International Journal of Organic Chemistry
Vol. 2 No. 1 (2012) , Article ID: 17863 , 4 pages DOI:10.4236/ijoc.2012.21012
Comparative Efficacy of Polyamine-Based Scavenger Resins
Bharavi Laboratories (P) Ltd., Bangalore, India
Email: *ravindranath@bharavilab.com
Received November 30, 2011; revised January 19, 2012; accepted February 2, 2012
Keywords: Acids; Acid Chlorides; Aldehydes; Isocyanates; Ethylenediamine; Diethylenetriamine; Triethylenetetramine; Tris(2-aminoethyl)amine; Scavenger Resins; Combinatorial Synthesis
ABSTRACT
Polyamine-based scavenger resins containing 2, 3 and 4 nitrogen atoms have been prepared and their comparative efficacy to scavenge appropriate electrophilic chemicals (acids, acid chlorides, isocyanates and aldehydes) from solutions has been studied. As expected, the scavenging efficiency is directly proportional to the number of nucleophic nitrogens present on the resin. The results have been compared with the performance of the popular scavenger resin, namely, tris(2-aminoethyl)amine resin, to conclude that the low-cost polyamine resins now prepared can be conveniently used as effectively as the expensive commercial product.
1. Introduction
In almost all synthetic organic reactions, one has undesirable products in addition to the desired ones. These may be un-reacted raw materials, byproducts, excess reagents, catalysts and, of course, solvents. Occasionally, excess reagents are intentionally used to hasten the reactions or to force them to go to completion. These need to be removed at the end of the reaction by different methods including filtration, evaporation, crystallization and/or chromatography. Use of scavengers is a technique by which the undesired components are removed by interaction of the scavenger that carries an appropriate functionality which can react with the unwanted component(s). Scavenger resins, in which the reactive functionality is immobilized on a polymer, are widely used for the workup of solution phase reactions to remove excess reagents, substrates or byproducts, often by simple filtration. Use of scavenger resins is particularly critical in parallel synthesis or combinatorial chemistry since a large number of compounds is usually present and selective removal of some of the reagents and components is often required [1,2]. Sometimes, the scavenger resins are also used to conveniently isolate the desired product from the complex mixture of reactants and byproducts [3].
The polymer-bound polyamines help in removal of acids from the solution by forming the corresponding salts, acid chlorides by forming amides, carbonyl compounds by forming Schiff bases and isocyanates by forming ureas. The most popular scavenger resin of this class is tris(2-aminoethyl)amine (TAEA) linked to polystyrene-co-divinylbenzene (PS-DVB) resin through a methylene bridge [4] which is very expensive. In the present work, we developed a simple and improved method of preparation of the related polyamine-functionalized resins which are much less expensive and made a comparative study of their efficacy as scavenger resins. They are the ethylenediamine (EDA), diethylenetriamine (DETA) and triethylenetetramine (TETA) linked to PS-DVB resins, carrying 2, 3 and 4 amino groups respectively. These resins have been made in multi-kilogram scale and are now available commercially from Bharavi Laboratories.
2. Results
The known method of attaching tris(2-aminoethyl)amine to polystyrene-based resin is treatment of chloromethylated PS-DVB resin (Merrifield resin) with excess of the polyamine in dimethylformamide at elevated temperatures [3]. However, we found that this method invariably gave products that showed an intense band around 1660 cm–1 in the infrared spectrum indicating retention of di- methylformamide in the beads. Washing of the resin with different solvents failed to remove the band indicating some kind of affinity between the resin and the formamide moiety. Therefore, nitrogen analysis for determination of the amine content for estimation of the scavenging capacity of the product would be undependable. Alternative solvents for the reaction were therefore examined. For reactions involving the cross-linked polymers, one of the important requirements is the swelling property of the resin in the solvent used for the reaction. Solvents like dichloromethane, tetrahydrofuran, dioxane, etc., in which the Merrifield resin swells, could possibly be used for the reaction under investigation. Dichloromethane or chloroform was unsuitable as the reaction was very slow even at their reflux temperatures. Among the other solvents that could be used, 1,4-dioxane was considered as it swells the resins and does not contain nitrogen. Reaction of Merrifield resin with different chlorine loading (1 - 3 mmol Cl/g) was examined in dioxane at various temperatures ranging from ambient to 100˚C; use of dioxane at 60˚C under nitrogen atmosphere proved to be safe and optimum. The molar ratio of the active chlorine on the resin to the amines was varied from 1:1, 1:2, 1:5 and 1:10 to determine that the ratio of 1:5 allowed the completion of the reaction in 8 - 10 h at 60˚C. Though it is economical to make these polyamines with high amine loading, the possibility of cross-linking within the beads at higher chlorine loading would cause the resins to become rigid or brittle. Therefore, the Merrifield resin of moderate loading (1.6 and 2.1 mmol Cl/g) was used in dioxane (10 volumes relative to the resin) with 5 equivalents of the polyamine; stirring the mixture at 60˚C for 8 h under nitrogen yielded the desired products. The extent of the reaction was determined by chlorine estimation using Volhard method [5] for the residual chlorine on the resin. When the chlorine content on the resin was negligible, the resin was filtered, washed as given under Experimental Section (below Scheme 1) and dried over potassium hydroxide under vacuum. The elemental analysis for nitrogen indicated lower nitrogen content (mmol N/g) than theoretical value, indicating a certain degree of cross linking but without the loss of swelling characteristics and reactivity (Table 1). It may be noted that while the maximum amine loading in the tris(aminoethyl)amine (TAEA) resin given in the literature [4] was 3.2 mmol N/g starting from Merrifield resin of 1.7 mmol Cl/g, the amine content of the resin obtained under the present conditions was 4.2 mmol N/g both for the tris(2-aminoethyl)amine and the triethylenetetramine, starting from Merrifield resin of 1.6 mmol Cl/g.
Scavenging or quenching capacity of the polyamine resins:
The polyamine resins, bearing ethylenediamine [EDA]; diethylenetriamine [DETA] triethylenete tramine [TETA] and tris(2-aminoethyl)amine (TAEA), showed a similar type of scavenging property when tested with various reagents, namely, 2-chlorobenzoyl chloride, phenylisocyanate and p-toluenesulfonyl chloride in dichloromethane and hydrogen chloride in dioxane. All the polyamines scavenged 2-chlorobenzoyl chloride and p-toluenesulfonyl chloride completely from the reaction mixtures within 15 minutes when the three equivalents of the polyamine resin was used; only two equivalents of polyamine resin were needed to quench all the hydrogen chloride during the same period. 2.5 equivalents of the polyamine resin could scavenge the phenylisocyanate in 1 h. Thus, comparative experiments showed that all the polyamines have the same or similar type of scavenging property. Table 2 shows the data for complete quenching of phenylisocyanate (1 g; 8.4 mmol) in dichloromethane (20 ml) in 1 h.
The swelling properties for all the four polyamines in different solvents also showed similar behavior (Table 3). The numbers indicate the volume of the resin bed in ml per gram of the dry weight of the resin.
3. Conclusion
Since all the polyamine resins behave in a similar manner with respect to the swelling and scavenging properties, it could be concluded that, in place of the expensive tris (2-aminoethyl)amine resin, triethylenetetramine [TETA] resin or equivalent quantities of the other polyamines like ethylenediamine [EDA], diethylenetriamine [DETA], which are much cheaper, can be effectively used. Use of dioxane as solvent is preferred over dimethylformamide for accurate determination of the nitrogen content on the resin.
4. Experimental Section
Preparation of polyamine scavenger resins (General method).
Merrifield resin (100 g; 2.1 mmol Cl/g; 0.210 mol) was swelled in dioxane (900 ml) and the polyamine (1.05 mol) was added to the swelled chloromethylpolystyrene resin; the reaction mixture was stirred and heated at 60˚C for 8 h under nitrogen atmosphere. The completion of reaction was checked by estimating the chlorine by Volhard’s method which showed that the residual chlorine was nil. The resin was filtered and successively washed with methanol (200 ml), then with 10% triethylamine in dichloromethane (1 L), methanol (200 ml), water till the pH = 7 and finally with the methanol (3 × 100 ml). The resin was dried in the vacuum oven at 50˚C for 24 h over potassium hydroxide pellets. Nitrogen content of the resin was determined by elemental analysis.
Quenching or scavenging capacity of the polyamine resins.
Phenylisocyanate (1 g; 8.4 mmol) was dissolved in dichloromethane (20 ml) and ethylenediamine resin of 3.2 mmol N/g (6.4 g; 20.5 mmol) was added to the solution and stirred. After 30 minutes, 10 µL of the solution from the reaction mixture was injected into the gas chromatograph to find that the peak intensity of the reaction mixture had decreased by 60% when compared to initial
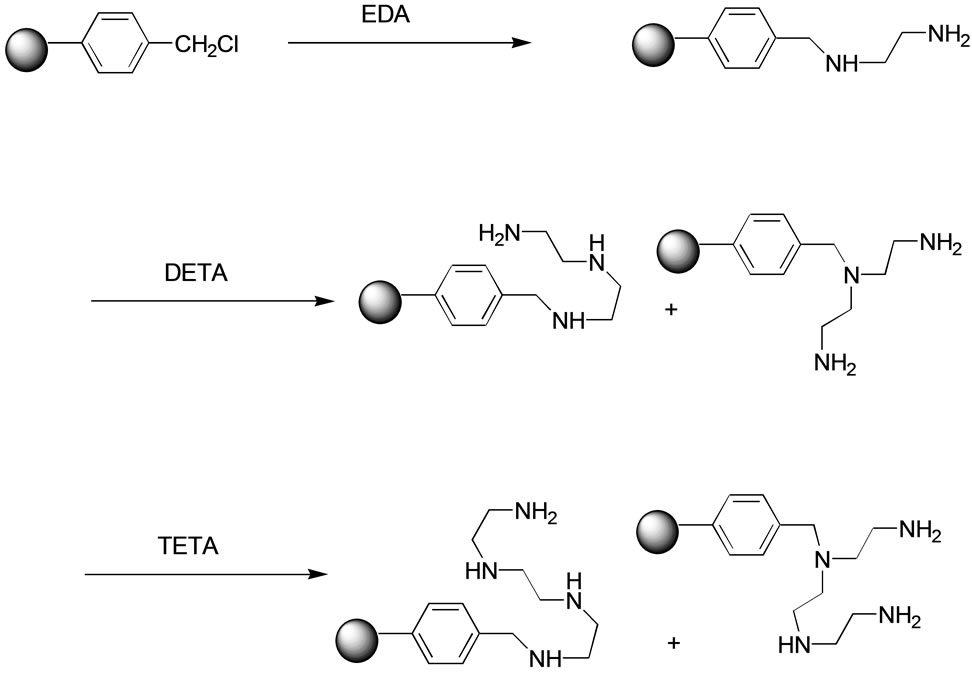
Scheme 1. Preparation of polyamine resins.

Table 1. Amine loading (mmol N/g) of different polyamine resins.

Table 2. Comparative quenching capacity of different polyamine resins toward phenylisocyanate (8.4 mmol).
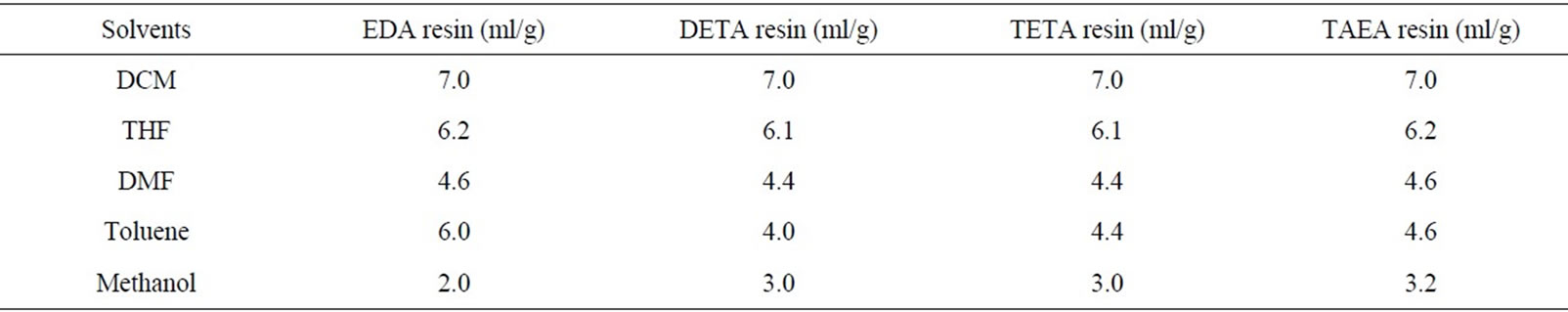
Table 3. Swelling property of four different polyamines 1 g = 1.6 ml/g dry weight.
concentration; on stirring for further 30 min, the gas chromatography of the sample showed no peak corresponding to the isocyanate. Therefore it was concluded that all the isocyanate is quenched by this polyamine within 1 h. Similar experiments were conducted with the other polyamine resins and data is summarized in Table 2.
The scavenging properties were similarly tested with different electrophilic reagents, namely, 2-chlorobenzoyl chloride and p-toluenesulfonyl chloride in dichloromethane and hydrogen chloride in dioxane. All the polyamines scavenged 2-chlorobenzoyl chloride and p-toluenesulfonyl chloride from the reaction mixtures within 15 minutes when the three equivalents of the polyamine resin was used; only two equivalents of polyamine resin were needed to quench all the hydrogen chloride during the same period.
REFERENCES
- J. C. Hodges, “Covalent Scavengers for Primary and Secondary Amines,” Synlett, Vol. 2000, No. 1, 2000, pp. 152-158. doi:10.1055/s-2000-6465
- J. G. Garcia, “Scavenger Resins in Solution Phase CombiChem,” Methods in Enzymology, Vol. 369B, 2003, pp. 391-412. doi:10.1016/S0076-6879(03)69021-X
- C. C. Tzschucke, C. Markert, W. Bannwarth, S. Rolter, A. Hebel and R. Haag, “Modern Separation Techniques for the Efficient Workup in Organic Synthesis,” Angewandte Chemie International Edition in English, Vol. 41, No. 21, 2002, pp. 3964-4000. doi:10.1002/1521-3773(20021104)41:21<3964::AID-ANIE3964>3.0.CO;2-3
- R. J. Booth and J. C. Hodges, “Polymer Supported Quenching Reagents for Parallel Purification,” Journal of the American Chemical Society, Vol. 119, No. 21, 1997, pp. 4882-4886. doi:10.1021/ja9634637
- O. Schales and S. S. Schales, “A Simple and Accurate Method for the Determination of Chloride in Biological Fluids,” Journal of Biological Chemistry, Vol. 141, No. 3, 1941, pp. 879-884.
NOTES
*Corresponding author.

