Paper Menu >>
Journal Menu >>
 Vol.2, No.1, 62-66 (2010) doi:10.4236/health.2010.21010 SciRes Copyright © 2010 Openly accessible at http://www.scirp.org/journal/HEALTH/ Health The central nucleus of amygdala is involved in tolerance to the antinociceptive effect of NSAIDs Merab G. Tsagareli1, Nana Tsiklauri1, Gulnazi Gurtskaia1, Ivliane Nozadze1, Elene Abzianidze2 1Beritashvili Institute of Physiology, Tbilisi, Georgia; tsagareli@biphysiol.ge 2State Medical University of Tbilisi, Tbilisi, Georgia Received 12 October 2009; revised 20 November 2009; accepted 27 November 2009. ABSTRACT Aim: Repeated microinjections of non-opioid an- algesics into the midbrain periaqueductal gray matter and rostral ventro-medial medulla induce antinociception with development of tolerance. Antinociception following systemic administra- tion of non-steroidal anti-inflammatory drugs (N SAIDs) also exhibit tolerance. Presently our aim was to investigate the development of tolerance to the antinociceptive effects of NSAIDs analgine, ketorolac, and xefocam microinjected into cen- tral nucleus of amygdala (Ce) in rats. Methods: Under anesthesia with thiopental a stainless steel guide cannula was stereotaxically implanted uni- laterally or bilaterally into the Ce using stereo- taxic atlas coordinates, and anchored to the cra- nium by dental cement. Five days after surgery, 3 µl of these NSAIDs were injected via the injec- tion cannula while the rat was gently restrained. Twenty min post microinjection, i.e. 10-min be- fore the peak of the drugs’ effect is normally rea- ched, animals were tested with tail flick (TF) and hot plate (HP) tests. On the 5th experimental day all animals received a Ce microinjection of mor- phine. Results: Daily microinjection of NSAIDs into the Ce uni- or bilaterally, produced antino- ciception with development of complete toler- ance over a 5-day period. Following the treat- ment period, morphine microinjection into the Ce failed to elicit antinociception, indicating cro- ss-tolerance to the antinociceptive effect of N SAIDs. In other words, the “non-opioid tolerant” rats showed cross-tolerance to morphine. Con- clusions: Our data confirmed the suggestion that NSAIDs interact with endogenous opioid systems, which likely play a key role in the development of tolerance to the antinociceptive effects of NSA IDs. Keywords: Descending Inhibition; Morphine Cross-Tolerance; Nociception 1. INTRODUCTION Microinjection of non-opioid analgesics metamizol, and lysine-acetylsalicylate (LASA) into certain brain areas, including the midbrain periaqueductal gray matter (PAG) and rostral ventro-medial medulla (RVM), produces an- tinociception with development of some degree of tol- erance [1-4]. We have also observed tolerance to the an- tinociceptive effects of analgine (metamizol), ketorolac, and xefocam administered systemically [5-7]. These stu- dies are consistent with the possibility that endogenous opioidergic mechanisms associated with descending pain modulation may partly mediate the tolerance observed with non-steroidal anti-inflammatory drugs (NSAIDs) [8]. The amygdala receives massive input from the hippo- campus and the neocortex and provides a major source of afferents to PAG [9]. Analgesia resulting from microin- jection of opioid agonists into the basolateral amygdala is blocked by lidocaine inactivation of, or opioid antago- nist injection into, the PAG [10-12]. Cortical afferents to the amygdala largely target its basolateral component. The basolateral amygdala then projects to the central nucleus of amygdala (Ce), which in turn projects densely to the PAG [13]. The Ce also receives nociceptive input, both directly from the spinal cord, and indirectly via a large projection from the dorsal horn to the parabrachial nucleus [14,15]. The Ce is an integral component of the endogenous pain-modulatory circuit and is critical for systemic morphine-induced suppression of spinal noci- ceptive reflexes [16]. The present study reports that microinjection of an- algine, ketorolac, and xefocam into the Ce of rats elicits antinociception with the development of tolerance. 2. MATERIALS AND METHODS The experiments were carried out using male Wistar rats, 200-250g in body weight, bred at the Beritashvili Insti- tute of Physiology. The animals were kept under standard housing conditions (22±2 C, 65% humidity, light from 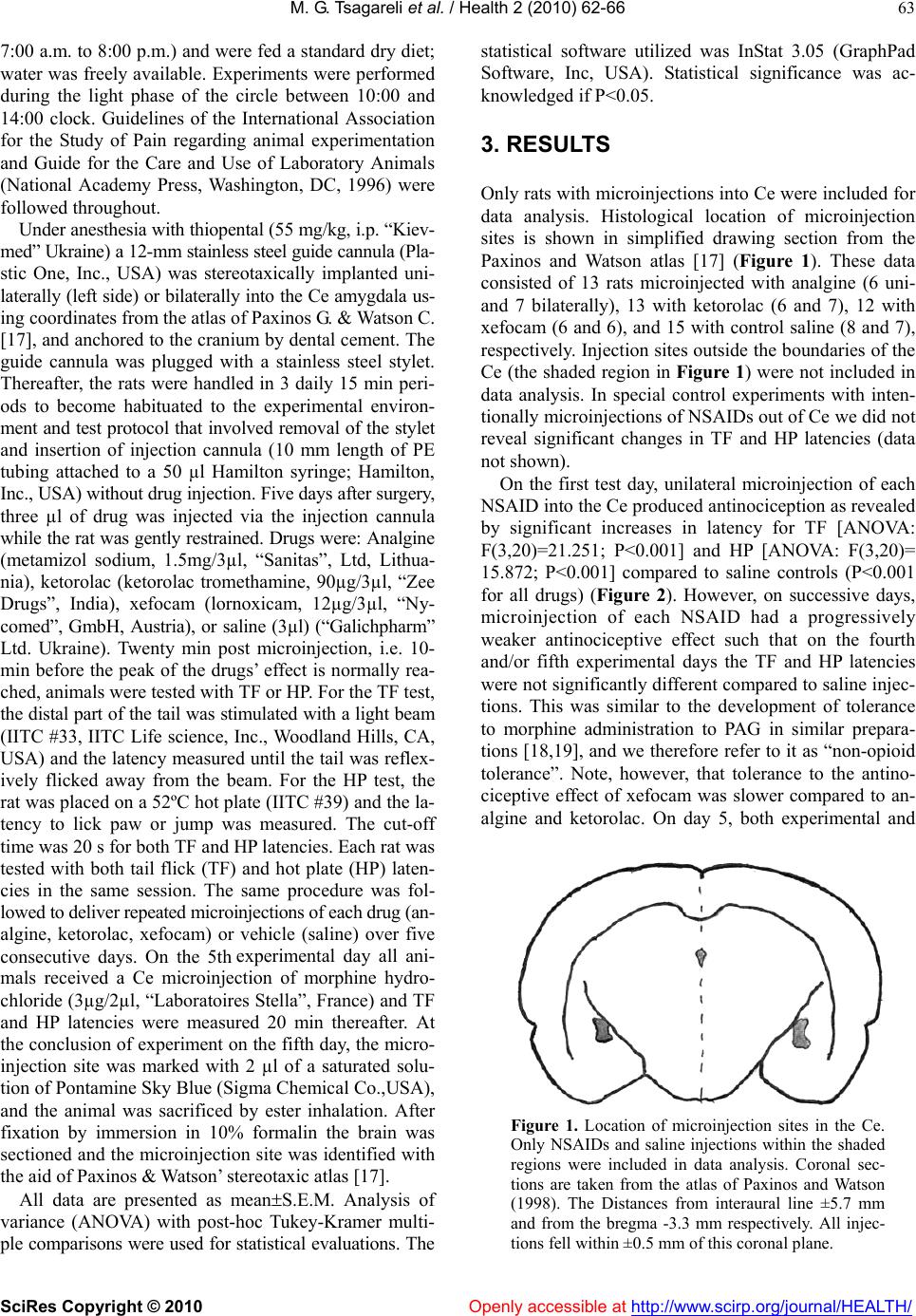 M. G. Tsagareli et al. / Health 2 (2010) 62-66 SciRes Copyright © 2010 Openly accessible at http://www.scirp.org/journal/HEALTH/ 63 7:00 a.m. to 8:00 p.m.) and were fed a standard dry diet; water was freely available. Experiments were performed during the light phase of the circle between 10:00 and 14:00 clock. Guidelines of the International Association for the Study of Pain regarding animal experimentation and Guide for the Care and Use of Laboratory Animals (National Academy Press, Washington, DC, 1996) were followed throughout. Under anesthesia with thiopental (55 mg/kg, i.p. “Kiev- med” Ukraine) a 12-mm stainless steel guide cannula (Pla- stic One, Inc., USA) was stereotaxically implanted uni- laterally (left side) or bilaterally into the Ce amygdala us- ing coordinates from the atlas of Paxinos G. & Watson C. [17], and anchored to the cranium by dental cement. The guide cannula was plugged with a stainless steel stylet. Thereafter, the rats were handled in 3 daily 15 min peri- ods to become habituated to the experimental environ- ment and test protocol that involved removal of the stylet and insertion of injection cannula (10 mm length of PE tubing attached to a 50 µl Hamilton syringe; Hamilton, Inc., USA) without drug injection. Five days after surgery, three µl of drug was injected via the injection cannula while the rat was gently restrained. Drugs were: Analgine (metamizol sodium, 1.5mg/3µl, “Sanitas”, Ltd, Lithua- nia), ketorolac (ketorolac tromethamine, 90µg/3µl, “Zee Drugs”, India), xefocam (lornoxicam, 12µg/3µl, “Ny- comed”, GmbH, Austria), or saline (3µl) (“Galichpharm” Ltd. Ukraine). Twenty min post microinjection, i.e. 10- min before the peak of the drugs’ effect is normally rea- ched, animals were tested with TF or HP. For the TF test, the distal part of the tail was stimulated with a light beam (IITC #33, IITC Life science, Inc., Woodland Hills, CA, USA) and the latency measured until the tail was reflex- ively flicked away from the beam. For the HP test, the rat was placed on a 52ºC hot plate (IITC #39) and the la- tency to lick paw or jump was measured. The cut-off time was 20 s for both TF and HP latencies. Each rat was tested with both tail flick (TF) and hot plate (HP) laten- cies in the same session. The same procedure was fol- lowed to deliver repeated microinjections of each drug (an- algine, ketorolac, xefocam) or vehicle (saline) over five consecutive days. On the 5th experimental day all ani- mals received a Ce microinjection of morphine hydro- chloride (3µg/2µl, “Laboratoires Stella”, France) and TF and HP latencies were measured 20 min thereafter. At the conclusion of experiment on the fifth day, the micro- injection site was marked with 2 µl of a saturated solu- tion of Pontamine Sky Blue (Sigma Chemical Co.,USA), and the animal was sacrificed by ester inhalation. After fixation by immersion in 10% formalin the brain was sectioned and the microinjection site was identified with the aid of Paxinos & Watson’ stereotaxic atlas [17]. All data are presented as meanS.E.M. Analysis of variance (ANOVA) with post-hoc Tukey-Kramer multi- ple comparisons were used for statistical evaluations. The statistical software utilized was InStat 3.05 (GraphPad Software, Inc, USA). Statistical significance was ac- knowledged if P<0.05. 3. RESULTS Only rats with microinjections into Ce were included for data analysis. Histological location of microinjection sites is shown in simplified drawing section from the Paxinos and Watson atlas [17] (Figure 1). These data consisted of 13 rats microinjected with analgine (6 uni- and 7 bilaterally), 13 with ketorolac (6 and 7), 12 with xefocam (6 and 6), and 15 with control saline (8 and 7), respectively. Injection sites outside the boundaries of the Ce (the shaded region in Figure 1) were not included in data analysis. In special control experiments with inten- tionally microinjections of NSAIDs out of Ce we did not reveal significant changes in TF and HP latencies (data not shown). On the first test day, unilateral microinjection of each NSAID into the Ce produced antinociception as revealed by significant increases in latency for TF [ANOVA: F(3,20)=21.251; P<0.001] and HP [ANOVA: F(3,20)= 15.872; P<0.001] compared to saline controls (P<0.001 for all drugs) (Figure 2). However, on successive days, microinjection of each NSAID had a progressively weaker antinociceptive effect such that on the fourth and/or fifth experimental days the TF and HP latencies were not significantly different compared to saline injec- tions. This was similar to the development of tolerance to morphine administration to PAG in similar prepara- tions [18,19], and we therefore refer to it as “non-opioid tolerance”. Note, however, that tolerance to the antino- ciceptive effect of xefocam was slower compared to an- algine and ketorolac. On day 5, both experimental and Figure 1. Location of microinjection sites in the Ce. Only NSAIDs and saline injections within the shaded regions were included in data analysis. Coronal sec- tions are taken from the atlas of Paxinos and Watson (1998). The Distances from interaural line ±5.7 mm and from the bregma -3.3 mm respectively. All injec- tions fell within ±0.5 mm of this coronal plane.  M. G. Tsagareli et al. / HEALTH 2 (2010) 62-66 SciRes Copyright © 2010 Openly accessible at http://www.scirp.org/journal/HEALTH/ 64 control groups received a morphine microinjection at the same Ce sites, and only the saline group exhibited anti- nociception (P<0.001). The latencies of the non-opioid tolerant rats were not altered by the morphine microin- jections, i.e. they showed cross-tolerance to morphine (Figure 2). Bilateral microinjections into the Ce also increased latency of TF [ANOVA: F(3,20)=8.873; P=0.006] and HP [ANOVA: F(3,20)=11.933; P<0.001] compared to control rats on the first day for these NSAIDs, in TF (P<0.01) and in HP (P<0.001) respectively (Figure 3). Similar to the unilateral drug injections, there was a pro- gressive decline in the antinociceptive effect elicited by bilateral injection of each drug over the 5-day period, 0 5 10 15 20 25 1 23 4 5morphine DAYS Tail flick latency (sec) Saline Analgine Ketorol ac Xefocam *** *** ** *** ** * A 0 5 10 15 20 25 12 3 45morphine DAYS Hot plate latency (sec) Sal i n e Analgi ne Ketoro la c Xe fo cam *** ** * B Figure 2. Response latencies in unilateral microinjections. Mean response latencies following unilateral microinjections of each NSAIDs are plotted over the 5-day period, followed by mor- phine, for TF (A) and HP (B) tests. Cutoff latency (20 sec) was reached in some animals for xefocam and morphine mi- croinjections. Asterisks mark significant difference of latency in comparison to saline control as analyzed by post hoc tests (*p<0.05, **p<0.01, ***p<0.001). 0 5 10 15 20 25 1 2 34 5morphine DAYS Tail flick latency (sec) Sal in e Analgine Ketor o l a c Xefocam ** *** * ** A 0 5 10 15 20 25 12345morphine DAYS Hot plate latency (sec) Saline Analgine Ketorol a c Xefocam *** *** ** * *** * B Figure 3. Response latencies in bilateral microinjections. Mean response latencies following bilateral microinjections of each NSAIDs are plotted over the 5-day period, followed by mor- phine, for TF (A) and HP (B) tests. Cutoff latency (20 sec) was reached in some animals for xefocam and morphine mi- croinjections. Asterisks mark significant difference of latency in comparison to saline control as analyzed by post hoc tests (*p<0.05, **p<0.01, ***p<0.001). such that TF and HP latencies were not significantly different from saline controls after the 4th-5th day (Fig- ure 3). Again, xefocam exhibited a slower time course for development of tolerance. Bilateral microinjection of each NSAIDs also exhibited cross-tolerance to morphine as compared with saline controls (P<0.001) (Figure 3). 4. DISCUSSION The present study revealed that microinjection of an- algine, ketorolac, and xefocam into the Ce induced anti- nociception in awake rats. This confirmed our previous results with systemic (i.p.) administration of NSAIDs  M. G. Tsagareli et al. / Health 2 (2010) 62-66 SciRes Copyright © 2010 Openly accessible at http://www.scirp.org/journal/HEALTH/ 65 [5-7], and results of others using microinjection of the same NSAIDs into the PAG [2-4]. Importantly, repeated microinjections of NSAIDs into the Ce resulted in a pro- gressive decrease in antinociceptive effectiveness (tol- erance) similar to that observed with intra-PAG injec- tions [2-4], and reminiscent of the effect of opiates. A major involvement of opioidergic mechanisms in tolerance to the analgesic effect of NSAIDs was surpris- ing, because traditionally the cellular and molecular ac- tions of opioids were thought to differ from those of nonopioid analgesics. One interesting aspect of NSAIDs administration, namely tolerance, emphasizes their simi- larity to opioid analgesics. Indeed, microinjection of meta- mizol [3,4,20], or LASA [2,20] into PAG or into Ce, pro- gressively led to a loss of their antinociceptive effects, i.e. produced tolerance. Furthermore, tolerance to meta- mizol or LASA was accompanied by cross-tolerance to morphine [2-4] as if opioid analgesics had been repeat- edly administered. Interestingly, tolerance to the effect of PAG-microinjected metamizol can, like tolerance to mor- phine, be reversed by microinjection of proglumide, a cholecystokinin antagonist, at the same PAG site [3]. The latter finding constituted additional evidence that the PAG effects of non-opioid analgesics are similar to those of morphine. Moreover, the data suggested that Ce should be incorporated into current models of endogenous pain con- trol circuitry [21]. It is well known that morphine injection after admini- stration of NSAIDs or in combination, morphine plus NSAIDs usually potentiates their own analgesic effects [8]. We have recently tested each of NSAIDs for cross- tolerance to morphine given over a 5-day period in two age groups of rats. There was a significant difference be- tween adult and juvenile rat groups for the degree of mor- phine analgesia, which was most marked on the first and second experimental days. Furthermore, morphine-tole- rant rats exhibited cross-tolerance to analgine, ketorolac, and xefocam for both TF and HP tests, respectively (data not shown). In conclusion, our data confirmed previous studies in- dicating that the antinociceptive action of NSAIDs may be closely related to that of endogenous opioids, includ- ing the development of tolerance. In addition, the Ce along with PAG and RVM represents an important com- ponent of the endogenous antinociceptive system. 5. ACKNOWLEDGEMENTS The authors would like to thank Professor E. Carstens for helpful comments and English revisions of the manuscript. This research was supported by the grant from Georgian National Science Foundation (GNSF/ST07/ 6-234). REFERENCES [1] Jones, S.L. (1996) Dipyrone into the nucleus raphe mag- nus inhibits the rat nociceptive tail flick reflex. Eur J Pharmacol, 318, 37-40. [2] Pernia-Andrade, A.J., Tortorici, V. and Venegas, H. (2002) Induction of opioid tolerance by lysine-acetylsalicylate in rats. Pain, 111, 191-200. [3] Tortorici, V., Nogueira, L., Salas, R. and Venegas, H. (2003) Involvement of local cholecystokinin in the tolerance induced by morphine microinjections into the periaque- ductal gray of rats. Pain, 102, 9-16. [4] Tortorici, V. and Vanegas, H. (2000) Opioid tolerance in- duced by metamizol (dipyrone) microinjections into the periaqueductal gray of rats. Eur J Neurosci, 12, 4074- 4080. [5] Tsagareli, M.G., Tsiklauri, N., Lagidze, T., Gurtskaia, G. and Abzianidze, E. (2005) Tolerance induction by non- opioid analgesics in rats. Proc Georgian Acad Sci Biol Series A, 31, 903-909. [6] Tsiklauri, N., Gurtskaia, G. and Tsagareli, M.G. (2006) Is endogenous opioid system involved in non-opioid anal- gesics tolerance? Georgian Med News, 8(137), 121-125. [7] Tsiklauri, N. and Tsagareli, M.G. (2006) Non-opioid- induced tolerance in rats. Neurophysiol, 38, 370-373. [8] Hernandez-Delgadillo, G.P. and Cruz, S.L. (2006) En- dogenous opioids are involved in morphine and dipyrone analgesic potentiation in the tail flick test in rats. Eur J Pharmacol, 546, 54-59. [9] Aggleton, J.P. (Ed) (1996) The amygdala: Neurobiologi- cal aspects of emotion, memory, mental dysfunction. Wil- ley-Liss, New York [10] Bodnar, R.J. (2000) Supraspinal circuitry mediating opioid antinociception: Antagonist and synergy studies in multi- ple sites. J Biomed Sci, 7, 181-194. [11] Helmstetter, F.J., Tershner, S.A. and Poore, L.H. (1998) Antinociception following opioid stimulation of the ba- solateral amygdala is expressed through the periaque- ductal grey and rostral ventromedial medulla. Brain Res, 779, 104-118. [12] Pavlovič, Z., Cooper, M. and Bodnar, R. (1996) Opioid antagonists in the periaqueductal gray inhibit morphine and beta-endorphin analgesia elicited from the amygdala of rats. Brain Res, 741, 13-26. [13] Rivzi, T.A., Ennis, M. and Behbehani, M.M. (1991) Con- nections between the central nucleus of the amygdala and the midbrain periaqueductal gray: Topography and re- ciprocity. J Comp Neurol, 303, 121-131. [14] Gauriau, C. and Bernard, J.F. (2002) Pain pathways and parabrachial circuits in the rat. Exp Physiol, 87, 251-258. [15] Gauriau, C. and Bernard, J.F. (2004) A comparative re- appraisal of projections from the uperficial laminae of dorsal horn in the rat: forebrain. J Comp Neurol, 468, 24-56. [16] Manning, B.H. and Mayer, D.J. (1995) The central nu- cleus of the amygdala contributes to the production of morphine antinociception in the tail-flick test. J Neurosci, 15, 8199-8213. [17] Paxinos, G. and Watson, C. (1998) The rat brain in stereo- taxic coordinates, 4th edition. Academic Press, New York. [18] Morgan, M.M., Clayton, C.C. and Boyer-Quick, J.S. (2005) Differential susceptibility of the PAG and RVM to tolerance to the antinociceptive effect of morphine in the rat. Pain, 113, 91-98. [19] Tortorici, V., Nogueira, L., Aponte, Y. and Vanegas, H.  M. G. Tsagareli et al. / HEALTH 2 (2010) 62-66 SciRes Copyright © 2010 http://www.scirp.org/journal/HEALTH/Openly accessible at 66 (2004) Involvement of cholecystokinin in the opioid tol- erance induced by dipyrone (metamizol) microinjections into the periaqueductal gray matter of rats. Pain, 112, 113-120. [20] Vanegas, H. and Tortorici, V. (2002) Opioidergic effects of non-opioid analgesics on the central nervous system. Cell Mol Neurobiol, 22, 655-661. [21] Price, D.D. (1999) Psychological mechanisms of pain and analgesia. IASP Press, Seattle. |

