence) and the missing indication do not justify the prescription of 1/2 quetiapine 25 mg tablet. If the physician prescribes deliberately 1/2 tablet in the setting of off-label prescribing, this could be expressly marked e.g., with a specific term on the prescription like “sic” in Switerzland or “@” in The Netherlands, as it is already common practice for the prescription of original products that should not be substituted by pharmacists.
Since therapeutic substitution is more complex than generic substitution, possibilities to by-pass the prescription of a split tablet are limited to drugs with identical active ingredient, identical pharmaceutical form and required strength. Nevertheless, we were able to find an alternative for 49% of the prescribed 1/2 tablets, which is similar to the 46% found by others [3]. Considering that the information on suitability of tablet splitting is available in the usual sources of information, computerised prescribing would instantly eliminate approximately half of the wrong prescriptions. Indeed, the introduction of computerised physician order entry (CPOE) containing splitting information in a German university hospital was able to halve the proportion of wrongly split drugs, mainly by intercepting undue fragmentation of unscored tablets [20]. Thus, electronically prescribing systems might be able to support physicians in reducing unintentional wrong prescription of tablet splitting.
A number of potential limitations of our study must be considered. First, we retrieved only bisection of tablets with the term “1/2”, and excluded “0.5” as well as other section like “1/4”, allowing to suppose that the rate of wrong prescription for split tablets is even higher than our findings. Further, the study was conducted in a single hospital and therefore results may not be extrapolated to other hospitals or cultures. However, our results are in accordance with former studies.
In conclusion, tablet splitting has a major role in dosage adjustment and should be limited to specific clinical situation e.g., titration of dose, paediatric and geriatric patients, and according to the recommendation of the product manufacturer. It should be cautiously ordered for drugs with narrow therapeutic range. Physicians who wrongly prescribe to split a tablet and pharmacists who dispense the drug accordingly should be aware that this renders the medication unlicensed. Since resolving the uncertainty about the prescription by the pharmacists results in much unnecessary work, splitting tablet is not suited as a method of general cost reduction. Taking into account all problems linked to the handling of a half tablet (patients’ dexterity and eyesight, conservation and confusion of the halves, therapeutic compliance), prescribing 1/2 tablet represents a safety issue. Pharmaceutical companies should be encouraged to introduce new strengths to an existing range of products.
5. Acknowledgements
The authors wish to thank Cindy Aeschlimann for collecting the data. The authors report no conflict of interest and confirm no plagiarism. They are responsible for the content and writing of the paper. The Pharmaceutical Care Research Group, University of Basel, funded this study.
REFERENCES
- B. Dean, N. Barber and M. Schachter, “What Is a Prescribing Error?” Quality in Health Care, Vol. 9, 2000, pp. 232-237. doi:10.1136/qhc.9.4.232
- R. Quinzler, C. Gasse, A. Schneider, P. Kaufmann-Kolle, J. Szecsenyi and W. Haefeli, “The Frequency of Inappropriate Tablet Splitting in Primary Care,” European Journal of Clinical Pharmacology, Vol. 62, No. 12, 2006, pp. 1065-1073. doi:10.1007/s00228-006-0202-3
- N. Rodenhuis, P. De Smet and D. Barends, “The Rationale of Scored Tablets as Dosage Form,” European Journal of Pharmaceutical Sciences, Vol. 21, No. 2-3, 2004, pp. 305-308. doi:10.1016/j.ejps.2003.10.018
- R. Quinzler and W. Haefeli, “Tabletten Teilen,” Therapeutische Umschau, Vol. 63, 2006, pp. 441-447. doi:10.1024/0040-5930.63.6.441
- R. Quinzler, W. Haefeli, “Zerkleinern von Tabletten,” TMJ, 2008, pp. 44-47.
- E. van Santen, D. Barends and H. Frijlink, “Breaking of Scored Tablets: A Review,” European Journal of Pharmaceutics and Biopharmaceutics, Vol. 53, No. 2, 2002, pp. 139-145. doi:10.1016/S0939-6411(01)00228-4
- P. Gupta and K. Gupta, “Broken Tablets: Does the Sum of the Parts Equal the Whole?” American Journal of Health-System Pharmacy, Vol. 45, No. 7, 1988, p. 1498.
- Australian Pharmaceutical Advisory Council, “Guidelines for Medication Management in Residential Aged Care Facilities,” 3rd Edition, Commonwealth of Australia, 2002.
- J. G. Schier, M. A. Howland, R. S. Hoffman and L. S. Nelson, “Fatality from Administration of Labetalol and Crushed Extended-Release Nifedipine,” Annals of Pharmacotherapy, Vol. 37, No. 10, 2003, pp. 1420-1423. doi:10.1345/aph.1D091
- “Summary of Product Information of Puri-Nethol® Tablets 50 mg,” 2012. www.compendium.ch
- D. Bates, D. J. Cullen, N. Laird, L. Petersen, S. Small, D. Servi, G. Laffel, B. Sweitzer, B. Shea and R. Hallisey, “Incidence of Adverse Drug Events and Potential Adverse Drug Events: Implications for Prevention,” Journal of American Medical Association, Vol. 274, No. 1, 1995, pp. 29-34. doi:10.1001/jama.274.1.29
- J. K. Aronson, “Medication Errors: Definitions and Classification,” British Journal of Clinical Pharmacology, Vol. 67, No. 6, 2009, pp. 599-604. doi:10.1111/j.1365-2125.2009.03415.x
- P. J. Lewis, T. Dornan, D. Taylor, M. P. Tully, V. Wass and D. M. Ashcroft, “Prevalence, Incidence and Nature of Prescribing Errors in Hospital Inpatients: A Systematic Review,” Drug Safety, Vol. 32, 2009, pp. 379-389. doi:10.2165/00002018-200932050-00002
- American Society of Health-System Pharmacists, “ASHP Standard Definition of a Medication Error,” American Journal of Hospital Pharmacy, Vol. 39, No. 2, 1982, p. 321.
- T. Lesar, L. Briceland and D. Stein, “Factors Related to Errors in Medication Prescribing,” Journal of the American Medical Association, Vol. 277, 1997, pp. 312-317. doi:10.1001/jama.277.4.312
- E. Arques-Armoiry, D. Cabelguenne, C. Stamm, A. Janoly-Dumenil, I. Grosset-Grange, N. Vantard, P. Maire and B. Charpiat, “Most Frequent Drug-Related Events Detected by Pharmacists during Prescription Analysis in a University Hospital,” La Revue de Médecine Interne, Vol. 31, No. 12, 2010, pp. 804-811. doi:10.1016/j.revmed.2010.08.001
- Arzneimittelkompendium der Schweiz Online, 2012. www.compendium.ch
- D. Bornand, “Zermörserbarkeit und Verabreichungshinweise von Tabletten,” 2008. www.spitalpharmazie-basel.ch/dienstleistungen/pdf/Zermoerserbarkeit_Tabletten.pdf
- Anonymous, “Teilbarkeit von Tabletten und Filmtabletten,” Swissmedic, Merkblatt, 2004.
- R. Quinzler, S. Schmitt, M. Pritsch, J. Kaltschmidt and W. E. Haefeli, “Substantial Reduction of Inappropriate Tablet Splitting with Computerised Decision Support: A Prospective Intervention Study Assessing Potential Benefit and Harm,” BMC Medical Informatics and Decision Making, Vol. 9, 2009, p. 30.
- T. Lesar, L. L. Briceland, K. Delcoure, J. Parmalee, V. Masta-Gornic and H. Pohl, “Medication Prescribing Errors in a Teaching Hospital,” Journal of the American Medical Association, Vol. 263, 1990, pp. 2329-2334. doi:10.1001/jama.263.17.2329
- N. Drewett, “Stop Regular Medicines Error,” Journal of Pharmacy Practice, Vol. 8, 1998, pp. 193-196.
- S. Dobrzanski, I. Hammond, G. Khan and H. Holdsworth, “The Nature of Hospital Prescribing Errors,” British Journal of Clinical Governance, Vol. 7, No. 3, 2002, pp. 187-193. doi:10.1108/14664100210438271
- S. O. Delgado, P. J. Nicolás, L. I. Martínez, F. A. Serrano, J. L. Anoz and C. F. Fernández, “Reconciliation Errors at Admission and Departure in Old and Polymedicated Patients. Prospective, Multicenter Randomized Study,” Medicina Clínica, Vol. 133, 2009, pp. 741-744.
- G. Schumock, A. Guenette, T. Keys and R. Hutchinson, “Prescribing Errors for Patients about to Be Discharged from a University Teaching Hospital,” American Journal of Hospital Pharmacy, Vol. 51, No. 18, 1994, pp. 2288-2290.
- K. B. Johnson, J. K. Butta, P. K. Donohue, D. J. Glenn and N. A. Holtzman, “Discharging Patients with Prescriptions Instead of Medications: Sequelae in a Teaching Hospital,” Pediatrics, Vol. 97, No. 4, 1996, pp. 481-485.
- R. Shawahna and Nisar-Ur-Rahman, “Prescribing Errors Involved Splitting of Tablets: A Two-Hospital Case Analysis,” Pakistan Journal of Medical and Health Sciences, Vol. 2, 2008, pp. 54-58.
- M. Rupp, M. DeYoung and S. Schondelmeyer, “Prescribing Problems and Pharmacist Interventions in Community Practice,” Medical Care, Vol. 30, No. 10, 1992, pp. 926-940. doi:10.1097/00005650-199210000-00005
- I. Arnet and K. Hersberger, “Misleading Score-Lines on Tablets: Facilitated Intake or Fractional Dosing?” Swiss Med Weekly, Vol. 140, 2010, pp. 105-110.
- J. Bachynsky, C. Wiens and K. Melnychuk, “The Practice of Splitting Tablets: Cost and Therapeutic Aspects,” PharmacoEconomics, Vol. 20, 2002, pp. 339-346. doi:10.2165/00019053-200220050-00005
- R. S. Stafford and D. C. Radley, “The Potential of Pill Splitting to Achieve Cost Savings,” American Journal of Managed Care, Vol. 8, 2002, pp. 706-712.
- Royal Pharmaceutical Society, “Guidance on the Pharmaceutical Issues When Crushing, Opening or Splitting Oral Dosage Forms,” 2011. www.rpharms.com/pharmacy-practice-resource/specials.asp
- A. Maher, M. Maglione, S. Bagley, M. Suttorp, J. Hu, B. Ewing, Z. Wang, M. Timmer, D. Sultzer and P. Shekelle, “Efficacy and Comparative Effectiveness of Atypical Antipsychotic Medications for Off-Label Uses in Adults: A Systematic Review and Meta-Analysis,” Journal of the American Medical Association, Vol. 306, 2011, pp. 1359-1369. doi:10.1001/jama.2011.1360
- J. Devlin, R. Roberts, J. Fong, Y. Skrobik, R. Riker, N. Hill, T. Robbins and E. Garpestad, “Efficacy and Safety of Quetiapine in Critically Ill Patients with Delirium: A Prospective, Multicenter, Randomized, Double-Blind, Placebo-Controlled Pilot Study,” Critical Care Medicine, Vol. 38, No. 2, 2010, pp. 419-427. doi:10.1097/CCM.0b013e3181b9e302
- B. Maneeton, N. Maneeton and M. Srisurapanont, “An Open-Label Study of Quetiapine for Delirium,” Journal of the Medical Association of Thailand, Vol. 90, 2007, pp. 2158-2163.
- C.-U. Pae, S.-J. Lee, C.-U. Lee, C. Lee and I.-H. Paik, “A Pilot Trial of Quetiapine for the Treatment of Patients with Delirium,” Human Psychopharmacology, Vol. 19, No. 2, 2004, pp. 125-127. doi:10.1002/hup.559
- Y. Sasaki, T. Matsuyama, S. Inoue, T. Sunami, T. Inoue, K. Denda and T. Koyama, “A Prospective, Open-Label, Flexible-Dose Study of Quetiapine in the Treatment of Delirium,” Journal of Clinical Psychiatry, Vol. 64, 2003, pp. 1316-1321. doi:10.4088/JCP.v64n1106
- M. Onor, M. Saina and E. Aguglia, “Efficacy and Tolerability of Quetiapine in the Treatment of Behavioral and Psychological Symptoms of Dementia,” American Journal of Alzheimer’s Disease & Other Dementias, Vol. 21, No. 6, 2007, pp. 448-453. doi:10.1177/1533317506294775

