Advances in Bioscience and Biotechnology
Vol.2 No.4(2011), Article ID:6868,4 pages DOI:10.4236/abb.2011.24039
Bioscience of ruminant intake evolution: feeding time models
![]()
Department of Animal Sciences, University of Zanjan, Zanjan, Iran.
E-mail: nikkhah@znu.ac.ir
Received 1 June 2011; revised 1 July 2011; accepted 18 July 2011.
Keywords: Chronobiolohy; Intake; Ruminant; Evolution; Nutrition
ABSTRACT
Ruminants have evolved to ruminate mostly overnight and graze during day. As such, rumen fermentation, post-rumen nutrient assimilation and peripheral metabolism have 24-h patterns. These evolutionary rhythms in eating behavior and metabolism have led to annual, seasonal, and circadian rhythms in ruminant endocrinology. Such natural patterns have encountered dramatic shifts in productivity in the last few decades. For optimum nutrient use and animal health, securing a synchrony between external cues and ruminant internal conditions is essential. Most recent discoveriess suggest alterations in postprandial intake patterns of non-grazing lactating cows by altered feeding time. Eating rate and feed intake within the first 3 h after feeding have been increased by evening instead of morning feeding. As a result, postprandial patterns in rumen fermentation and peripheral blood levels of metabolites and hormones have been altered. These findings and insights establish a chronological nature for intake regulation in modern ruminants. Feeding time is a major external cue that affects eating extent, rate and efficiency in ruminants. Time of feeding requires special consideration and more mechanistic evaluations for animals and humans.
1. INTRODUCTION
Feeding timing has most recently been found to alter ruminant chronobiology [1-4]. The time of feed delivery and the time of most intense eating activity determine postprandial rhythms and daily amount of intake [1-4]. Chronobiologically, for maximum nutrient efficiency and health, nutrient supply to reticulorumen, splanchnic and peripheral tissues should be synchronized with endogenous rhythms in ruminant endocrinology and metabolism [5]. Endogenous rhythms are controlled mainly by biological clocks located in the hypothalamic suprachiasmatic nuclei, and to some extent by photoperiod and feeding cues [3,6]. Glucose tolerance and blood glucose in humans and rats have diurnal endogenous rhythmcity [7]. Exogenous rhythms are controlled mainly by external factors, including feeding time and feed type. Blood urea is for instance exogenously regulated [5]. Knowledge on how feeding and eating times affect diurnal and postprandial intake patterns will enable predicting diurnal patterns in rumen, post-rumen and peripheral nutrient assimilation. These will suggest optimal, suboptimal, and unfavorable times of nutrient supply. The primary objective is to delineate an evolutionary basis of ruminant nutrition through describing the most recent chronological discoveries of feeding time effects on diurnal rhythms in intake, rumen fermentation and metabolism of high-producing ruminants. In addition, a new integrative evolutionary theory will be constructed.
2. SCIENCE OF FEED INTAKE EVOLUTION
Based on an evolutionary concept, rumination occurs mostly overnight [1,8,9]. This implies a greater rumen digestion capacity during night than day [8]. Such a greater overnight rumen volume and fermentation has been shown in grazing cows [8] and intensively housed lactating cows [4,9]. If the increased rumen fermentation capacity is concurrent with increased rumination, optimum rumen pH and microbial metabolism may be easier to secure with evening vs. morning feeding. Through an increased rumen absorption capacity, evening feeding would not likely depress rumen pH below the threshold for microbial rupture, endotoxin release, and proinflammatory responses (e.g., <5.2 - 5.5; [10,11]).
3. CHRONOBIOLOGY, EVOLUTION, AND INSULIN
Insulin stimulates peripheral nutrient storage. Insulin has limited effects on propionate-driven hepatic gluconeogenesis and mammary nutrient uptake [12,13]. Increased peripheral blood levels of energy-producing substrates by altered feeding time would increase both mammary and non-mammary nutrient uptakes. As a result, milk secretion and peripheral nutrient retention can simultaneously improve. Recent chronological perspectives into human metabolism have revealed evolutionary facts on the etiology of diabetes [6,7,14-16]. The new vision underlines the significance of feed presentation time on diurnal patterns in ingestion, digestion and metabolism that can be optimally synchronized with endogenous physiological rhythms to improve nutrient use and organ health. Such information will contribute to improving metabolic and health predictions using mathematical models.
4. POSTPRANDIAL INTAKE PATTERNS FOR EVENING VS. MORNING FEEDING
Nikkhah et al. [3] discovered that the proportion of daily feed intake ingested within 3 h post-feeding was 55% for cows fed at 2100 h and only 46% for cows fed at 0900 h (Figure 1). As a result, rumen pH was lower at nadir, and VFA levels were higher at zenith (Figure 2). Cumulatively, the feed amounts consumed between 0 - 6 h and 0 - 9 h post-feeding were similar between the two groups. Parity and dietary concentrate did not interact with feeding time on postprandial intake patterns.
The stimulatory effects of fresh feed presentation on eating activity may persist even with 4 times daily feeding [17]. Group-housed cows fed once daily at 0530 h spent less time eating compared to cows fed twice daily at 0530 and 1515 h. The prolonged eating of twice fed cows was mostly due to prolonged eating between 2000 to 0600 h. When fresh mixed feed was fed once at 0600 h vs. 4 times at 0600, 1000, 1400 and 1900 h, total eating time of early lactation cows in free stalls was similar [18]. Cows fed 4 times daily tended to spend more time eating during 1600 - 2000 h vs. 0400 - 1200 h.
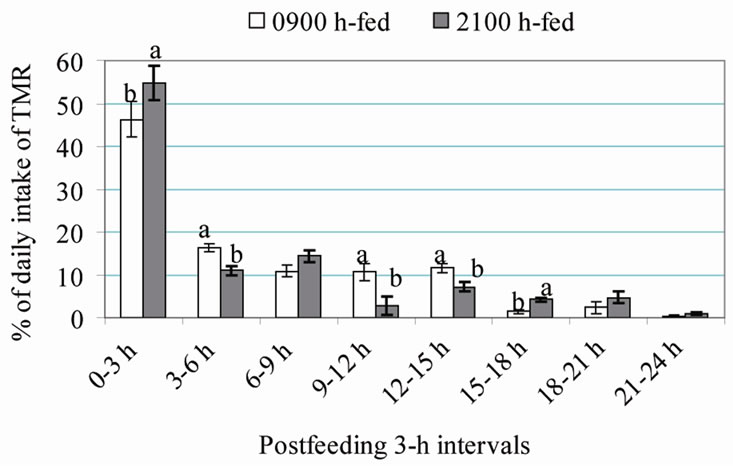
Figure 1. Post-feeding patterns of feed intake in cows fed either at 0900 h or at 2100 h. Different superscripts within each 3-h interval differ at P < 0.05, except for 0 - 3 h with P = 0.06 [1,3].
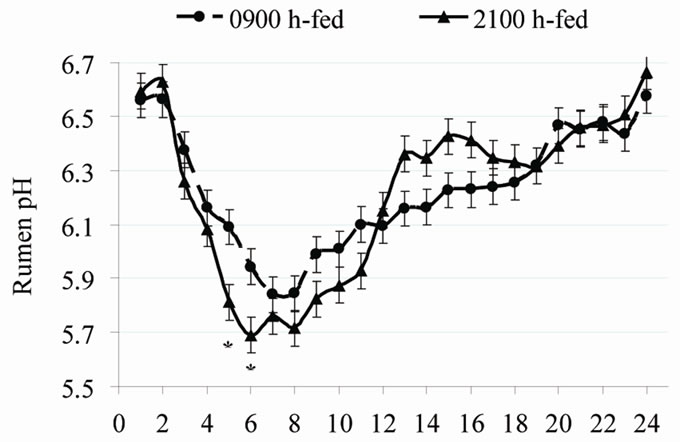
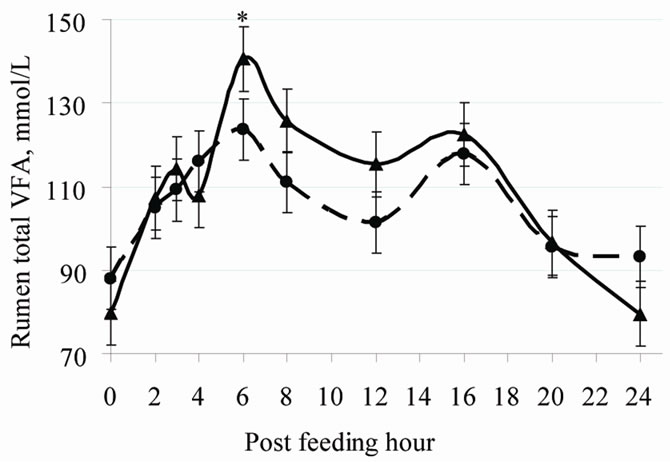
Figure 2. Post-feeding patterns of rumen pH (top) and total VFA concentrations (bottom) in cows fed either at 0900 h or at 2100 h. Within each hour, * =P < 0.05 [4].
These suggest that dairy cows’ intake shortly postfeeding depends on time of day. In the study of Nikkhah et al. [4], the proportion of daily intake consumed within 3 h post-feeding was 55% in 2100 h-fed cows and only 46% in 0900 h-fed cows (Figure 1). Nikkhah et al. [2,3] found that 2100 h-fed cows ate 37% but 0900 h-fed cows ate only 26% of their daily intake within 3-h of feeding. In beef cattle [19], evening instead of morning feeding of high-concentrate diets altered feed intake post-feeding patterns. Schwartzkopf-Genswein et al. [20] found a positive response in feedlot cattle feed intake to feeding at 2100 vs. 0900 h, but did not monitor intake diurnal patterns. Energy-corrected milk yield was increased by 2.1 kg/d in primiparous cows and by 1.3 kg/d in multiparous cows, when feed was presented at 2100 vs. 0900 h [4]. These suggest that time of feeding and thus time of most intense eating affect nutrient efficiency in lactating cows.
5. ANTICIPATION OF FEED PROVISION
Anticipating feed presentation can elongate the eating peak after feeding [18]. The greater intake of cows fed at 2100 h vs. 0900 h within 3-h of feeding [3,4,10] suggests that cows anticipate evening feeding better than morning feeding. Plasma insulin was higher and plasma glucose was lower at 2 h post-feeding in cows fed at 2100 h vs. 0900 h [21]. Higher insulin could weaken glucagon and reduce gluconeogenesis [12]. The intravenous glucagon has reduced feed intake in sheep [22]. It is likely that the higher blood insulin and lower glucose at 2 h postfeeding in evening fed cows may have delayed a glucagon-driven satiety. This could contribute to increased intake within 3 h of evening feeding.
6. GENEARAL INTUITIONS AND DISCUSSION
The greater appetite for evening vs. morning feeding could partly be due to photoperiod. Lights were turned on at 0345 h and turned off at 2245 h or at 1:45 h after evening feeding [4]. The possible anticipation of the light-off by evening fed cows might contribute to their rapid eating upon feeding. Moreover, melatonin is known to regulate glucose metabolism in humans and rats [14,23,24]. Melatonin may contribute to feed intake regulation. Exogenous melatonin consumed via drinking water boosts postprandial insulin response [14]. Also, melatonin secretion is known to be induced by dark exposure. In 2100 h-fed cows, feeding occurred shortly before lights-off (2245 h) when melatonin secretion was expected to be high [3,4]. In humans, reduced glucose tolerance is linked to elevated melatonin secretion [15]. Reduced glucose tolerance is due to reduced insulin responsiveness and peripheral glucose uptake, which reflects a reduction in glucose and insulin demands [16,25]. With such simultaneously reduced glucose tolerance and elevated melatonin secretion occurring in cows, evening eating would increase blood insulin and glucose. These would suggest a decline in peripheral glucose uptake and insulin turnover [24].
Peripheral metabolites such as glucose and VFA depress feed intake through cell entry and not only by staying in the blood [9]. Thus, factors reducing peripheral metabolite uptake can consequently attenuate the metabolite-driven satiety. As such, the expected rise in evening blood melatonin could reduce peripheral metabolites use in favor of milk secretion. Thus, a higher nocturnal melatonin might weaken the feed-driven satiety in evening fed cows. This may have led eveningfed cows to consume more feed shortly post-feeding compared to morning-fed cows [3,4,10]. Such altered regulation of feed intake would allow a possibility for night-time glucose intolerance in lactating cows [21]. Milk energy output was greater in 2100 h-fed cows than in 0900 h-fed cows, suggesting that at times of high feed intake and hepatic metabolite output shortly post-feeding, milk precursors (e.g., lactate, glucose, and BHBA; Figure 3) were used rather by the mammary gland. The greater rumen VFA concentration shortly post-feeding by evening vs. morning feeding supports the increased milk output (Figure 2).
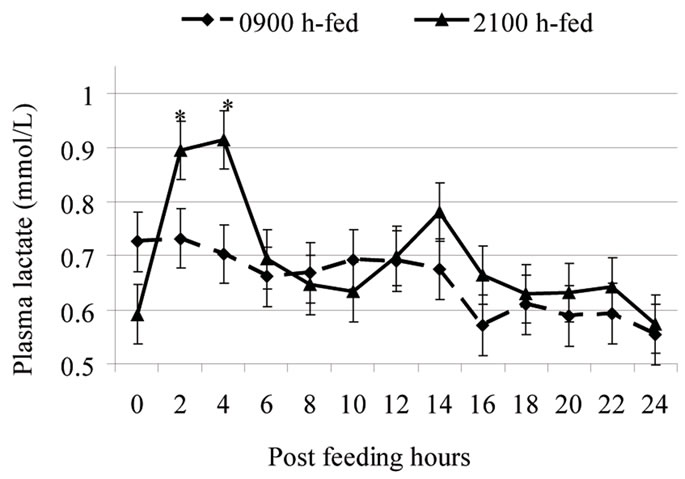
Figure 3. Post-feeding patterns of plasma lactate in cows fed either at 0900 h or at 2100 h. Within each sampling hour, * = P < 0.05 [1,2]
7. IMPLICATIONS
Evolutionary feeding management related factors (e.g., feeding and eating timing) are among the key missing modules of the accurate global predictions of nutrient intake, rumen health and intermediary metabolism. Most recent discoveries have revealed the timing of ruminant feeding as a significant determinant of postprandial patterns in feed intake and cow metabolism. Altered feeding and eating behaviors and nutrient utilization efficiency will be contemplated in light of ruminant evolution. Such chronobiological insights into modern ruminant production will offer prospects to improve rumen and animal health. Ruminant feed intake is an evolutionary bioscience interfacing viable ruminant longevity, adequately safe and secure food supply, and quality environments.
8. ACKNOWLEDGEMENTS
The Ministry of Science, Research and Technology and University of Zanjan (Zanjan, Iran) are acknowledged for supporting the author’s programs of optimizing science education policies in the third millennium.
REFERENCES
- Nikkhah, A., Furedi, C.J., Kennedy, A.D., et al. (2008) Effects of feed delivery time on feed intake, rumen fermentation, blood metabolites and productivity of lactating cows. Journal of Dairy Science, 91, 1-12.
- Nikkhah, A., Plaizier, J.C., Furedi, C.J., et al. (2007) Time of feeding: A determinant of post feeding patterns in feed intake of lactating cows. Journal of Dairy Science, 90, 559.
- Nikkhah, A., Plaizier, J.C., Furedi, C.J., et al. (2006) Response in diurnal variation of circulating blood metabolites to nocturnal vs diurnal provision of fresh feed in lactating cows. Journal of Animal Science, 84, 111.
- Nikkhah, A., Furedi, C.J., Kennedy, A.D., et al. (2011) Feed delivery at 2100 h vs. 0900 h for lactating dairy cows. Canadian Journal of Animal Science, 91, 113-122. doi:10.4141/CJAS10012
- Piccione, G. and Caola, G. (2002) Review: Biological rhythms in livestock. Journal of Veterinary Science, 3, 145-157.
- Sehgal, A. (2004) Molecular biology of circadian rhythms. John Wiley & Sons, Inc., Hoboken. doi:10.1002/0471459186
- La Fleur, S.E., Kalsbeek, A., Wortel, J., et al. (2001a) A daily rhythm in glucose tolerance: A role for the suprachiasmatic nucleus. Diabetes, 50, 1237-1243. doi:10.2337/diabetes.50.6.1237
- Taweel, H.Z., Tas, B.M., Smit, H.J., et al. (2006) A note on eating behaviour of dairy cows at different stocking systems-diurnal rhythm and effects of ambient temperature. Applied Animal Behavior Science, 98, 315-322. doi:10.1016/j.applanim.2005.10.010
- Forbes, J.M. (1995) Voluntary food intake and diet selection in farm animals. CABI Int., Wallingford.
- Nikkhah, A., (2011) Postprandial rhythms of circulating urea in lactating dairy cows: Feeding timing and diet effects. Biological Rhythm Research, in press.
- Krause, K.M. and Oetzel, G.R. (2006) Understanding and preventing subacute ruminal acidosis in dairy herds: A review. Animal Feed Science and Technology, 126, 215-236. doi:10.1016/j.anifeedsci.2005.08.004
- Brockman, R.P. (1978) Roles of glucagons and insulin in the regulation of metabolism in ruminants: A review, Canadian Veterinary Journal, 19, 55-62.
- Brockman, R.P. (1990) Effect of insulin on the utilization of propionate in gluconeogenesis in sheep. British Journal of Nutrition, 64, 95-101. doi:10.1079/BJN19900012
- La Fleur, S.E., Kalsbeek, A., Wortel, J., et al. (2001b) Role for the pineal and melatonin in glucose homeostasis: Pinealectomy increases night-time glucose concentrations. Journal of Neuroendocrinology, 13, 1025-1032. doi:10.1046/j.1365-2826.2001.00717.x
- Van Cauter, E., Moreno-Reyes, R., Akseki, E.L., et al. (1998) Rapid phase advance of the 24-h melatonin profile in response to afternoon dark exposure. American Journal of Physiology: Endocrinology and Metabolism, 275, E48-E54.
- Van Cauter, E., Biackman, J., Roland, D., et al. (1991) Modulation of glucose regulation and insulin secretion by circadian rhythmicity and sleep. Journal of Clinical Investigations, 88, 934-942. doi:10.1172/JCI115396
- DeVries, T.J., Von Keyserlingk, M.A.G. and Beauchemin, K.A. (2005) Frequency of feed delivery affects the behavior of lactating dairy cows. Journal of Dairy Science, 88, 3553-3562. doi:10.3168/jds.S0022-0302(05)73040-X
- Phillips, C.J. and Rind, M.I. (2001) The effects of frequency of feeding a total mixed ration on the production and behavior of dairy cows. Journal of Dairy Science, 84, 1979-1987. doi:10.3168/jds.S0022-0302(01)74641-3
- Pritchard, R.H. and Knutsen, J.S. (1995) Feeding frequency and timing, Intake by feedlot cattle. Oklahoma Agricultural Experiment Station, Oklahoma State University, Stillwater, 162-166.
- Schwartzkopf-Genswein, K.S., Beauchemin, K.A., McAllister, T.A., et al. (2004) Effect of feed delivery fluctuations and feeding time on ruminal acidosis, growth performance, and feeding behavior of feedlot cattle. Journal of Animal Science, 82, 3357-3365.
- Furedi, C., Kennedy, A.D., Nikkhah, A., et al. (2006) Glucose tolerance and diurnal variation of circulating insulin in evening and morning fed lactating cows. Advances in Dairy Technology, 18, 356.
- Deetz, L.E. and Wangsness, P.J. (1981) Influence of intrajugular administration of insulin, glucagon and propionate on voluntary feed intake of sheep. Journal of Animal Science, 53, 427-433.
- Lima, F.B., Machado, U.F., Bartol, L., et al. (1998) Pinealectomy causes glucose intolerance and decreases adipose cell responsiveness to insulin in rats. American Journal of Physiology, 275, E934-E941.
- Picinato, M.C., Haber, E.P., Carpinelli, A.R., et al. (2002) Daily rhythm of glucose-induced insulin secretion by isolated islets from intact and pinealectomized rat, Journal of Pineal Research, 33, 172-177. doi:10.1034/j.1600-079X.2002.02925.x
- Nikkhah, A., Plaizier, J.C., Furedi, C.J., et al. (2006) Response in diurnal variation of circulating blood metabolites to nocturnal vs diurnal provision of fresh feed in lactating cows. Journal of Animal Science, 84, 111.

