Advances in Bioscience and Biotechnology
Vol.3 No.3(2012), Article ID:20005,3 pages DOI:10.4236/abb.2012.33035
Agrobacterium-mediated transient transformation of Pentalinon andrieuxii Müll. Arg.
![]()
1Centro de Investigación Científica de Yucatán, A.C. Laboratorio de Química Orgánica, Unidad de Biotecnología, Mérida, México
2Unidad de Bioquímica y Biología Molecular, Mérida, México
3Departamento de Microbiología Ambiental y Biotecnología, Universidad Autónoma de Campeche, Campeche, México
Email: *ggodoy@cicy.mx, alexqbb@cicy.mx, avibeli@cicy.mx, manjchan@mail.uacam.mx, lmanuel@cicy.mx
Received 13 March 2012; revised 20 April 2012; accepted 30 April 2012
Keywords: β-Glucuronidase; Agrobacterium tumefaciens; Kanamycin; Apocynaceae
ABSTRACT
Sections of hypocotyls, roots and leaves from Pentalinon andrieuxii plantlets were transiently transformed with Agrobacterium tumefaciens LBA4404 bearing the binary plasmid pCAMBIA2301 with an interrupted β-glucuronidase (GUS) gene. Histochemical GUS assays showed transient gene expression in all infected tissues, being older roots those which displayed the most intense GUS staining. To our knowledge, this is the first report of Pentalinon andrieuxii susceptibility to Agrobacterium tumefaciens-mediated genetic transformation.
1. INTRODUCTION
Pentalinon andrieuxii Müll. Arg. (Apocynaceae), a plant commonly named “contrayerba” in the Yucatan Peninsula, is used in the treatment of Leishmania’s skin lesions (“chiclero’s ulcer”). Mayan healers also recommend chewing their roots and leaves to relieve ailments derived from snakebites, and the stem-collected latex to alleviate headaches and nervous disturbances [1,2]. Cardenolides, pyrrolizidine alkaloids, steroidal compounds and betulinic acid derivatives with different physiological activities have been found in Pentalinon tissues [3-6]. Furthermore, two physiologically inactive, but structurally unusual trinorsesquiterpenoids, named urechitols A and B have been described in this species. Urechitols include the novel bicyclic campechane skeleton, which is formed by two cyclic nuclei of five and seven carbon units, respectively [7]. Although, the synthesis of racemic mixtures of urechitol A was recently reported [8], the biosynthetic origin of urechitols or the campechane skeleton remains unknown. The availability of in vitro culture systems of P. andrieuxii tissues, including those used for genetic transformation, may allow not only the controlled production of these compounds, but also the development of the basic tools for functional genetics applied to the identification of genes involved in the biosynthetic pathway of urechitols. In here, we report the development of a protocol for the transient transformation of P. andrieuxii explants with Agrobacterium tumefaciens.
2. MATERIALS AND METHODS
2.1. Plant Material
P. andrieuxii seeds were collected from mature dehiscent siliques in February 2009 from a population located 3.5 km northeast from Campeche City, Mexico (19˚51'0"N, 90˚31'50"W). A voucher specimen was deposited in the Herbarium of Centro de Investigación Científica de Yucatán (P. Sima 2503). Seeds were surface sterilized with 5% Extran and 70% ethanol for 5 min each, followed by immersion in a 50% bleach solution (3% sodium hypochlorite) for 20 min. Disinfested seeds were germinated in modified hormone-free PC-L2 medium [9] pH 5.5, supplemented with 2.5% sucrose and 1% agar. Eight seeds per container were incubated at 25˚C ± 2˚C for two weeks in the dark, and then on, under continuous light (40 - 50 mmol·m2·seg–1).
2.2. Agrobacterium tumefaciens Strains and Vectors
Agrobacterium tumefaciens LBA4404 strain and the binary vector pCAMBIA 2301 (Center for the Applications of Molecular Biology to International Agriculture, Canberra, Australia (http://www.cambia.org/daisy/bioforge_ legacy/3724.html)) were used in all experiments. The pCAMBIA 2301 plasmid contains the neomycin phosphotransferase II (nptII) gene for kanamycin selection, in addition to the uidA gene encoding the β-glucuronidase gene (GUS), interrupted by the catalase intron, for the phenotypical selection of transformed tissues. Both genetic markers are driven by the CaMV 35S promotor. The presence of an intron in uidA ensures expression in eukaryotic cells. A. tumefaciens was cultured in liquid yeast extract and beef (YEB) medium, pH 5.6, containing 100 mg·l–1 rifampicin and streptomycin each (Sigma, St. Louis, MO). Cultures were kept in the dark at 28˚C for 48 h. Bacteria were made competent with CaCl2 and transformed with the plasmid via heat shock [10]. Transformed cells, harboring pCAMBIA 2301, were screened on semisolid YEB media with 100 mg·l–1 rifampicin and streptomycin each, and 50 mg·l–1 kanamycin (YEB-AB), and then, cultured in 10 ml of liquid YEB-AB medium (pH 5.6). Cultures were kept at 28˚C for 48 h in a rotatory shaker (200 rpm). A 200-μl aliquot of this suspension was diluted in 10 ml of YEB-AB, and further incubated for 24 h as described. Culture volume was then completed to 20 ml with YEB-AB, and added with 200 μM acetosyrin-gone (AS). This was incubated up to 5 h, prior to tissue inoculation.
2.3. Genetic Transformation
Sections of hypocotyls, roots (10 mm length) and leaves (ca. 0.25 cm2) were excised from 15 day-old plantlets and superficially wounded in a longitudinal manner with a scalpel prior to infection with A. tumefaciens. Explants were vacuum infiltrated with a 20 ml of a 0.1 OD600 bacterial suspension in PC-L2 medium for 20 min. Tissues were then blotted with sterile filter paper in order to eliminate the bacterial excess, and placed on semisolid PC-L2 medium for 72 h. After 3 days of cocultivation, at 28˚C in the dark, explants were transferred to semisolid PC-L2 medium supplemented with 100 mg·l–1 cefotaxime and 10 mg·l–1 kanamycin for further development. Transient GUS expression was histochemically assayed 3, 6 and 21 days after infection by staining transformed tissues with 5-bromo-4-chloro-3-indolyl-β-D-glucuronide (XGLUC) [11]. Briefly, 50 explants were vacuum-infiltrated in the buffer solution for 5 min, and thereafter incubated for 24 h at 37˚C in the dark [12]. GUS activity was estimated by the number of blue spots per explant after washing them in a 3:1 (v/v) mixture of methanol: acetone. A blue spot was considered as a single transient GUS-expression focus.
3. RESULTS AND DISCUSSION
Transient Transformation of Pentalinon andrieuxii
After 21 days of infection, transient GUS expression was found in both root and leaf sections (Figure 1). Positive GUS activity could be solely attributed to the expression and correct splicing of the inserted uidA in eukaryotic plant cells. P. andrieuxii does not present endogenous GUS activity as reported for carrot, celery, parsley, rice [13], Ricinus communis [14], Agave fourcroydes [15] and Capsicum chinense [16]. Older roots displayed a higher GUS activity than younger root explants and leaf sections. Interestingly, even when hypocotyls’ explants did not show GUS activity during the first 20 days of infection, after a total of 40 days and once tissues had turned into undifferentiated calli, significant activity was observed (Figure 1). Older root tissues (two months) were the most susceptible to infection with a 26% of positive transformation events, followed by leaves (15%) and younger roots (3.3%) (Table 1). Furthermore, older roots also presented the highest foci number per explant in comparison to leaf sections (7.83 vs 3.96).
4. CONCLUSION
In conclusion, we have developed the first protocol for transient genetic transformation of P. andrieuxii. At the present time, this method is being used to probe a protocol for its stable genetic transformation, which in combination with the already developed plant regeneration system [17], will allow to assay the functional role of genes putatively involved in secondary metabolism.
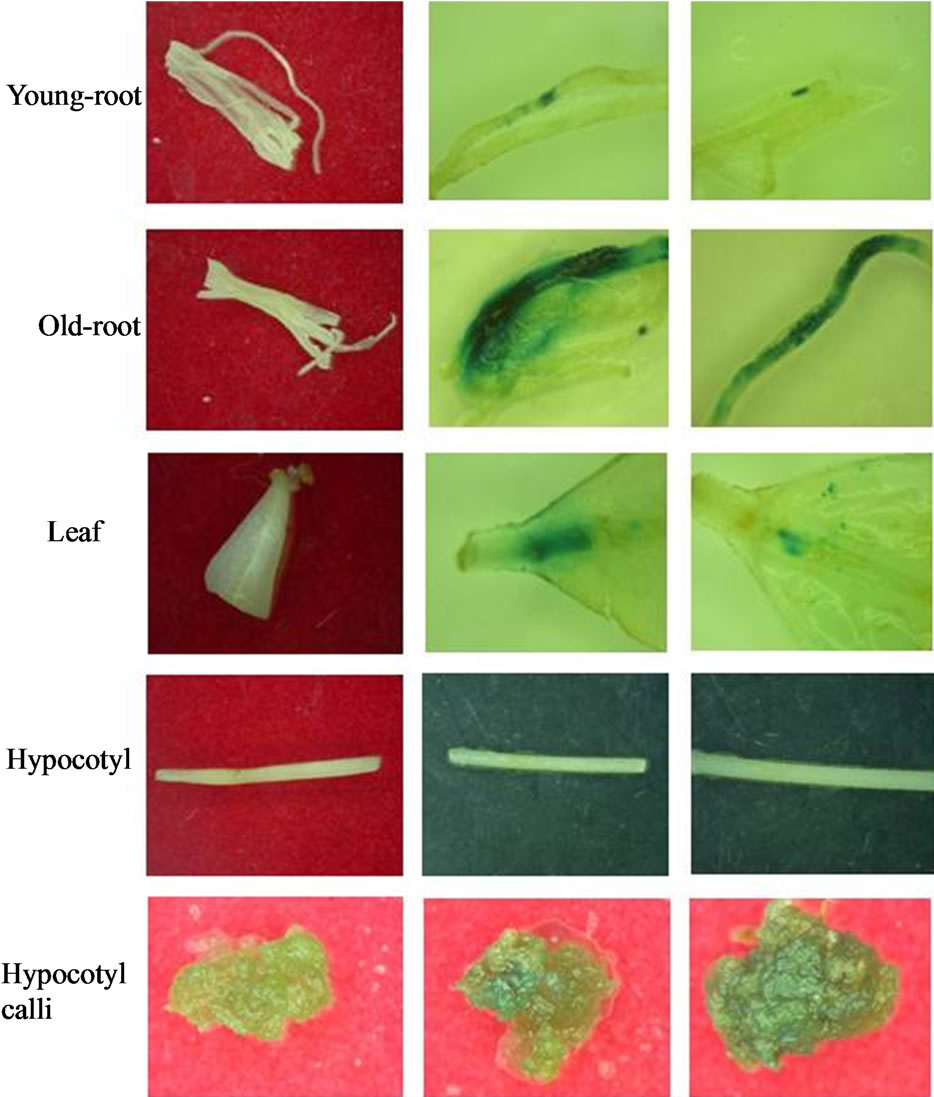
Figure 1. Histochemical GUS assay performed on tansformed explants of Pentalinon andrieuxii Müll. Arg. Explants tansformed with Pcambia2301 via Agrobacterium tumefaciens stain LBA4404.
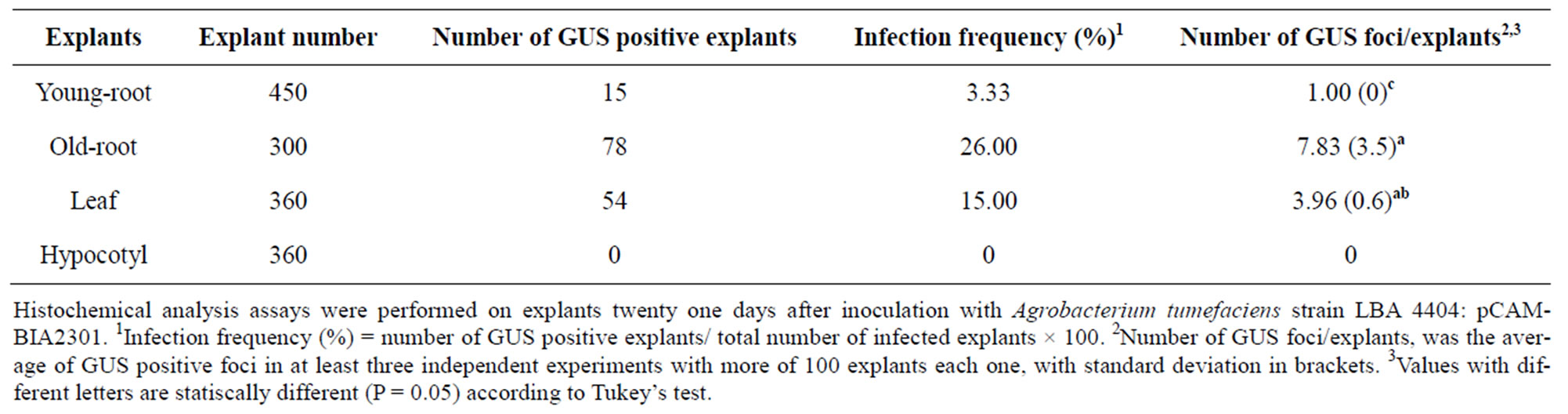
Table 1. Infection frequency and number of GUS foci in P. andrieuxii explants.
5. ACKNOWLEDGEMENTS
The authors acknowledge receiving financial support from the National Council for Science and Technology-Mexico (CONACYT) (Project No. 59695-Z).
REFERENCES
- Chan-Bacab, M.J., Balanza, E., Deharo, E., Muñoz, V., García-Durán, R. and Peña-Rodriguez, L.M. (2003) Variation of leishmanicidal activity in four populations of Urechites andrieuxii. Ethnopharmacology, 86, 243-247. doi:10.1016/S0378-8741(03)00011-4
- Lezama-Dávila, C.M., Isaac-Márquez, A.P., Zamora-Cresencio, P., Uc-Encalada, M.R., Justiniano-Apolinar, S.Y., Angel-Robles, R., del., Satoskar, A. and Hernández-Rivero, L. (2007) Leishmanicidal activity of Urechites andrieuxii. Fitoterapia, 78, 255-257. doi:10.1016/j.fitote.2006.12.005
- Tillequin, F., Michael, S. and Seguin, I. (1993) Alkaloids and sulphur compounds. Academic Press, San Diego.
- Yam-Puc, A., Chee-González, L., Escalante-Erosa, F., Arunachalampilai, A., Wendt, O.F., Sterner, O., GodoyHernández, G. and Peña-Rodríguez, L.M. (2012) Steroids from the root extract of Pentalinon andrieuxii. Phytochemistry Letters, 5, 45-48. doi:10.1016/j.phytol.2011.09.004
- Domínguez-Carmona, D.B., Escalante-Erosa, F., GarcíaSosa, K., Ruíz-Pinelli, G., Gutiérrez-Yapu, D., Chan-Bacab, M.J., Giménez-Turba, A. and Peña-Rodríguez, L.M. (2010) Antiprotozoal activity of Betulinic acid derivatives. Phytomedicine, 17, 379-382. doi:10.1016/j.phymed.2009.08.002
- Yogeeswari, P. and Sriram, D. (2005) Betulinic acid and its derivatives: A review on their biological properties. Current Medicinal Chemistry, 12, 657-666. doi:10.2174/0929867053202214
- Yam-Puc, A., Escalante-Erosa, F., Pech-López, M., ChanBacab, M.J., Arunachalampilai, A., Wendt, O.F., Sterner, O. and Peña-Rodríguez, L.M. (2009) Trinorsesquiterpenoids from the root extract from Pentalinon andrieuxii Muell-Arg. Journal of Natural Products, 72, 745-748. doi:10.1021/np800554n
- Sumiya, T., Ishigami, K. and Watanabe, H. (2010) Stereoselective total synthsis of (±)—Urechitol A. Angewandte Chemie International Edition, 49, 5527-5528. doi:10.1002/anie.201002505
- Phillips, G.C. and Collins, G.B. (1979) In vitro tissue culture of selected legumes and plant regeneration from callus culture of red clover. Crop Science, 19, 59-64. doi:10.2135/cropsci1979.0011183X001900010014x
- Zhang, H.X. and Zeevaart, J.A.D. (1999) An efficient Agrobacterium tumefaciens-mediated transformation and regeneration system for cotyledons of spinach (Spinacia oleracea L.). Plant Cell Reports, 18, 640-645. doi:10.1007/s002990050635
- Jefferson, R. (1987) Assaying chimeric genes in plants: The GUS gene fusion system. Plant Molecular Biology Reporter, 5, 387-405. doi:10.1007/BF02667740
- Humara, J.M., López, M. and Ordas, R.J. (2003) Agrobacterium tumefaciens-mediated transformation of Pinus pinea L. cotyledons: An assessment of factors influencing the efficiency of uidA gen transfer. Plant Cell Report, 19, 51-58. doi:10.1007/s002990050709
- Hu, C.Y., Chee, P.P., Chesney, R.H., Zhou, J.H., Miller, P.D. and O’Brien, W.T. (1990) Intrinsic GUS-like activities in seed plants. Plant Cell Reports, 9, 1-5. doi:10.1007/BF00232123
- Rezmer, C., Schlichting, R., Wachter, R. and Ullrich, C.I. (1999) Identification and localization of transformed cells in Agrobacterium tumefaciens-induced plant tumors. Planta, 209, 399-405. doi:10.1007/s004250050742
- Godoy-Hernández, G., Avilés-Berzunza, E., Carrillo-Pech M. and Vázquez-Flota, F. (2008) Agrobacterium-mediated transient transformation of Mexican prickly poppy (Argemone mexicana L.). Electronic Journal of Biotechnology, 11, 1-5. http://158.251.16.248/content/vol11/issue1/full/3/3.pdf
- Solís-Ramos, L.Y., González-Estrada, T., Andrade-Torres, A., Godoy-Hernández, G. and Castaño de la Serna, E. (2010) Endogenous GUS-like activity in Capsicum chinense Jacq. Electronic Journal of Biotechnology, 13, 1-7. http://redalyc.uaemex.mx/redalyc/pdf/1733/173316385002.pdf
- Martin-Acosta, J.C., Avilés-Berzunza, E. and GodoyHernández, G. (2012) In vitro plant regeneration from explants of Pentalinon andrieuxii (Müll. Arg.). Hansen & Wunderlin, Unpublished.
NOTES
*Corresponding author.

