Paper Menu >>
Journal Menu >>
 Vol.2, No.1, 38-44 (2010) Health doi:10.4236/health.2010.21007 SciRes Copyright © 2010 Openly accessible at http://www.scirp.org/journal/HEALTH/ Gait spectral index (GSI): a new quantification method for assessing human gait Rodolphe Héliot1, Christine Azevedo-Coste2, Laszlo Schwirtlich3, Bernard Espiau4 1Inria, Montbonnot, France; rodolphe.heliot@gmail.com 2 Demar Inria/Lirmm, Montpellier, France; azevedo@lirmm.fr 3 Institute for Rehabilitation “Dr Miroslav Zotovic”, Belgrade, Serbia; laslo@bitsyu.net 4 Inria, Montbonnot, France; espiau@inrialpes.fr Received 25 November 2009; revised 7 December 2009; accepted 12 December 2009. ABSTRACT This paper introduces a simple, quantitative as- sessment tool to follow up the recovery of gait. Today, micro-electro-mechanical systems (MEMS) technology provides with small, simple, low-pow- er consuming and easy to don and doff sensors. In our approach we have selected an accelerome- ter and introduced a new quantity that charac- terizes the gait pattern in the frequency domain, we term it Gait Spectral Index (GSI). GSI allows assessing gait quality and closely relates to the speed and cadence of gait (dynamics). We have tested the GSI approach to quantify the quality of the gait of healthy young and elderly, and post- stroke hemiplegic individuals. We investigated the repeatability and coherence of GSI in healthy individuals (young and elderly) and contrasted this to the post-stroke hemiplegic individuals. We found that high correlation of the GSI with conventional gait parameters. This suggests that GSI, which needs only data from one acceler- ometer, could be an objective quantitative mea- sure of the quality of the walking thereby a sim- ple yet reliable measure of the recovery of func- tion during neuronrehabilitation. Keywords: Gait Evaluation; Walk Training; Accelerometer; Spectral Analysis 1. INTRODUCTION Clinical methods for gait evaluation consist in standard- ized functional tests (Barthel Index, Rivermead Mobility Index (Bohannon et al. 1987), Functional Ambulation Category (FAC)) are qualitative; thereby, somewhat sub- jective because they depend on individual observation skills of the rater. We developed a simple instrument that after appropriate processing provides objective quantita- tive measure of the gait performance. This new instru- ment is of great importance for clinical use for assessing the gait pattern in all, including elderly and individuals with disabilities (Sekine et al., 2002). The heart of the instrument is the accelerometer that is being mounted on the leg, does not need calibration and provides data that is highly reproducible. The development of this instrument follows the fast growth of the filed of Micro-electro-mechanical systems (MEMS). The MEMS sensors have been applied for the assessment of physical activities using body-mounted systems (Jasiewicz et al., 2006; Luinge and Veltink, 2004 ; Pappas et al., 2002). MEMS based accelerometers pro- vide a good representation of movement dynamics (Ja- siewicz et al., 2006; Hester et al., 2006; Brandes et al., 2005; Luinge and Veltink, 2004). The typical application of multiple accelerometers is for reconstruction of the kinematics of the gait (Wagenaar et al., 1992; Bussmann et al., 2000). Reconstruction of gait kinematics is difficult since the integration of data captured by accelerometers results with drift and substantial errors. The alternative is to use the acceleration data directly as a measure of the quality of gait. Acceleration patterns of trunk have ex- tensively been employed since they have been shown to comprise low intra-variability; thereby suitable for de- tection of phases of the gait cycle (Ziljstra et al., 2003; Brandes et al., 2006). The reproducibility of trunk ac- celerations declined as gait speed decreases which is the case when analyzing the gait of elderly and even more of subjects with gait disabilities (Saremi et al., 2006). Acceleration can be analyzed in the frequency domain. The fundamental frequency is equivalent to the stride frequency, and the harmonics amplitudes vary depending on the gait pattern. It is important to notice that the posi- tion of the accelerometer on a body segment does not affect the frequency content of the signal. One of the best confirmations was presented by Waarsing et al. (1996) showing that the power contained in the peaks is in- versely proportional to stability performance. Here, we show that only one accelerometer can be used  R. Héliot et al. / HEALTH 2 (2010) 38-44 SciRes Copyright © 2010 Openly accessible at http://www.scirp.org/journal/HEALTH/ 39 as a good source of data for gait characterization. One of the features of this gait assessment system is that it does not need to be calibrated to each individual, and provides with easy donning and doffing. The major novelty that allows the use of this simple hardware is the Gait Spectral Index (GSI). The GSI is the new measure that character- izes the gait pattern in the frequency domain. We hy- pothesized that the GSI is highly correlated with the conventional gait parameters. Based on proved hypothe- sis we performed the experiments in which we compared the GSI determined from analysis of the gait of healthy young and elderly subjects with the GSI determined for the gait of individuals of hemiplegia. 2. METHODS 2.1. Subjects Six healthy young (HY) (age 31. 8 years ± 7. 2), and ten healthy elderly (HE) (age 67 years ± 6. 9) individuals participated in the study to confirm the high correlation of the GSI and conventional gait parameters. 19 post-stroke hemiplegic individuals (SP) (age 58. 6 years ± 10.18) also participated in the study in order to analyze the differ- ences between healthy and pathological gait. Basic tada on study the subjects is summarized in Tables 1-4. The HY group was formed from volunteers from our laboratory in LIRMM, France. Two subjects, out of ten, in the HE group were recruited in an elderly cultural asso- ciation while the other eight from a local waling club. Hemiplegic individuals were recruited from the inpa- tient population of the Institute for Rehabilitation “Dr Miroslav Zotovic” in Belgrade. The hemiplegic gait abili- ties were assessed by using the Functional Ambulation Category scale (Holden et al, 1984). The FAC scale has five grades: 1 - person needs to be physically supported for any ambulation (the worst), and 5 - the person can walk independently anywhere (the best). All hemiplegic individuals could walk with their usual walking aid (13 patients used cane or tripod, 4 patients had also an ankle or foot orthosis) as shown in Tables 3 and 4. The (SP) group was divided into two sub-groups: (SP1) includes the eight individuals with a 5 FAC rank and (SP2) with the 11 remaining individuals. This study was approved by the local ethics Committee, and all study participants signed the informed consent. 2.2. Equipment We used a uniaxial-accelerometer (ADXL-203, Analog Devices) positioned on the shank close to the ankle. The accelerometer axis was directed along the shank with the positive direction pointing upwards (Figure 1). The alig- nment was performed visually, except from this con- straint, no specific care was needed for the positioning of the accelerometer. In the case of hemiplegic individuals the accelerometer was positioned on the non-paretic leg. In order to determine the correlation with the conven- tional gait parameters subjects were also equipped with two 3-contact point insoles used to detect gait phases, gait cadence and assess the averaged stride length when as- sociated to chronometric recordings. The sampling rate was 100 samples per second based on known low fre- quency content of the signal. 2.3. Protocol Subjects were asked to walk 10 meters at their normal self-paced speed. Depending on the individual ability to Table 1. Healthy young subject group (HY) gait description. # Age Cadence 6 30 0.90 1 46 0.96 2 28 0.78 3 32 1.05 5 26 0.99 4 29 0.84 Mean 31.8 0.92 Std 7.2 0.1 Table 2. Healthy elderly subject group (HE) gait description. # Age Walking speed Cadence Stride length 5 73 1.39 1.02 1.36 1 67 1.49 0.93 1.61 8 65 1.35 0.97 1.39 7 63 1.24 0.92 1.36 6 61 1.19 0.9 1.31 2 63 1.06 0.92 1.53 3 60 1.23 0.91 1.36 4 62 1.22 0.86 1.4 10 79 1.36 1.05 1.29 9 77 1.24 1.09 1.14 Mean 67 1.27 0.96 1.38 Std 6.87 0.12 0.07 0.13 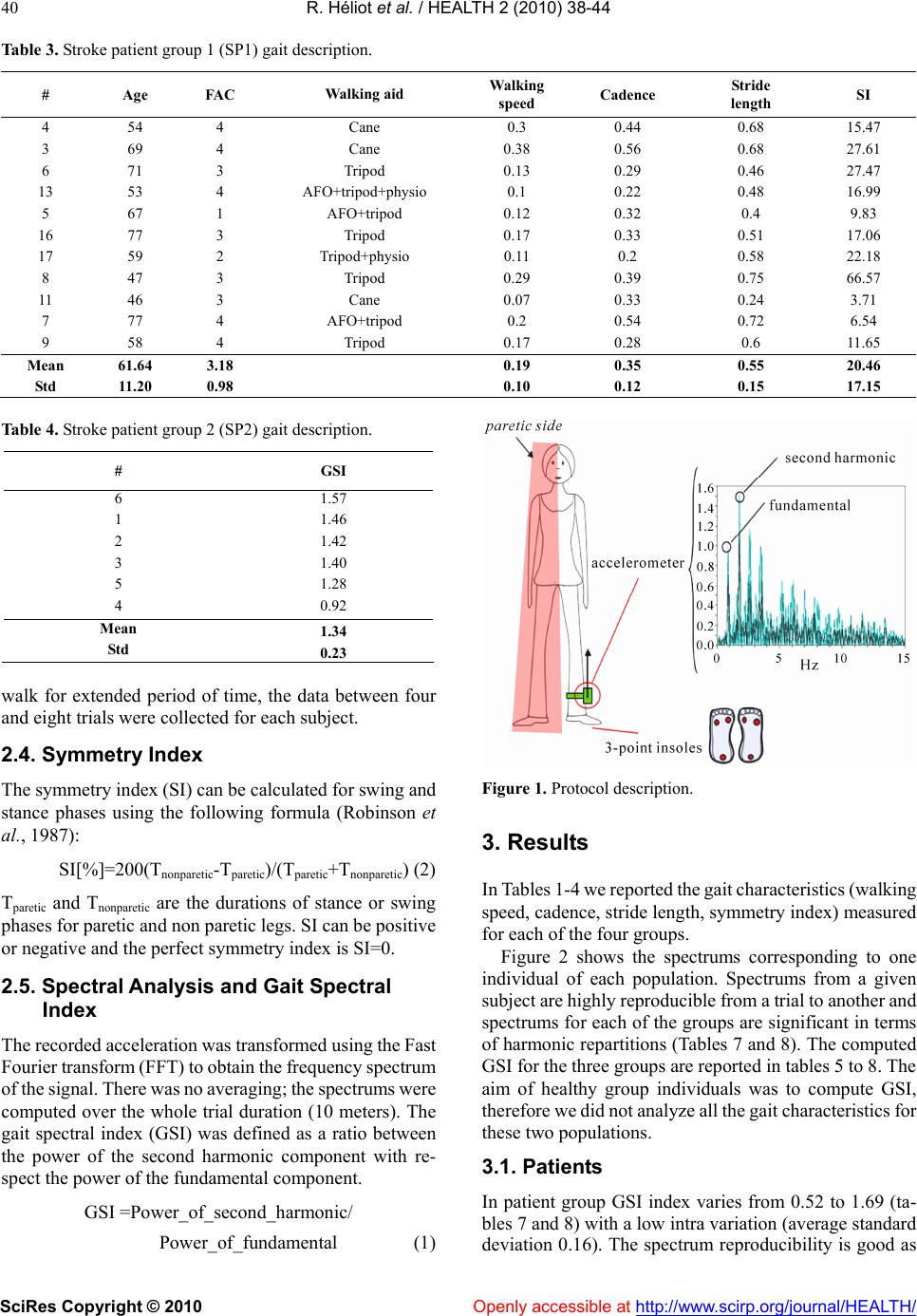 R. Héliot et al. / HEALTH 2 (2010) 38-44 SciRes Copyright © 2010 Openly accessible at http://www.scirp.org/journal/HEALTH/ 40 Table 3. Stroke patient group 1 (SP1) gait description. # Age FAC Walking aid Walking speed Cadence Stride length SI 4 54 4 Cane 0.3 0.44 0.68 15.47 3 69 4 Cane 0.38 0.56 0.68 27.61 6 71 3 Tripod 0.13 0.29 0.46 27.47 13 53 4 AFO+tripod+physio 0.1 0.22 0.48 16.99 5 67 1 AFO+tripod 0.12 0.32 0.4 9.83 16 77 3 Tripod 0.17 0.33 0.51 17.06 17 59 2 Tripod+physio 0.11 0.2 0.58 22.18 8 47 3 Tripod 0.29 0.39 0.75 66.57 11 46 3 Cane 0.07 0.33 0.24 3.71 7 77 4 AFO+tripod 0.2 0.54 0.72 6.54 9 58 4 Tripod 0.17 0.28 0.6 11.65 Mean 61.64 3.18 0.19 0.35 0.55 20.46 Std 11.20 0.98 0.10 0.12 0.15 17.15 Table 4. Stroke patient group 2 (SP2) gait description. # GSI 6 1.57 1 1.46 2 1.42 3 1.40 5 1.28 4 0.92 Mean 1.34 Std 0.23 walk for extended period of time, the data between four and eight trials were collected for each subject. 2.4. Symmetry Index The symmetry index (SI) can be calculated for swing and stance phases using the following formula (Robinson et al., 1987): SI[%]=200(Tnonparetic-Tparetic)/(Tparetic+Tnonparetic) (2) Tparetic and Tnonparetic are the durations of stance or swing phases for paretic and non paretic legs. SI can be positive or negative and the perfect symmetry index is SI=0. 2.5. Spectral Analysis and Gait Spectral Index The recorded acceleration was transformed using the Fast Fourier transform (FFT) to obtain the frequency spectrum of the signal. There was no averaging; the spectrums were computed over the whole trial duration (10 meters). The gait spectral index (GSI) was defined as a ratio between the power of the second harmonic component with re- spect the power of the fundamental component. GSI =Power_of_second_harmonic/ Power_of_fundamental (1) Figure 1. Protocol description. 3. Results In Tables 1-4 we reported the gait characteristics (walking speed, cadence, stride length, symmetry index) measured for each of the four groups. Figure 2 shows the spectrums corresponding to one individual of each population. Spectrums from a given subject are highly reproducible from a trial to another and spectrums for each of the groups are significant in terms of harmonic repartitions (Tables 7 and 8). The computed GSI for the three groups are reported in tables 5 to 8. The aim of healthy group individuals was to compute GSI, therefore we did not analyze all the gait characteristics for these two populations. 3.1. Patients In patient group GSI index varies from 0.52 to 1.69 (ta- bles 7 and 8) with a low intra variation (average standard deviation 0.16). The spectrum reproducibility is good as  R. Héliot et al. / HEALTH 2 (2010) 38-44 SciRes Copyright © 2010 Openly accessible at http://www.scirp.org/journal/HEALTH/ 41 stated by the low intra variability associated to funda- mental and 2nd harmonic amplitudes (tables 7 and 8). For each individual, we have also estimated the correlation between each trial spectrum and the average spectrum for this given individual. The good correlation (r=0.78) vali- dates the hypothesis of the spectrum reproducibility. There is a moderate correlation between GSI and FAC rank (r= 0.66). In figure 3 we plotted the GSI values obtained for the two patient groups in function of FAC rank. In table 7, the patients are classified in terms of GSI index values. The higher GSI values correspond to best walkers in FAC quotation (level 5-group SP1), they vary from 1.22 to 1.69. The mean value is 1.21. In SP2 group, GSI varies from 0.52 to 0.89 (table 8). For lower levels, the GSI does not seem to allow to discriminate between the different levels. The mean value is 0.71. There is a good correlation between GSI and gait speed (r=0.81) and gait cadence (r=0.74). Correlation between GSI and stride length is moderate (r=0.66). Correlation with symmetry index is not significant. The mean value of symmetry index is 21%. It is interesting to notice that FAC rank and symmetry index are not correlated (r=0.03). FAC rank is correlated with gait speed (r=0.7), gait cadence (r=0.72) and stride length (r=0.66). 3.2. Elderly In elderly subject group GSI index varies from 0.4 to 1.3 (table 5). The mean value is 1.02. In healthy elderly group, only stride length appeared to be significant in terms of correlation with GSI (r= 0.72). The two older subjects (77 and 79) have the lower GSI scores. 3.3. Young In young subject group GSI index varies from 0.92 to 1.57 (table 6). The mean value is 1.34. 4. Discussion The results presented document that the spectrum of the shank acceleration is highly reproducible from trial to trial in the same subject and can be considered as an individual signature pattern. We noticed the presence of strong high frequencies components in the spectrums of healthy and patient best walkers. High frequencies components are likely present due to the richer dynamics of healthy gait compared to the gait of hemiplegic and poor walkers. The proposed GSI ratio compares fundamental am- plitude with second harmonic amplitude; it correlates with gait speed which is classically considered as an indirect measurement of gait quality. It is important to notice that to be valuable speed should be evaluated through distances long enough to assess adaptation and gait efficiency. The low correlation between FAC score and GSI should be counterbalanced by the fact that the higher GSI values correspond to best classification in FAC. But. FAC classes are not varying linearly with gait quality and the number of patients in each of the categories is not similar enough to conclude. The GSI scores in elderly population are higher com- pared to our expectations. We suggest that this is due to the fact that the population included in the study was mainly recruited in a gait club. Hence, they are not rep- resentative for typical aged walkers with limited sen- sory-motor capacity. Nor GSI, nor FAC are correlated with symmetry of gait in patients. This should be an improvement of GSI in future. 5. CONCLUSIONS The spectral analysis of the shank acceleration, in the direction aligned with the shank can characterize the gait. Two main observations can be made: 1) dealing with a given subject: high reproducibility of spectrums from trial to trial, and 2) presence of strong high frequencies components in the spectrums of healthy and patients who are good walkers. We found out that placing one accelerometer on the “healthy” leg of post-stroke hemiplegic patient allows assessing the effects of gait reeducation. The instrumen- tation and processing that we described are simple and they comprise light and non expensive setup, including wireless communication of sensory output to computer. At this point the GSI index is mainly correlated with gait speed and gait cadence and better than what FAC rank. This is an interesting result as our approach is technically less constraining than classical methods to Figure 1. Hemiparetic stroke GSI. 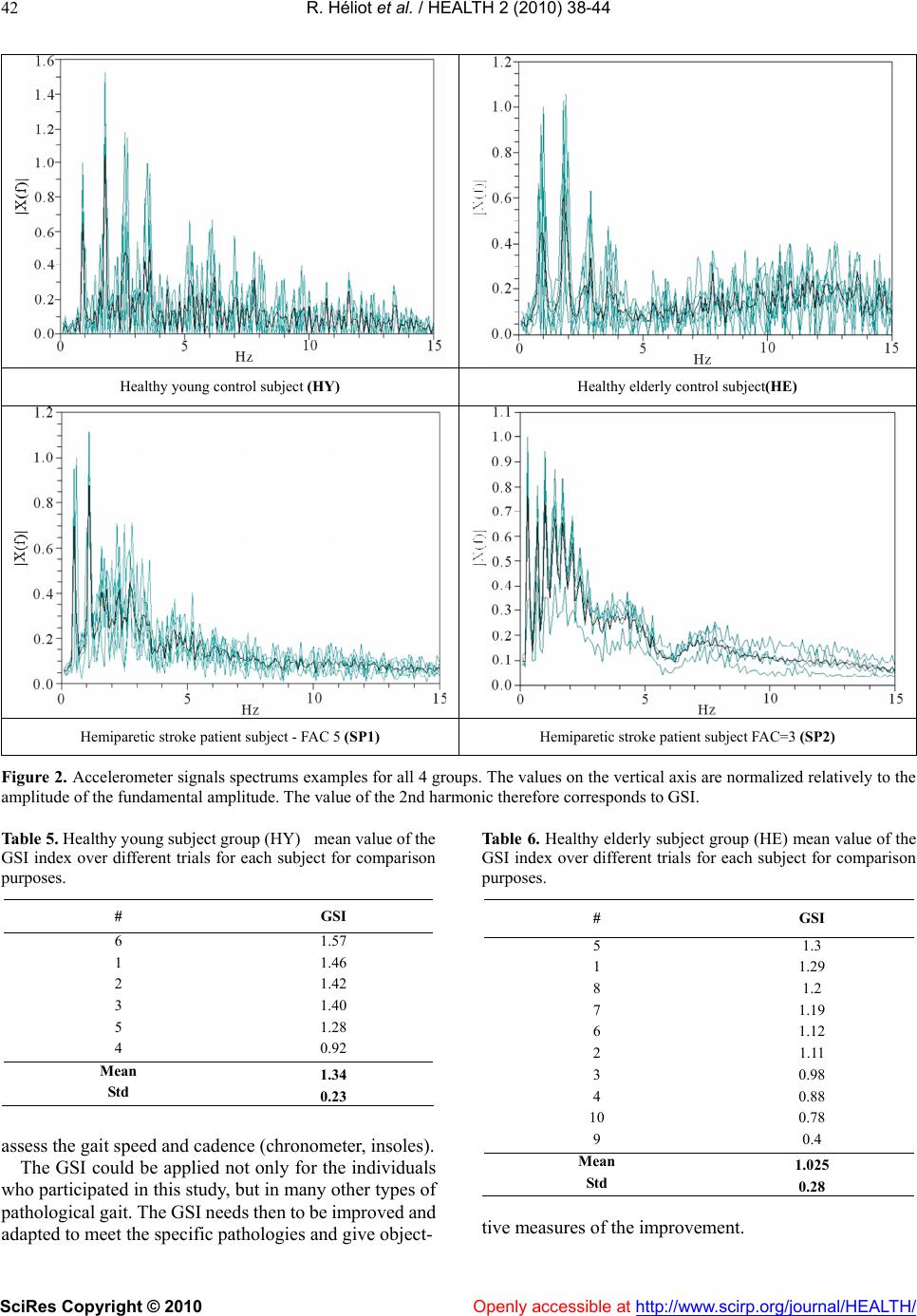 R. Héliot et al. / HEALTH 2 (2010) 38-44 SciRes Copyright © 2010 Openly accessible at http://www.scirp.org/journal/HEALTH/ 42 Figure 2. Accelerometer signals spectrums examples for all 4 groups. The values on the vertical axis are normalized relatively to the amplitude of the fundamental amplitude. The value of the 2nd harmonic therefore corresponds to GSI. Table 5. Healthy young subject group (HY) mean value of the GSI index over different trials for each subject for comparison purposes. # GSI 6 1.57 1 1.46 2 1.42 3 1.40 5 1.28 4 0.92 Mean 1.34 Std 0.23 assess the gait speed and cadence (chronometer, insoles). The GSI could be applied not only for the individuals who participated in this study, but in many other types of pathological gait. The GSI needs then to be improved and adapted to meet the specific pathologies and give object- Table 6. Healthy elderly subject group (HE) mean value of the GSI index over different trials for each subject for comparison purposes. # GSI 5 1.3 1 1.29 8 1.2 7 1.19 6 1.12 2 1.11 3 0.98 4 0.88 10 0.78 9 0.4 Mean 1.025 Std 0.28 tive measures of the improvement. Healthy young control subject (HY) Healthy elderly control subject(HE) Hemiparetic stroke patient subject - FAC 5 (SP1) Hemiparetic stroke patient subject FAC=3 (SP2) 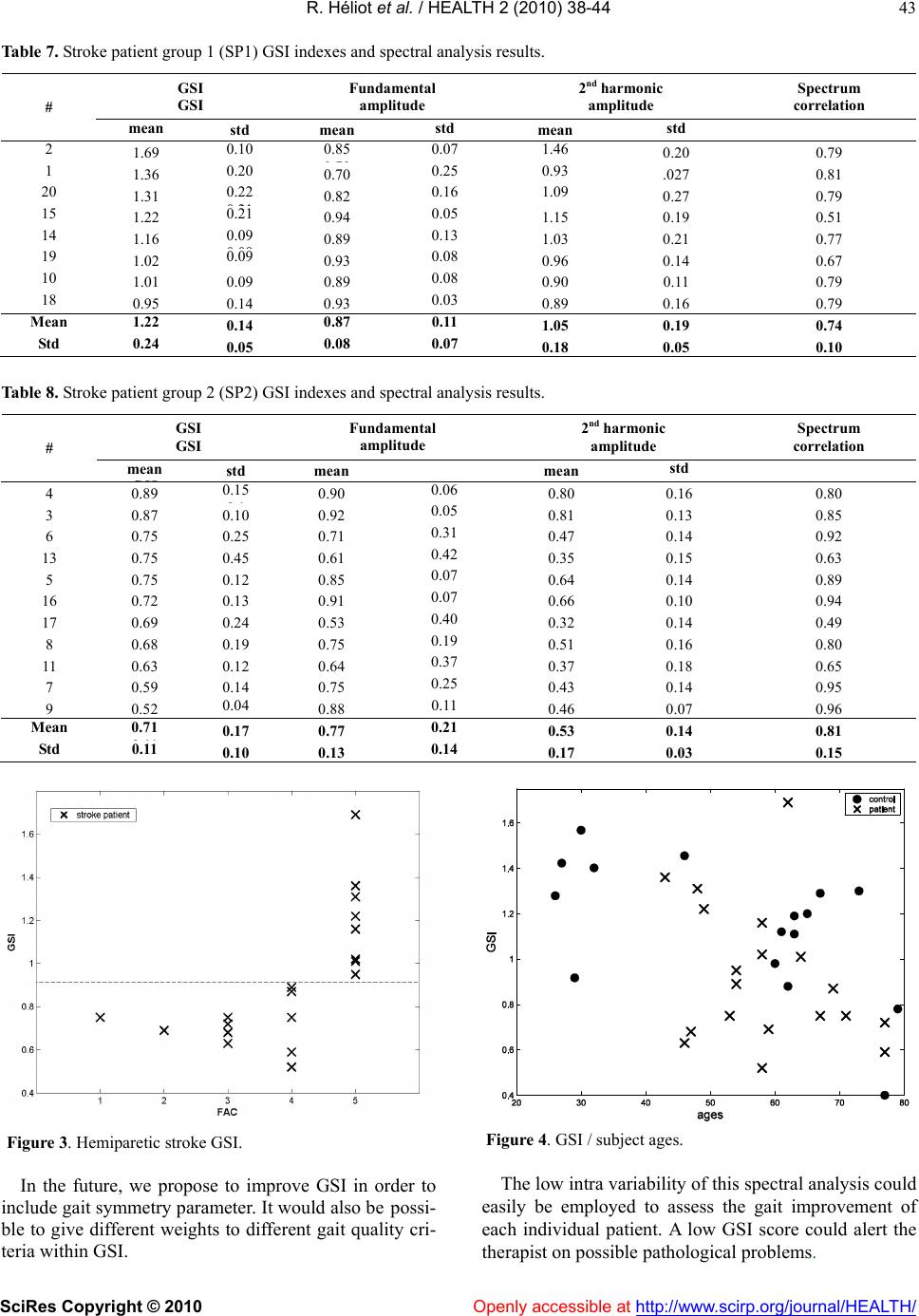 R. Héliot et al. / HEALTH 2 (2010) 38-44 SciRes Copyright © 2010 Openly accessible at http://www.scirp.org/journal/HEALTH/ 43 Table 7. Stroke patient group 1 (SP1) GSI indexes and spectral analysis results. GSI GSI Fundamental amplitude 2nd harmonic amplitude Spectrum correlation # mean GSI std mean std mean std 2 1.69 0.10 0.85 0 . 70 0.07 1.46 0.20 0.79 1 1.36 0.20 . 0.70 0.25 0.93 .027 0.81 20 1.31 0.22 0 . 21 0.82 0.16 1.09 0.27 0.79 15 1.22 0.21 0.94 0.05 1.15 0.19 0.51 14 1.16 0.09 0 . 09 0.89 0.13 1.03 0.21 0.77 19 1.02 0.09 0.93 0.08 0.96 0.14 0.67 10 1.01 0.09 0.89 0.08 0.90 0.11 0.79 18 0.95 0.14 0.93 0.03 0.89 0.16 0.79 Mean 1.22 0.14 0.87 0.11 1.05 0.19 0.74 Std 0.24 0.05 0.08 0.07 0.18 0.05 0.10 Table 8. Stroke patient group 2 (SP2) GSI indexes and spectral analysis results. GSI GSI Fundamental amplitude 2nd harmonic amplitude Spectrum correlation # mean GSI std mean mean std 4 0.89 0.15 0 . 1 0.90 0.06 0.80 0.16 0.80 3 0.87 0.10 0.92 0.05 0.81 0.13 0.85 6 0.75 0.25 0.71 0.31 0.47 0.14 0.92 13 0.75 0.45 0.61 0.42 0.35 0.15 0.63 5 0.75 0.12 0.85 0.07 0.64 0.14 0.89 16 0.72 0.13 0.91 0.07 0.66 0.10 0.94 17 0.69 0.24 0.53 0.40 0.32 0.14 0.49 8 0.68 0.19 0.75 0.19 0.51 0.16 0.80 11 0.63 0.12 0.64 0.37 0.37 0.18 0.65 7 0.59 0.14 0.75 0.25 0.43 0.14 0.95 9 0.52 0.04 0.88 0.11 0.46 0.07 0.96 Mean 0.71 0 . 11 0.17 0.77 0.21 0.53 0.14 0.81 Std 0.11 0.10 0.13 0.14 0.17 0.03 0.15 Figure 3. Hemiparetic stroke GSI. In the future, we propose to improve GSI in order to include gait symmetry parameter. It would also be possi- ble to give different weights to different gait quality cri- teria within GSI. Figure 4. GSI / subject ages. The low intra variability of this spectral analysis could easily be employed to assess the gait improvement of each individual patient. A low GSI score could alert the therapist on possible pathological problems.  R. Héliot et al. / HEALTH 2 (2010) 38-44 SciRes Copyright © 2010 Openly accessible at http://www.scirp.org/journal/HEALTH/ 44 REFERENCES [1] Bohannon, R.W., Smith, M.B. (1987) Interrater reliability of modified Ashworth scale of muscle spasticity. Phys Ther; 67, 206-7. [2] Bonnet, S. and Heliot, R. (2007) A magnetometer-based approach for studying human movements to appear. IEEE Trans. Biomedical Engineering. [3] Brandes, M., Zijlstra, W., Heikens, S.R., Lummel, R. Van and Rosenbaum, D. (2005) Accelerometry based assess- ment of gait parameters in children. Gait & Posture, 24, 482-486. [4] Bussmann, J.B. and Damen, L. and Stam, H.J. (2000) Analysis and decomposition of signals obtained by thigh-fixed uni-axial accelerometry during normal gait. Med Biol Eng Comput, 38, 632-638. [5] Hester, T., Hughes, R., Sherrill, D., Knorr, B., Akay, M., Stein J., and Bonato, P. (2006) Using wearable sensors to measure motor abilities following stroke. International Workshop on Wearable and Implantable Body Sensor Networks, Massachusetts. [6] Holden, M.K., Gill, K.M., Magliozzi, M.R. and et al. (1984) Clinical gait assessment in the neurologically im- paired, reliability and meaningfulness. Physical Therapy, 64, 35-40. [7] Jasiewicz, J.M., Allum, J. H.J., Middleton, J. W., Bar- riskill, A., Condie, P., Purcell, B. and Che Tin Li, R.C.T. (2006) Gait event detection using linear accelerometers or angular velocity transducers in able-bodied and spi- nal-cord injured individuals. Gait & Posture, 24, 502-509. [8] Lee, J.A. and Cho, S.H. and Lee, J.W. and Lee, K.H. and. Yang, H.K. (2007) Wearable Accelerometer System for Measuring the Temporal Parameters of Gait. Engineering in Medicine and Biology Society EMBS, 483-486. [9] Luinge, H. and Veltink, P. (2004) Inclination measure- ment of human movement using a 3d accelerometer with autocalibration. IEEE Trans. Neural Sys. and Rehabil. Eng., 12, 112-121. [10] Montoye, H.J., Washburn, R., Servais, S., Ertl, A., Web- ster, J.G. and Nagle, F.J. (1983) Estimation of energy ex- penditure by a portable accelerometer. Medi. Sci. Sport Exercise, 15, 403-407. [11] Pappas, I.P.I., Keller, T. and Mangold, S. A reliable, gy- roscope based gait phase detection sensor embedded in a shoe insole. (2002) In Proceedings of IEEE Sensors 002, First IEEE International Conference on Sensors, 2, 1085-1088. [12] Robinson, R.O., Herzog, W., Bigg, B.M. (1987) Use of platform variables to quantify the effects of chiropractors manipulation on gait symmetry. J Manipulative Physiol Ther. 10, 172-176. [13] Roth, E.J. Merbitz, C., Mroczek, K., Dugan, and Suh, W.W. (1997) Hemiplegic gait. relationships between gait speed and other temporal parameters. Am J Phys Med Rehabil., 76(2) , 128-133. [14] Saremi, K., Marehbian, J., Yan, X., Regnaux, J.P., Elashoff, R., Bussel, B. and Dobkin, B.H. (2006) Reli- ability and validity of bilateral thigh and foot acceler- ometric measures in healthy and hemiparetic subjects. The American Society of Neurorehabilitation, 20(2). [15] Sekine, M., Tamura, Akay, M.T., Fujimoto, T., Togawa, and Fukui, Y. (2002) Discrimination of gait patterns using wavelet-based fractal analysis. IEEE transactions on Neural Systems and Rehabilitation Engineering, 10-3, 188-196. [16] Waarsing, J.H., Mayagoitia, R.E. and Veltink, P.H (1996) Quantifying the stability of gait using accelerometers. In 18th Annual International Conference of the IEEE Engi- neering in Medicine and Biology Society, Amsterdam. utilisation d'accéléros pour quantifier la marche. [17] Wagenaar, R.C. and Beek, W.J. (1992) Hemiplegic gait, a kinematic analysis using gait speed as a basis. J Bio- mech.25, 1007-1115. [18] Ziljstra, W. and Hof, A.L. (2003) Assessment of spa- tio-temporal gait parameters from trunk accelerations during human gait. Gait & Posture, 4, 212-221. |

