International Journal of Clinical Medicine
Vol.3 No.2(2012), Article ID:17886,6 pages DOI:10.4236/ijcm.2012.32029
Tako-Tsubo Cardiomyopathy—The Role of Physical and Emotional Stressors: Two Case Report
![]()
Internal Medicine Ward, Coimbra University Hospital, Coimbra, Portugal.
Email: *pms.carvalho@gmail.com
Received December 16th, 2011; revised January 24th, 2012; accepted February 5th, 2012
Keywords: Tako-Tsubo; Left Ventricle; Postmenopausal; Stress
ABSTRACT
Introduction: Tako-Tsubo cardiomyopathy describes a form of acute and reversible left ventricular dysfunction with a clinical presentation, ECG and cardiac biomarkers that makes it indistinguishable from acute coronary syndrome. Case Presentation: The authors present two cases of tako-tsubo cardiomyopathy in postmenopausal women, the first case following an emotional stressful event and a second case following a blood transfusion and probably associated with intravenous catecholamine perfusion. Both had unobstructed coronary arteries and regional wall-motion abnormalities typical of this syndrome. Conclusions: Tako-tsubo cardiomyopathy is a condition often misdiagnosed. A clinical presentation suggestive of acute coronary syndrome in a postmenopausal woman without history of coronary disease and in whom a precipitating stressful event can be found should lead the physician to suspect the diagnosis of tako-tsubo cardiomyopathy. This syndrome associated with blood transfusion has rarely been described.
1. Introduction
Tako-Tsubo cardiomyopathy (TC), also known as “transient left ventricular apical ballooning syndrome”, “broken heart syndrome” or “stress induced cardiomyopathy”, was first described under this terminology in 1990 in the Japanese population, owing its name to the resemblance between the Japanese octopus trap and the shape of the left ventricle (LV) during systole [1].
This condition describes a form of acute, often severe and reversible LV dysfunction, with a clinical presentation indistinguishable from an acute coronary syndrome, electrocardiographic changes that mimic ST segment elevation myocardial infarction (STEMI) and minimal increases in the cardiac enzymes, incongruent with the degree of LV dysfunction, in the absence of coronary lesions visible in coronary angiography [2,3].
It is well recognized that TC is frequently precipitated by a stressful emotional or physical event [1,3-5], affects mainly postmenopausal women (90% of the cases) and accounts for about 1% - 2% of cases presenting as STEMI [1-3].
The exact cause of this cardiomyopathy is unknown, but several hypotheses have been proposed: catecholamine excess and sympathetic nervous system hyperactivity, transient epicardial coronary spasm, transient coronary artery occlusion/reperfusion episodes, coronary microvascular spasm, oxidative stress in response to catecholamine excess or relative deficiency of oestrogen [2,3].
The presence of regional wall-motion abnormalities, with hypokinesis, akinesis or dyskinesis of the mid ventricular and apical segments and hyperkinesis of the basal left ventricular segments, found on transthoracic echocardiogram or left ventriculogram can suggest the diagnosis [3,6].
2. Case Presentation
2.1. Case 1
A 62-year-old white woman was admitted to the emergency department with sudden onset of chest pain and dyspnea, immediately after she had witnessed her husband’s death.
From her past history, the only remarkable event was a transient ischemic attack 6 years prior; she suffered from high blood pressure and depression and was under therapy with lisinopril 20 mg, amlodipine 5 mg, indapamide 2.5 mg, acetylsalicylic acid 150 mg, dipyridamole 75 mg, trimetazidine 35 mg, amitriptyline 10 mg, alprazolam 0.25 mg, paroxetine 20 mg and simvastatin 20 mg.
At presentation she was hemodynamically stable (blood pressure—BP—130/70 mmHg, heart rate 100 bpm, oxygen saturation of 92%, with oxygen delivery 24%), her cardiac auscultation was unremarkable and her pulmonary auscultation revealed rales in the lower third bilaterally.
The blood analysis showed elevated troponin I (0.34 ng/ml, normal range < 0.2 ng/ml) and myoglobin (214 ng/ml, normal range between 9 - 82 ng/ml); the creatinine kinase (CK) and CK-MB isoenzyme were only slightly above normal range (167 U/L and 4 ng/ml, normal range < 145 U/L and 3.6 ng/ml, respectively).
Her chest X-ray revealed an enlarged cardiothoracic index, with bilateral basal infiltrates, suggestive of interstitial edema (Figure 1 (a)).
The ECG showed sinus rhythm, 101 bpm, normal QRS axis, with inferior ST depression and lateral ST elevation, inferior to 1 mm. (Figure 2(a)).
She developed hypotension (BP 70/45 mmHg), with good response to volume. The ECG was repeated and pointed out to anterior and lateral ST elevation (still inferior to 1 mm), inferior ST depression and T Wave inversion, changes more prominent than before (Figure 2(b)).
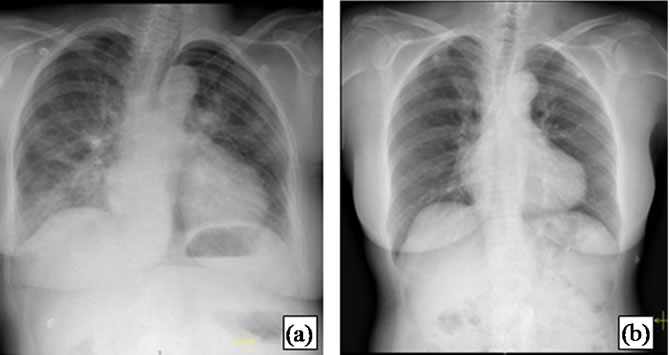
Figure 1. First case report X-ray. (a) At admission, the X-ray revealed an enlarged cardiothoracic index, with bilateral infiltrates, suggestive of intersticial edema; (b) X-ray repeated during the second admission showing improvement, with a smaller cardiothoracic index and without infiltrates.
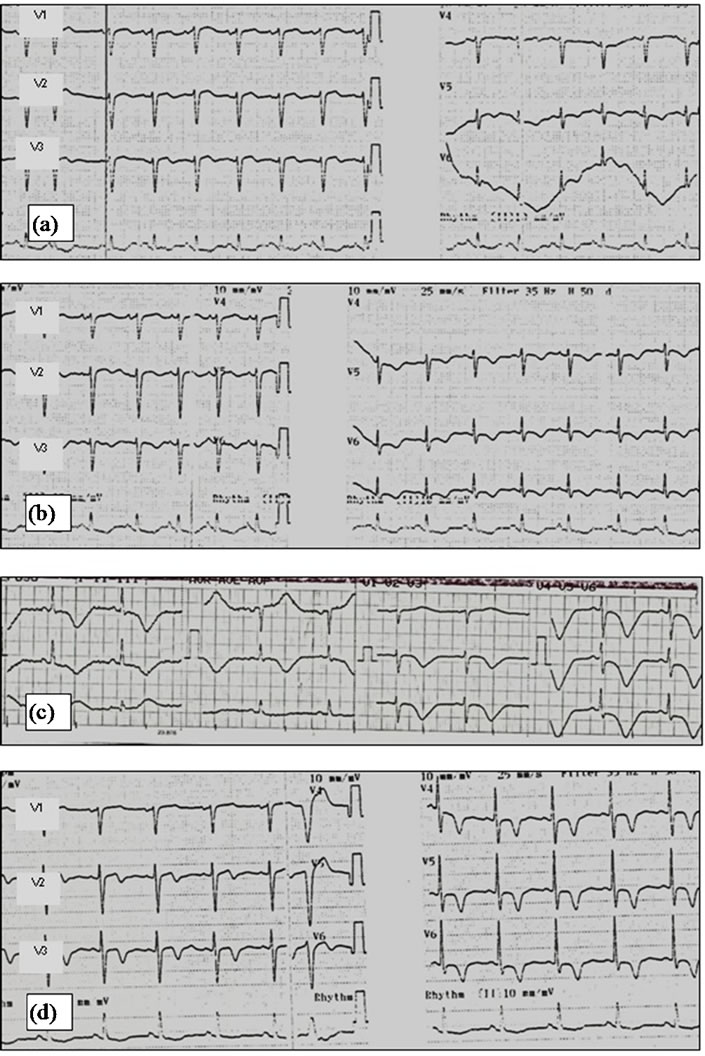
Figure 2. First case report serial ECGs. (a) ECG at admission, showing sinus rhythm, 101 bpm, normal QRS axis, with inferior ST depression and lateral ST elevation; (b) Later, there were evolutive changes in the ECG, with more prominent ST elevation and T wave inversion; (c) ECG three days later showing diffuse and deep T wave inversion and prolonged QT (0.6 s, with heart rate 60 bpm); (d) After five weeks, the patient maintained T wave inversion.
A transthoracic echocardiogram was performed, showing mid ventricular and apical hypokinesis with apical ballooning, and preserved contraction of the basal segments.
At this time TC was suspected. The patient underwent cardiac catheterization with left ventriculography, which demonstrated unobstructed coronary arteries (Figure 3), with spiral morphology, and a normal dimensioned LV, with antero-lateral, apical and infero-apical akinesis, with global function preserved (ejection fraction—EF—52%) (Figure 4).
She was then admitted to the coronary intensive care unit. The peak troponin I concentration was 1.88 ng/ml, with rapid descending profile. The ECG repeated three days later pointed out diffuse T wave inversion, and prolonged QT (0.6 sec, heart rate 60 bpm) (Figure 2(c)). She was discharged after five days under therapy with acetylsalicylic acid, clopidogrel and atorvastatin. Four weeks later she was observed at a follow up appointment; she had abandoned all therapeutics at this point.
She was readmitted to the emergency department five weeks later with headache, chest pain and dysarthric; the
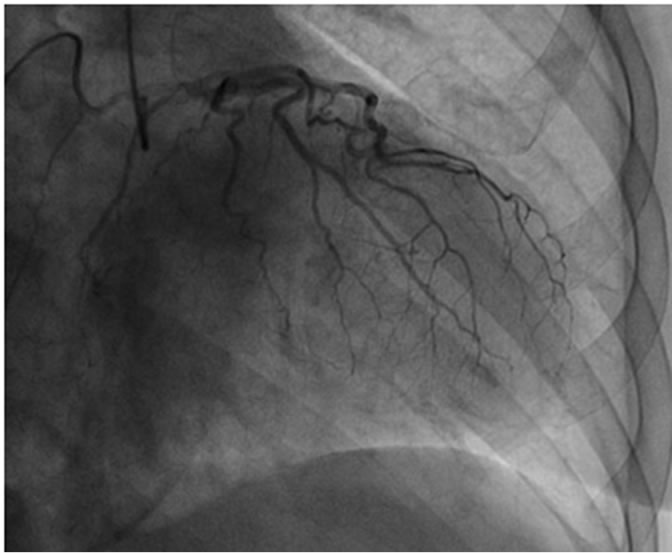
Figure 3. Coronary angiography (first case). Unobstructed coronary arteries with spiral morphology.
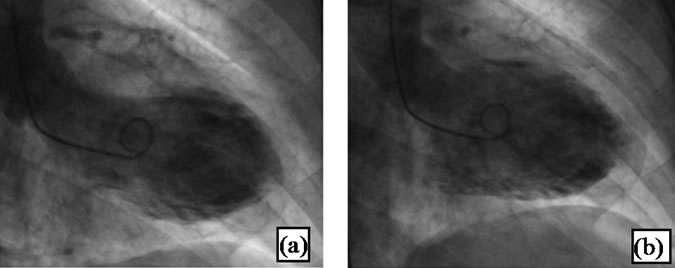
Figure 4. Left ventriculography. (a) During systole showing no contraction in the apical region, with apical ballooning; (b) During diastole there was no difference in the apical shape in comparison to systole.
neurological exam and head CT excluded a cerebrovascular event; serial cardiac enzymes determination were negative; the ECG showed sinus rhythm, 80 bpm, with antero-lateral T wave inversion (Figure 2(d)); the chest X-ray no longer showed bilateral infiltrates and the cardiothoracic index was smaller (Figure 1(b)). The echocardiogram was repeated, showing complete resolution of the hypokinesis observed before. Her complaints were attributed to anxiety.
TC was confirmed.
2.2. Case 2
A 84-year-old white woman presented to the emergency department with dyspnea, diaphoresis and pallor, without chest pain, beginning a few hours after she had been transfused with two units of packed red blood cells.
Her medical history included cerebrovascular disease, stroke with left hemiparesis, congestive heart failure and chronic anemia. She was under therapy with triflusal 300 mg, amiodarone 200 mg, enalapril 5 mg, iron polymaltose complex, sertraline 50 mg, mirtazapine 15 mg, lorazepam 1 mg, omeprazole 20 mg and multivitamin complex.
On physical exam, she was normotensive, apyretic, with a mitral systolic murmur (II/VI) and bilateral rales.
Her cardiac enzymes were increased: troponin I of 1.37 ng/ml, myoglobin of 220 ng/ml and CK MB of 6.8 ng/ml; her creatinine level was also raised (2.18 mg/dl, with normal range between 0.55 and 1.02 mg/dl). The peak troponin I concentration was 1.94 ng/ml, five hours after admission.
The ECG revealed sinus rhythm, 87 bpm, with nonspecific lateral ST changes (Figure 5(a)). Nine hours after admission, she became hypotensive (BP 67/47 mmHg), with partial response to volume. Intravenous administration of dopamine was started at 5 mg/Kg/min and she was admitted to the internal medicine ward.
After thirty six hours she maintained hypotension, despite dopamine perfusion at 10 mg/Kg/min. The ECG revealed diffuse ST segment elevation (Figure 5(b)) and cardiac catheterization was performed, revealing unobstructed coronary arteries (Figure 6). The next day, she underwent transthoracic echocardiogram, showing hypokinesis of the mid-apical segments of the lateral wall and apical segment of the remaining walls, with hyperkinesis of the basal segments, with ejection fraction of 50%; a left intraventricular pressure gradient wasn’t found; there was moderate mitral valve regurgitation and severe tricuspid regurgitation present. The ECG was repeated, showing sinus rhythm, 81 bpm, with non-specific antero-lateral ST changes and prolonged QT (0.6 sec, heart rate 81 bpm) (Figure 5(c)).
TC was suspected.
Dopamine was discontinued. As she presented refractory
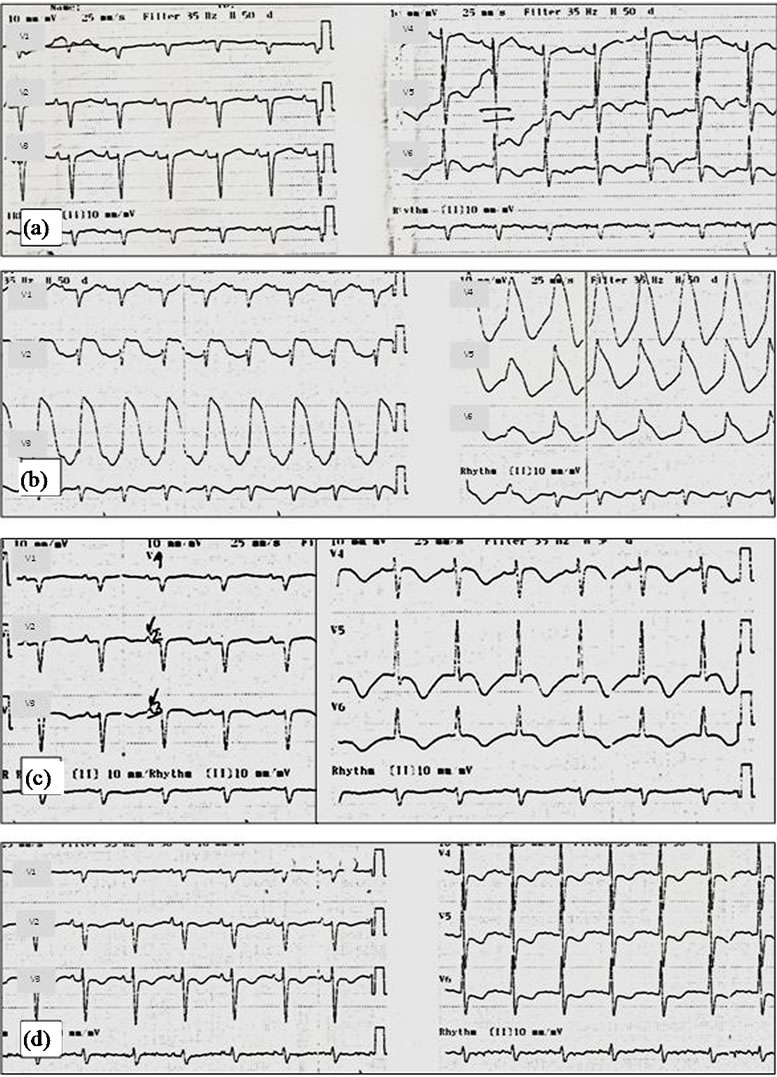
Figure 5. Second case report serial ECGs. (a) ECG at admission showing non-specific lateral ST changes; (b) Thirty-six hours later, the ECG showed diffuse ST segment elevation; (c) Twenty-four hours later, the ST segment elevation had resolved; there was prolonged QT; (d) After 17 days, the patient maintained T wave inversion.
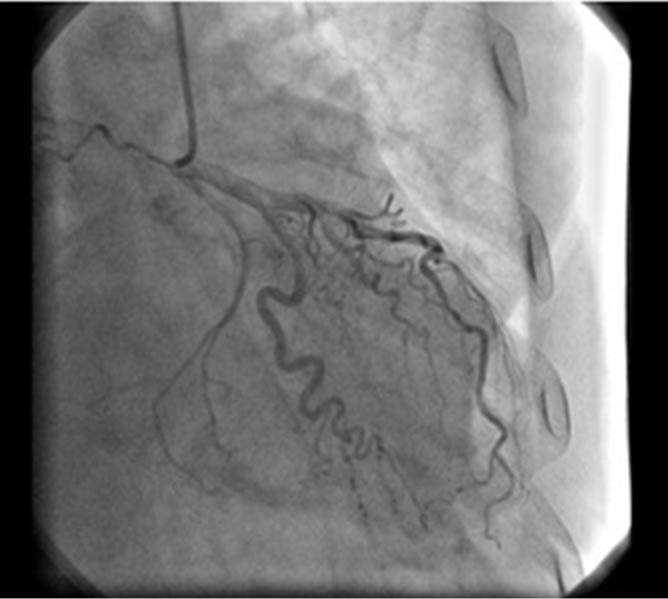
Figure 6. Coronary angiography (second case). Unobstructed coronary arteries.
hypotension in the context of cardiogenic shock, intravenous perfusion of dobutamine was initiated, with good response.
She was discharged after six days under therapy with triflusal and atorvastatin, and readmitted two days after with health care associated pneumonia; she had no complains of chest pain during the latter. The ECG repeated after 17 days still showed T wave inversion (Figure 5(d)). A control echocardiogram showed complete resolution of the regional wall motion abnormalities. TC was confirmed.
3. Discussion
The majority of patients with TC present with chest pain and dyspnea; some cases of syncope and ventricular arrhythmias have been described [3,4,6]. Although usually hemodinamically stable, some patients can develop congestive heart failure; hypotension may occur duo to reduced stroke volume or dynamic left ventricular outflow tract obstruction [3,6]. Cardiogenic shock, albeit rare, may occur.
Our first case has a typical clinical presentation, precipitated by an emotional stressful event, known to be associated to TC in other cases previously described on literature [5,7]. She had prior history of depression and some studies suggest that, besides the important role that psychiatric disorders may have in coronary artery disease, they may also contribute to the cardiac syndrome during acute stress in patients with TC [8].
Our second case, an elderly woman with valvular heart disease, presented with dyspnea without chest pain, and hypotension in the context of cardiogenic shock, following a blood transfusion; Wever-Pinzon et al. describe a similar case [9]. The administration of dopamine in our patient may have had a role in perpetuating hypotension, although an intraventricular pressure gradient wasn’t detected on echocardiogram. The administration of catecholamine in patients with TC may cause further hemodynamic deterioration in the presence of high intraventricular pressure gradient [6,10]; besides, catecholamine are thought to be involved in the pathogenesis of the syndrome and should be avoided in these patients [1,2,7].
The most frequent electrocardiographic abnormalities in TC are ST segment elevation in the precordial leads, usually with evolutionary changes in a matter of two to three days: resolution of the ST segment elevation with subsequent development of diffuse and deep T wave inversion in most leads, the appearance of new pathologic Q waves, and prolongation of the corrected QT interval. The T wave inversion may persist beyond the complete resolution of the LV dysfunction [2-4,6]. In both our patients some of this evolutionary changes were observed, namely the appearance of T wave inversion and prolongation of the QT interval; in the first case the T wave inversion persisted despite the normalization of the regional wall-motion abnormalities noticed after five weeks; in the second case, the ST segment elevation resolved in a matter of twenty four hours; T wave inversion persisted after 17 days.
The cardiac biomarkers, including troponin I, show small and rapid increase and decrease, kinetics different from that generally observed in myocardial infarction, with higher concentrations and slower increase and decrease [2,6]. This was observed in our cases.
Both our patients had unobstructed coronary arteries and typical wall motion abnormalities, detected on ventriculogram and echocardiogram.
The optimal treatment is still questionable, so supportive therapy is mandatory in the period of left ventricular dysfunction, including acetylsalicylic acid, β-blockers, heparin and ACE inhibitors. There are no studies to guide the optimal dose and duration of therapy, although some suggest that β-blockers may be continued, because TC may be related to abnormal response to excessive catecholamines [2,3].
The prognosis is generally good and the regional wallmotion abnormalities usually resolve completely after a small period of time (days to weeks) [3].
4. Conclusions
Until recently, TC was an entity frequently misdiagnosed; increasing awareness and better access to coronary angiography will allow early recognition and proper treatment.
The onset of chest pain and dyspnea in postmenopausal women, without history of coronary disease or cardiovascular risk factors, particularly if a prior emotional or physical stressor can be identified, should prompt the physician to suspect of TC.
REFERENCES
- Y. J. Akashi, et al., “Takotsubo Cardiomyopathy: A New Form of Acute, Reversible Heart Failure,” Circulation, Vol. 118, No. 25, 2008, pp. 2754-2762. doi:10.1161/CIRCULATIONAHA.108.767012
- M. Zeb, et al., “Takotsubo Cardiomyopathy: A Diagnostic Challenge,” Postgraduate Medical Journal, Vol. 87, No. 1023, 2010, pp. 51-59. doi:10.1136/pgmj.2010.102475
- A. Prasad, “Apical Ballooning Syndrome: An Important Differential Diagnosis of Acute Myocardial Infarction,” Circulation, Vol. 115, No. 5, 2007, pp. e56-e59. doi:10.1161/CIRCULATIONAHA.106.669341
- M. Gianni, et al., “Apical Ballooning Syndrome or Takotsubo Cardiomyopathy: A Systematic Review,” European Heart Journal, Vol. 27, No. 13, 2006, pp. 1523- 1529. doi:10.1093/eurheartj/ehl032
- W. V. Vieweg, et al., “Depression, Stress, and Heart Disease in Earthquakes and Takotsubo Cardiomyopathy,” American Journal of Medicine, Vol. 124, No. 10, 2011, pp. 900-907. doi:10.1016/j.amjmed.2011.04.009
- K. A. Bybee, et al., “Systematic Review: Transient Left Ventricular Apical Ballooning: A Syndrome That Mimics ST-Segment Elevation Myocardial Infarction,” Annals of Internal Medicine, Vol. 141, No. 11, 2004, pp. 858-865.
- I. S. Wittstein, et al., “Neurohumoral Features of Myocardial Stunning Due to Sudden Emotional Stress,” New England Journal of Medicine, Vol. 352, No. 6, 2005, pp. 539-548. doi:10.1056/NEJMoa043046
- S. B. Nguyen, et al., “Do Comorbid Psychiatric Disorders Contribute to the Pathogenesis of Tako-Tsubo Syndrome? A Review of Pathogenesis,” Congestive Heart Failure, Vol. 15, No. 1, 2009, pp. 31-34. doi:10.1111/j.1751-7133.2008.00046.x
- O. Wever-Pinzon and L. Tami, “Takotsubo Cardiomyopathy Following a Blood Transfusion,” Congestive Heart Failure, Vol. 16, No. 3, 2010, pp. 129-131. doi:10.1111/j.1751-7133.2009.00134.x
- Y. Abe, A. Tamura and J. Kadota, “Prolonged Cardiogenic Shock Caused by a High-Dose Intravenous Administration of Dopamine in a Patient with Takotsubo Cardiomyopathy,” International Journal of Cardiology, Vol. 141, No. 1, 2001, pp. e1-e3. doi:10.1016/j.ijcard.2008.11.123
NOTES
*Corresponding author.

