Advances in Bioscience and Biotechnology
Vol.4 No.3A(2013), Article ID:29306,6 pages DOI:10.4236/abb.2013.43A064
Vaccination against urinary tract infections caused by E. coli
![]()
Urological Clinic Maximilian, Urological Clinic LM Universitiy München, München, Germany
Email: mail@praxis-bauer.de
Received 5 January 2013; revised 7 February 2013; accepted 23 February 2013
Keywords: Pyelonephritis; Outer Membrane Antigen; Iron Compound Receptors
ABSTRACT
Uropathogenic Escherichia coli (UPEC) causes uncomplicated urinary tract infection (UTI) depicts a prevalent and potentially uncompromising infectious disease. In this analysis, we explained the functions of an immunoproteomics concept to vaccine development that has been successfully employed to recognize vaccine targets in other pathogenic bacteria. Pyelonephritis strains E. coli CFT073 are used for outer membrane isolation mimics urinary tract environment in which iron limitation, osmotic stress, human urine, and exposure to uroepithelial cells are included. During experiments of UTI, the antigens that induce the humoral immune response is to identified, two-dimensional gel electrophoresis are employed for the isolation of outer membrane protein and probed using pooled antisera from 20 CBA/J mice chronically infected with E. coli CFT073. 23 total outer membrane antigens, in which a unique iron compound receptor is included, are reacted with antisera and were identified by mass spectrometry. These antigens comprises of proteins with known functions in UPEC pathogenesis such as, ChuA, IroN, IreA, Iha, IutA, and FliC. These all information and data elaborate that these factors are associated with virulence during UTI are directed by antibody response. We also represents that the genes encoding ChuA, IroN, hypothetical protein c2482, and IutA are significantly more prevalent among UPEC strains than among fecal-commensal E. coli isolates. Therefore we concluded that, the outer membrane antigens are identified in this study are conserved, could be reflective part for the UTI vaccine generated to induce protective immunity against UPEC infections.
1. INTRODUCTION
Urinary tract infection (UTI) is a widespread infectious disease having possibly uncompromising complexities. Almost 11.3 million communities suffered from UTIs come off in United States each year, with an annual cost of $1.6 billion [1]. If UTI left untreated, they can lead to more extreme conditions including acute pyelonephritis, bacteremia, and renal scarring. Furthermore, increasing rates of antimicrobial resistance among uropathogens will likely complicate future treatment of these infections. [2,3] Consequently, there is an urgent public health need to develop an efficacious vaccine to prevent UTI.
The most common etiological agent of UTIs accounts for greater than 80% of these infections is Uropathogenic Escherichia coli (UPEC) [4]. Most of the virulence determinants expedite the ability of UPEC to colonize the urinary tract and exert degenerative changes in cell, including Type 1 fimbriae, [5] P fimbriae, [6] Dr adhesins, [7] hemolysin, [8,9] cytotoxic necrotizing Factor 1, [10] flagella, [11] capsule polysaccharide, [12] lipopolysaccharide O antigen, [13] and TonB-dependent iron transport systems [14].
currently, the perseverance of the in vivo transcriptome—the set of all RNA molecule including mRNA, tRNA, rRNA and other no coding RNAs, of UPEC more accentuated the significance of adhesion and iron salvage during UTI, because genes associated in these processes were exceeding in the number of receptors on the surface of target cells, making the cells more sensitive to the agent during experimental infection. [15] Most of the factors associated with virulence have been experimented as vaccine target due to the medical and economic impact of UPEC and UTI. For instance, immunization with FimH—the type 1 fimbrial adhesin, specifically decreased bladder colonization in C3H/J mice [16] and explained shield in a primate model of UTI. [17] Furthermore, a subunit vaccine using PapG—the P fimbrial adhesin, complexed with its periplasmic chaperone, PapD, specifically shielded primates from histological indications of pyelonephritis, [18] as periplasmic chaperone used to enhance functional secretion of proteins in E. coli.
The success rate is found to be very limited by using hemolysin, [19] Dr Fimbriae, [20] and the siderophore receptor IroN [21] in some experiments for the induction of protective immunity against uropathogenic E. coli. Uropathogens mixture that is deactivated by heat treatment and mucosal immunization specifically decreased urinary tract infection events among in women. This is occurred recent clinical trials of Phase II [22]. Although these vaccine preparations are not considered to be the best for providing long term protection. So, it is necessary to identify some other antigens, which is mainly employed for the production of vaccine against UPEC.
Former endeavors for vaccine production against UPEC were relied initially on some specific virulence factors or whole cells, genomic and proteomic process provide an extensive approach in order to design vaccine. Reverse vaccinology, the most advance technique used for screening of genome of serogroup B Neisseria meningitidis that exhibit a conserved surface exposed antigens among Neisseria meningitidis strains [23]. Antibody response that is activated in immunized animals by the antigens, were employed successfully to produce a universal multivalent vaccine against UPEC. Meanwhile, the immunoproteomics methods that is the isolation and mass spectrometric identification of MHC (major histocompatibility complex) binding peptides, purification and identification of protein antigens binding specific antibodies (or other affinity reagents, and comparative immunoproteomics to identify proteins and pathways modulated by a specific infectious organism, disease or toxin. These methods are linked with the bacterial proteomes screening by using sera from infected individuals, are mainly employed for antigen identification in pathogens particularly Campylobacter jejuni [24], Anaplasma marginale, [25] Bartonella henselae, [26] and Klebsiella pneumonia [27]. The addition of novel proteins and non virulence factors as candidate for immunization is the main advantage of these genomics and proteomics techniques that is proteins which are particularly eliminated from the strategies used for traditional vaccine design.
Innate and adaptive immune mechanisms are involved for immune response to UTI. Toll-like receptors (TLRs) are a class of proteins that play a key role in the innate immune system [28,29]. They are single, membranespanning, non-catalytic receptors that recognize structurally conserved molecules derived from microbes. Once these microbes have breached physical barriers such as the skin or intestinal tract mucosa, they are recognized by TLRs, which activate immune cell responses. [30] neutrophil infiltration is considered to be the primary mechanism of the innate immune response to control UTI [31]. Because, the mice have lack of neutrophils contains degenerated capability to eradicate UPEC infection compared to neutrophil-replete animals. Nevertheless, adaptive immune responses also integrate to immunity against UPEC. High susceptibility to infection with UPEC are found in severe combined immunodeficient mice that implicating B and T cell mediated immunity take part in eradication of bacteria particularly. As a result, the natural infections with UPEC are elicited by many aspects of immune response. And this clue declared that the hummoral response induced by the vaccine is actually protected from uncomplicated UTI.
Because an antibody response is probably a particular component of the adaptive immune response to UPEC, [32,33] ideal vaccine targets should be surface exposed and accessible to circulating immunoglobulins. Surface exposed proteins in E. coli are attached in the outer membrane. So the group of anticipated vaccine candidates is depicted on the outer membrane protein (OMPs) of UPEC. Meanwhile it is necessary that ideal vaccine candidates must be particular to pathogenic E. coli to refrain cross reactivity with commensal strains. In this analysis, powerful vaccine targets in UPEC are being identified by immunoproteomic methods. E. coli CFT073 grown under certain conditions and OMPs has been purified that mimics the urinary tract with antisera from the chronically infected mice that shows 23 antigenic OMPs are found that elicit the strong immune response during infection. Several OMPs are UPEC specific and novel ironinduced protein as well. This indicated that these antigenic OMPs represents novel identified targets for the production of multivalent vaccine against UTI agents (Figures 1 and 2).
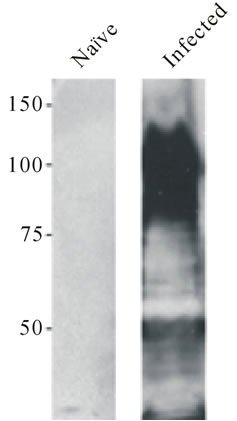
Figure 1. Specificity of antisera from chronically infected mice: Specificity of antisera generated against E. coli CFT073. Western blots of a CFT073 whole-cell lysate electrophoresed on a 12% polyacrylamide gel and probed with nonimmune sera from naïve CBA/ J mice (left) or antisera from chronically infected mice (right) are shown. Molecular mass standards are shown in kDa.
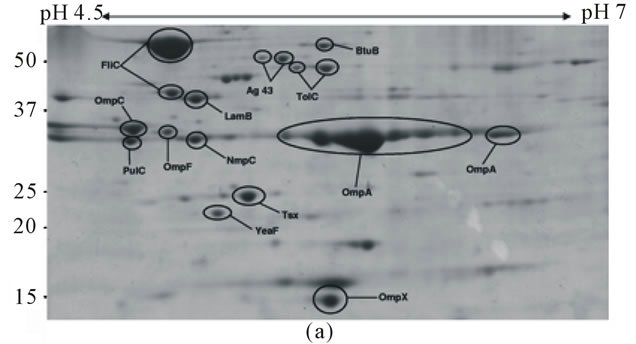

Figure 2. Outer membrane antigens from UPEC are antigenic: Identification of antigenic OMPs of E. coli CFT073. (a) Colloidal Coomassie-stained 2D-PAGE gel of outer membrane fractions isolated from CFT073 cultured in rich medium; (b) Western blot of 2D-PAGE gel probed with pooled antisera from chronically infected CBA/J mice. Proteins annotated in A are seroreactive and were identified by mass spectrometry. Molecular mass standards are shown in kDa.
2. DISCUSSION
OMP antigens that are produced during UPEC infection is being recognized by immunoproteomic approach and depicts the first wide screened for vaccine targets for this UPEC. In vivo studies shows that 23 outer membrane antigens from UPEC are expressed and capable of eliciting the hummoral response by using immunoreactive antisera from chronically infected mice. Hypothetical protein c2482 has been identified in this screen. This novel antigen expressed in iron limitation under certain conditions. Moreover, it is also discussed that the genes encoding at least four of these OMPs, chuA, c2482, iroN, and iutA are the most predominate genes among UPEC strains as compare to fecal commensal UPEC strains. This shows that these conserved antigenic OMPs may be useful and great target for a vaccine against UPEC.
There has been wide investigation for the determent of UTI by evaluating the virulence factors of UPEC as protected immunogens for provoking adaptive immune response. Though, the disparate trait of UPEC isolates proposes that increased vaccine targets will be attained to secure protection against a widespread series of strains.
Therefore, there is main focus on identifying conserved outer membrane antigens of UPEC that may be used in vaccine against UTI.
In addition to Porins and adhesins—a beta barrel protein that forms channels that allow certain cellular material to pass through cell membranes. Any type of material, below a certain size, may pass through the porins, which join together to create cylindrical passageways that allow various molecules to passively move from one side of a membrane to another, screen recognized a new outer membrane of antigen of UPEC. c2482, a speculated iron receptor, was identified during iron limitation conditions, proliferation in human urine and association with the cells of bladder epithelial. These recommendations conclude the anticipated outer membrane localization of this protein and accommodate indirect affirmation of its engrossment in iron redemption as well. Current studies show the high expression of c2482 during murine UTI [15] and this protein is considered to be the most eliciting proteins during the growth of CFT073 in human urine [34], proposing that this immunogen is a promising vaccine target.
This analysis also suggests during experimental UTI E. coli CFT073 expresses proteins. However, the transcript level gene expression is discussed by transcriptome of murine UTI in vivo during infection, [15] the recent outcomes assist and boost former research by observing in vivo protein expression. As a result, the 23 seroreactive OMPs identified are expressed in the UPEC outer membrane during infection. This data coincide with the former work, as 17 of these 23 seroreactive OMPs were among the top 30% of CFT073 transcripts detected in vivo. Indeed, 11 of these 17 OMPs were upregulated at least twofold during experimental UTI. Besides, these discoveries aid in understanding of the pathogenesis of UPEC as well.
The most outstanding benefit of this recent analysis study over other immunoproteomics analyses is the involvement of several optimum cultured conditions, has designed to mimic natural environment of the infectious agent. While these different conditions only modestly extend the number of antigens recognized, the approach nevertheless enhances our confidence that few major outer membrane antigens were excluded from the screen. This method also declared different iron-related antigens, ChuA, Iha, IroN, c2482, and IreA, which were identified during growth under only three culture conditions: iron limitation, human urine, and exposure to bladder epithelial cells. As these environments likely elicit iron loss, it is not astounding that they also activate the expression of additional proteins involved in iron acquisition. A recent study from our laboratory concealing these outcomes, showing the induction of these five OMPs during culture in human urine as well as their repression during growth in iron-replete medium [34]. It is interesting that the genes encoding three of the iron-related antigens, chuA, iroN, and c2482, were also found to be both conserved and UPEC specific by dot blot hybridization. Given the well-established role of iron acquisition in pathogenesis, we speculate that UPEC, compared to commensal E. coli, expresses a greater range of iron receptors in response to iron-limiting environments.
Fimbriae—the anchored outer membrane surface structures were thoroughly missing by western blots that predict the infected mouse antisera used in the screen consists of fimbriae-specific antibodies. In the sera of primates infected with UPEC, [18] anti-P fimbria immunoglobulin G (IgG) was discovered undoubtedly. Nevertheless, fimbrial proteins mostly need some more steps for solubilization [35] and during preparation they are clipped easily from the surface. These proteins are probably absent from 2D gel electrophoresis. Additionally, an absolute 2D-polyacrylamide gel electrophoresis analysis of the E. coli outer membrane proteome also deficit the apprehension of fimbriae, [36] this finding shows that this is the most expected outcome.
The identification of important outer membrane antigens expressed by E. coli CFT073 during UTI with limitations of several congenital stipulations with this technique is considered to be the major breakthrough. OMPs are seroreactive that are less abundantly found, could have been below the limits of detection of 2D-PAGE colloidal Coomassie staining and mass spectrometry analyses. IgG response induced by primary antigens during infection are detected as well. Secretory IgG is considered to play a pivotal role in the clearance of UTI [37] as UPEC is a mucosal pathogen but these limitations do not affect harshly on our breakthrough.
Many OMPs recognized as being powerful protective antigens have roles in the virulence of UPEC and other pathogens. E. coli strains lacking the heme/hemoglobin receptor ChuA [14], the aerobactin receptor IutA, [14]
the salmochelin receptor IroN, [38] the iron-responsive element IreA, [39] or the iron-regulated adhesin Iha [40] were particularly outcompeted by the wild-type strain in a mouse model of UTI, determining the significance of iron possession to the fitness of this pathogen in vivo. Moreover, isogenic mutants lacking the major flagellum subunit FliC were similarly outcompeted by wild-type E. coli CFT073 [41]. Therefore, this data provide evidence that a humoral response is generated against these virulence-associated factors during murine infection. Furthermore, while a role in pathogenesis is likely not a requirement for a vaccine target, it may be beneficial, as neutralizing antibodies may block critical functions of such targets upon infection.
In recent studies the outer membrane antigen are identifies contain additional features that assist in assumed roles as vaccine candidate. β-barrel structures are formed by several outer membrane antigens including the iron compound receptors. Although, most of the β-barrel proteins will be plunged in the membrane, extracellular loops accommodate surface exposed regions due to the neutrophils, as they are the most pivotal constituent of immune response to UTI, [31] opsonizing antibodies against such surface-exposed proteins may be essential to enhance phagocytosis at the site of infection. Furthermore, some of the OMPs are identified in this screen are associated in cellular processes that are typical for in vivo bacterial growth like iron redemption. Bacterial clearance from urinary tract can also be possible by ceasing the function of these proteins through neutralizing antibodies. Eventually, four of the antigens that are ChuA, c2482, IutA, and IroN are conserved among most UPEC strains and mostly absent in some fecal commensal-E. coli strains. Some genetic assortments at the amino acid level are detected in each of these antigens, among sequenced strains of pathogenic E. coli. Each antigen is 90 to 100% similar between strains. This concludes as these proteins may produce protection against a wide series of pathogenic strains, if attained as vaccine target, which is least cross react with normal flora.
3. CONCLUSION
Uncomplicated UTI that is secured by a vaccine would have outstanding public health advantages. The information and data existed in this study depicts a first step towards the development of such a broadly protective vaccine against UPEC. Some of the outer membrane antigens have not observed yet as vaccine targets for UPEC. Moreover, c2482, a unique antigen recognized in this screen shows not only a novel vaccine target but a newly recognized OMP as well that could function as an iron compound receptor. Although, lots of examinations are required to enhance these observations and investigate immunization with these antigens before an efficient UTI vaccine can be cultivated.
REFERENCES
- Foxman, B., Barlow, R., D’Arcy, H., Gillespie, B. and Sobel, J.D. (2000) Urinary tract infection: Self-reported incidence and associated costs. Annals of Epidemiology, 10, 509-515. doi:10.1016/S1047-2797(00)00072-7
- Gupta, K., Hooton, T.M. and Stamm, W.E. (2001) Increasing antimicrobial resistance and the management of uncomplicated community-acquired urinary tract infections. Annals of Internal Medicine, 135, 41-50.
- Ishikawa, K., Hayakawa, S., Miyakawa, S., Kusaka, M., Shiroki, R. and Hoshinaga, K. (2005) Survey of the susceptibility of urinary isolates to antibacterial agents in 2003. Journal of Infection and Chemotherapy, 11, 44-47. doi:10.1007/s10156-004-0356-9
- Mobley, H.L.T. and Warren, J.W. (1996) Urinary tract infections: Molecular pathogenesis and clinical management. ASM Press, Washington DC.
- Connell, I., Agace, W., Klemm, P., Schembri, M., Marild, S. and Svanborg, C. (1996) Type 1 fimbrial expression enhances Escherichia coli virulence for the urinary tract. Proceedings of the National Academy of Sciences of the United States of America, 93, 9827-9832. doi:10.1073/pnas.93.18.9827
- Roberts, J.A., Marklund, B.I., Ilver, D., Haslam, D., Kaack, M.B., Baskin, G., Louis, M., Mollby, R., Winberg, J. and Normark, S. (1994) The Gal(alpha 1-4)Gal-specific tip adhesin of Escherichia coli P-fimbriae is needed for pyelonephritis to occur in the normal urinary tract. Proceedings of the National Academy of Sciences of the United States of America, 91, 11889-11893. doi:10.1073/pnas.91.25.11889
- Goluszko, P., Moseley, S.L., Truong, L.D., Kaul, A., Williford, J.R., Selvarangan, R., Nowicki, S. and Nowicki, B. (1997) Development of experimental model of chronic pyelonephritis with Escherichia coli O75:K5:H-bearing Dr fimbriae: Mutation in the dra region prevented tubulointerstitial nephritis. Journal of Clinical Investigation, 99, 1662-1672. doi:10.1172/JCI119329
- van den Bosch, J.F., Emody, L. and Ketyi, I. (1982) Virulence of haemolytic strains of Escherichia coli in various animal models. FEMS Microbiology Letters, 13, 427-430. doi:10.1111/j.1574-6968.1982.tb08300.x.
- Welch, R.A., Dellinger, E.P., Minshew, B. and Falkow, S. (1981) Haemolysin contributes to virulence of extra-intestinal E. coli infections. Nature, 294, 665-667. doi:10.1038/294665a0
- Rippere-Lampe, K.E., O’Brien A.D., Conran, R. and Lockman, H.A. (2001) Mutation of the gene encoding cytotoxic necrotizing factor type 1 (cnf1) attenuates the virulence of uropathogenic Escherichia coli. Infection and Immunity, 69, 3954-3964. doi:10.1128/IAI.69.6.3954-3964.2001
- Lane, M.C., Lockatell, V., Monterosso, G., Lamphier, D., Weinert, J., Hebel, J.R., Johnson, D.E. and Mobley, H.L. (2005) Role of motility in the colonization of uropathogenic Escherichia coli in the urinary tract. Infection and Immunity, 73, 7644-7656. doi:10.1128/IAI.73.11.7644-7656.2005
- Bahrani-Mougeot, F.K., Buckles, E.L., Lockatell, C.V., Hebel, J.R., Johnson, D.E., Tang, C.M. and Donnenberg, M.S. (2002) Type 1 fimbriae and extracellular polysaccharides are preeminent uropathogenic Escherichia coli virulence determinants in the murine urinary tract. Molecular Microbiology, 45, 1079-1093. doi:10.1046/j.1365-2958.2002.03078.x
- Schilling, J.D., Mulvey, M.A., Vincent, C.D., Lorenz, R.G. and Hultgren, S.J. (2001) Bacterial invasion augments epithelial cytokine responses to Escherichia coli through a lipopolysaccharide-dependent mechanism. The Journal of Immunology, 166, 1148-1155.
- Torres, A.G., Redford, P., Welch, R.A. and Payne, S.M. (2001) TonB-dependent systems of uropathogenic Escherichia coli: Aerobactin and heme transport and TonB are required for virulence in the mouse. Infection and Immunity, 69, 6179-6185. doi:10.1128/IAI.69.10.6179-6185.2001
- Hagberg, L., Engberg, I., Freter, R., Lam, J., Olling, S. and Eden, C.S. (1983) Ascending, unobstructed urinary tract infection in mice caused by pyelonephritogenic Escherichia coli of human origin. Infection and Immunity, 40, 273-283.
- Langermann, S., Palaszynski, S., Barnhart, M., Auguste, G., Pinkner, J.S., Burlein, J., Barren, P., Koenig, S., Leath, S., Jones, C.H. and Hultgren, S.J. (1997) Prevention of mucosal Escherichia coli infection by FimH-adhesinbased systemic vaccination. Science, 276, 607-611. doi:10.1126/science.276.5312.607
- Langermann, S., Mollby, R., Burlein, J.E., Palaszynski, S.R., Auguste, C.G., DeFusco, A., Strouse, R., Schenerman, M.A., Hultgren, S.J., Pinkner, J.S., Winberg, J., Guldevall, L., Soderhall, M., Ishikawa, K., Normark, S. and Koenig, S. (2000) Vaccination with FimH adhesin protects cynomolgus monkeys from colonization and infection by uropathogenic Escherichia coli. The Journal of Infectious Diseases, 181, 774-778. doi:10.1086/315258
- Roberts, J.A., Kaack, M.B., Baskin, G., Chapman, M.R., Hunstad, D.A., Pinkner, J.S. and Hultgren, S.J. (2004) Antibody responses and protection from pyelonephritis following vaccination with purified Escherichia coli PapDG protein. Journal of Urology, 171, 1682-1685. doi:10.1097/01.ju.0000116123.05160.43
- O’Hanley, P., Lalonde, G. and Ji, G. (1991) Alpha-hemolysin contributes to the pathogenicity of piliated digalactoside-binding Escherichia coli in the kidney: Efficacy of an alpha-hemolysin vaccine in preventing renal injury in the BALB/c mouse model of pyelonephritis. Infection and Immunity, 59, 1153-1161.
- Goluszko, P., Goluszko, E., Nowicki, B., Nowicki, S., Popov, V. and Wang, H.Q. (2005) Vaccination with purified Dr fimbriae reduces mortality associated with chronic urinary tract infection due to Escherichia coli bearing Dr adhesin. Infection and Immunity, 73, 627-631. doi:10.1128/IAI.73.1.627-631.2005
- Russo, T.A., McFadden, C.D., Carlino-MacDonald, U.B., Beanan, J.M., Olson, R. and Wilding, G.E. (2003) The siderophore receptor IroN of extraintestinal pathogenic Escherichia coli is a potential vaccine candidate. Infection and Immunity, 71, 7164-7169. doi:10.1128/IAI.71.12.7164-7169.2003
- Hopkins, W.J., Elkahwaji, J., Beierle, L.M., Leverson, G.E. and Uehling, D.T. (2007) Vaginal mucosal vaccine for recurrent urinary tract infections in women: Results of a phase 2 clinical trial. Journal of Urology, 177, 1349- 1353, 1591. doi:10.1016/j.juro.2006.11.093
- Pizza, M., Scarlato, V., Masignani, V., Giuliani, M.M., Arico, B., Comanducci, M., Jennings, G.T., Baldi, L., Bartolini, E., Capecchi, B., Galeotti, C.L., Luzzi, E., Manetti, R., Marchetti, E., Mora, M., Nuti, S., Ratti, G., Santini, L., Savino, S., Scarselli, M., Storni, E., Zuo, P., Broeker, M., Hundt, E., Knapp, B., Blair, E., Mason, T., Tettelin, H., Hood, D.W., Jeffries, A.C., Saunders, N.J., Granoff, D.M., Venter, J.C., Moxon, E.R., Grandi, G. and Rappuoli, R. (2000) Identification of vaccine candidates against serogroup B meningococcus by whole-genome sequencing. Science, 287, 1816-1820. doi:10.1126/science.287.5459.1816
- Prokhorova, T.A., Nielsen, P.N., Petersen, J., Kofoed, T., Crawford, J.S., Morsczeck, C., Boysen, A. and SchrotzKing, P. (2006) Novel surface polypeptides of Campylobacter jejuni as traveller’s diarrhoea vaccine candidates discovered by proteomics. Vaccine, 24, 6446-6455. doi:10.1016/j.vaccine.2006.05.085
- Lopez, J.E., Siems, W.F., Palmer, G.H., Brayton, K.A., McGuire, T.C., Norimine, J. and Brown, W.C. (2005) Identification of novel antigenic proteins in a complex Anaplasma marginale outer membrane immunogen by mass spectrometry and genomic mapping. Infection and Immunity, 73, 8109-8118. doi:10.1128/IAI.73.12.8109-8118.2005
- Chenoweth, M.R., Greene, C.E., Krause, D.C. and Gherardini, F.C. (2004) Predominant outer membrane antigens of Bartonella henselae. Infection and Immunity, 72, 3097- 3105. doi:10.1128/IAI.72.6.3097-3105.2004
- Kurupati, P., The, B.K., Kumarasinghe, G. and Poh, C.L. (2006) Identification of vaccine candidate antigens of an ESBL producing Klebsiella pneumoniae clinical strain by immunoproteome analysis. Proteomics, 6, 836-844. doi:10.1002/pmic.200500214
- Schilling, J.D., Martin, S.M., Hung, C.S., Lorenz, R.G. and Hultgren, S.J. (2003) Toll-like receptor 4 on stromal and hematopoietic cells mediates innate resistance to uropathogenic Escherichia coli. Proceedings of the National Academy of Sciences of the United States of America, 100, 4203-4208. doi:10.1073/pnas.0736473100
- Zhang, D., Zhang, G., Hayden, M.S., Greenblatt, M.B., Bussey, C., Flavell, R.A. and Ghosh, S. (2004) A toll-like receptor that prevents infection by uropathogenic bacteria. Science, 303, 1522-1526. doi:10.1126/science.1094351
- Chromek, M., Slamova, Z., Bergman, P., Kovacs, L., Podracka, L., Ehren, I., Hokfelt, T., Gudmundsson, G.H., Gallo, R.L., Agerberth, B. and Brauner, A. (2006) The antimicrobial peptide cathelicidin protects the urinary tract against invasive bacterial infection. Nature Medicine, 12, 636-641. doi:10.1038/nm1407
- Haraoka, M., Hang, L., Frendeus, B., Godaly, G., Burdick, M., Strieter, R. and Svanborg, C. (1999) Neutrophil recruitment and resistance to urinary tract infection. The Journal of Infectious Diseases, 180, 1220-1229. doi:10.1086/315006
- Thumbikat, P., Waltenbaugh, C., Schaeffer, A.J. and Klumpp, D.J. (2006) Antigen-specific responses accelerate bacterial clearance in the bladder. The Journal of Immunology, 176, 3080-3086.
- Uehling, D.T., Johnson, D.B. and Hopkins, W.J. (1999) The urinary tract response to entry of pathogens. World Journal of Urology, 17, 351-358. doi:10.1007/s003450050160
- Alteri, C.J. and Mobley H.L. (2007) Quantitative profile of the uropathogenic Escherichia coli outer membrane proteome during growth in human urine. Infection and Immunity, 75, 2679-2688.
- Hanson, M.S., Hempel, J. and Brinton Jr., C.C. (1988) Purification of the Escherichia coli type 1 pilin and minor pilus proteins and partial characterization of the adhesin protein. Journal of Bacteriology, 170, 3350-3358.
- Molloy, M.P., Herbert, B.R., Slade, M.B., Rabilloud, T., Nouwens, A.S., Williams, K.L. and Gooley, A.A. (2000) Proteomic analysis of the Escherichia coli outer membrane. European Journal of Biochemistry, 267, 2871- 2881. doi:10.1046/j.1432-1327.2000.01296.x
- Hopkins, W.J. and Uehling, D.T. (1995) Resolution time of Escherichia coli cystitis is correlated with levels of preinfection antibody to the infecting Escherichia coli strain. Urology, 45, 42-46. doi:10.1016/S0090-4295(95)96444-4
- Russo, T.A., McFadden, C.D., Carlino-MacDonald, U.B., Beanan, J.M., Barnard, T.J. and Johnson, J.R. (2002) IroN functions as a siderophore receptor and is an urovirulence factor in an extraintestinal pathogenic isolate of Escherichia coli. Infection and Immunity, 70, 7156-7160. doi:10.1128/IAI.70.12.7156-7160.2002
- Russo, T.A., Carlino, U.B. and Johnson, J.R. (2001) Identification of a new iron-regulated virulence gene, ireA, in an extra intestinal pathogenic isolate of Escherichia coli. Infection and Immunity, 69, 6209-6216. doi:10.1128/IAI.69.10.6209-6216.2001
- Johnson, J.R., Jelacic, S., Schoening, L.M., Clabots, C., Shaikh, N., Mobley, H.L. and Tarr, P.I. (2005) The IrgA homologue adhesin Iha is anEscherichia coli virulence factor in murine urinary tract infection. Infection and Immunity, 73, 965-971. doi:10.1128/IAI.73.2.965-971.2005
- Lane, M.C., Lockatell, V., Monterosso, G., Lamphier, D., Weinert, J., Hebel, J.R., Johnson, D.E. and Mobley, H.L. (2005) Role of motility in the colonization of uropathogenic Escherichia coli in the urinary tract. Infection and Immunity, 3, 7644-7656. doi:10.1128/IAI.73.11.7644-7656.2005

