Advances in Biological Chemistry
Vol.4 No.1(2014), Article ID:43307,5 pages DOI:10.4236/abc.2014.41012
Essential metal Zn in sponge Callyspongia aerizusa from Spermonde Archipelago
![]()
1Doctoral Programme, Chemistry Department, Hasanuddin University, Makassar, Indonesia
2Chemistry Department, Hasanuddin University, Makassar, Indonesia
3Netherlands Center for Biodiversity, Naturalis, Leiden,The Netherlands
Email: *lylamety@yahoo.com
Copyright © 2014 Lydia Melawaty et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In accordance of the Creative Commons Attribution License all Copyrights © 2014 are reserved for SCIRP and the owner of the intellectual property Lydia Melawaty et al. All Copyright © 2014 are guarded by law and by SCIRP as a guardian.
Received 13 January 2014; revised 16 February 2014; accepted 22 February 2014
KEYWORDS:Essential Metal; Sponge; Callyspongia aerizusa; Filter Feeder; Spermonde Archipelago
ABSTRACT
Spermonde Archipelago has been found to have a high sponge biodiversity. Sponges that have feeding behavior as filter feeder can be exposed to heavy metals, including Zn metal. Zn is an essential metal that is physiologically needed by sponge to catalyze chemical reactions and generally operates on enzymes. A study on Zn pollution level in Spermonde Archipelago from zone I to Zone IV was conducted by analyzing Zn concentration in sponges, sediments, and seawater. Physicochemical parameters measurement in Spermonde Archipelago indicates that Spermonde Archipelago meets the requirements for sponge growth. Callyspongia aerizusa can be used as metal pollution indicator because this sponge can accumulate heavy metal Zn in high level (99.30 mg/kg dry weight). This sponge was derived from Kondingareng Lompo island at zone III. In addition, Kodingareng Lompo island waters had the highest Zn concentration compared to other islands. Sediments from Lae-Lae island at zone I had the highest Zn metal concentration. Lae-Lae island is the nearest island to Makassar coastal line.
1. INTRODUCTION
Porifera are highly vulnerable to metal contamination because they filter most of the water (0.002 - 0.84 mL/ second/cm3 sponge tissue). These filter feeder invertebrates live in sublittoral rock biotopes where they are exposed to various chronic contamination. Heavy metals can endanger the sublittoral biota at various levels, depending on the metal concentration and the physiology of organism. Even at low concentration, metals can result in adverse effects in the early life of sponge such as in larva and juvenile phase [1].
Sponges have much higher filtering capacity compared to bivalves and are capable of processing thousands of liters of seawater per hour. Sponge community is sensitive to disorders but can be stable in very long time, making them suitable for metal biomonitoring. The ability of sponge in accumulating heavy metals depends on the metal types and the sponge species [2,3].
Molecular and biochemical mechanisms have been developed to counter the metal adverse effects. Long exposure to metal has been found to alter the sponge health, including its immunity system [4,5].
The waste disposal system of Makassar city involves the city channels (canals) and rivers flowing to the sea. Most of the waste products are derived from civil activities including households, hotels, restaurants, agricultures, and smaller industries in the vicinity of rivers/ canals or the coast. These waste products contain heavy metals that produce accumulation in waters environment, marine biota, and sediments.
Spermonde Archipelago is divided into 4 zones, stretching from North to South and comprising small islands groups with white sandy beast and coral reef ecosystem that have been a habitat to marine biota. The first zone (zone I) is the nearest zone from main coastal line of Sulawesi Island, with average depth of 10 m. The second zone (zone II) distances about 5 km from Sulawesi terrestrial, with an average depth of 30 m. The third zone (zone III) starts from 12.5 km off the Sulawesi coast with depth of 20 - 50 m. The fourth zone or outer zone is the barrier reef zone and distances 30 km from mainland of Sulawesi. To the eastern side of these coral islands the depth ranges from 40 to 50 m. While to the western side, the depth is more than 100 m.
This study was aimed to determine the distribution of heavy metal Zn in sponge, sponge-associated sediments, and seawater in coastal area of Spermonde (Figure 1) from zone I (Lae-Lae), zone II (Samalona, Barrang Lompo, and Barrang Caddi), zone III (Kodingareng Keke and Kodingareng Lompo) and zone IV (Kapoposang). Findings of this study are expected to be a great contribution to the biomonitoring of heavy metal Zn level in Spermonde coastal area.
2. EXPERIMENTAL
2.1. Determination of Essential Metal Zn in Sponge [6]
Each of the sponge samples was divided into two parts: 1 part for treatment in skeleton and 1 part for total treatment. The dry sample of the sponge was weighted 0.5 g carefully in beaker glass, and then was added with 5 mL HNO3 and heated at 150˚C for 2 hours. After cooling at room temperature, samples were added into measuring flask 25 mL and its volume was rounded with aquabidest and shaken until homogeneous. The solution was filtered using Whatman filter paper and ready for analysis with Inductively Couple Plasma—Optical Emission Spectrophotometer (ICP-OES) Optima 2000 DV Perkin Elmer.
2.2. Determination of Essential Metal Zn in Sediment [7]
Five grams of dried sediment sample was destructed in teflon beaker with HNO3 at temperature of ±100˚C for 8 hours. After cooling at room temperature, sample was then added into measuring flask 100 mL and its volume was rounded by aquabidest and was shaken until homogenous. The solution was filtered using Whatman filter paper and ready for analysis with ICP-OES Optima 2000 DV Perkin Elmer.
2.3. Determination of Heavy Metal Zn in Sea Water [8]
Sea water with volume of 250 mL was taken and then the pH was adjusted to 3 - 3.5 with HNO3 or NaOH. The solution was poured into teflonseparaing funnel and then added with 2.5 mL Ammonium Pirolydine Dithyocarbamate (APDC) 4%, shaken for ±1 minute. 25 mL of Methyl Isobuthyl Keton (MIBK) was added into it, shaken for ±1 minute. It was left in place for ±5 minutes, and the organic phase was harvested. The organic phase was then flushed with 10 mL aquadest and then left in place for ±5 minutes. Again, the organic phase formed was then harvested and added with 0.25 mL concentrated HNO3 and was shaken. It was left in place for 20 minutes and then added with 9.75 mL aquadest, shaken for ±1 minute. The organic phase was harvested again and stored in polyethilene vial. The heavy metal Zn level was measured using ICP-OES Optima 2000 DV PerkinElmer.
3. RESULTS AND DISCUSSION
The conditions of Kodingareng Lompo and Barrang Caddi islands during the sampling period can be seen from Table 1.
The physicochemical parameters have been found to meet the requirements for sponge growth. Optimum temperature for sponge growth is 25˚C - 29˚C. Bioaccumulation depends on the temperature, where the accumulated metal concentration increases with temperature. The temperature effect presumably involves ion transport
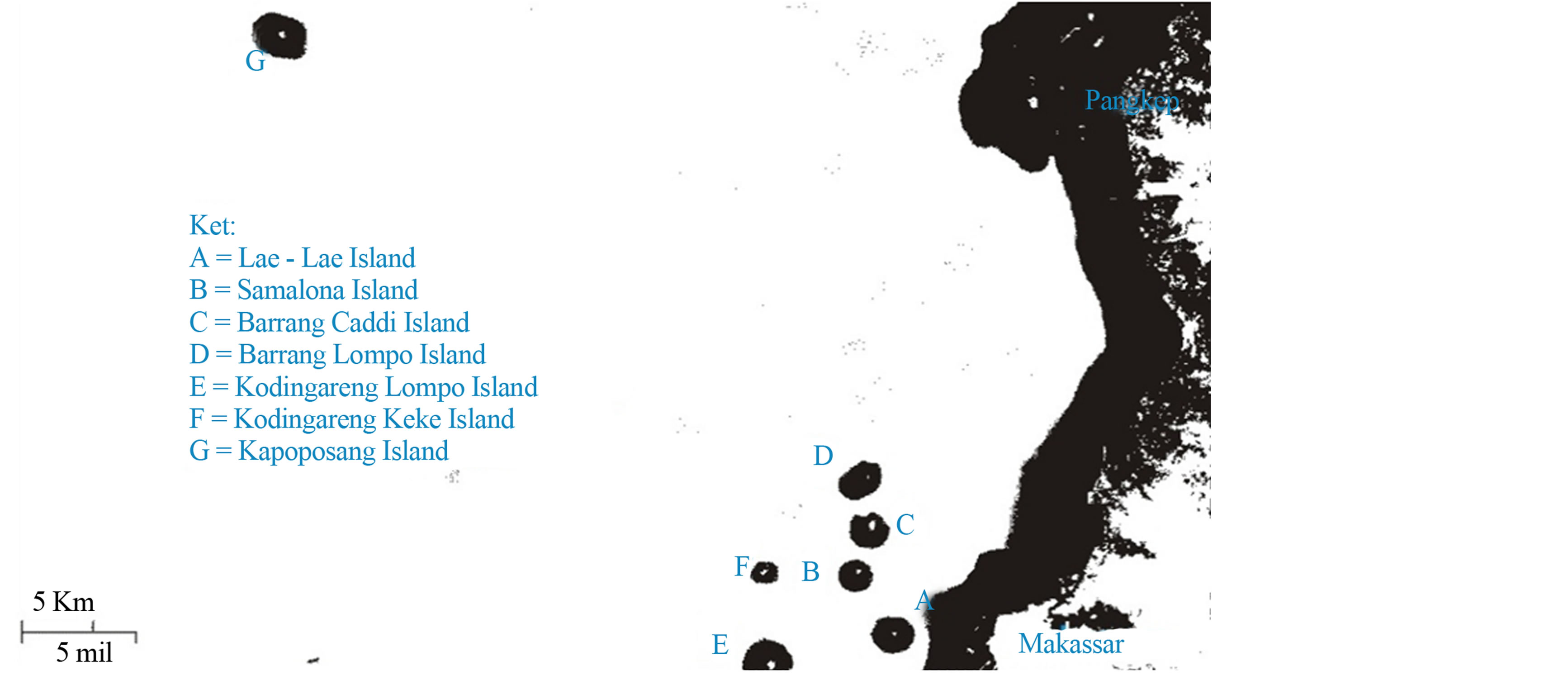
Figure 1. The map of sampling locations in Spermonde Archipelago [10].

Table 1. Conditions of waters for sponge sampling.
mechanism on membrane surface. Bioaccumulation of zinc depends on temperature only when associated to water flow with very low salt content [9].
Salinity of waters of the three islands also met the requirement for sponge growth (29‰ - 36‰). Generally, biotic metal concentrations increase with decreased salt level. In low salt level condition, concentration of the directly absorbed free metal ions such as Zn2+ is maximum. Different metal levels can cause different metal absorption acceleration due to physiological alterations in the organism itself such as water drinking rate or water filtration rate [9].
The pH of water environment that meets the growing requirement of sponge ranges from 6 to 8 and has influence on the new metal species formation in water. The effect of pH depends also on the type of metal [9].
Sponge Callyspongia aerizusa derived from Kodingareng Lompo island (Figure 1) was able to accumulate higher heavy metal Zn (99.3 mg/kg) compared to Clathria reinwardtii and Melophlussarassinorum. Callyspongia aerizusa was capable of accumulating Zn twice higher than Melophlussarassinorum and once higher than Clathria reinwardtii (Figure 2).
Single factor ANOVA analysis indicated a significant (F (9.849) > Ftable (2.759)) Zn concentration difference in body, skeleton, seawater, and sediment of Spermonde Archipelago. Correlation test between body Zn and skeleton Zn(r (0.668) < r table (0.811)) and between seawater Zn and sediment Zn(r (−0.112) < r table (0.811)) indicated negative correlations. Body Zn and seawater Zn alterations were followed by skeleton Zn and sediment Zn alterations with opposite directions.
Zinc (Zn) is an essential metal required in trace level for an organism growth. Zn is widely used in paint, ceramic, rubber, wood, and dye industries. The use of Zn as an agent in anti-fouling paint is one of the most important anthropogenic source in seawater [9]. Maximum Zn concentration derived from anthropogenic activities in biota, including sponge, is not more than 5 × 10−2 mg/L [11].
Metals are not commonly found in isolated form in nature. The presence of copper and manganese can reduce the bioaccumulation of Zn. This is probably due to presence of semi-selective binding site that can be occupied by specific metals, depending on their concentration relative to other metals that are also capable of occupying the binding site [9].
The relative abundance of essential metals in a living organism reflects the required level to maintain biochemical functions. When the essential metals uptake exceeds the relative abundance, homeostatic mechanism controls the level of the metals in the body and their transfer to tissues. When the uptake is excessive, the homeostatic mechanism is inhibited and bioaccumulation ensues because the uptake rate exceeds the reduction rate [12].
Silicase enzyme has been identified in marine sponge S. domuncula. Silicase is able to depolimerize amorphous silica [13]. Expression of gene that code for this silica-catabolic enzyme increases significantly as a response to higher silicone concentration, such as siicateine and collagen expression. The obtained cDNA silicase polypeptide is very similar to carbonic anhydrase, a family of zinc metal enzyme. The three characteristic residues of histidine for these enzymes bind to a zinc ion (Figure 3) [14].
It was assumed that the reaction mechanism of the sponge enzyme is analogue to other zinc-dependent enzymes involved in ester hydrolysis. Zinc ion (Lewis acid) interacts with water (Lewis base). The hydroxide ion formed by water molecule splitting is bind to zinc ion. This hydroxide ion then launches a nucleophylic attack on a silicone atom of polymer silicate. In the next step, zinc complex is bound to silicone in the presence of oxygen bond breaking in polymeric silicate. The intermediate zinc-bound silicate is then hydrolyzed by water, resulting in silicate acid release and regeneration of zincbound hydroxide [14].
Metal element is acquired by the aquatic biota in passive process with decreasing concentration gradient toward the tissue. Mechanism to counter the metal toxicity potential in sponge tissue involves elements absorption in the form of metallothionein or in the form of granules in water species tissue. Sponge uses regulation and detoxification processes presenting various capacities to ac-

Figure 2. Zn concentration in body, skeleton, seawater, and sediments.
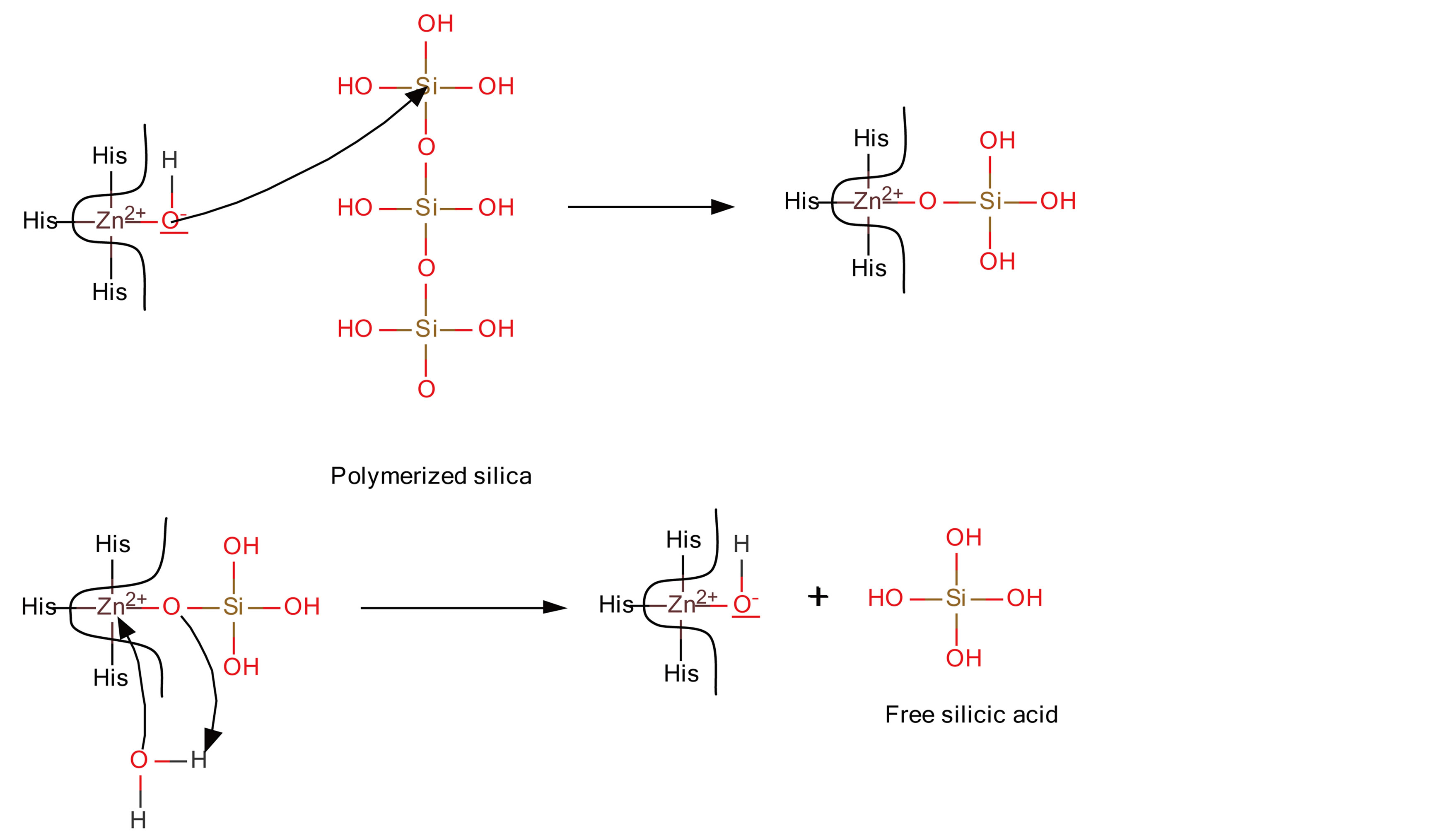
Figure 3. Proposed mechanism for silicase [13-15].
cumulate metals, from the weak to strong capacity. Trace metal accumulation by sponge depends on physicochemical properties of the metal, in addition to the organism itself and biological strategy for metal detoxification [16].
4. CONCLUSION
Callyspongia aerizusa can accumulate essential metal Zn in high concentration and is able to survive. This was due to the fact that essential metal Zn is required by silicase enzyme in the sponge to depolimerize amorphous silica in sponge. Single-factor ANOVA analysis indicated a significant Zn concentration difference in sponge body, sponge skeleton, seawater, and sediment among seven islands in Spermonde Archipelago.
ACKNOWLEDGEMENTS
Thank you to the Ministry of Education and Culture that has provided research grants BPPS.
REFERENCES
- Cebrian, E. and dan Uriz, M.J. (2007) Do heavy metals play an active role insponge cell behaviour in the absence of calcium? Consequences in larval settlement. Journal of Experimental Marine Biology and Ecology, 346, 60-65. http://dx.doi.org/10.1016/j.jembe.2007.02.010
- Vogel, S. (1977) Current-induced flow through living sponges in nature. Proceedings of the National Academy of Sciences of the United States of America, 74, 2069- 2071.
- Cebrian, E., et al. (2003) Sublethal effects of contamination on the Mediterranean sponge Crambecrambe: Metal accumulation and biological responses. Marine Pollution Bulletin, 46, 1273-1284. http://dx.doi.org/10.1016/S0025-326X(03)00190-5
- Saby, E., et al. (2009) In vitro effects of metal pollution on Mediterranean sponges: Species-specific inhibition of 2,5-oligoadenylatesynthetase. Aquatic Toxicology, 94, 204- 210. http://dx.doi.org/10.1016/j.aquatox.2009.07.002
- Carballo, J.L. and danNaranjo, S. (2002) Environmental assessment of a large industrial marine complex based on a community of benthic filter feeders. Marine Pollution Bulletin, 44, 605-610. http://dx.doi.org/10.1016/S0025-326X(01)00295-8
- Müller, et al. (1998) Accumulation of cadmium and zinc in the marine sponge Suberites domuncula and its potential consequences on single-strand breaks and on expression of heat-shock protein: A natural field study. Marine Ecology Progress Series, 167, 127-135. http://dx.doi.org/10.3354/meps167127
- Loring, D.H. and Rantala, R.T.T. (1992) Manual for the geochemical analyses of marine sediments and suspended particulate matter. Earth-Science Reviews, 32, 235-316. http://dx.doi.org/10.1016/0012-8252(92)90001-A
- Magnusson, W. and Westerlund, S. (1981) Solvent extraction procedures combined with back-extraction for trace metal determinations by atomic absorption spectrometry. Analytica Chimica Acta, 131, 63-72. http://dx.doi.org/10.1016/S0003-2670(01)93534-2
- Connell, D.W. (1990) Xenobiotic compound bioaccumulation. UI-Press, Jakarta.
- Google map. https:/maps. google.co.id/maps
- Eisler, R. (1993) Report of contaminant hazard review. Patuxent Wildlife Research Center, Maryland.
- Connell, D.W. and dan Miller, G.J. (1995) Pollution chemistry ecotoxicology. UI-Press, Jakarta.
- Schröder, H.C., et al. (2003) Silicase, an enzyme which degrades biogenous amorphous silica: Contribution to the metabolism of silica deposition in the demosponge Suberites domuncula. Progress in Molecular and Subcellular Biology, 33, 250-268.
- Schröder, H.C., et al. (2007) Silicateins, silicase and spicule-associated proteins: Synthesis of demosponge silica skeleton and nanobiotechnological applications. Porifera Research: Biodiversity, Innovation and Sustainability, Série Livros 28, Museu Nacional, Rio de Janeiro, 581- 592.
- Ehrlich, H., et al. (2010) Modern views on desilification: Biosilica and abiotic silica dissolution in natural and artificial environments. Chemical Reviews, 110, 4656-4689. http://dx.doi.org/10.1021/cr900334y
- Phillips, D.J.H. (1995) The chemistries and environmental fates of trace metals and organochlorines in aquatic ecosystems. Marine Pollution Bulletin, 31, 193-200.
NOTES
*Corresponding author.

