Open Journal of Stomatology
Vol. 3 No. 1 (2013) , Article ID: 29432 , 7 pages DOI:10.4236/ojst.2013.31012
Efficacy of a triclosan/copolymer dentifrice and a toothbrush with tongue cleaner in the treatment of oral malodor: A monadic clinical trial
![]()
1Colgate Palmolive Company, Piscataway, USA
2University at Buffalo, School of Dental Medicine, Buffalo, USA
Email: *jjzambon@buffalo.edu
Received 20 January 2013; revised 28 February 2013; accepted 9 March 2013
Keywords: Halitosis; Oral Malodor; Triclosan; Tongue
ABSTRACT
Aim: Oral malodor (halitosis) is a widespread condition caused by oral bacteria, particularly sulfur compound-producing species. This study assessed the effect of a triclosan/copolymer-containing dentifrice and a novel toothbrush with attached tongue cleaner on oral malodor and on the bacteria colonizing the dorsal surface of the tongue. Materials and Methods: 14 adult subjects with oral malodor defined as organoleptic scores ≥ 3 (scale 0 - 5) and mouth air sulfur levels ≥ 250 ppb participated in this study. Subjects were examined at baseline and after 28 days use of the triclosan dentifrice and toothbrush/tongue cleaner for: 1) organoleptic assessment; 2) mouth air sulfur levels; 3) tongue coating; and, 4) dorsal tongue surface microorganisms. Total bacterial numbers were assayed by microscopy. Dot-blot hybridization was used to assess a panel of 20 oral bacteria. Results: After 28 days, all subjects had significantly reduced organoleptic scores and mouth air sulfur levels compared to baseline (p < 0.0001). There was >70% reduction in microbial numbers (p < 0.001) on the dorsal tongue and significant reductions in Enterococcus faecalis (p < 0.003), Neisseria sp. (p < 0.008), Peptostreptococcus micros (p < 0.0007), Prevotella melaninogenica (p < 0.02), Porphyromonas gingivalis (p < 0.0001), Solobacterium moorei (p < 0.04), and Streptococci sp. (p < 0.03). Conclusions: Oral malodor was successfully treated in all subjects following 28 days use of the triclosan/copolymer-containing dentifrice and toothbrush with a tongue cleaner as determined by significant reductions in clinical measures of halitosis and a corresponding reduction in microbiological measures including sulfur compound-producing oral bacteria.
1. INTRODUCTION
Oral malodor, also referred to as oral halitosis, is an unpleasant or offensive odor emanating for the oral cavity. It is a common world-wide complaint and a significant reason for professional consultation and referrals [1]. Halitosis is frequently self-diagnosed and is a source of social embarrassment, psychological discomfort, and altered behavior and it can influence diet and habits [2,3]. It is estimated that 85% of oral malodor originates in the oral cavity with the remainder associated with a number of extra-oral sources including systemic diseases. Thus, oral halitosis represents a multi-factorial problem straddling several clinical and scientific disciplines [1,4,5].
Descriptions of oral halitosis and potential treatments can be traced back to antiquity [6]. Despite a paucity of epidemiological studies, halitosis has been reported in populations and cultures across the world such as China, Brazil, Israel, Japan, Switzerland and the USA. Furthermore, it is estimated that approximately 50% of the population may suffer from recurring episodes of halitosis with clinical symptoms in 75% of examined individuals [5,7]. Halitosis was reported in approximately one-third of 419 subjects from a randomly selected population [8]. The overall prevalence of halitosis has led to its identification as an important social problem [4].
Based on the effects of halitosis on patient health and well-being, research has examined the causes of oral malodor. It is now widely accepted that tongue coating represents an important factor with contributions from other conditions such as periodontitis, gingivitis, caries, oral ulceration and necrosis, and food debris [9]. Microbiological studies indicate proteolytic and other metabolic features of some tongue surface microorganisms that facilitate production of volatile sulfur compounds (VSC’s) resulting in halitosis. Recent clinical investigations utilizing nucleic acid analysis of oral bacteria [10- 12] have found distinct microorganisms amongst subjects with clinical halitosis. One microorganism, Solobacterium moorei, was isolated primarily from the dorsal tongue surface of subjects with halitosis [10,11] suggesting that this bacterium is closely associated with the etiology of oral malodor [1,7].
A recent review highlights the lack of clinical studies evaluating the effects of antimicrobials in oral health care products in conjunction with mechanical cleansing in mitigating halitosis [9]. Triclosan is a substituted phenol with an extended history of safe and effective use in oral hygiene formulations [13]. A toothpaste formulated with triclosan/copolymer has been widely evaluated for clinical efficacy [13,14] and demonstrates significant inhibitory effects on the bacteria found in dental plaque, in saliva and on the tongue [15]. The present clinical investigation evaluated the effects of mechanical tongue cleaning using a specially designed toothbrush with a tongue cleaner in conjunction with a dentifrice formulated with triclosan/copolymer on clinical measures of halitosis and on the microbes colonizing the dorsal tongue surface.
2. MATERIALS AND METHODS
2.1. Subjects
Adult subjects from the Buffalo, NY, USA area were screened for study criteria. The nature of the study was explained to prospective subjects who completed and signed an informed consent statement. Adults with 20 natural teeth including at least one molar and one bicuspid in each quadrant of the dentition, in good oral and general health and who were not undergoing medical treatment were scheduled for a baseline visit. Exclusion criteria included; subjects with dental prostheses, rampant caries or other conditions requiring immediate dental care; subjects who required antibiotic pre-medication prior to dental treatment or who had taken antibiotics within 3 months or who had received periodontal therapy within 6 months. All subjects indicated routine use of a toothbrush and toothpaste and were enrolled based on the following inclusion criteria: 1) organoleptic scores of ≥3 on a 5-point scale assessed by a single trained examiner [16]; 2) halimeter scores ≥ 250 ppb utilizing a portable sulfide monitor (Halimeter®, Chatsworth, CA); and 3) assessments for tongue coating utilizing a 0 - 5 scale [2, 17].
2.2. Clinical Examinations and Treatments
After screening, subjects were scheduled for baseline clinical evaluations for halitosis that included organoleptic, halimeter and tongue coat assessments. Following these evaluations, an approximately 2 cm2 area of the posterior dorsal tongue surface about the midline was gently scraped with a sterile wooden tongue depressor and the sample was placed in 3 ml of pre-reduced, anaerobically sterilized Ringer’s solution.
Subjects were given a commercially available dentifrice formulated with triclosan/copolymer (Colgate Total) and a soft-bristled toothbrush with a tongue cleaner. Subjects were instructed in oral hygiene using the sulcular method. Subjects were instructed to brush their teeth for at least two minutes—once in the morning after breakfast and once at night before bed—with the assigned toothbrush and dentifrice. After toothbrushing, they were instructed to use the tongue cleaner on the dorsal and lateral surfaces of their tongue for at least ten seconds. Subjects were instructed to refrain from chewing gum or using any other oral hygiene products such as mouthwash during the course of the study.
Post-treatment clinical examinations and sample collections were conducted 28 days later. Dorsal tongue samples were collected approximately 12 hours after final use of the assigned toothbrush and dentifrice. A dentist conducted an evaluation of oral soft and hard tissue health for each enrolled subject at the conclusion of the study.
2.3. Microbiological Analysis
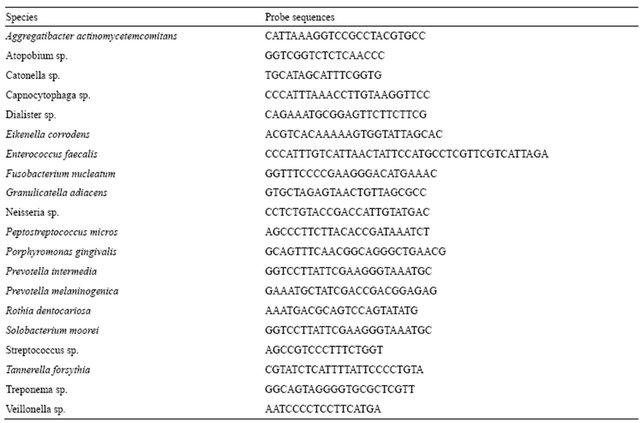
The dorsal tongue bacterial samples were dispersed by vortexing at maximal setting for 1 min. The number of tongue surface bacteria was assayed by phase-contrast microscopy as previously described [18]. Dot-blot hybridization was used to enumerate a panel of 20 target oral microorganisms (Table 1) utilizing digoxigenin-11- ddUTP labeled oligonucleotide probes (Genius 5, Boehringer Mannheim, Indianapolis, IN). Dorsal tongue surface samples were blotted onto charged nylon membranes (Hybond-N, Amersham, Arlington Heights, IL) using 10× standard salt phosphate EDTA buffer (SSPE) and hybridized with the digoxigenin-labeled oligonucleotide probes at the appropriate temperature for each probe. Known amounts of each microorganism were included in the dot blot assays as standards to determine the number of target microorganisms in each sample with results reported as log counts per ml of sample.
All chemicals were analytical grade or better. Buffers and saline were obtained from Invitrogen, Carlsbad, CA. Reagents for nucleic acid blotting and labeling were obtained from Boehringer Mannheim, Indianapolis, IN and Amersham, Arlington Heights, IL, respectively.
2.4. Statistical Analysis
Clinical, microscopic and microbiological results from the study were each evaluated by paired t-tests with p values of <0.05 reported as significant. Analyses were conducted with JMP software, Cary, NC.
Table 1. Target microorganisms and probe sequences.
3. RESULTS
The demographic characteristics of subjects enrolled in this study are presented in Table 2. All 14 subjects completed the study—five males and nine females—with a mean age of 55.3 years.
Figure 1 shows the effects of treatment on organoleptic scores and tongue coating index. Use of the triclosan/copolymer toothpaste and tongue cleaner resulted in significant reductions in each clinical parameter compared to their corresponding baselines (p < 0.0001). The mean organoleptic score at baseline was 4.14 and fell to 1.43 after 28 days. The mean tongue coating index score was 3.85 at baseline and fell to 1.42 after 28 days.
Scores from the halimeter evaluations are shown in Figure 2. The mean halimeter value at baseline was 423 and fell to 94 after 28 days. Statistical analysis demonstrated significant reductions for halimeter scores compared to baseline (p < 0.0001).
Phase-contrast microscopy was used to enumerate microorganisms on the dorsal tongue surface at baseline and at the post-treatment evaluation (Figure 3). Results (log counts/ml) indicated that compared to baseline, the post-treatment samples demonstrated significantly fewer microorganisms (p < 0.001). Post-treatment samples demonstrated a greater than 70% reduction in the number of microorganisms.
Dot-blot assays for a panel of target microorganisms (Table 1) in samples from the dorsal tongue surface at baseline and at the post-treatment evaluation are shown in Figures 4(a)-(c). There were significant reductions after 28 days in the number of target pathogens including the periodontal pathogens A. actinomycetemcomitans (p < 0.001), P. gingivalis (p < 0.0001), P. intermedia (p < 0.01), P. melaninogenica (p < 0.02), and T. forsythia (p < 0.001), microorganisms associated with gingivitis such as F. nucleatum (p < 0.01) and veillonella (p < 0.01), and microorganisms associated with endodontitic and other oral infections such as Enterococcus faecalis (p < 0.003), neisseria (p < 0.008), P. micros (p < 0.0007), and streptococci (p < 0.03). S. moorei, a bacterium previously shown to be associated with halitosis demonstrated significant reduction from baseline following treatment (p < 0.04). All other target microorganisms also demonstrated significant reductions at the post-treatment evaluation compared to baseline (p < 0.05).
4. DISCUSSION
Clinical indices for halitosis such as organoleptic measures and tongue coating indicies are frequently used to evaluate the efficacy of tongue cleaning or the effects of
Table 2. Subject demographics.
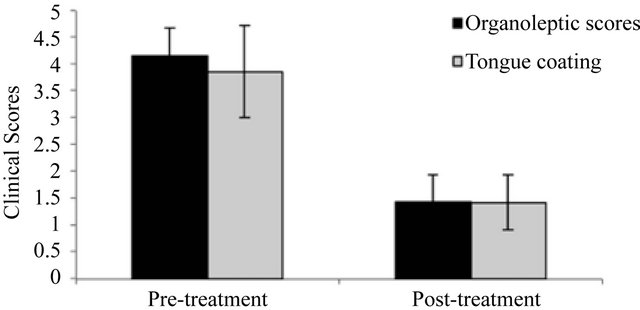
Figure 1. The effect of treatment on organoleptic scores and tongue coating index.
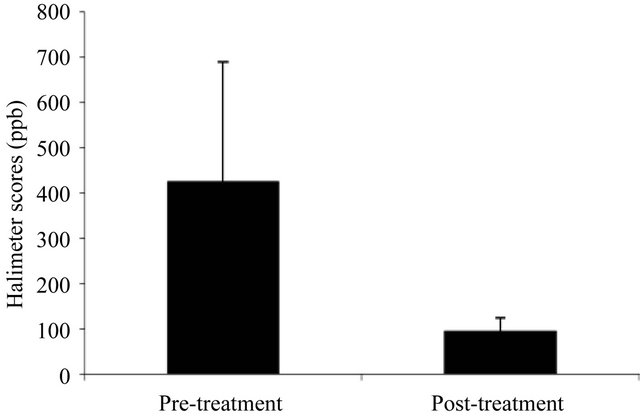
Figure 2. The effect of treatment on mouth air sulfur levels as determined by Halimeter.
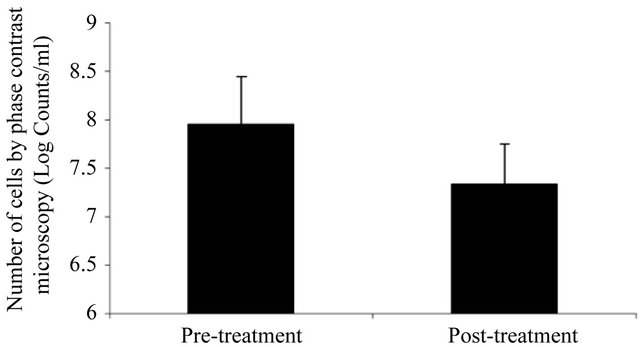
Figure 3. Total bacterial numbers from dorsal tongue samples assayed by phase contrast microscopy.
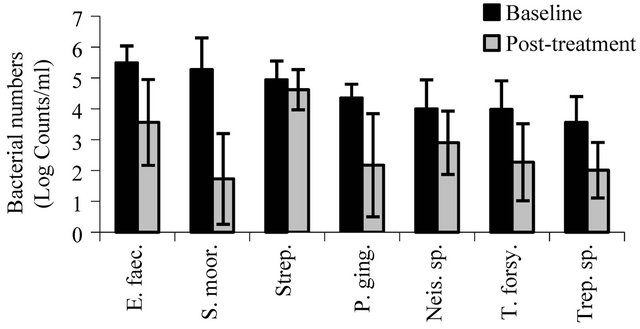 (a)
(a)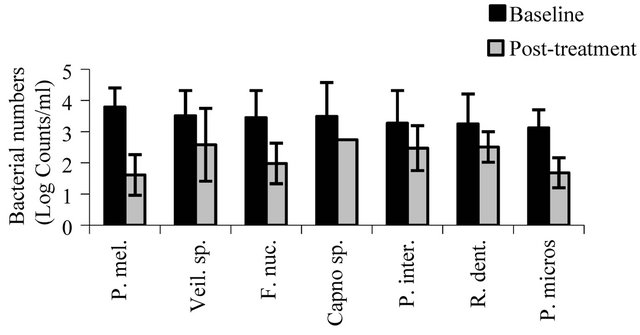 (b)
(b)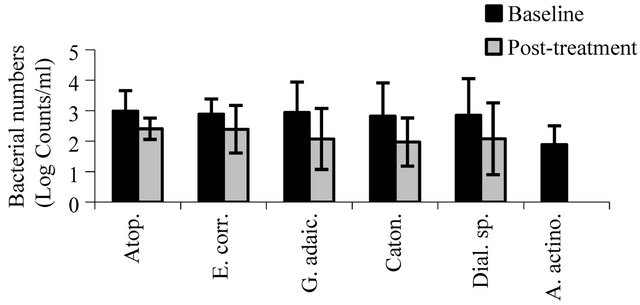 (c)
(c)
Figure 4. The effect of treatment on oral bacteria. Results represent the mean ± standard deviation at baseline and posttreatment (log counts/ml). All reductions from baseline to posttreatment are statistically significant.
oral hygiene formulations on oral malodor [4,9]. Fewer studies use more objective measures such as measurement of VSC’s to assess halitosis. Although it has been determined that tongue microorganisms are important sources of oral malodor, few studies have examined the effect of oral hygiene on the numbers and kinds of tongue surface microorganisms. Even fewer studies have used both clinical measures of halitosis together with measures of VSC’s and microbiological analysis of tongue surface microorganisms. This investigation utilized a variety of widely accepted clinical and microbiological assessments to evaluate the effects of oral hygiene with an antimicrobial dentifrice [18] in conjunction with a toothbrush designed with a tongue cleaner on oral halitosis.
Amongst olfactory techniques for malodor assessments, the organoleptic method has a long history of applications to both treatment and research [2,9,17]. Although widely considered a gold standard, malodor judges need training and calibration [16]. While organoleptic methods are subjective requiring qualified judges, the sensitivity of the human nose and its ability to detect 10,000 odoriferous compounds supports the value of this method [19]. Despite these limitations, this method is widely utilized to evaluate breath malodor. At baseline, mean organoleptic scores for subjects enrolled in the present study was >4 with a corresponding mean halimeter score of >250 ppb and a tongue coating score of ≈4. Together these parameters defined the study population as having significant oral halitosis.
The efficacy of the treatment protocol in eliminating oral halitosis—twice daily toothbrushing with a triclosan/ copolymer dentifrice and brushing the dorsal and lateral surfaces of the tongue for at least ten seconds with the tongue cleaner—was clearly demonstrated in these subjects. All subjects responded to the treatment with significant reductions in the organoleptic scores—average organoleptic scores were 1.4 representing values at the lower end of the 5-point scale and indicating that these subjects no longer had oral halitosis [9,20]. While the specific contribution of either the triclosan/copolymer dentifrice or the tongue cleaner in eliminating halitosis cannot be defined from this study, both treatments are known to reduce oral bacteria colonizing the tongue [15, 17].
Corresponding to the organoleptic assessments, the halimeter data also demonstrated significant reductions from baseline. However, in contrast to the subjective organoleptic assessments, the halimeter provides quantitation of three volatile sulfur compounds (VSC’s)- dimethyl sulfide ((CH3)2S), hydrogen sulfide (H2S), and methyl mercaptan (CH3SH). Limitations of the halimeter include the need for frequent calibration and decreased sensitivity for dimethyl sulfide, the more odoriferous of the three target VSC’s [21]. Furthermore, it is unclear from the literature what the upper range of halimeter scores is associated with “good” breath [20,22]. Investigators have utilized values from 75 to 110 ppb as normal [22,23] with values below 110 ppb recently recommended as normal and associated with superior oral hygiene [22]. On average, halimeter values for this cohort demonstrated a reduction from pre-treatment levels of 329 ppb to less than 100 ppb at the post-treatment evaluations and correspond with values reported for good breath.
Anatomic features of the tongue surface include a papillary structure with numerous irregularities including grooves, crypts, fissures and depapillated areas. Surface irregularities along with differences in redox potential and an abundance of nutrients, epithelial cells and other debris within these regions, which are frequently sheltered from the effects of oral hygiene or the flushing actions of saliva, favor microbial adhesion and growth [24]. Periodontal disease and systemic factors increase tongue coating but the mechanism that leads to tongue coating amongst healthy individuals remains unknown. In this study, numerical values for the tongue coating index paralleled organoleptic values and demonstrated similar post-treatment reductions compared to baseline. Previous studies suggest that there are transient reductions in oral malodor following removal of tongue coating [25] by virtue of reduced VSC levels and altered H2S/CH3SH ratios. In the current study, subjects demonstrated a considerable reduction in oral malodor during the posttreatment evaluations that were conducted 12 hours after oral hygiene.
Microbiological studies of tongue surface microorganisms have utilized microscopy, bacterial culture and nucleic acid-based methods [2,17]. Previous studies with phase contrast microscopy, for example, demonstrate daily and intra-individual variations of tongue surface microorganisms over the course of a month [17]. In comparative terms, the tongue represents an understudied organ compared to the greater emphasis on treatments for the hard dental tissues and supporting anatomical regions. The tongue is estimated to be the largest niche for oral microorganisms [17] with approximately one-third of the microbial population [2,11] that may populate other intra-oral sites [9,17]. In the present investigation, phase-contrast microscopy of tongue samples demonstrated a post-treatment reduction of 70% compared to baseline. These results indicate an overall reduction for the entire microbial load of tongue surface samples and correspond to the post-treatment reductions in tongue coating scores and halitosis.
Microbial culture-based analyses presently indicate 82 different types of microorganisms on the tongue including anaerobic microorganisms which metabolize dietary or host derived components to produce VSCs, fatty acids and other malodorous compounds. Large densities of evaluated organisms including periodontal pathogens and S. moorei, an organism found in halitosis [26], were observed in the baseline samples by dot-blot hybridization assay using specific oligonucleotide probes. Advantages of this assay include analysis of multiple microorganisms within a small sample volume. Another advantage of this assay is the ability of the probe to bind multiple genetic types of the target organism. The triclosan/copolymer dentifrice and tongue cleaner treatment protocol resulted in a greater than 1 log reduction (>90%) in the number of P. gingivalis, T. forsythia, P. melaninogenica, veillonella, fusobacteria and P. micros on the dorsal tongue surface. S. moorei, the microorganism prevalent in halitosis subjects, demonstrated a greater than 3 log reduction representing a greater than 99.9% reduction in this population. Post-treatment reductions in the number of these microorganisms are consistent with the decreased microbial load found by microscopy and reductions in clinical measures of halitosis.
Treatment to reduce microbial load on the tongue as well as other areas in the oral cavity such as the teeth have benefits in addition to eliminating oral halitosis. For instance, the metabolic components of bacteria responsible for bad breath i.e. the VSCs, are reportedly toxic to gingival cells and may facilitate progression of periodontal disease [1]. These features of VSCs are of greater significance amongst those with periodontal disease and the elderly who are prone to tongue coating [16].
In conclusion, this study used clinical evaluations together with microbiological analyses of tongue surface bacteria to examine a treatment protocol for oral halitosis. Accordingly, the study provides both clinical and microbiological rationales for the post-treatment reductions seen following use of the triclosan/copolymer toothpaste in conjunction with mechanical oral hygiene and tongue brushing with a specifically designed toothbrush with a tongue cleaner.
![]()
![]()
REFERENCES
- Scully, C. and Greenman, J. (2008) Halitosis (breath odor). Periodontology, 48, 66-75. doi:10.1111/j.1600-0757.2008.00266.x
- Roldán, S., Herrera, D. and Sanz, M. (2003) Biofilms and the tongue: Therapeutical approaches for the control of halitosis. Clinical Oral Investigations, 7, 189-197. doi:10.1007/s00784-003-0214-7
- Amiraian, D.E. and Sobal, J. (2009) Dating and eating: Beliefs about dating foods among university students. Appetite, 53, 226-232. doi:10.1016/j.appet.2009.06.012
- Rayman, S. and Almas, K. (2008) Halitosis among racially diverse populations: An update. International Journal of Dental Hygiene, 6, 2-7. doi:10.1111/j.1601-5037.2007.00274.x
- Quirynen, M., Dadamio, J., Van den Velde, S., De Smit, M., Dekeyser, C., Van Tornout, M. and Vandekerckhove, B. (2009) Characteristics of 2000 patients who visited a halitosis clinic. Journal of Clinical Periodontology, 36, 970-975. doi:10.1111/j.1600-051X.2009.01478.x
- Krespi, Y.P., Shrime, M.G. and Kacker, A. (2006) The relationship between oral malodor and volatile sulfur compound-producing bacteria. Otolaryngology-Head and Neck Surgery, 135, 671-676. doi:10.1016/j.otohns.2005.09.036
- Porter, S.R. and Scully, C. (2006) Oral malodour (halitosis). British Medical Journal, 333, 632-635. doi:10.1136/bmj.38954.631968.AE
- Bornstein, M.M., Kislig, K., Hoti, B.B., Seemann, R. and Lussi, A. (2009) Prevalence of halitosis in the population of the city of Bern, Switzerland: A study comparing selfreported and clinical data. European Journal of Oral Sciences, 117, 261-267. doi:10.1111/j.1600-0722.2009.00630.x
- Van den Broek, A.M., Feenstra, L. and de Baat, C. (2008) A review of the current literature on management of halitosis. Oral Diseases, 14, 30-39.
- Haraszthy, V.I., Zambon, J.J., Sreenivasan, P.K., Zambon, M.M., Gerber, D., Rego, R. and Parker, C. (2007) Identification of oral bacterial species associated with halitosis. Journal of the American Dental Association, 138, 1113- 1120.
- Kazor, C.E., Mitchell, P.M., Lee, A.M., Stokes, L.N., Loesche, W.J., Dewhirst, F.E. and Paster, B.J. (2003) Diversity of bacterial populations on the tongue dorsa of patients with halitosis and healthy patients. Journal of Clinical Microbiology, 41, 558-563. doi:10.1128/JCM.41.2.558-563.2003
- Riggio, M.P., Lennon, A., Rolph, H.J., Hodge, P.J., Donaldson, A., Maxwell, A.J. and Bagg, J. (2008) Molecular identification of bacteria on the tongue dorsum of subjects with and without halitosis. Oral Diseases, 14, 251-258. doi:10.1111/j.1601-0825.2007.01371.x
- Blinkhorn, A., Bartold, P.M., Cullinan, M.P., Madden, T.E., Marshall, R.I., Raphael, S.L. and Seymour, G.J. (2009) Is there a role for triclosan/copolymer toothpaste in the management of periodontal disease? British Dental Journal, 207, 117-125. doi:10.1038/sj.bdj.2009.669
- Gunsolley, J.C. (2006) A meta-analysis of six-month studies of antiplaque and antigingivitis agents. Journal of the American Dental Association, 137, 1649-1657.
- Fine, D.H., Furgang, D., Markowitz, K., Sreenivasan, P.K., Klimpel, K. and De Vizio, W. (2006) The antimicrobial effect of a triclosan/copolymer dentifrice on oral microorganisms in vivo. Journal of the American Dental Association, 137, 1406-1413.
- Rosenberg, M. and McCulloch, C.A.G. (1992) Measurement of oral malodor: Current methods and future prospects. Journal of Periodontology, 63, 776-782. doi:10.1902/jop.1992.63.9.776
- Danser, M.M., Gómez, S.M. and Van der Weijden, G.A. (2003) Tongue coating and tongue brushing: A literature review. International Journal of Dental Hygiene, 1, 151- 158. doi:10.1034/j.1601-5037.2003.00034.x
- Zambon, J.J., Reynolds, H.S., Dunford, R.G., DeVizio, W., Volpe, A.R., Berta, R., Tempro, J.P. and Bonta, Y. (1995) Microbial alterations in supragingival dental plaque in response to a triclosan-containing dentifrice. Oral Microbiology and Immunology, 10, 247-255. doi:10.1111/j.1399-302X.1995.tb00150.x
- Hatt, H. (2004) Molecular and cellular basis of human olfaction. Chemistry & Biodiversity, 1, 1857-1869. doi:10.1002/cbdv.200490142
- Vandekerckhove, B., Van den Velde, S., De Smit, M., Dadamio, J., Teughels, W., Van Tornout, M. and Quirynen, M. (2009) Clinical reliability of non-organoleptic oral malodour measurements. Journal of Clinical Periodontology, 36, 964-969.
- Furne, J., Majerus, G., Lenton, P., Springfield, J., Levitt, D.G. and Levitt, M.D. (2002) Comparison of volatile sulfur compound concentrations measured with a sulfide detector vs. gas chromatography. Journal of Dental Research, 81, 140-143. doi:10.1177/154405910208100211
- Nalcaci, R. and Baran, I. (2008) Oral malodor and removable complete dentures in the elderly. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology, 105, e5-e9. doi:10.1016/j.tripleo.2008.02.016
- Yaegaki, K. and Sanada, K. (1992) Volatile sulfur compounds in mouth air from clinically healthy subjects and patients with periodontal disease. Journal of Periodontal Research, 27, 233-238. doi:10.1111/j.1600-0765.1992.tb01673.x
- DeBoever, E.H. and Loesche, W.J. (1995) Assessing the contribution of anaerobic microflora of the tongue to oral malodor. Journal of the American Dental Association, 126, 1384-1393.
- Seemann, R. (2006) Tongue scrapers may reduce halitosis in adults. Evidence-Based Dentistry, 7, 78. doi:10.1038/sj.ebd.6400433
- Haraszthy, V.I., Gerber, D., Clark, B., Moses, P., Parker, C., Sreenivasan, P.K. and Zambon, J.J. (2008) Characterization and prevalence of Solobacteriummoorei associated with oral halitosis. Journal of Breath Research, 2, Article ID: 017002. doi:10.1088/1752-7155/2/1/017002
NOTES
*Corresponding author.

