Soft Nanoscience Letters
Vol.3 No.4A(2013), Article ID:40524,3 pages DOI:10.4236/snl.2013.34A012
Interaction of Boron-Nitrogen Substitued Graphene Nanoribbon with Nucleobases: The Idea of Biosensor
![]()
Department of Physics, Assam University, Silchar, India.
Email: *utpalchemiitkgp@yahoo.com
Copyright © 2013 Barnali Bhattacharya et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received October 1st, 2013; revised November 12th, 2013; accepted November 19th, 2013
Keywords: Biosensor; DFTB; Nucleobases; Transport Properties; Nanoribbon
ABSTRACT
In this paper we have designed a biosensor device built from B-N substituted graphene nanoribbon within density functional based tight-binding (DFTB) framework. We have investigated the interaction of the nucleobases adenine (A), Guanine (G), Cytosine (C) and Thymine (T) with device. Our calculation suggests that all the nucleobases have different interaction strength when they interact with device and shows that guanine has stronger interaction with device than other nucleobases. It reveals that the absorption energy shows the hierarchy: G > C > T > A. Our results also demonstrate the transport properties of the device and how the transport properties change due to the absorption of nucleobases on the device.
1. Introduction
In order to improve the sequencing technologies, the interaction of nucleobases with nanostructure has attracted much attention [1-3]. It has been found that some nano structured systems are capable to detect the biomolecules. Recently the graphene nanoribbons (GNR) are treated as a suitable system for sensing small molecules such as H2O, CO [4]. Recently GNR with pores has been used to design biosensors [5]. Again it was observed that nuclobases can introduce a distinguishable signal in current through a GNR [6] even at room temperature in water.
Due to the rapid progress in nano-technology, graphene nanoribbons become the promising nano-material for its extraordinary mechanical, electronic, and transport properties. Graphene nanoribbon gives the evidence of having large density of state at the edge near the Fermi level; due to this, it provides a golden opportunity to explore distinctive transport properties of GNR and has been investigated by many groups [7]. The nature of edge often plays an important role in properties of GNR. In case of zigzag edge GNR the unpaired π and π* electron localized near the edge. The properties of ZGNR depend on many factors, such as doping, external field, defects, molecule adsorption. This provides us golden opportunities for tuning them as a sensor. It was already investigated by many groups that substitutional doping of GNR by B or N changes the conducting characteristics and has opened a great possibility for electronic device fabrication [8,9].
In this letter, we have designed a device with BN doped graphene nanoribbon. In order to avoid the contact resistance we have used graphene nanoribbon as electrode here. Our specific aim is to study the interaction of nucleobases with BN doped graphene and for this, we have chosen four nucleobases adenine (A), guanine (G), cytosine (C), and thymine (T). We placed these nucleobases (A, G, C, T) in the centre of the device parallel to ribbon surface. Our specific aim is to calculate the absorption strength of these nucleobases placed on device and explore the change on transport properties of B-N doped graphene. This will give the clear idea about the ability of B-N substituted graphene for sensing biomolecules.
2. Computational Details
All the calculations have done within the density functional based tight-binding (DFTB) framework [10]. We have optimized the geometry of B-N substituted graphene with nucleobases using the 4 × 4 × 6 grid for k point sampling of the Brillouin zone and DFTB parameter which have been derived by M. Elstner et al. [11].
3. Results and Discussions
In order to calculate the stability of device with different nucleobases we have calculated the adsorption energy of each nucleobases on device. It is found that the adsorption energy of these four fundamental nucleobases while interact with device shows the hierarchy G (0.59977 eV) > C (0.25791) > T (0.257323) > A (0.07599 eV).
We can explain this on the basis of their configuration. It was found that biomolecules arrange themselves on device by π-π staking interaction between the aromatic rings of them and nanoribbon where access of electron participate. Both the nucleobases and BN substituted graphene have a tendency to minimise the overlap of π- orbital, as a result their geometry shifted from perfect AB-stacking. The adenine and thymine exhibit least deviation, while guanine deviated highest.
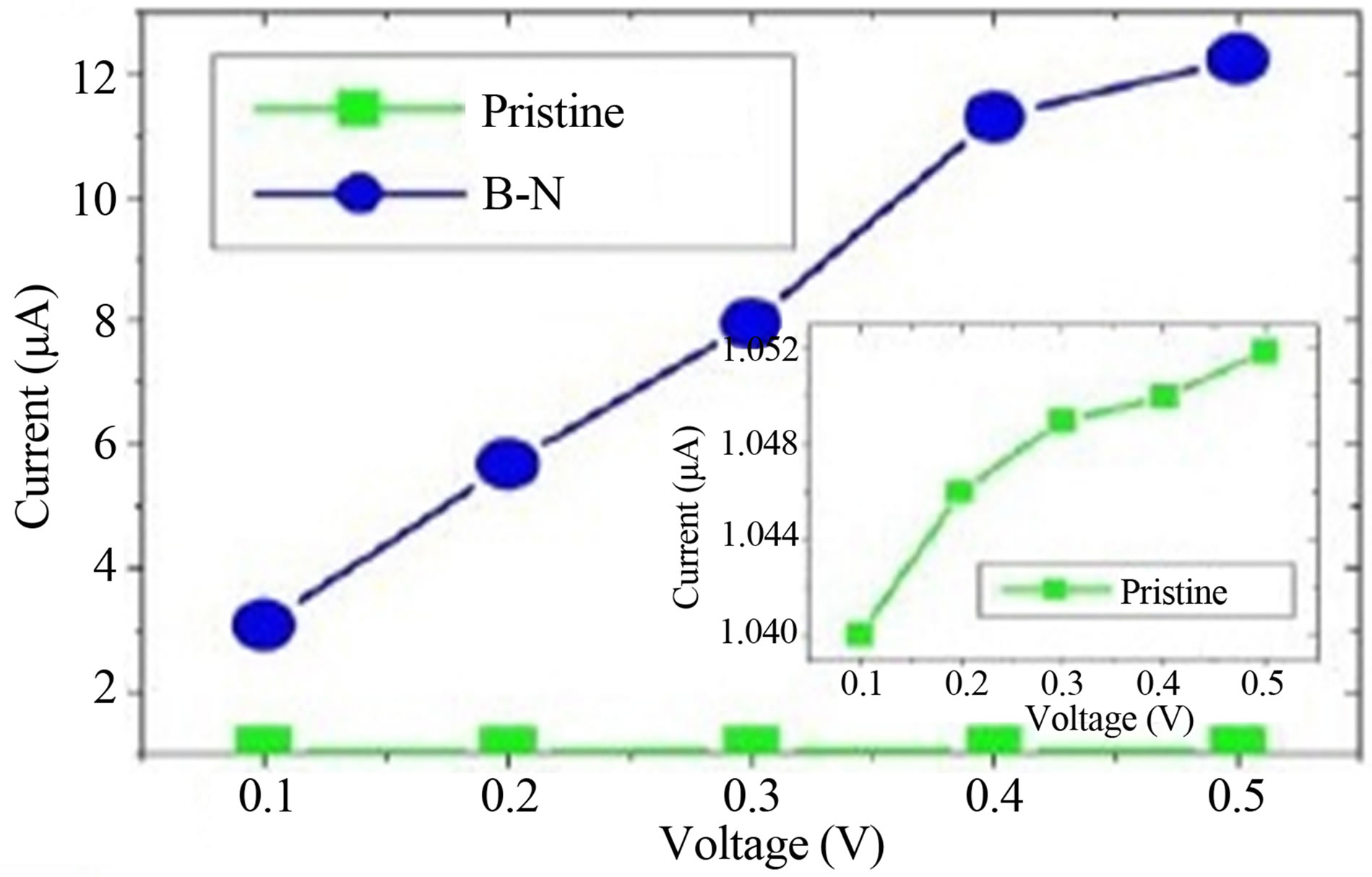
The current and transmission curves of “pristine graphene” device and device with B-N substituted graphene are shown in Figures 1(a) and (b) respectively. It is clear from I - V characteristic (Figure 1(a)) that the substitution of one layer of carbon atom by B-N increases the electron transport ability of GNR. From the transmission curve (Figure 1(b)) it is also clear that the transmission dips at −0.002 eV below the Fermi level and 0.006 eV above the Fermi level, due to the substitution of one layer of graphene by alternative arrangement of B and N. It has been found that the substitutional doping by boron and nitrogen reduces the band gap of zigzag GNR for both the spins [8] and make the semiconductor ZGNR to a half metallic one.
Figure 1(c) represents transmission curves of device with different nucleobases. It is clear that the presence of nucleobases on device increases the transmission. At certain bias voltage the device with different nucleobases shows different transmission. At the Fermi energy (E − EF = 0) the transmission (see Figure 1(c)) of G, C, A, T and B-N are 0.917, 0.584, 0.568 and 0.565 and 0.171 respectively. The increase in transmission due to presence of nucleobases can be explained from their binding on device through non-covalent bond that means π-π stacking interaction between the aromatic rings of them and nanoribbon. Due to the lone pair of nitrogen (N) and oxygen (O) atoms and the presence of aromatic rings in nucleobases, the deformability of charge distribution increases which in turns increases the adsorption energy and transmission and is highest for Guanine. The high conductivity of guanine, cytosine, and thymine can be understood from this point of view. These three bases (G, C and T) contain oxygen in their configuration, which induce an extra signal in current through device.
 (a)
(a)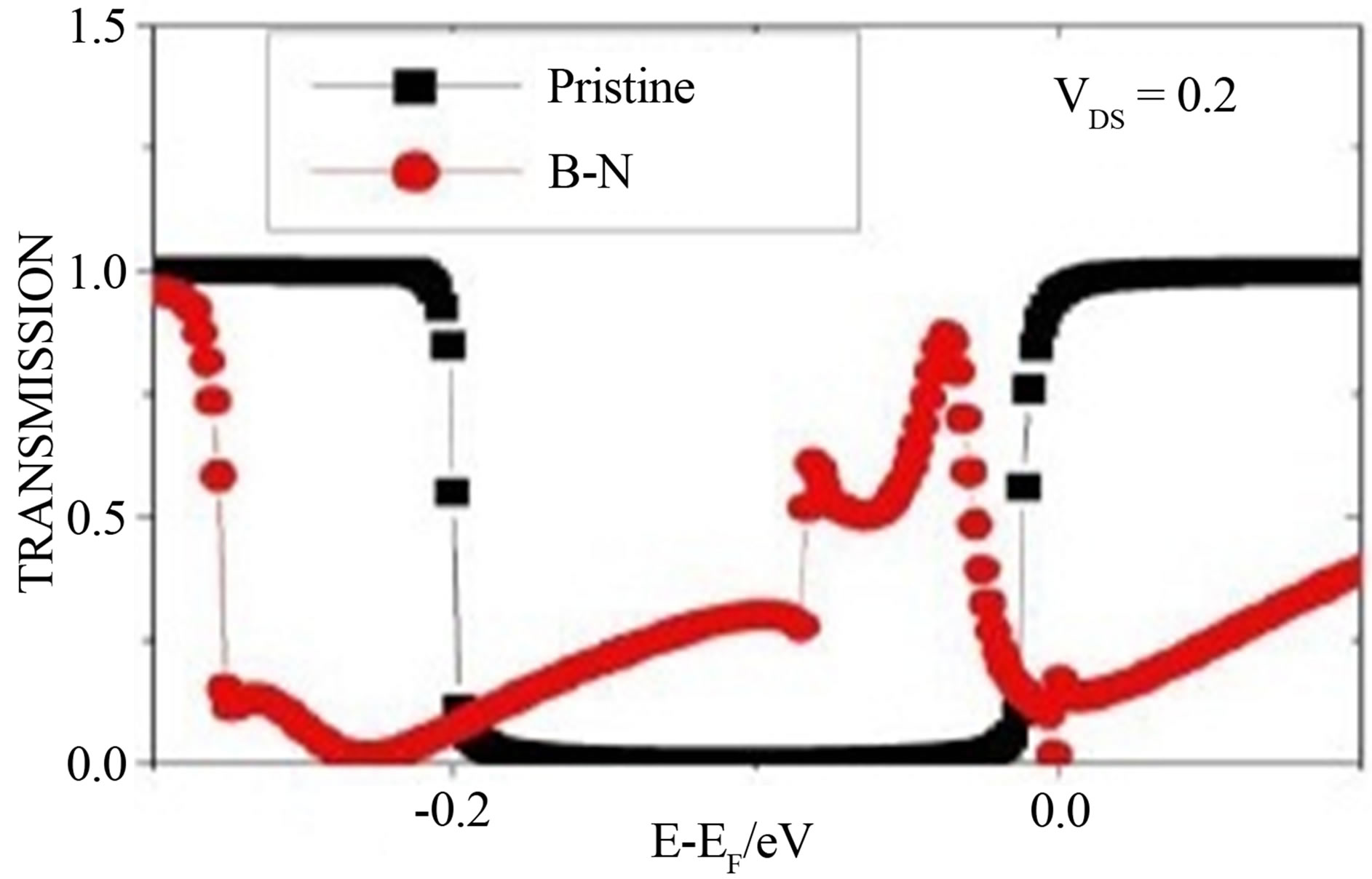 (b)
(b)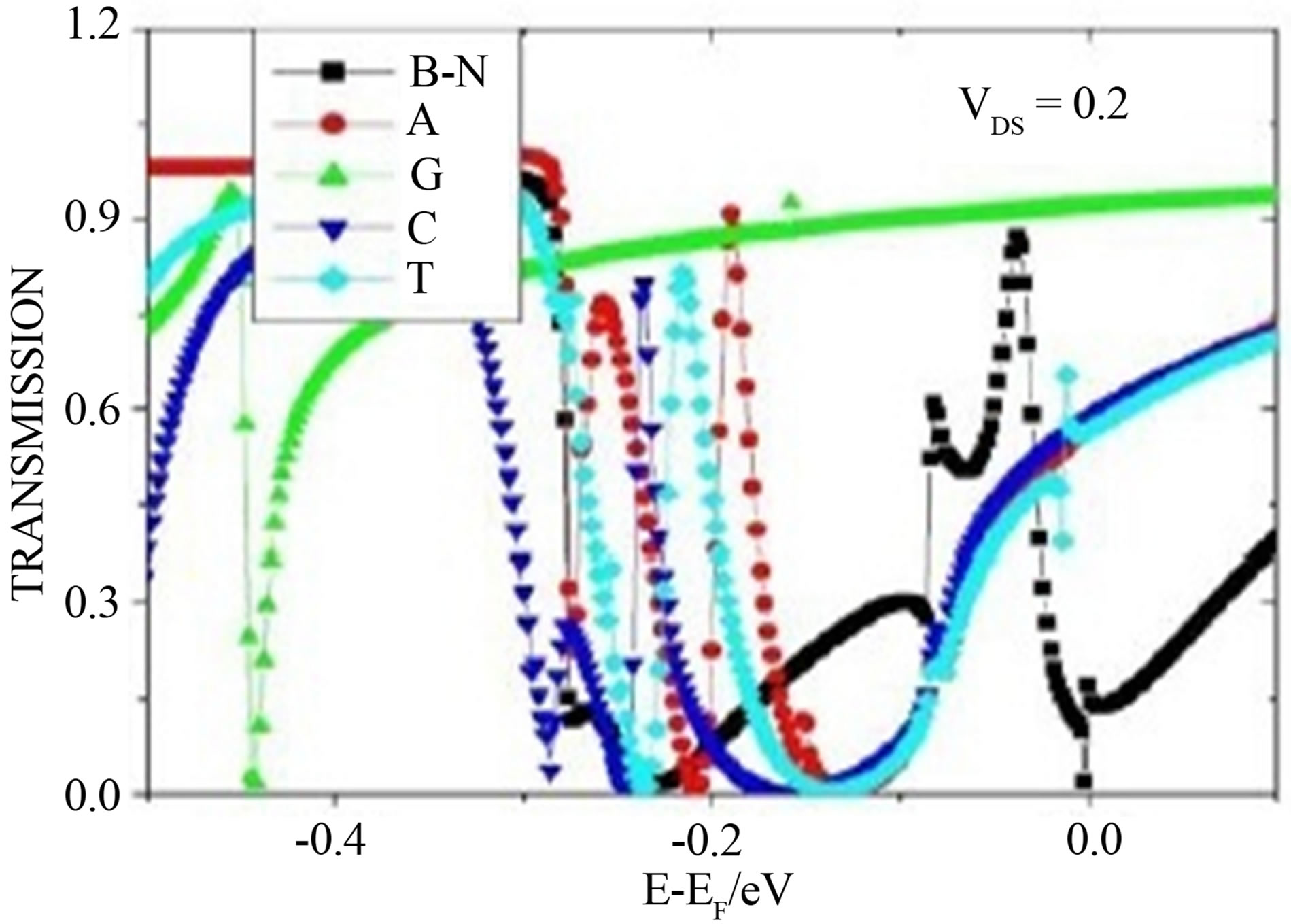 (c)
(c)
Figure 1. (a) and (b) represent the Current-Voltage curve and transmission curve for pristine zigzag graphene nanoribbon (ZGNR) and B-N substituted graphene whereas (c) represents the transmission curve of B-N substituted graphene with nucleobases.
4. Conclusion
The effect of the different nucleobases on transport property of B-N substituted graphene has been demonstrated here within density functional tight-binding approach. Our result demonstrates that the nucleobases exhibit different adsorption strength on device. Guanine adsorbed more strongly on device than other nucleobases. Adsorption of guanine, cytosine, thymine introduces an extra signal in electric current through device, hence they are detectable in this manner.
5. Acknowledgements
Dr. Utpal Sarkar would like to acknowledge Prof. Somnath Dasgupta, Vice Chancellor, Assam University, for his inspiration and help in various ways.
REFERENCES
- S. Gowtham, R. H. Scheicher, R. Ahuja, R. Pandey and S. P. Karna, “Physisorption of Nucleobases on Graphene,” Physical Review B, Vol. 76, No. 3, 2007, Article ID: 033401. http://dx.doi.org/10.1103/PhysRevB.76.033401
- S. Mukhopadhyay, S. Gowtham, R. H. Scheicher, R. Pandey and S. P. Karna, “Theoretical Study of Physisorption of Nucleobases on Boron Nitride Nanotubes: A New Class of Hybrid Nano-Biomaterials,” Nanotechnology, Vol. 21, No. 16, 2010, Article ID: 165703. http://dx.doi.org/10.1088/0957-4484/21/16/165703
- X. Zhong, W. J. Slough, R. Pandey and C. Friedrich, “Interaction of Nucleobases with Silicon Nanowires: A First Principle Study,” Chemical Physics Letters, Vol. 553, 2012, pp. 55-58. http://dx.doi.org/10.1016/j.cplett.2012.10.011
- J. Dai, J. Yuan and P. Giannozzi, “Gas Adsorption on Graphene Doped with B, N, Al and S: A Theoretical Study,” Applied Physics Letters, Vol. 95, No. 23, 2009, Article ID: 232105. http://dx.doi.org/10.1063/1.3272008
- O. F. Ping, P. S. Lin, Z. Hua, W. L. Bo and X. Hui, “A Biosensor Based on Graphene Nanoribbon with Nanopores: A First Principles Devices-Design,” Chinese Physics, Vol. 20, No. 5, 2011, Article ID: 058504. http://dx.doi.org/10.1088/1674-1056/20/5/058504
- B. Song, G. Cuniberti, S. Sanvito and H. Fang, “Nucleobase Adsorbed at Graphene Devices: Enhanced Bio-Sensorics,” Applied Physics Letters, Vol. 100, No. 6, 2012, Article ID: 063101. http://dx.doi.org/10.1063/1.3681579
- Y. T. Zhang, Q. M. Li, Y. C. Li, Y. Y. Zhang and F. Zhai, “Band Structures and Transport Properties of Zigzag Graphene Nanoribbons with Antidot Arrays,” Journal of Physics: Condensed Matter, Vol. 22, No. 31, 2010, Article ID: 315304. http://dx.doi.org/10.1088/0953-8984/22/31/315304
- Z. Wang, J. Xiao and X. Li, “Effects of Heteroatom (Boron or Nitrogen) Substitutional Doping on the Electronic Properties of Graphene Nanoribbons,” Solid State Communications, Vol. 152, No. 2, 2012, pp. 64-67. http://dx.doi.org/10.1016/j.ssc.2011.11.002
- X. H. Zhengh, I. Rungger, Z. Zeng and S. Sanvito, “Effects Induced by Single and Multiple Dopents on the Transport Properties in Zigzag-Edged Graphene Nanoribbons,” Physical Review B, Vol. 80, No. 23, 2009, Article ID: 235426. http://dx.doi.org/10.1103/PhysRevB.80.235426
- B. Aradi, B. Hourahine and Th. Frauenheim, “DFTB+, a Sparse Matrix-Based Implementation of the DFTB Method,” The Journal of Physical Chemistry A, Vol. 111, No. 26, 2007, pp. 5678-5684. http://dx.doi.org/10.1021/jp070186p
- M. Elstner, D. Porezag, G. Jungnickel, J. Elsner, M. Haugk, T. Frauenheim, S. Suhai and G. Seifert, “Self-ConsistentCharge Density-Functional Tight-Binding Method for Simulations of Complex Materials Properties,” Physical Review B, Vol. 58, No. 11, 1998, pp. 7260-7268. http://dx.doi.org/10.1103/PhysRevB.58.7260
NOTES
*Corresponding author.

