Journal of Environmental Protection
Vol.06 No.12(2015), Article ID:62527,18 pages
10.4236/jep.2015.612126
Synthesis of Nano-Titanium Tannate as an adsorbent for Crystal Violet Dye, Kinetic and Equilibrium Isotherm Studies
Taha M. Elmorsi
Chemistry Department, Faculty of Science, Al-Azhar University, Cairo, Egypt

Copyright © 2015 by author and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 15 December 2015; accepted 28 December 2015; published 31 December 2015
ABSTRACT
The purpose of this study was to prepare nano-titanium tannate complex (TTC) and to investigate its adsorption capacity for removal of cationic dyes such as crystal violet (CV) dye. The morphology and the main elements of TTC adsorbent were characterized by scanning electron microscopy (SEM-EDS), while its crystal structure was characterized by X-ray diffraction (XRD). Also, FT-IR spectroscopy study structural aspects of TTC. A “cotton-ball”-like and porous surface structure of titanium tannate complex (TTC) with nanoparticle size of 16.18 nm show high capability for absorbing crystal violet dye. The effect of several parameters such as contact time, initial concentrations of CV, solution pH and the amount of TTC was investigated. Three different kinetic equations such as pseudo-first order, pseudo-second order and intraparticle diffusion were used to study the order and the mechanism of the adsorption process. The adsorption of CV dye followed pseudo- second order equation. Moreover, equilibrium data were tested with four adsorption isotherm models namely, Langmuir, Freundlich, Temkin and Dubinin-Radushkevich (D-R). Langmuir was the best fit for the data with maximum capacity as 58.8 mg/g. The results of Separation factor, Surface coverage and standard free energy (DG˚) indicated that adsorption of CV onto TTC was favorable with fast rate and spontaneous physical adsorption process.
Keywords:
Titanium(III) Tannate Complex (TTC), Crystal violet, Adsorption kinetics, Adsorption isotherms

1. Introduction
Due to the rapid growth of the need for water in different industries, huge number of different pollutants accumulated in water. In many cases, these pollutants make water unsuitable to be reused and become wastewater [1] . Presence of these pollutants in water such as organic dyes and heavy metals became a major concern not only for the human health but for the entire environment quality [2] [3] . In addition, water polluted with low concentration of organic dyes [4] [5] or even trace levels of heavy metals can cause serious health problems to human body [6] . Hence, there is an urgent need for water treatment using different techniques. Currently, several processes such as chemical, biological and/or physical processes [7] -[12] have been used for the treatment of different pollutants. Physical processes such as adsorption method are a unique technique used for accumulating the pollutant molecules onto the surface of the adsorbent to remove it from the aqueous or organic phase. Adsorption is not only a least complex technique but it is cheap and effective method for removal of different pollutants [13] [14] . Although activated carbon is the effective adsorbent used in adsorption method, its production is highly coast [15] and requires complex care [16] . Production of an alternative effective adsorbent which does not require many processing is of great interest. There are many low-coast adsorbents (also called green adsorption), used for removal of different pollutants [16] . However, the adsorption capacity of many of them is not large [17] . Therefore, other adsorbents with better adsorption capacities and good performance are still under development [17] . It is known that several industries including textiles, plastics and printing use different dyes such as cationic, anionic and non-ionic dyes for coloring [18] . Cationic dyes were used extensively due to their good characteristics such as low coast production, solubility, bright color and applying to fabric. It was reported that from 0% to 5% of the produced cationic dyes were released directly to the wastewater [19] . Crystal violet (CV) a type of a synthetic basic triphenylmethane dyes which can exist in the form of colored cations [20] . CV is mainly used in textile and medicine industries [21] . Aqueous effluents of CV are toxic and may absorb through skin causing different health problems [15] [22] . CV was chosen as a pollutant model to evaluate the efficiency of the studied adsorbent. To the best of our knowledge, the research of R. Zhang et al., [23] was the only work on an optimized method for synthesizing ferric tannate complex by using tannic acid. However, no work has been done to synthesis titanium(III) tannate complex (TTC) by using tannic acid and Ti(III) chloride. In this study, we reported the synthesis and characterization of titanium(III)-tannate complex (TTC) in addition to investigating its adsorption capacity for adsorption of cationic dyes such as CV dye. Several parameters were investigated such as adsorbent dosage, contact time, initial CV dye concentration and temperature. In addition, adsorption isotherm kinetics of the adsorption process were studied.
2. Experiment
2.1. Chemicals
Analytical-grad that chemicals were used in this work without further purification. Crystal violet, a type of triphenylmethane dyes with molecular formula (C25H30ClN3), (aq. solubility 16 g/l; purity 80%) as shown in Figure 1 was received from LOBA Chemie Company, India and used as received. To evaluate the significant of the adsorbent, stock solutions (1000 mg/L) of CV was prepared in double distilled water and further diluted to perform adsorption experiments in presence of titanium(III)-tannate complex (TTC) as an adsorbent. Dilute solutions of 1M HNO3 and 1M NaOH (BDH) were used to adjust pH of CV solutions using a pH meter.
Figure 1. Crystal violet structure (mol. formula C25H30ClN3).
2.2. Synthesis of Titanium(III) tannate Complex (Adsorbent)
A Beaker with an amount of 50 ml (4%:1.0%, ethanol: water) of tannic acid (0.1 M) was placed in ultrasonic bath and sonicated for few minutes. Then a cretin volume of TiCl3 was added drop wise with continuous sonication for 8.0 minutes. The molar ratio of tannic acid to titanium(III) chloride was chosen to be 1:20 according to R. Zhang et al., [23] . The pH of the mixture was adjusted to 3.5 - 4.0 using tris-HCl to avoid precipitation of titanium ion in the form of hydroxide. The colorless solution of tannic acid turned to orange precipitate at the beginning of the addition of titanium(III) chloride then changed into dark red precipitate. Complete precipitation was tested by adding few drops of both tannic acid solution and titanium ion solution respectively. There was no more precipitate formed to indicate that the chosen molar ratio of tannic acid to titanium(III) chloride was optimal. The obtained product was separated through precipitation and washed several times with 30%:70% ethanol: water then dried in the oven overnight at 60˚C before use in the adsorption experiments.
2.3. Characterization of Titanium(III) tannate
To study the surface morphology and main elements present in the complex of titanium tannate, scanning electron microscopy (SEM) using SEM model Quanta 250 FEG attached with an energy dispersive X-ray spectroscopy (EDS) unit with accelerating voltage 30 K.V., magnification14x up to 1000,000 and resolution for Gun.1n was used. The crystal structure of titanium tannate was analyzed by X-ray diffraction (XRD) in scan mode with Cu-Ka radiation (l = 0.01542 nm) in the 2q range of 10˚ to 80˚. The functional group in the synthesized complex was analyzed through the analysis of Fourier transform infrared spectrometry (FTIR) using KBr pellet in the range of 400 to 4000 cm−1.
2.4. Adsorption Experiments
To evaluate the significant of titanium(III) tannate complex (TTC) as an adsorbent, CV dye solution was used in this study as an environmental pollutants. Stock solutions (1000 mg/L) of CV was prepared in distilled water and further diluted to the required concentration to perform the calibration curves and adsorption experiments. Serial dilutions (1.0 to 6.0 mg/L) of CV stock solutions were used to prepare the calibration curves. Batch adsorption experiments were performed by introducing known amounts of TTC into several glass tubes, each containing 5.0 mL (to minimize the waste) of CV solution with an initial concentration ranging from 40 to 150 mg/L. Tubes were agitated at 120 rpm using an electric shaker at room temperature. To ensure performing the adsorption experiments at known pH value, the pH was adjusted before starting the experiment using solution of either NaOH or HCl. At interval times, CV solutions were withdrawn and introduced into a centrifuge to separate TTC powder from samples. CV solutions of 50 mg/L with different amount of TTC powder (0.5 to 2.5 g/L) were used to study the effect of adsorbent dose on the adsorption process. The effect of pH was studied by performing similar experiments with 2.0 g/L of TTC powder at different pHs from 2.75 to 10. A known amount of TTC powder (2.0 g/L) were added to 5.0 mL of CV solution with different concentrations (40 to 150 mg/L) at room temperature to calculate the adsorption isotherm.
2.5. Analytical Methods
UV-vis spectrophotometer (Perkin Elmer) was used to determine the concentrations of CV solution before and after the adsorption experiments. Calibration curves were obtained and used to calculate dye concentrations during adsorption experiments at any time (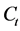 ).
).
The amount of CV dye sorbed onto TTC powder at any time, 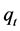 (mg/g), was calculated from;
(mg/g), was calculated from;
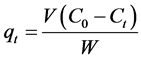 (1)
(1)
At equilibrium, 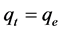 and
and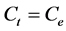 ; therefore the amount of sorbed CV dye,
; therefore the amount of sorbed CV dye, 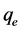 , was calculated from
, was calculated from
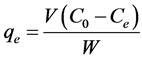 (2)
(2)
where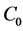 ,
, 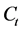 and
and 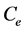 are the initial concentration, concentration at any time and equilibrium concentrations of CV dye solution (mg/L), respectively, V is the volume of the dye solution (L), and W is the mass of TTC (g). Dye removal percentage can be calculated as follows:
are the initial concentration, concentration at any time and equilibrium concentrations of CV dye solution (mg/L), respectively, V is the volume of the dye solution (L), and W is the mass of TTC (g). Dye removal percentage can be calculated as follows:
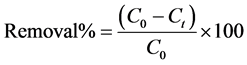 (3)
(3)
2.6. Adsorption kinetics
Kinetic Study will allow the determination of the rate of CV uptake by TTC adsorbent which in turn led to measuring the efficiency of the adsorption process. Three different kinetic models namely a pseudo-first order [24] -[26] , a pseudo-second order [27] [28] and an intraparticle diffusion [29] were used in the current study. The linear form of Lagergren pseudo-first order model is represented as:
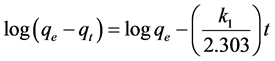 (4)
(4)
where  and
and 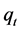 are the amounts of the CV adsorbed (mg/g) at equilibrium and at time t (min), respectively, and
are the amounts of the CV adsorbed (mg/g) at equilibrium and at time t (min), respectively, and 
While, the linear form of pseudo second-order developed by Ho [27] based on equilibrium adsorption can be expressed as:

where 
Also, intraparticle diffusion equation can be described by the following equation:

where, 

2.7. Adsorption isotherm
The equilibrium state between the amount of adsorbed onto the adsorbent surface (

3. Results and Discussion
3.1. Characterization of Titanium(III) Tannate
SEM images of the morphology of the synthesized titanium tannate complex (TTC) is shown in Figure 2 at two different magnification view as low (30,000×) and high (100,000×). The images show bladed crystals for TTC with “cotton-ball”-like morphology and porous surface structure. In addition, the average particle size was estimated from SEM image (Figure 2(b)) as 16.18 nm. The main elements present in TTC is determined by SEM- EDS spectrum and shown in Figure 3. The results indicated that the carbon, oxygen, titanium were the main elements present in TTC structure with percent composition as 21.07%, 48.95% and 28.75% by weight respectively. The calculated percent composition from the SEM-EDS spectrum shows that the ratio of carbon to oxygen is different from their ratio in the chemical formula of tannic acid (C76H52O46). Although previous research [23] proposes a possibility of hydrolysis of metal ions such as Ti(III) which led to increase the % of oxygen, further research is required to prove that. Figure 4 shows the XRD patterns of 16.18 nm TTC with a diffraction peaks appear in the range of 2q = 30˚ to 35˚ indicated that titanium tannate complex has an amorphous structure.
Figure 2. (a) Low (30,000×) and (b) high (100,000×) magnification SEM images of TTC.
Figure 3. SEM-EDS spectrum of TTC.
Figure 4. XRD spectra of the titanium tannate complex.
The results in Figure 5 show the FTIR spectra of tannic acid (TA) and titanium tannate complex (TTC). The results show that the main bands appeared in the spectrum of tannic acid are the stretching vibrations band of hydroxyl group (OH) of the phenolic ring which appeared as abroad band at 3700 cm−1 to 3000 cm−1 range due to the hydrogen bonding between OH. Also, stretching vibration carbonyl groups (C=O) appeared at 1730 - 1705 cm−1, in addition to C-O bands at 1100 - 1300 cm−1 as reported also previously [23] [37] . For FTIR spectra of TTC (solid line in Figure 5), the major difference was in the OH group region in addition to the C-O region where absorption was much less indicating that the C-O bonds had been weakened as would occur when chelated with titanium ion. Bonding therefore occurred between the metal ion and the hydroxyl groups. Accordingly, the suggested structure of titanium tannate complex is shown in Figure 6.
3.2. Effect of Contact Time and initial concentrations of CV Dye
A known amount of ttc powder 0.010 g (2.0 g/L) was added to 0.005 L of different concentrations of CV (from 40 - 150 mg/L). For a total period of 120 min at a temperature of 298 K, experiments were performed to evaluate the effect of contact time (0 - 120 min) and different initial concentrations (40, 50, 70, 90 and 150 mg/L) of CV dye on the adsorption process as shown in Figure 7. The obtained results in indicated that the curve of the adsorption rate of CV onto TTC powder was very rapid in the first 10 min confirming the presence of large number of readily accessible adsorption sites. For example at 40 mg/L of CV the adsorbed amount (
Figure 5. FTIR of tannic acid (TA) and titanium tannate complex (TTC).
Figure 6. Suggested chemical structure of titanium tannate complex.
Figure 7. Effect of initial concentrations and contact time on the adsorption of crystal violet dye onto TTC powder (T = 298 K, time = 120 min, [CV]0 = 40 - 150 mg/L, pHi = 7, V = 0.005 L, TTC dosage = 2.0 g/L).
16.85 mg/g, which represented about 78.11%, comparing to 19.91 mg/g (92.3%) at 120 min. Then, the adsorption of CV dye increased gradually during the following 40 min until reached equilibrium at about 120 min. Furthermore, the sorbed amount of CV dye was also increased from 16.85 to 42.10 mg/g by increasing the initial concentrations from 40 to 150 mg/L respectively. The results showed that the uptake of CV dye by TTC powder depends on the contact time and the initial concentration. This may be due to the time required for the CV dye to encounter the boundary layer effect, then diffuse to the surface of TTC powder and finally diffuse to the porous structure [38] . Also, it was noted that the time profile of the adsorption of CV dye reached saturation in a single, smooth and contentious curve which indicated the possible formation of monolayer of CV dye onto the surface of TTC powder [39] . To ensure complete equilibrium of the data, adsorption samples were collected at 120 min.
3.3. Effect of Solution pH on adsorption of CV
Adsorption rate can be affected by the degree of ionization of adsorbate and the surface charge of adsorbent [40] -[42] . However, these two parameters were controlled mainly by the initial solution pH. To evaluate the effect of pH on the equilibrium adsorption capacity (
Figure 8. Effect of solution pH on the adsorption of crystal violet dye onto TTC (T = 298 K, time = 120 min, [CV]0 = 50 mg/L, V = 0.005 L, TTC dosage = 2.0 g/L).
3.4. Effect of Adsorbed Amount
To investigate the effect of adsorbent dosage on the adsorption of CV, different tubes filled with a specific volume of CV dye solution with [CV]0 (50 mg/L) at different amounts of TTC powder (0.25 to 2.5 g/L) at room temperature. Figure 9 showed a general trend for the adsorption of CV onto the surface of TTC. The percent removal of CV increased rapidly in the first stage with the increase in the adsorbent dose then increased slowly until reached equilibrium with the further increase in the adsorbent dose. The solution of CV dye is decolorized by 31.63% by using 0.5 g/L of TTC dosage. The decolonization rate of CV solution increases dramatically by introducing more dosage of TTC to reach 96.27% at dosage of 2.0 g/L TTC. Furthermore, increasing the dosage of TTC to 2.5 g/L leads to decrease the % removal to reach 92.95%. Thus in further experiments 2.0 g/L of TTC were chosen as the optimum dose. It is clear that the increase in both the surface area and the adsorption sites which resulted by introducing more dosage of TTC adsorbent, lead to increase the % removal of CV from aqueous solution. On the other hand, % removal of CV decreased from 96.27% to 92.95% when introducing more amount of TTC as 2.5 g/L. It was reported that [46] increasing the amount of the adsorbent may led to increase the overlapping and/or aggregation of the adsorption sites on the adsorbent surface which can decrease the adsorption rate.
3.5. Adsorption Kinetics (Determining adsorption rate Constant)
Determining the kinetic parameter (such as the rate constant) and predicting information about adsorbent/adsorbate interaction it is significant for any adsorption experiments. As discussed above, three different models were used to study the kinetic of adsorption of CV onto TTC powder.
3.5.1. Pseudo First-Order Equation
Equation (4) expressed the linear form of Langergren pseudo first-order for the adsorption process of CV dye onto TTC adsorbent. Different concentrations of CV dye (40 - 150 mg/L) were used to calculate 






3.5.2. Pseudo-Second Order Rate Equation
The kinetic data of the adsorption of CV onto TTC were further analyzed by the linear form of pseudo-second
order model (Equation (5)). The slope and the intercept of the plot of 
Figure 9. Effect of adsorbent dose on the adsorption of crystal violet onto TTC (T = 298 K, time = 120 min, [CV]0 = 50 mg/L, pHi = 7, V = 0.005 L, ttc dosage = 0.5.0 - 2.5 g/L).
Figure 10. Pseudo-first order (a) and Pseudo-second order (b) kinetics for the adsorption of CV onto TTC (T = 298 K, time = 120 min, [CV]0 = 40 - 150 mg/L, pHi = 7, V = 0.005 L, TTC dosage = 2.0 g/L).
Table 1. Adsorption kinetic parameters for the adsorption of CV onto TTC (T = 298 K, time = 120 min, [CV]0 = 40 - 150 mg/L, pHi = 7, V = 0.005 L, TTC dosage = 2.0 g/L).
of both 






3.5.3. Intra-Particle Diffusion Study
It is proposed that the uptake of the adsorbate (such as CV dye) by the adsorbent (such as TTC) varies almost proportionately with the square root of the contact time (t1/2) according to the Equation (6) [13] . In order to investigate the mechanism of CV dye adsorption onto TTC, intra-particle diffusion based mechanism was studied. The results indicated that the plot in Figure 11 was not linear over the whole time range. Also, the plot indicated that the external surface adsorption (stage 1) is absent and it is finished before 10 min. Then after 10 min, the intra-particle diffusion control (stage 2) started and kept on to 60 min. Finally, the equilibrium step (stage 3) appeared after 60 min up to 120 min. It can be seen that the adsorption of CV dye onto TTC involved more than one process, and the intra-particle transport is not the rate-limiting step.
3.6. Adsorption isotherms for Crystal Violet
The affinity of TTC adsorbent for the adsorption of CV dye was evaluated by using different adsorption isotherms such as Langmuir, Freundlich, Temkin and Dubinin-Radushkevich (D-R).
3.6.1. Langmuir Isotherm
The Langmuir isotherm model assumes that a monolayer of adsorbed material (in liquid, such as CV is adsorbed over a uniform adsorbent surface such as TTC. The Langmuir-I equation is derived by some mathematical manipulation as:

where 




Figure 11. Intra-particle diffusion plot for the adsorption of CV onto TTC (T = 298 K, time = 120 min, [CV]0 = 40, 70 and 150 mg/L (from bottom to top respectively), pHi = 7, V = 0.005 L, TTC dosage = 2.0 g/L).
Figure 12(a) shows the linear fit of Langmuir-I for the adsorption of CV onto TTC at room temperature. The value of





Figure 12. Adsorption isotherm models, (a) Langmuir-I, (b) Freundlich, (c) Temkin and (d) D-R for the adsorption of CV onto TTC (T = 298 K, time = 120 min, [CV]0 = 40 - 150 mg/L, pHi = 7, V = 0.005 L, TTC dosage = 2.0 g/L).
Table 2. Langmuir, Freundlich, Temkin and D-R constants for the adsorption of CV onto TTC (T = 298 K, time = 120 min, [CV]0 = 40 - 100 mg/L, pHi = 7, V = 0.005 L, dosage = 2.0 g/L).
Table 3. Comparison of the maximum monolayer adsorption of CV dye onto various adsorbents.
Separation Factor and Surface Coverage (θ)
There are two factors such separation factor (

where 









In addition, the surface coverage (θ) of the adsorbent (TTC) is related to the initial concentration of CV dye - (


where 


3.6.2. Freundlich Isotherm
The adsorption process takes place on a heterogeneous surface when the resulted data followed Freundlich isotherm model. Freundlich equations (linear and nonlinear) can be expressed as:
Figure 13. Separation Factor (RL) and Surface Coverage (θ) for the adsorption of CV onto TTC (T = 298 K, time =120 min, [CV]0 = 40 - 150 mg/L, pHi = 7, V = 0.005 L, dosage = 2.0 g/L).


where 

indicates both the relative distribution of energy and the heterogeneity of the adsorbent sites. Plot of
versus 


determined from the intercept and the slope respectively. Although, the value of 

Recent reports for the adsorption of crystal violet dye onto different adsorbents found similar trends indicating that the equilibrium data were described well by Langmuir model and did not fit well with Freundlich model [54] .
3.6.3. Temkin isotherms
Equation (14) represented the linear form of Temkin isotherm model which can be used to test the adsorption potential of adsorbent to adsorbate. This equation supposes that increasing the coverage layer of adsorbate onto the surface of adsorbent makes the heat of adsorption (

where, R is common gas constant (0.008314 kJ/mol K), T is the absolute temperature (K), 



culated from the liner plots of 





3.6.4. Dubinin-Radushkevich (D-R) Isotherm Model
The adsorption data of CV dye onto TTC were also tested by Dubinin-Raduskevich (D-R) isotherm model in order to investigate the characteristic porosity and the apparent free energy of adsorption. Isotherm model of D-R (Equation (15)) does not assume constant adsorption potential or homogeneous surface for the adsorbent [13] .

where, 



where R is common gas constant (8.314 J/mol K) and T(K) is the absolute temperature. Figure 12(d), re- presenting the plot of 





The results in Table 2 show that the value of E is equal to 0.475 kJ/mol which is lower than 8 kJ/mol. It indicates that the adsorption of CV dye onto TTC complex is a physical adsorption process. Similar results were obtained by T. Chinniagounder et al. [52] for the adsorption of CV dye onto Cocoa (Theobroma cacao) Shell Activated Carbon (CSAC).
3.7. Standard Gibbs free Energy Change (DG˚)
The degree of spontaneity of the adsorption process is mainly determined from standard Gibbs free energy (DG˚) equation. Increase the negative value of DG˚ represents the increase in the spontaneity and the favorability of the adsorption [55] . Equations (18) and (19) were used to calculate DG˚.


where; T is the temperature (K), R is gas constant (kJ/mol・K) and 



4. Conclusion
The main conclusions of this investigation indicated that Ti-tannate complex (TTC) could be used as an effective adsorbent for the removal of CV dye from aqueous solution. Also, the adsorption of CV dye onto TTC powder was found to depend on the contact time and the initial concentration in addition to the dosage of TTC. Furthermore, the electrostatic interactions between CV and TTC surface controlled by the value of pH and the maximum removal were observed at pH 7. Kinetic study of adsorption process showed that pseudo-second order was the best model to describe the rate of removal of CV dye with the correlation coefficient (


Acknowledgements
Author appreciated the great help and valuable discussion from Mr. Mohamed Hammad and Mr. Mahmoud Baseem, Al-Azhar University, Cairo, Egypt. Also, the author would like to thank Dr. El-Sayed Abd El-Monem Waly, Atomic Energy Authority, Egypt for great help in sample analysis.
Cite this paper
Taha M.Elmorsi, (2015) Synthesis of Nano-Titanium Tannate as an Adsorbent for Crystal Violet Dye, Kinetic and Equilibrium Isotherm Studies. Journal of Environmental Protection,06,1454-1471. doi: 10.4236/jep.2015.612126
References
- 1. Grover V.I. (2006) Water: Global Common and Global Problems. Science Publisher, Enfield.
http://dx.doi.org/10.1201/b11005 - 2. Elmorsi, T.M., Mohamed, Z.H., Shopak, W. and Ismaiel A.M. (2014) Kinetic and Equilibrium Isotherms Studies of Adsorption of Pb(II) from Water onto Natural Adsorbent. Journal of Environmental Protection, 5, 1667-1681.
http://dx.doi.org/10.4236/jep.2014.517157 - 3. Bhattacharyya, K.G. and Sharma, A. (2004) Adsorption of Pb(II) from Aqueous Solution by Azadirachta indica (Neem) Leaf Powder. Journal of Hazardous Materials, B113, 97-109.
http://dx.doi.org/10.1016/j.jhazmat.2004.05.034 - 4. Sharma, P., Kaur, H., Sharma, M. and Sahore, V. (2011) A Review on Applicability of Naturally Available Adsorbents for the Removal of Hazardous Dyes from Aqueous Waste. Environmental Monitoring and Assessment, 183, 151-195.
http://dx.doi.org/10.1007/s10661-011-1914-0 - 5. Attia, A.A., Girgis, B.S. and Fathy, N.A. (2008) Removal of Methylene Blue by Carbons Derived from Peach Stones by H3PO4 Activation: Batch and Column Studies. Dyes and Pigment, 76, 282-289.
http://dx.doi.org/10.1016/j.dyepig.2006.08.039 - 6. Moyo, M. and Chikazaza, L. (2013) Bioremediation of Lead(II) from Polluted Wastewaters Employing Sulphuric Acid Treated Maize Tassel Biomass. American Journal of Analytical Chemistry, 4, 689-695.
http://dx.doi.org/10.4236/ajac.2013.412083 - 7. Fil, B.A., Yilmaz, M.T., Bayar, S. and Elkoca, M.T. (2014) Investigation of Adsorption of the Dyestuff Astrazon Red Violet 3RN (Basic Violet 16) on Montmorillonite Clay. Brazilian Journal of Chemical Engineering, 31, 171-182.
http://dx.doi.org/10.1590/S0104-66322014000100016 - 8. Man, L.W., Kumar, P., Teng, T.T. and Wasewar, K.L. (2012) Design of Experiments for Malachite Green Dye Removal from Wastewater Using Thermolysis-Coagulation-Flocculation. Desalination and Water Treatment, 40, 260-271.
http://dx.doi.org/10.1080/19443994.2012.671257 - 9. van Leeuwen, J., Sridhar, A., Esplugas, M., Onuki, S., Cai, L. and Koziel, J.A. (2009) Ozonation within an Activated Sludge System for Azo Dye Removal by Partial Oxidation and Biodegradation. Ozone: Science & Engineering, 31, 279-286.
http://dx.doi.org/10.1080/01919510902907720 - 10. Aoudj, S., Khelifa, A., Drouiche, N., Hecini, M. and Hamitouche, H. (2010) Electrocoagulation Process Applied to Wastewater Containing Dyes from Textile Industry. Chemical Engineering and Processing: Process Intensification, 49, 1176-1182.
http://dx.doi.org/10.1016/j.cep.2010.08.019 - 11. Rondón, W., Freire, D., de Benzo, Z., Sifontes, A.B., González, Y., Valero, M. and Brito, J.L. (2013) Application of 3A Zeolite Prepared from Venezuelan Kaolin for Removal of Pb(II) from Wastewater and Its Determination by Flame Atomic Absorption Spectrometry. American Journal of Analytical Chemistry, 4, 584-593.
http://dx.doi.org/10.4236/ajac.2013.410069 - 12. Elmorsi, T.M., Riyad, Y.M., Mohamed, Z.H. and Abd El Bary, H.M. (2010) Decolorization of Mordant Red 73 Azo Dye in Water Using H2O2/UV and Photo-Fenton Treatment. Journal of Hazardous Materials, 174, 352-358.
http://dx.doi.org/10.1016/j.jhazmat.2009.09.057 - 13. Kan, T.T., Jiang, X.H., Zhou, L.M., Yang, M., Duan, M., Liu, P.L. and Jiang, X.M. (2011) Removal of Methyl Orange from Aqueous Solutions Using a Bentonite Modified with a New Gemini Surfactant. Applied Clay Science, 54, 184-187.
http://dx.doi.org/10.1016/j.clay.2011.07.009 - 14. Fungaro, D.A. and Magdalena, C.P. (2012) Adsorption of Reactive Red 198 from Aqueous Solution by Organozeolite from Fly Ash: Kinetic and Equilibrium Studies. International Journal of Chemical and Environmental Engineering Systems, 3, 74-83.
- 15. Mittal, A., Mittal, J., Malviya, A., Kaur, D. and Gupta, V.K. (2010) Adsorption of Hazardous Dye Crystal Violet from Wastewater by Waste Materials. Journal of Colloid and Interface Science, 343, 463-473.
- 16. Chakraborty, S., Chowdhury, S. and Saha, P.D. (2011) Adsorption of Crystal Violet from Aqueous Solution onto NaOH-Modified Rice Husk. Carbohydrate Polymers, 86, 1533-1541.
http://dx.doi.org/10.1016/j.carbpol.2011.06.058 - 17. Pei, Y.Y., Wang, M., Tian, D., Xu, X.F. and Yuan, L.J. (2015) Synthesis of Core-Shell SiO2@MgO with Flower like Morphology for Removal of Crystal Violet in Water. Journal of Colloid and Interface Science, 453, 194-201.
http://dx.doi.org/10.1016/j.jcis.2015.05.003 - 18. Kyzas, G.Z. and Kostoglou, M. (2014) Green Adsorbents for Wastewaters: A Critical Review. Materials, 7, 333-364.
http://dx.doi.org/10.3390/ma7010333 - 19. Blackburn, R.S. (2004) Natural Polysaccharides and Their Interactions with Dye Molecules: Applications in Effluent Treatment. Environmental Science and Technology, 38, 4905-4909.
http://dx.doi.org/10.1021/es049972n - 20. Fry, B.A. (1957) Basic Triphenylmethane Dyes and the Inhibition of Glutamine Synthesis by Staphylococcus uureus. Journal of General Microbiology Society, 16, 341-349.
- 21. Ayed, L., Chaieb, K., Cheref, A. and Bakhrouf, A. (2009) Biodegradation of Triphenylmethane Dye MG by Sphingomonas paucimobilis. World Journal of Microbiology and Biotechnology, 25, 705-711.
http://dx.doi.org/10.1007/s11274-008-9941-x - 22. Bertolini, T.C.R., Izidoro, J.C., Magdalena, C.P. and Fungaro, D.A. (2013) Adsorption of Crystal Violet Dye from Aqueous Solution onto Zeolites from Coal Fly and Bottom Ashes. Orbital—The Electronic Journal of Chemistry, 5, 179-191.
- 23. Zhang, R.N., Li, L. and Liu, J.X. (2015) Synthesis and Characterization of Ferric Tannate as a Novel Porous Adsorptive-Catalyst for Nitrogen Removal from Wastewater. Advances—Royal Society of Chemistry, 5, 40785-40791.
- 24. Hashem, A. and El-Khiraigy, K. (2013) Bioadsorption of Pb(II) onto Anethum graveolens from Contaminated Wastewater: Equilibrium and Kinetic Studies. Journal of Environmental Protection, 4, 108-119.
http://dx.doi.org/10.4236/jep.2013.41012 - 25. Miah, M.Y., Volchek, K., Kuang, W. and Tezel, F.H. (2010) Kinetic and Equilibrium Studies of Cesium Adsorption on Ceiling Tiles from Aqueous Solutions. Journal of Hazardous Materials, 183, 712-717.
http://dx.doi.org/10.1016/j.jhazmat.2010.07.084 - 26. Qiu, H., Lv, L., Pan, B.-C., Zhang, Q.-J., Zhang, W.-M. and Zhang, Q.-X. (2009) Critical Review in Adsorption Kinetic Models. Journal of Zhejiang University Science A, 10, 716-724.
http://dx.doi.org/10.1631/jzus.A0820524 - 27. Ho, Y.S. and McKay, G. (1999) Pseudo-Second Order Model for Sorption Processes. Process Biochemistry, 34, 735-742. http://dx.doi.org/10.1016/S0032-9592(98)00112-5
- 28. Hameed, B.H., Din, A.T.M. and Ahmad, A.L. (2009) A Novel Agricultural Waste Adsorbent for the Removal of Cationic Dye from Aqueous Solutions. Journal of Hazardous Materials, 162, 305-311.
http://dx.doi.org/10.1016/j.jhazmat.2008.05.036 - 29. Priyantha, N. and Bandaranayaka, A. (2011) Investigation of Kinetics of Cr(VI)-Fired Brick Clay Interaction. Journal of Hazardous Materials, 188, 193-197.
http://dx.doi.org/10.1016/j.jhazmat.2011.01.090 - 30. Li, W., Zhang, L., Peng, J., Li, N., Zhang, S. and Guo, S. (2008) Tobacco Stems as a Low Cost Adsorbent for the Removal of Pb(II) from Wastewater: Equilibrium and Kinetic Studies. Industrial Crops and Products, 28, 294-302.
http://dx.doi.org/10.1016/j.indcrop.2008.03.007 - 31. Boamah, P.O., Zhang, Q., Hua, M., Huang, Y., Liu, Y., Wang, W. and Liu, Y. (2014) Lead Removal onto Cross-Linked Low Molecular Weight Chitosan Pyruvic Acid Derivatives. Carbohydrate Polymers, 110, 518-527.
http://dx.doi.org/10.1016/j.carbpol.2014.03.034 - 32. Feng, N.C., Guo, X.Y., Liang, S., Zhu, Y.S. and Liu, J.P. (2011) Biosorption of Heavy Metals from Aqueous Solutions by Chemically Modified Orange Peel. Journal of Hazardous Materials, 185, 49-54.
http://dx.doi.org/10.1016/j.jhazmat.2010.08.114 - 33. Singha, B., Naiya, T.K., Bhattacharya, A.K. and Das, S.K. (2011) Cr(VI) Ions Removal from Aqueous Solutions Using Natural Adsorbents—FTIR Studies. Journal of Environmental Protection, 2, 729-735.
http://dx.doi.org/10.4236/jep.2011.26084 - 34. Pahlavanzadeh, H., Keshtkar, A.R., Safdari, J. and Abadi, Z. (2010) Biosorption of Nickel(II) from Aqueous Solution by Brown Algae: Equilibrium, Dynamic and Thermodynamic Studies. Journal of Hazardous Materials, 175, 304-310.
http://dx.doi.org/10.1016/j.jhazmat.2009.10.004 - 35. Wang, Y., Lu, Y.F., Chen, R.Z., Ma, L., Jiang, Y. and Wang, H. (2014) Lead Ions Sorption from Waste Solution Using Aluminum Hydroxide Modified Diatomite. Journal of Environmental Protection, 5, 509-516.
http://dx.doi.org/10.4236/jep.2014.56054 - 36. Pokhrel, D. and Viraraghavan, T. (2008) Arsenic Removal from an Aqueous Solution by Modified A. niger Biomass: Batch Kinetic and Isotherm Studies. Journal of Hazardous Materials, 150, 818-825.
http://dx.doi.org/10.1016/j.jhazmat.2007.05.041 - 37. Pantoja-Castro, M.A. and González-Rodríguez, H. (2011) Study by Infrared Spectroscopy and Thermogravimetric Analysis of Tannins and Tannic Acid. Revista latinoamericana de química, 39, 108-112.
- 38. Senthilkumaar, S., Varadarajan, P.R., Porkodi, K. and Subbhuraam, C.V. (2005) Adsorption of Methylene Blue onto Jute Fiber Carbon: Kinetics and Equilibrium Studies. Journal of Colloid and Interface Science, 284, 78-82.
http://dx.doi.org/10.1016/j.jcis.2004.09.027 - 39. Senthilkumaar, S., Kalaamani, P. and Subburaam, C.V. (2006) Liquid Phase Adsorption of Crystal Violet onto Activated Carbons Derived from Male Flowers of Coconut Tree. Journal of Hazardous Materials B, 136, 800-808.
http://dx.doi.org/10.1016/j.jhazmat.2006.01.045 - 40. Heidari, H.Y., Mehraban, Z. and Heikkinen, H. (2013) Selective Adsorption of Pb(II), Cd(II), and Ni(II) Ions from Aqueous Solution Using Chitosan-MAA Nanoparticles. International Journal of Biological Macromolecules, 61, 251-263.
http://dx.doi.org/10.1016/j.ijbiomac.2013.06.032 - 41. Giraldo, L. and Moreno-Piraján, J.C. (2008) Pb2+ Adsorption from Aqueous Solutions on Activated Carbons Obtained from Lignocellulosic Residues. Brazilian Journal of Chemical Engineering, 25, 143-151.
- 42. Shouman, M.A., Khedr, S.A. and Attia, A.A. (2012) Basic Dye Adsorption on Low Cost Biopolymer: Kinetic and Equilibrium Studies. IOSR Journal of Applied Chemistry (IOSR-JAC), 2, 27-36.
- 43. George, Z.K. and Nikolaos, K.L. (2009) Reactive and Basic Dyes Removal by Sorption onto Chitosan Derivates. Journal of Colloid and Interface Science, 331, 32-39.
http://dx.doi.org/10.1016/j.jcis.2008.11.003 - 44. Elmorsi, T.M. (2011) Equilibrium Isotherms and Kinetic Studies of Removal of Methylene Blue Dye by Adsorption onto Miswak Leaves as a Natural Adsorbent. Journal of Environmental Protection, 2, 817-827.
http://dx.doi.org/10.4236/jep.2011.26093 - 45. Sahu, M.K., Mandal, S., Yadav, L.S., Dash, S.S. and Patel, R.K. (2015) Equilibrium and Kinetic Studies of Cd(II) Ion Adsorption from Aqueous Solution by Activated Red Mud. Desalination and Water Treatment, June 26, 1-15.
http://dx.doi.org/10.1080/19443994.2015.1062428 - 46. Manzoor, Q., Nadeem, R., Iqbal, M., Saeed, R. and Ansari, T.M. (2013) Organic Acids Pretreatment Effect on Rosa bourbonia Phyto-Biomass for Removal of Pb(II) and Cu(II) from Aqueous Media. Bioresource Technology, 132, 446-452.
http://dx.doi.org/10.1016/j.biortech.2013.01.156 - 47. Ho, Y.-S. (2006) Review of Second-Order Models for Adsorption Systems. Journal of Hazardous Materials B, 136, 681-689.
- 48. Patil, S., Deshmukh, V., Renukdas, S. and Patel, N. (2011) Kinetics of Adsorption of Crystal Violet from Aqueous Solutions Using Different Natural Materials. International Journal of Environmental Sciences, 1, 1116-1134.
- 49. Gandhimathi, R., Ramesh, S.T., Sindhu, V. and Nidheesh, P.V. (2012) Single and Tertiary System Dye Removal from Aqueous Solution Using Bottom Ash: Kinetic and Isotherm Studies. Iranica Journal of Energy & Environment, 3, 52-62.
http://dx.doi.org/10.5829/idosi.ijee.2012.03.01.0113 - 50. Lim, L.B.L., Priyantha, N., Zehra, T., Then, C.W. and Chan, C.M. (2015) Adsorption of Crystal Violet Dye from Aqueous Solution onto Chemically Treated Artocarpus odoratissimus Skin: Equilibrium, Thermodynamics, and Kinetics Studies. Desalination and Water Treatment, 53, 1-15.
- 51. Neupane, S., Ramesh, S.T., Gandhimathi, R. and Nidheesh, P.V. (2015) Pineapple Leaf (Ananas comosus) Powder as a Biosorbent for the Removal of Crystal Violet from Aqueous Solution. Desalination and Water Treatment, 54, 2041-2054.
http://dx.doi.org/10.1080/19443994.2014.903867 - 52. Chinniagounder, T., Shanker, M. and Nageswaran, S. (2011) Adsorptive Removal of Crystal Violet Dye Using Agricultural Waste Cocoa (Theobroma cacao) Shell. Research Journal of Chemical Sciences, 1, 38-45.
- 53. Malarvizhi, R. and Ho, Y.-S. (2010) The Influence of pH and the Structure of the Dye Molecules on Adsorption Isotherm Modeling Using Activated Carbon. Desalination, 264, 97-101.
http://dx.doi.org/10.1016/j.desal.2010.07.010 - 54. Depci, T., Kul, A.R., Onal, Y., Isli, E.D., Alkan, S. and Turkmenoglu, Z.F. (2012) Adsorption of Crystal Violet from Aqueous Solution on Activated Carbon Derived from Golbasi Lignite. Physicochemical Problems of Mineral Processing, 48, 253-270.
- 55. Jirekar, D.B., Pramila, G. and Farooqui, M. (2014) Kinetics and Isotherm Studies on Crystal Violet Dye Adsorption onto Black Gram Seed Husk. International Journal of ChemTech Research, 15, 427-434.
















