Paper Menu >>
Journal Menu >>
 Vol.2, No.1, 20-23 (2010) doi:10.4236/health.2010.21004 SciRes Copyright © 2010 http://www.scirp.org/journal/HEALTH/ Health Openly accessible at Does conventional abdominal preoperative ultrasonography have a role in the detection of pancreatic insulinoma? A case report L. Spadaro, O. Magliocco, G. Trovato, F. Purrello Dpt di Medicina Interna e Medicina Specialistica, Università degli Studi di Catania, Ospedale Garibaldi, Italy; fpurrell@unict.it Received 2 September 2009; revised 14 October 2009; accepted 16 October 2009. ABSTRACT Combinations of strategies as MRI, endoscopic sonography, selective arteriography, were the first choice for the detection of pancreatic insu- linoma. In these proposal, therefore, abdominal was not in- cluded at all. Case presentation: a 78-year-old no diabetic women was referred to us because fasting hypoglycemic symptoms. The clinical and laboratory findings suggested an insulinoma. Abdominal ultrasound showed a small solid mass in the head-istmus pancreatic tract. Conclusion: the solid mass was confirmed with a contrast-enhanced- computed tomogra- phy of the abdomen. A surgical enucleation of the tumor was achieved by laparoscopy and histological examination of the specimen estab- lished a diagnosis of insulinoma. This case re- inforced the value of the conventional trans- abdomen ultrasound in addition to accuracy of anamneses and biochemical tests as the first step in the hospital clinical setting for man- agement of pancreatic insulinoma, reserving as the second step more expensive and invasive- ness techniques. Keywords: Lunch Time Napping; Mental Work Efficiency Insulinomas are rare endocrine tumors of the pancreas. Their incidence was estimated to be four cases per 1 million person-years [1]. In the earliest stages of the tu- mor’s clinical history, they usually manifest because of hypersecretion of insulin, responsible for the typical hypoglycemic symptoms. The three criteria (Whipple's triad or Whipple's criteria) on which hypoglycemia due to pancreatic islet-cell disease (most commonly insuli- noma) is diagnosed are: 1) symptoms known or likely to be caused by hypoglycemia, 2) low blood glucose levels associated with clinical signs, and 3) reversal of clinical signs with the administration of glucose. The optimal therapy is curative surgical removal of the tumor. There- fore, preoperative localization of the lesion is crucial for determining the proper surgical treatment. However, in several cases, insulinoma identification remains ex- tremely difficult and there was a continuing debate on the optimal strategy for his localization. Some authors have proposed MR as the first choice for preoperative imaging [2,3], whereas recent reports suggested a com- bined imaging protocol that consists of both dual-phase thin-section multi-detector CT and endoscopic sonogra- phy [4,5]. Actually, conventional abdominal ultrasono- graphy was not included in the imaging protocol due to his limited successful reports. We wish to present our own experience with abdominal conventional ultrasound in a case of insulinoma. A non-diabetic 78-year-old woman was brought to our attention because of hypoglycaemic symptoms. The pa- tient had a history of mild and transient sweating, anxi- ety and trembling recurring for about 2 years during her activities of ordinary life. These symptoms usually dis- appeared after food ingestion. Ten days before her refer- ral to our hospital, she had a more severe episode with sweating, trembling, anxiety, palpitations, nausea, dizzi- ness with tiredness and a severe headache crisis. She had been seen by her family physician. The practitioner had ordered general blood tests that had revealed low serum glucose level (2.2 mmol/L, 40 mg/dL). Therefore, he suggested the admission to the hospital, to determine the cause of hypoglycaemia. She had a history of hypertension and dyslipidemia on pharmacological treatment. She didn’t have any treat- ment or drugs that might have induced hypoglycaemia. On admission to our hospital, on examination, the pa- tient was overweight; her height was 165 cm and body weight 94,5 Kg, BMI 34,74, CV 118 cm. Blood pressure was 140/80 mmHg and the pulse was 68. Neurological examination and an electrocardiogram were normal. Laboratory tests were performed: urinalysis, blood cell counts, and biochemical tests (e.g. the liver and kidney function tests) other than plasma glucose concentration 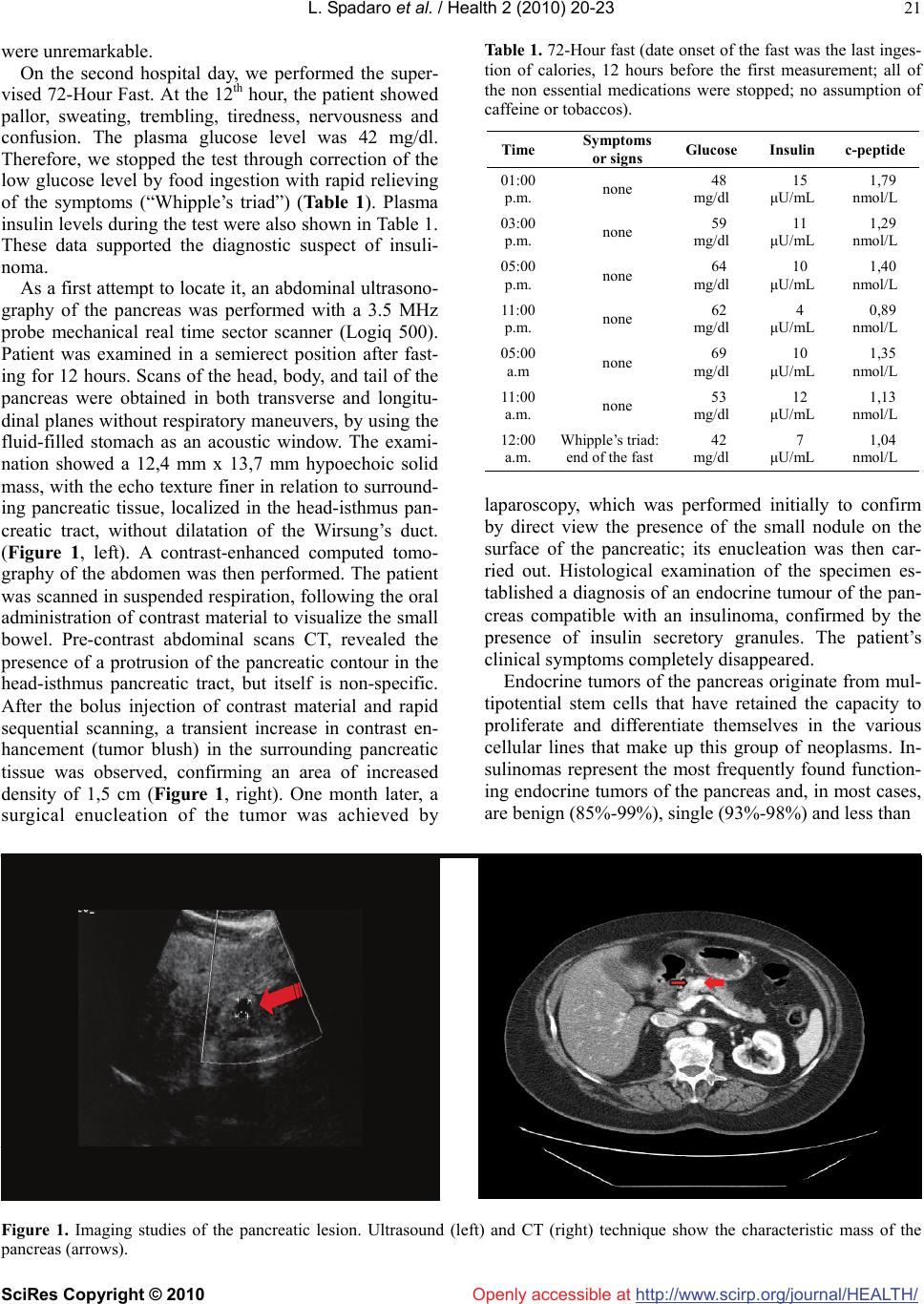 L. Spadaro et al. / Health 2 (2010) 20-23 SciRes Copyright © 2010 http://www.scirp.org/journal/HEALTH/Openly accessible at 21 were unremarkable. On the second hospital day, we performed the super- vised 72-Hour Fast. At the 12th hour, the patient showed pallor, sweating, trembling, tiredness, nervousness and confusion. The plasma glucose level was 42 mg/dl. Therefore, we stopped the test through correction of the low glucose level by food ingestion with rapid relieving of the symptoms (“Whipple’s triad”) (Table 1). Plasma insulin levels during the test were also shown in Table 1. These data supported the diagnostic suspect of insuli- noma. As a first attempt to locate it, an abdominal ultrasono- graphy of the pancreas was performed with a 3.5 MHz probe mechanical real time sector scanner (Logiq 500). Patient was examined in a semierect position after fast- ing for 12 hours. Scans of the head, body, and tail of the pancreas were obtained in both transverse and longitu- dinal planes without respiratory maneuvers, by using the fluid-filled stomach as an acoustic window. The exami- nation showed a 12,4 mm x 13,7 mm hypoechoic solid mass, with the echo texture finer in relation to surround- ing pancreatic tissue, localized in the head-isthmus pan- creatic tract, without dilatation of the Wirsung’s duct. (Figure 1, left). A contrast-enhanced computed tomo- graphy of the abdomen was then performed. The patient was scanned in suspended respiration, following the oral administration of contrast material to visualize the small bowel. Pre-contrast abdominal scans CT, revealed the presence of a protrusion of the pancreatic contour in the head-isthmus pancreatic tract, but itself is non-specific. After the bolus injection of contrast material and rapid sequential scanning, a transient increase in contrast en- hancement (tumor blush) in the surrounding pancreatic tissue was observed, confirming an area of increased density of 1,5 cm (Figure 1, right). One month later, a surgical enucleation of the tumor was achieved by Table 1. 72-Hour fast (date onset of the fast was the last inges- tion of calories, 12 hours before the first measurement; all of the non essential medications were stopped; no assumption of caffeine or tobaccos). Time Symptoms or signs Glucose Insulin c-peptide 01:00 p.m. none 48 mg/dl 15 μU/mL 1,79 nmol/L 03:00 p.m. none 59 mg/dl 11 μU/mL 1,29 nmol/L 05:00 p.m. none 64 mg/dl 10 μU/mL 1,40 nmol/L 11:00 p.m. none 62 mg/dl 4 μU/mL 0,89 nmol/L 05:00 a.m none 69 mg/dl 10 μU/mL 1,35 nmol/L 11:00 a.m. none 53 mg/dl 12 μU/mL 1,13 nmol/L 12:00 a.m. Whipple’s triad: end of the fast 42 mg/dl 7 μU/mL 1,04 nmol/L laparoscopy, which was performed initially to confirm by direct view the presence of the small nodule on the surface of the pancreatic; its enucleation was then car- ried out. Histological examination of the specimen es- tablished a diagnosis of an endocrine tumour of the pan- creas compatible with an insulinoma, confirmed by the presence of insulin secretory granules. The patient’s clinical symptoms completely disappeared. Endocrine tumors of the pancreas originate from mul- tipotential stem cells that have retained the capacity to proliferate and differentiate themselves in the various cellular lines that make up this group of neoplasms. In- sulinomas represent the most frequently found function- ing endocrine tumors of the pancreas and, in most cases, are benign (85%-99%), single (93%-98%) and less than Figure 1. Imaging studies of the pancreatic lesion. Ultrasound (left) and CT (right) technique show the characteristic mass of the pancreas (arrows).  L. Spadaro et al. / HEALTH 2 (2010) 20-23 SciRes Copyright © 2010 http://www.scirp.org/journal/HEALTH/Openly accessible at 22 2.5 cm in diameter [6,7]. Age of onset is usually 40-60 years, ranging from 6 weeks to 70 years. No sex differ- ence has been reported [8]. In our case, the patient was a 78-year-old female. Insulinoma become clinically evident because of signs and symptoms linked to hypoglycemia due to the hyper- secretion of insulin. The symptoms of hypoglycemia arise from the autonomic nervous system and from an insufficient supply of glucose to the brain (neuroglyco- penia). During acute insulin-induced hypoglycemia in healthy persons, symptoms have been recognized at plasma glucose levels of approximately 60 mg per deci- liter as measured in arterialized venous blood, and im- pairment of brain function has occurred at approximately 50 mg per deciliter [9-10-11]. In our patient, the first report of low plasma glucose levels (40 mg/dL) was as- sociated with typical hypoglycemic symptoms, sweating, trembling, anxiety, palpitations, nausea, dizziness with tiredness and headache crisis. Moreover, her past history was positive for sporadic and vague symptoms, recurring for about 2 years, but no plasma glucose levels was measured before. It has been reported that the time in- terval from the onset of symptoms to diagnosis ranges from 10 days to 30 years (median 2 years), and that the hypoglycemic episodes may happen at irregular intervals with a varying duration [8]. The symptoms intermittency and the multifaceted characters delay the diagnosis, although all signs and symptoms are usually reverted rapidly by oral or par- enteral glucose administration. Obesity or weight gain is present in 25% of patients, justified, at least in part, by the need for frequent feeding. Accordingly, our patient was overweight, and she reported a weight gain (over 20 kg) in the past two years. Because of the lack of speci- ficity of the symptoms, it is necessary to measure a low glucose level at the time that the spontaneous symptoms occur and that the symptoms are relieved through cor- rection of the low glucose level (“Whipple’s triad”) be- fore concluding that the patient have a hypoglycemic disorder. The supervised 72-hour fast is the classic di- agnostic test for hypoglycemia. In our case, the super- vised 72-hour fast confirmed a hypoglycemic disorder. In addition, the appearance of the Whipple’s triad asso- ciated with plasma glucose level ≤ 45 mg/dl, insulin level ≥ 6 /mL and c-peptide level ≥0.2 mmol/L sug- gested the diagnosis of insulinoma [12]. When the diagnosis of pancreatic insulinoma has been established clinically and biochemically, the precise lo- calization of the tumor is crucial, to facilitate the surgical resection and to prevent a blind partial or subtotal pan- createctomy [13]. Insulinomas are usually small and difficult to detect. The main problem in detecting a pan- creatic insulinoma lies in the organ’s anatomic structure and the tumor’s small dimensions at clinical appearance. Most of the tumors are in the body-tail area of the pan- creas and are less than 2 cm in diameter [14]. Many procedures have been proposed for their localization. These imaging techniques vary in accuracy, invasiveness, operator dependence, and cost. Recent protocols propose the combination of advanced imaging techniques as well as MRI, endoscopic sonography, and CT as the first choice for preoperative detection of insulinomas [2,4]. In these proposals, therefore, abdominal ultrasonography is not included at all. However, ultrasonography is a non- invasive and relatively inexpensive technique, and actu- ally it is easily available everywhere. In the case report we discuss here, we performed abdominal ultrasonogra- phy as the imaging technique of first choice because it was immediately available in our unit. In our patient, this technique correctly localized a pancreatic solid mass as a well-defined, hypoechoic lesion, without calcifica- tions or necrosis. This finding was then confirmed by CT examination. Early studies of preoperative ultrasound localization primarily used static scanners, yielding disappointing results. More recently, a higher sensitivity (approxi- mately 60-80%) has been reported using real-time US equipment [7,15,16]. This level of accuracy was highly dependent on meticulous attention to scanning technique [6]. Ultrasonographic detection of insulinomas was dif- ficult because of the frequent location at the body-tail area of the pancreas and the small sizes of the tumours in generally obese patients. In our patient insulinoma was small, < 1.5 cm, and was localized in head-isthmus pan- creatis tract. It is important to emphasize the improve- ment in ultrasonographic technology in terms of spatial resolution with increased diagnostic sensitivity and ac- curacy. In addition, ultrasonography technique enabled us to view continuous images of the lesion from several angles, with relative ease compared to other imaging technique. In the current case, after correct diagnosis and local- ization of pancreatic insulinoma, surgical resection was performed successfully and without complication. Actu- ally, after one year, the patient was free from symptoms. In conclusion, the present case supported the value of the conventional abdomen ultrasonography as the first step in the preoperative localization of pancreatic insu- linomas, reserving as the second step more expensive and invasiveness techniques. REFERENCES [1] Service, F.J., McMahon, M.M., O’Brien, P.C., et al. (1991) Functioning insulinoma: incidence, recurrence, and long term survival of patients: a 60-years study. Mayo Clin Proc, 66, 711-719. [2] Virally, M.L., Guillausseau, P.J. (1999) Hypoglycemia in adults. Diabetes e Metabolism, 25, 477-490.  L. Spadaro et al. / Health 2 (2010) 20-23 SciRes Copyright © 2010 http://www.scirp.org/journal/HEALTH/Openly accessible at 23 [3] Catalano, C., Pavone, P., Laghi, A., et al. (1999) Local- ization of pancreatic insulinomas with MR imaging at 0.5 T. Acta Radiol, 39, 644-648. [4] Moore, N.R., Rogers, C.E., Britton, B.J. (1995) Magnetic resonance imaging of endocrine tumors of the pancreas. Br J Radiol, 68, 341-347. [5] Gouya, H., Vignaux, O., Augui, J., Dousset, B., Palazzo, L., Louvel, A., Chaussade, S., Legmann, P. (2003) CT, endoscopic sonography, and a combined protocol for preoperative evaluation of pancreatic insulinomas. AJR, 181, 987-992. [6] Schumacher, B., Lubke, H.J., Frieling, T., Strohmeyer, G., Starke, A.A. (1996) Prospective study on the detection of insulinomas by endoscopic ultrasonography. Endo scopy, 28, 273-276. [7] Liu, T.H., Tseng, H.C., Zhu, Y., Zhong, S.X., Chen, J., Cui, Q.C. (1985) Insulinoma: an immunohistochemical and morphologic analysis of 95 cases. Cancer, 56, 1420-1429. [8] Stefanini, P., Carboni, M., Patrassi, N., Basoli, A. (1974) Beta islet-cell tumors of the pancreas: result of a study on 1067 cases. Surgery, 75, 597-609. [9] Villary, M.L., Guillausseau, P.J. (1999) Review: Hypoglycemia in adults. Diabetes & Metabolism, 25, 477-490. [10] Schwartz, N.S., Clutter, W.E., Shah, S.D., Cryer, P.E. (1987) Glycemic thresholds for activation of glucose counterregulatory systems are highter than the threshold for symptoms. J Clin Invest, 79, 777-781. [11] Mitrakou, A., Ryan, C., Veneman, T., et al. (1991) Hier- rarchy of glycemic thresholds counterregulatory hormone secretion, symptoms, and cerebral dysfunction. Am J Physiol, 260, E67-E74. [12] Cyper, P.E. (1993) Glucose counterregulation: the physiological mechanisms that prevent or correct hypoglycaemia. In: Frier, B.M., Fisher, B.M., (eds.), Hypoglycaemia and Diabetes: Clinical and Physiological Aspects. Edward Arnold, London, 34-55. [13] Service, F.J. (1995) Review: Hypoglycemic disorders.. NEJM, 332, 1144-1152. [14] Placzkowski, K.A., Vella, A., Thompson, G.B., Grant, C.S., Reading, C.C., Charboneau, J.W., Andrews, J.C., Lloyd, R.V., Service, F.J. (2009) Secular trends in the presentation an management of functioning insulinoma at the majp clinic, 1987-2007. J Clin Endocrinol Metab. [15] D’Onofrio, M., Mansueto, G., Vasori, S., Falconi, M., Procacci, C. (2003) Contrast-enhanced ultrasonographic detection of small pancreatic insulinoma. J Ultrasound Med, 22, 413- 417. [16] Angeli, E., vanzulli, A., Castrucci, M., Venturini, M., Sironi, S., Zerbi, A., Di Carlo, V., Del Maschio, A. (1997) Value of abdominal sonography and MR imaging at 0.5 T in preoperative detection of pancreatic insulinoma: a comparison with dynamic CT and angiography. Abdom Imaging, 22(3), 295-303. [17] Galiber, A.K., Reading, C.C., Charboneau, J.W., Sheedy, P.F., James, E.M., Gorman, B., Grant, C.S., van Heerden, J.A., Telander, R.L. (1998) Localization of pancreatic insulinoma: comparison of pre-and intraoperative US with CT and angiography. Radiology, 166(2), 405-8. |

