Food and Nutrition Sciences
Vol.4 No.8(2013), Article ID:35196,11 pages DOI:10.4236/fns.2013.48108
Functional Foods: Can Food Technology Help in the Prevention and Treatment of Diabetes?
![]()
Institute of Food Science, National Research Council, Avellino, Italy.
Email: *rgiacco@isa.cnr.it
Copyright © 2013 Rosalba Giacco et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received February 20th, 2013; revised March 20th, 2013; accepted March 27th, 2013
Keywords: Diabetes; Functional Food; Glycemic Index; Whole Grains; Food Technologies
ABSTRACT
Recently, the interest in functional foods has increased dramatically thanks to some scientific evidences demonstrating that, regardless of their nutritional value, some foods components beneficially influence several functions of the body, making them “functional” for the prevention and treatment of degenerative diseases, such as type 2 diabetes and cardiovascular diseases. In particular, the marketplace offers conventional and modified functional foods which acting on intestine, glucose and lipid metabolism are potentially useful for diabetic patients. The field of functional foods, however, presents some criticalities. Up to now, only few functional foods have obtained the statement of a “Health Claim”, either because there are not sufficient scientific evidences based on human intervention studies, that justify a “Health Claim”, or because data are not consistent with the proposed claims. In addition, some functional foods show a low palatability and a lack of variety. Both these issues limit their ordinary intake. This critical aspect also involves the lack of a possible synergistic effect deriving from the contemporary consumption in the diet of different functional foods, as observed with some healthy dietary pattern. Functional foods represent, then, a challenge for the food industry, which needs to implement and/or develop innovative technologies able to create new foods with improved healthy properties, but also with better organoleptic properties suitable for a daily consumption.
1. Introduction
The interest in functional foods has increased dramatically in the last two decades, especially in North America, thanks to their capacity in beneficially influencing metabolic parameters and chronic degenerative diseases such as type 2 diabetes (T2DM), cardiovascular disease (CVD) and some types of cancer [1].
The results of several epidemiological and clinical studies indicate that the consumption of soft drinks, red meat, sausages, saturated and trans fats is closely associated with an increased risk of developing T2DM [2,3]. By contrast, the use of whole grains, legumes, fruit, vegetables, nuts, tea and coffee decreases that risk [4-6] (Table 1). It is also known that the combination of these types of food in a diet model such as the “Mediterranean diet”, or so-called “Prudent” diet, is even more effective in preventing T2DM than the consumption of the single food [1]. In fact, the food synergy of the Mediterranean diet maximizes the effect of all the mechanisms involved in the prevention of type 2 diabetes such as weight loss, improved insulin sensitivity, reduction of oxidative stress and of subclinical inflammation. We can therefore say that the consumption of those foods, associated with a lower risk of developing type 2 diabetes, allows not only the intake of nutrients essential for life, but also of other components (e.g., vegetable fiber, oligosaccharides, phytosterols, oligo-mineral elements, vitamins and various molecules with antioxidant activity) capable of positively influencing several functions of the body, regardless of their nutritional value, making them “functional” for the prevention and treatment of T2DM.
Over the past two decades, due to evidences of this type, the landscape of the food and nutrition field has changed constantly. Therefore, food is no longer solely viewed in terms of macro or micronutrients, but it is now a real way to achieve the so-called state of “wellness”. The possibility of other potential health promoting components found in foods has increased the interest in functional foods due not only to the results of scientific researches, which link the diet to the development of
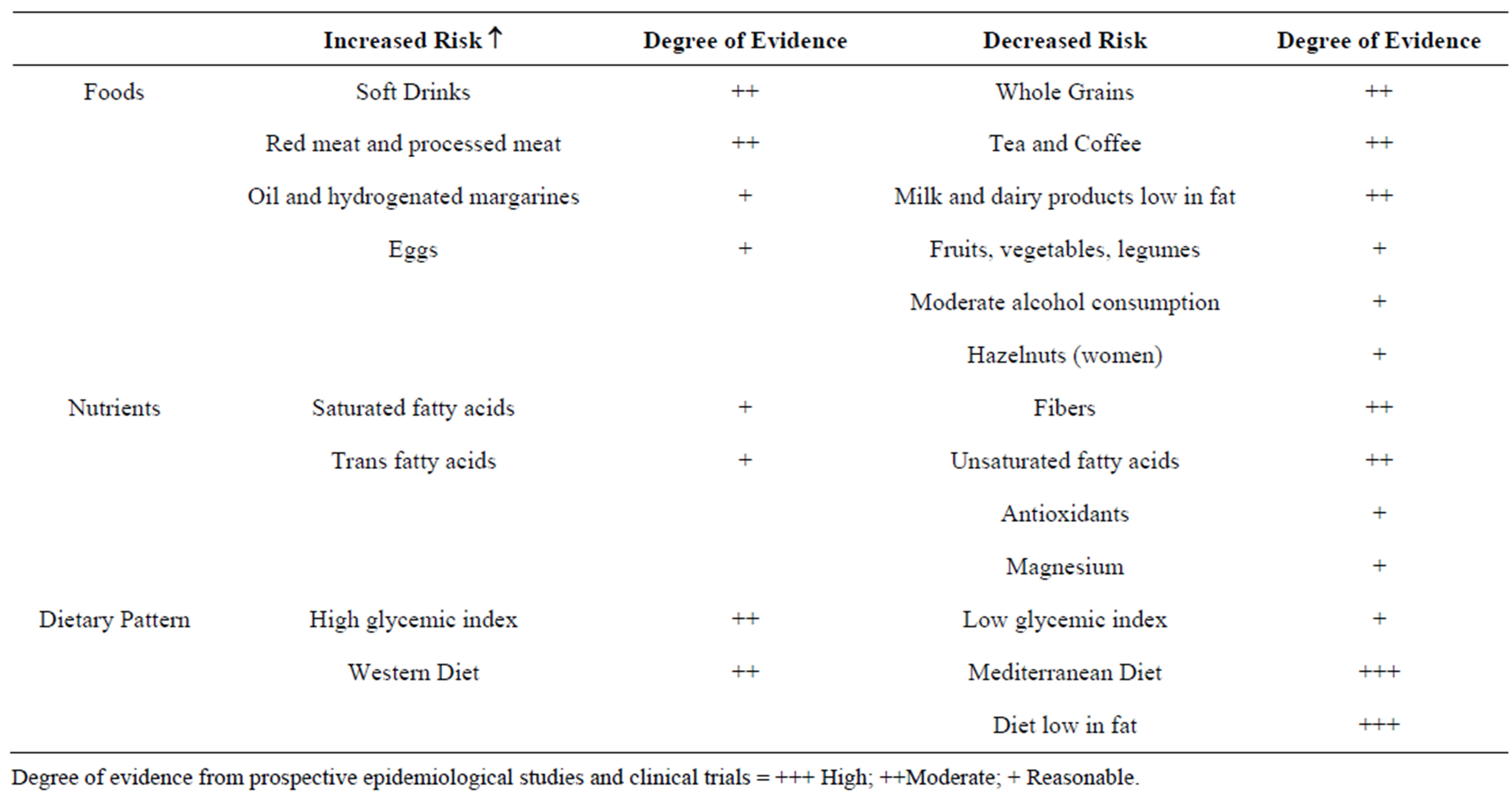
Table 1. Foods, nutrients and dietary patterns associated with risk of developing type 2 diabetes.
chronic diseases, but also to other factors, such as a greater attention of consumers to their health, the increase of health costs, the high competitiveness of the food marketplace, the new legislation governing food and the technology advancements such as biotechnology.
Although several functional foods are available on the marketplace, only few have a “health claim” due to scant scientific evidence based on human intervention studies [7]. Functional foods represent a challenge for the food industry, which needs to implement and/or develop innovative technologies able to create new foods with improved healthy properties, but also with better organoleptic properties suitable for an everyday consumption.
2. Definition of “Functional Food”
In the United States the functional foods are not officially recognized as a regulatory category by the American Food and Drug Administration (FDA) [8]. Thus, the term functional food is currently a marketing rather than a regulatory idiom.
Therefore there is not a universal and unambiguous definition of “functional food”, but several working definitions have been put forward by various organizations, such as the American Dietetic Association (ADA) [9], the International Food Information Council (IFIC) [10] or the Institute of Food Technologists [11]. All these definitions, beyond small differences, specify, however, that a food to be considered “functional”, in addition to fulfil nutritional needs, must have further properties beneficial to the body.
The European Commission Concerted Action on Functional Food Science in Europe (FUFOSE) regards a food as functional if it is satisfactorily demonstrated to affect beneficially one or more target functions of the body, in a way that it is relevant to an improved state of health and well-being and/or a reduction of disease risk, beyond adequate nutritional effects [12]. In this context, functional foods must demonstrate their effects with an ordinary intake normally expected to be consumed in a diet.
This latter feature distinguishes the functional food from the nutraceutical food, with which it is often confused. Nutraceuticals exhibit, by definition, their effects in doses much higher than those usually assumed in a diet, i.e. pharmacological doses and not necessarily contained in the food [13].
As a matter of fact, having also become very popular in recent years, unlike functional foods, nutraceuticals are pharmaceutical preparations (tablets, capsules, pills, etc.) containing the physiologically active components naturally present in foods, but extracted, purified, concentrated, and, therefore, undetectable.
3. Functional Food Categories
There are different types of “functional foods”, but basically they can be divided into conventional and modified foods. Examples of conventional foods are those in which the components with positive effects are already naturally present, such as whole grains, nuts, soybean, tomatoes etc. Modified foods are those that have been transformed by technology to obtain a functional food, by enriching or adding one or more components with beneficial effects, by removing one or more components with adverse effects from the food (e.g., allergenic proteins, lactose-free milk), by replacing a component with another that has beneficial effects (e.g., fat replaced by HP inulin) or by improving the bioavailability of some molecules, which show potential health benefits.
Whole grains represent a particularly interesting example of conventional functional food. The most consumed cereal in the world is certainly wheat, which is the main energy source in human nutrition. Wheat, as other cereals intended for human consumption, can be used either as whole or refined.
In particular, consumers of whole grains have shown a lower risk of developing type 2 diabetes than those consuming refined grains [4]. In order to explain the mechanisms that support this observation, it is necessary to fully understand what happens during the refining process of the caryopsis, the structure of which is formed by the following three different layers:
1) the external part, more commonly known as bran, is rich in fibres, group B vitamins, phytonutrients (flavonoids, indoles), antioxidants, minerals and contains also a small amount of proteins;
2) the central stratum, the most important from a nutritional point of view, is the endosperm, which is rich in starch and proteins but poor in micronutrients;
3) the inner part is represented by the embryo or germ, and contains a high amount of lipids and proteins, minerals (Fe, Zn), vitamins B group, phytosterols, antioxidants and vitamin E [14].
During the refining process, the cereal is crushed and deprived of the bran and the germ to produce the so-called “refined grain flour.” When the bran and germ components are not removed, the flour obtained is defined as “whole grain flour.” Refining process depletes the cereal by many nutrients and biologically active components such as fibres, oligosaccharides, vitamins, minerals, phytosterols, phytoestrogens, and polyphenols. In fact, refined grain flours contain about 78% less total fiber and over 50% less minerals and vitamins. As regards the polyphenols, refined grain flour contains 91% less ferulic acid, 51% less betaine and 78% less alkilresorcinol [15]. There are many experimental evidence showing that the components removed from the refining process are able to regulate different important functions of the body [14,15].
It is well known, in fact, that the vegetable fiber modulates some digestive enzymes, reduces the absorption of fats and slows down the absorption of carbohydrates, with beneficial effects both on the lipid and glucose profile. It also improves gut peristalsis and stimulates the selective growth of colonic bacterial flora, crucial for the production of short-chain fatty acids (SCFAs): acetate, propionate and butyrate. These molecules, by acting on different organs with multiple mechanisms, are capable of preserving the colonic mucosa, counteracting the subclinical inflammation and improving insulin sensitivity. In addition, SCFAs, produced by bacterial flora, influence body fat stores by sending satiety signals to hypothalamic nuclei, which regulate the sense of hunger and satiety, inhibiting the food intake. This mechanism contributes to the prevention of overweight/obesity, an important risk factor for the T2DM and CVD [16].
The polyphenols, flavonoids, vitamins and some minerals, such as selenium and zinc, possess remarkable antioxidant, chemo-preventive and also hormonal regulation properties, as in the case of phytoestrogens [15].
Therefore, the lowest risk of developing T2DM, observed in consumers of whole grains compared to those consuming refined grains, depends on the ability of the fibers, polyphenols, phytoestrogens, sterols and oligomineral elements to improve the insulin resistance, which represents the pathogenic factor of T2DM. Probably all these components act with different mechanisms giving rise to a synergistic effect.
4. Food Technology Applied to Cereals: Advantages and Disadvantages
Scientific evidence suggests that the refining of wheat is an example of food technology unfavourable to human health [17,18]. Therefore, the researchers’ interest, both in the medical field and in the field of food technology, has been stimulated to investigate the mechanisms of action by which the consumption of whole grains produces beneficial effects and to develop innovative techniques, able to preserve, as much as possible, the functional components of whole grain products. This concern has led to the implementation of a European project, HealthGrain, the purpose of which was to improve the nutritional value and health benefits of whole grains and to optimize their use in the diet. For this purpose, the organoleptic qualities of cereal-based foods have been studied and new technologies have been developed in order to obtain the production of cereal foods, that still contain the healthy components [19].
Within this project, for example, a less aggressive method for the peeling of the wheat caryopsis has been developed. This new peeling removes only the outermost layer of the pericarp and so the whole wheat flour obtained has higher contents of folic acid, choline, tocols and alkilresorcinol than the traditional flour. This is an emblematic example of how technology is able to preserve the functional components naturally present in cereals [20].
5. Functional Foods and Their Targets
Functional foods may act on different targets; the largest segment of the marketplace for functional products in Europe, Japan and Australia is certainly represented by those which better the gut environment, but many are also those that act by improving glucose and lipid metabolism.
5.1. Intestine
The interest for this type of functional foods in relation to metabolic diseases is growing because results of studies have shown that gut microbiota may influence metabolism and body composition [21]. In particular, gut microbiota seems to be involved in the genesis of excessive adipose tissue development, chronic inflammation and insulin resistance [22,23].
This first category of functional foods includes prebiotics, probiotics and synbiotics. “Prebiotic” is a component or a food ingredient which is fermented by the gut bacterial flora and is able to induce the selective growth of one or more bacterial strains, with positive effects on human health. “Probiotic” is a living and vital organism which, if ingested in sufficient quantities, has beneficial effects on human health. “Synbiotic” consists, instead, of a blend of prebiotics and probiotics, which improves the survival and engraftment of living microorganisms ingested in the gastrointestinal tract [24].
Prebiotics used in the food industry are polymers of fructose with glucosidic β (2→1) bonds that cannot be hydrolyzed by human digestive enzymes and, therefore, reach the colon undigested, where they are fermented by intestinal flora to short chain fatty acids. Prebiotics according to their polymerization degree are classified into:
Fructo-oligosaccharides (FOS), are a class of short chain polymers of fructose—consisting of one molecule of glucose and at least 2 of fructose (GFn), up to a maximum of 4 molecules (Degree polymerization, DP-4)— and are synthesized from sucrose;
Oligofructose (OFS), is a polymer of fructose (Fn) containing a maximum of 10 units and is produced by the partial hydrolysis of inulin;
Inulin is a polymer with a long chain (DP ≥ 10) which is obtained from various vegetal sources by aqueous extraction and is not subjected to further processes (mixture of GFn and Fn);
HP inulin, is a long chain polymer, with a high molecular weight, from which the polymers of fructose with DP < 10 units have been removed (mixture of GFn and Fn) [25].
FOS, OFS and inulin are water highly soluble, nonviscous, odourless and have approximately 30% to 50% of the sweetness of sucrose molecules. Thanks to these characteristics they can partially replace sucrose in food industry, particularly in baked products. HP inulin is characterized, on the other hand, by a low water solubility for which, when mixed with water or milk, it forms micro-crystals that impart a creamy appearance to the mixture; it can, therefore, substitute fat in the food industry for the production of low fat spreads.
There is a substantial scientific evidence about the fact that a daily consumption of 10 - 20 g of prebiotics induces selective growth of Lactobacilli and Bifidobacteria, increases stool volume, raises the number of evacuations, stimulates the production of SCFAs and immunoglobulin synthesis [26]. Thus, the habitual consumption of prebiotics regularizes the bowel, increases gut resistance to infection by pathogenic bacteria, helps to prevent and treat gut inflammatory diseases and reduces the risk of developing colorectal cancer [27-30]. Further prebiotics consumption benefits are the increase in absorption of some minerals (Ca, Mg, Zn, Fe) and the enhancement of vitamin B synthesis [31]. The benefits of regular consumption of prebiotics on insulin sensitivity are less clear. The experimental evidences in animal models suggest that the beneficial effect of prebiotics on insulin sensitivity and risk of type 2 diabetes is largely mediated by a reduced weight gain [32]. In fact, prebiotics are fermented in the colon by intestinal bacteria with the production of acetic, propionic and butyric acids and may influence body weight regulation by at least three processes: 1) by decreasing gastric emptying rate by SCFA, as demonstrated in animals and humans [33], thus increasing the satiating effect of the meal; 2) by using the ability of propionate and acetate to reduce non esterified fatty acids (NEFA) plasma levels, responsible for peripheral and hepatic insulin resistance [34,35]; and finally, 3) by exploiting the ability of propionate to modulate glucose metabolism by increasing hepatic glycolysis and decreasing hepatic glucose production, as shown in isolated rat hepatocytes [36], as well as by stimulating Glucagon Like Peptide-1 (GLP-1) secretion [37].
At present, the consumption of functional foods enriched and/or added with prebiotics in the context of a healthy diet can, however, be recommended both to general population and to patients with diabetes, on the basis of the beneficial effects on the gut, consistently demonstrated in clinical trials and on the potential benefits in improving insulin resistance [38].
Probiotic is a living and vital organism, usually belonging to the group of lactic acid bacteria and bifidobacteria. The strains selection is based on the following characteristics: human intestinal environment; bile, acids enzymes and oxygen stability; ability of adhesion to the gut mucosa; aptitude to colonize the human gastrointestinal tract; production of antimicrobial substances and, finally, demonstrated efficacy and safety. Recently, the ability to ferment oligosaccharides, especially FOS (prebiotic), has been considered as an additional criterion of selection. This ability may confer a competitive advantage against the gut microbial population.
Probiotics are mainly added to yoghurt, fermented milks and cheeses, but other fortified foods, such as ice creams, spreads and meat products, are under investigation. A crucial aspect of functional foods based on probiotics, in the light of the labelling legislation and the consumer information, concerns their capability to survive the technological processes and, in particular, the preparation and storage steps. Another important feature regards the minimum number of probiotic cells that are useful for the food production. Effectively, the survival to the technological processes has been only sporadically considered, even if such a characteristic constitutes the limiting factor for the production of some commercial products. As for as cheese and fermented milks are concerned, it is necessary to establish and ensure proper shelf life, which is a suitable interval within which the declared functional properties do not decrease below a predefined level of acceptability.
In this respect, there is no comprehensive information regarding the minimum number of probiotic cells necessary to ingest to get a significant effect on the different categories of consumers. However, despite the great variability, both in the literature and in practice, this number refers to “recommended” values [39].
Therefore, the technological problems related to these functional foods are focused on the preservation of cell viability during their production and their storage. Among the technologies that were developed to prolong the cells life, a good example is the microencapsulation in protective matrices [40,41].
Combining prebiotics and probiotics in one type of food a “synbiotic food” is obtained, the expected benefit of which is to improve both the survival of bacterial strains in the upper gastrointestinal tract and the adhesion of such microorganisms in the colon [42-44].
The quality of scientific data behind the claims attributed to the pro and prebiotics is variable and probably lower than expected, largely because of a lack of studies and a poor definition of the microorganisms and ingredients used in the clinical and biological trials.
In light of recent information, suggesting that the gut bacterial flora may be involved in the development of obesity and metabolic syndrome, the marketplace of functional products aimed at improving the gut environment is intended, however, to grow. In fact, some studies have shown that these diseases are associated to profound microbioma alterations. The induction of metabolic syndrome by faecal transplants reinforces the importance of gut flora in the pathogenesis of this disease. However, much of the knowledge in this area is based on studies in animals, therefore, their biological significance requires further studies to be performed in humans [32, 45].
5.2. Glucose Metabolism
Observational studies have shown that the glycemic index (GI), an indicator of the carbohydrates ability to raise blood sugar, is directly associated with the risk of developing T2DM [46,47] and coronary artery disease [48]. Therefore, low-GI foods can be considered as a class of functional foods with protective effects against both the T2DM and the coronary heart disease.
Several factors can contribute to reducing the GI of foods rich in carbohydrates, first of all the content of viscous fiber and prebiotics (inulin, FOS etc.), the type of starch (ratio of amylose/amylopectin, Resistant Starch), the physical structure of the food and the type of leavening [49].
The oat is, among the cereals, the richer in β-glucan, which is a highly viscous soluble fiber, capable of reducing the influence of the food on postprandial glucose with beneficial effects on glucose metabolism [50]. This effect is related to some characteristics of soluble fibers and, among them, the viscosity surely plays a significant role. In fact, by slowing the gastric emptying, the digestion and the absorption of nutrients in the intestine, it limits the increase of postprandial glucose levels. The enrichment of breakfast cereals with fractions of oat fiber, obtained by an extrusion process, has been shown to reduce the glycemic response from 33% to 62% in type 2 diabetic patients [51].
The addition of β-glucan or other viscous fibers in food products has, therefore, become a common practice in the food industry, as it can broaden the range of low GI foods on the marketplace. Foods particularly rich in fiber, however, are not very palatable and, moreover, more viscous is the fiber less palatable the food is. Therefore, the addition of viscous fiber cannot exceed a certain threshold.
Another way to reduce the food GI, without interfering with the palatability, is the partial replacement of glucose with fructose, which has a GI of 29% compared to glucose, the GI of which is 84%. However, even such a replacement should be done in moderation, because the fructose in high doses has detrimental effects on lipid metabolism, since it increases both the levels of triglycerides and low density lipoprotein cholesterol (LDLc) [52]. The simultaneous approaches described above, that is the increase of β-glucan amount and the substitution of sucrose with fructose, allow to reduce the GI value of a food product of about 40%, which is a clinically significant effect [53].
This example shows how, acting on the amount of one or more ingredients, it is possible to produce a functional food with beneficial properties on glucose and insulin metabolism and with a palatability accepted by the consumer in a daily diet.
Another strategy to reduce the foods GI is to use technologies capable of modifying the starch digestibility, such as that employed to produce the parboiled rice (Table 2). This technology involves a treatment of the rice grains with steam at high pressure. This process is able to lead to the formation of an outer layer of retrograded starch, also known as resistant starch, that is capable of resisting to the hydrolysis by amylases of the human gut and of not increasing the blood sugar level [54]. Therefore, the parboiled rice has a lower GI compared to superfine rice [55]. In the case of pasta, instead, the GI is about 35% - 40% lower than that of white bread, because of the extrusion process which is applied to the mixtures. The extrusion, in fact, is able to structure the proteins of the dough (gliadin) in a network, which is insoluble in water. In the mesh of this network the starch is incorporated making it less accessible to the hydrolysis by the gut amylases. This fact slows down the digestion and lowers the GI (Table 2) [56].
The importance of the physical structure of a food in determining GI is largely demonstrated by one of our studies, the aim of which was the evaluation of the GI of the potato dumplings with respect to that of bread and other leavened foods made by wheat flour, such as pizza and toast bread [57]. Potato dumplings give rise to a glycemic response of 30% lower than that of white bread, toasted bread and pizza, at the same carbohydrate content, fiber, resistant starch and viscosity. The lower potato dumplings GI is attributable to the different physical structure of that type of food. In fact, at electron microscopy white bread and other leavened foods present a structure rich in vacuoles, in which the starch granules are surrounded by gas bubbles and, therefore, easily accessible to amylases. On the contrary, potato dumplings structure appears compact, with the starch granules closely linked to the proteins and, then, less reachable by amylolytic enzymes (Figure 1) [57].
The production of bread by acid leavening (realized with starter such as Lactobacillus plantarum P1, L. and L.

Figure 1. Electron microscopy: Physical structure of four different glycemic index foods [57].

Table 2. Examples of technological processes able to modify the starch digestibility.
plantarum DC400 brevis P2 rather than Saccharomyces cerevisiae) is another process used for the GI reduction. In this case, the fermentation of the lactic acid bacteria produces organic acids (lactic and acetic acids) that lowers the pH of dough and increases the formation of resistant starch, with a consequent decrease of the GI [58]. If the acid leavening fermentation is associated with the use of a flour enriched with β-glucan, it is possible to obtain a bread with a GI further reduced, compared to that of a conventional leavening bread, prepared without the addition of viscose fiber. In fact, the addition of β-glucan contributes, together with the viscosity, to further slowdown the starch hydrolysis [58]. In addition, both the rheological characteristics of this type of bread, such as volume, mass and structure and the sensory properties, are almost all enhanced against those of conventional bread [58]. Since in most European countries bread is the main source of carbohydrates, the inclusion of bread with a lower GI in a daily diet presents undoubted metabolic advantages in a population strategy aimed at the T2DM and CVD prevention.
This type of processes which do not add, not remove nor replace a component with another with functional properties do not lead to the production of functional foods in sensu strictu.
However, in terms of health impact, these processes, by reducing the GI of foods, exert substantial benefits in both the prevention and therapy of T2DM.
For what concerns the soluble fiber used to produce functional foods it is to underline that, despite several scientific indications support the viscous fibers ability of reducing foods GI and, then, the risk of developing CVD, the FDA has recognized a “Health Claim” to the fiber of oats and psyllium not for the benefits on blood glucose, but only for their demonstrated capability of reducing cholesterol plasma levels [59]. Therefore, it is crucial to produce further scientific data to support the capability of viscous fibers of improving not only the lipid metabolism, but also the glucose and insulin metabolism. On the other hand, researchers should co-operate with industry, in order to make it to invest in new biotechnology for the production of different types of low GI functional foods, with better acceptability by the consumers.
5.3. Lipid Metabolism
It is widely known that in diabetic patient the risk for CVD is 2 - 4 times higher than the general population [60]. This higher risk is not justified only by high plasma glucose levels but also by association with high plasma cholesterol concentrations. Therefore, the management of diabetes needs to control either blood glucose and lipid metabolism. In clinical practice, pharmacological intervention with statins has been demonstrated to be successful in reducing blood cholesterol and cardiovascular mortality. But we must consider that treatment with statins have also some disadvantages: 1) statins have a cost for the National Health Service and are provided free of charge only to those subjects presenting a CVD high risk, 2) they are drugs and so, in addition to having beneficial effects, also cause drawback side effects that contraindicate their use in some individuals, and 3) treatment with statins could be insufficient alone to control cholesterol. However, several components of foods are able to reduce plasma cholesterol levels. The National Cholesterol Education Program (NCEP) Expert Panel for the treatment of hypercholesterolemia advices not only to limit the diet intake of saturated fats and cholesterol, but also to increase the consumption of viscous fiber and phytosterols, because of their lipid-lowering action [61]. Besides viscous fiber (contained in oats, barley and psyllium) and phytosterols, also soy proteins and nuts have a proven cholesterol-lowering effect [62]. The literature reports that each of these components reduces the levels of LDLc from 2% to 20% but, in clinical practice, it is likely that the magnitude of the effect can be lower, equal to 3% - 7%. However, the simultaneous presence of all these components in the same diet, thanks to a synergistic action, can make the cholesterol lowering effect much greater and clinically relevant. A reduction in LDLc by about 22.5% has been estimated to be achieved if, in the same diet, the consumption of viscous fiber, plant sterols (fortified margarine), soy proteins (e.g. soy milk, tofu etc.) and nuts is combined [62].
The simultaneous use of these functional components is particularly interesting, because in this case the obtained cholesterol lowering effect is additive, since each component acts by a different mechanism. The viscous fiber increases the loss of bile, the phytosterols reduce the absorption of dietary cholesterol, soy proteins tend to inhibit the synthesis of cholesterol and Apo B and nuts act by a combination of these mechanisms. The effectiveness of this type of diet on the reduction of LDLc is equivalent to that obtained by associating a treatment with 20 mg/day of lovastatin to a low fat diet (recommended by the NCEPAdult Treatment Panel III) [63]. It should be noted that the cholesterol lowering effect of this type of diet based on functional foods also shows the advantage of reducing the C-reactive protein (CRP) plasma concentrations, another cardiovascular risk factor [63].
One of the limitations of some functional foods is, however, the low palatability, that, associated with the lack of variety, prevents optimal compliance to diet. The side effects are, instead, scarce. The larger volume of the products rich in fiber can be a problem, but it can also offer the big advantage of limiting the energy intake in overweight/obese. Furthermore, in some cases, flatulence induced by the ingestion of these foods limits their consumption making them less attractive.
In any case, based on this evidence, the FDA has extended the Health Claim of cardiovascular protection conferred to the viscous fibers also to phytosterols, soy and nuts [64,65]. However, other authorities, who have expertise in this area, have not yet expressed an explicit opinion about the use of functional foods or about food components with cholesterol-lowering properties.
6. Conclusions
Functional foods, consumed in a context of a mixed diet and a healthy lifestyle, can represent an additional opportunity for T2DM and CVD prevention for general population, while for those with clinical disease, this type of foods can help in controlling the metabolic alterations thereto associated. However, before recommending the use of functional foods, a nutritional assessment, which demonstrates the health benefit or that the risk reduction of diseases resulting from their use is clinically relevant, well tolerated and free of side effects, is required. In fact, among all the functional foods available on the marketplace, to date only a few of them have obtained the statement of a “Health Claim”. This is due to the fact that, in most cases, there is not any scientific evidence based on human intervention studies that justify a “Health Claim”. In other cases, instead, the available data are not consistent with the proposed claims.
In order to encourage the competent authorities to certify the Health Claim of a food, it is of primary importance to make “ad hoc”, controlled and randomized clinical trials, which unequivocally demonstrate the effectiveness of a particular food component in improving of a biological function or in the prevention of any disease.
On the other hand, functional foods are also a challenge for the food industry, which is called upon to implement and/or develop new technologies useful for the creation of new foods, which, besides enhanced health properties, also have better organoleptic properties suitable to be consumed in a daily diet.
Finally, consumer education to functional foods consumption should be supported by adequate information, able to dispel prejudices about processed food, usually negatively perceived by the consumer when compared to the natural ones.
REFERENCES
- J. Salas-Salvadó, M. Á. Martinez-González, M. Bulló and E. Ros, “The Role of Diet in the Prevention of Type 2 Diabetes,” Nutrition, Metabolism and Cardiovascular Diseases, Vol. 21, No. 2, 2011, pp. B32-B48. doi:10.1016/j.numecd.2011.03.009
- A. K. Thompson, A. M. Minihane and C. M. Williams, “Trans Fatty Acids, Insulin Resistance and Diabetes,” European Journal of Clinical Nutrition, Vol. 65, No. 5, 2011, pp. 553-564. doi:10.1038/ejcn.2010.240
- S. M. Hofmann and M. H. Tschöp, “Dietary Sugars: A Fat Difference,” Journal of Clinical Investigation, Vol. 119, No. 5, 2009, pp. 1089-1092. doi:10.1172/JCI39332
- M. G. Priebe, J. J. van Binsbergen, R. de Vos and R. J. Vonk, “Whole Grain Foods for the Prevention of Type 2 Diabetes Mellitus,” John Wiley & Sons, Ltd., Hoboken, 2008, doi:10.1002/14651858.CD006061
- K. A. Harris and P. M. Kris-Etherton, “Effects of Whole Grains on Coronary Heart Disease Risk,” Current Atherosclerosis Reports, Vol. 12, No. 6, 2010, pp. 368-376. doi:10.1007/s11883-010-0136-1
- P. Carter, L. J. Gray, J. Troughton, K. Khunti and M. J. Davies, “Fruit and Vegetable Intake and Incidence of Type 2 Diabetes Mellitus: Systematic Review and Meta-Analysis,” BMJ, Vol. 341, 2010, pp. c4229-c4236. doi:10.1136/bmj.c4229
- M. B. Roberfroid, “Concepts and Strategy of Functional Food Science: The European Perspective,” American Journal of Clinical Nutrition, Vol. 71, No. 6, 2000, pp. 1660S-1664S.
- S. Ross, “Functional Foods: The Food and Drug Administration Perspective,” American Journal of Clinical Nutrition, Vol. 71, No. 6, 2000, pp. 1735S-1738S.
- American Dietetic Association, “Position of the American Dietetic Association: Functional Foods,” Journal of the American Dietetic Association, Vol. 109, No. 4, 2009, pp. 735-746. doi:10.1016/j.jada.2009.02.023
- International Food Information Council, “Functional Foods: Attitudinal Research,” 2009. http://www.ific.org/research/funcfoodsres02.cfm
- Institute of Food Technologists, “Functional Foods: Opportunities and Challenges,” 2009. http://members.ift.org/IFT/Research/IFTExpertReports/functionalfoodsreport.htm
- A. T. Diplock, P. J. Aggett, M. Ashwell, F. Bornet, E. B. Fern and M. B. Roberfroid, “Scientific Concepts of Functional Foods in Europe. Consensus Document,” British Journal of Nutrition, Vol. 81, No. 1, 1999, pp. S1-S27.
- E. K. Kalra, “Nutraceutical-Definition and Introduction,” AAPS PharmSci, Vol. 5, No. 3, 2003, pp. 27-28. doi:10.1208/ps050325
- A. Fardet, “New Hypotheses for the Health-Protective Mechanisms of Whole-Grain Cereals: What Is beyond Fibre?” Nutrition Research Reviews, Vol. 23, No. 1, 2010, pp. 65-134. doi:10.1017/S0954422410000041
- J. L. Slavin, M. C. Martini, D. R. Jacobs Jr. and L. Marquart, “Plausible Mechanisms for the Protectiveness of Whole Grains,” American Journal of Clinical Nutrition, Vol. 70, No. 3, 1999, pp. 459S-463S.
- R. Giacco, G. Della Pepa, D. Luongo and G. Riccardi, “Whole Grain Intake in Relation to Body Weight: From Epidemiological Evidence to Clinical Trials,” Nutrition, Metabolism & Cardiovascular Diseases, Vol. 21, No. 12, 2011, pp. 901-908. doi:10.1016/j.numecd.2011.07.003
- P. G. Williams, “Evaluation of the Evidence between Consumption of Refined Grains and Health Outcomes,” Nutrition Reviews, Vol. 70, No. 2, 2012, pp. 80-99. doi:10.1111/j.1753-4887.2011.00452.x
- R. Giacco, G. Clemente, D. Cipriano, D. Luongo, D. Viscovo, L. Patti, L. Di Marino, A. Giacco, D. Naviglio, M. A. Bianchi, R. Ciati, F. Brighenti, A. A. Rivellese and G. Riccardi, “Effects of Regular Consumption of Wholemeal Wheat Foods on Cardiovascular Risk Factors in Healthy People,” Nutrition, Metabolism & Cardiovascular Diseases, Vol. 20, No. 3, 2010, pp. 186-194. doi:10.1016/j.numecd.2009.03.025
- J. A. Delcour, X. Rouau, C. M. Courtin, K. Poutanen and R. Ranieri, “Technologies for Enhanced Exploitation of the Health-Promoting Potential of Cereals,” Trends in Food Science & Technology, Vol. 25, No. 2, 2012, pp. 78-86. doi:10.1016/j.tifs.2012.01.007
- Y. Hemery, X. Rouau, V. Lullien-Pellerin, C. Barron and J. Abécassi, “Dry Processes to Develop Wheat Fractions and Products with Enhanced Nutritional Quality,” Journal of Cereal Science, Vol. 46, No. 3, 2007, pp. 327-347. doi:10.1016/j.jcs.2007.09.008
- A. Vrieze, F. Holleman, E. G. Zoetendal, W. M. de Vos, J. B. Hoekstra and M. Nieuwdorp, “The Environment within: How Gut Microbiota May Influence Metabolism and Body Composition,” Diabetologia, Vol. 53, No. 4, 2010, pp. 606-613. doi:10.1007/s00125-010-1662-7
- R. Burcelin, L. Garidou and C. Pomié, “Immuno-microbiota Cross and Talk: The New Paradigm of Metabolic Diseases,” Seminars in Immunology, Vol. 24, No. 1, 2012, pp. 67-74. doi:10.1016/j.smim.2011.11.011
- J. M. Wong, A. Esfahani, N. Singh, C. R. Villa, A. Mirrahimi, D. J. Jenkins and C. W. Kendall, “Gut Microbiota, Diet, and Heart Disease,” Journal of AOAC International, Vol. 95, No. 1, 2012, pp. 24-30. doi:10.5740/jaoacint.SGE_Wong
- J. Schrezenmeir and M. de Vrese, “Probiotics, Prebiotics, and Synbiotics-Approaching a Definition,” American Journal of Clinical Nutrition, Vol. 73, No. 2, 2001, pp. 361S-364S.
- G. Kelly, “Inulin-Type Prebiotics—A Review: Part 1,” Alternative Medicine Review, Vol. 13, No. 4, 2008, pp. 315-329.
- B. Watzl, S. Girrbachn and M. Roller, “Inulin, Oligofructose and Immunomodulation,” British Journal of Nutrition, Vol. 93, No. 1, 2005, pp. S49-S55. doi:10.1079/BJN20041357
- M. Roberfroid, G. R. Gibson, L. Hoyles, A. L. McCartney, R. Rastall, I. Rowland, D. Wolvers, B. Watzl, H. Szajewska, B. Stahl, F. Guarner, F. Respondek, K. Whelan, V. Coxam, M. J. Davicco, L. Léotoing, Y. Wittrant, N. M. Delzenne, P. D. Cani, A. M. Neyrinck and A. Meheust, “Prebiotic Effects: Metabolic and Health Benefits,” British Journal of Nutrition, Vol. 104, No. 2, 2010, pp. S1-S63. doi:10.1017/S0007114510003363
- D. Meyer and M. Stasse-Wolthuis, “The Bifidogenic Effect of Inulin and Oligofructose and Its Consequences for Gut Health,” European Journal of Clinical Nutrition, Vol. 63, No. 11, 2009, pp. 1277-1289. doi:10.1038/ejcn.2009.64
- M. A. Looijer-van Langen and L. A. Dieleman, “Prebiotics in Chronic Intestinal Inflammation,” Inflammatory Bowel Diseases, Vol. 15, No. 3, 2009, pp. 454-462. doi:10.1002/ibd.20737
- A. R. Lomax and P. C. Calder, “Prebiotics, Immune Function, Infection and Inflammation: A Review of the Evidence,” British Journal of Nutrition, Vol. 101, No. 5, 2009, pp. 633-658. doi:10.1017/S0007114508055608
- K. E. Scholz-Ahrens, P. Ade, B. Marten, P. Weber, W. Timm, Y. Açil, C. C. Glüer and J. Schrezenmeir, “Prebiotics, Probiotics, and Synbiotics Affect Mineral Absorption, Bone Mineral Content, and Bone Structure,” Journal of Nutrition, Vol. 137, No. 3, 2007, pp. 838S-846S.
- H. Tilg and A. Kaser, “Gut Microbiome, Obesity, and Metabolic Dysfunction,” Journal of Clinical Investigation, Vol. 121, No. 6, 2011, pp. 2126-2132. doi:10.1172/JCI58109
- C. Cherbut, “Motor Effects of Short-Chain Fatty Acids and Lactate in the Gastrointestinal Tract,” Proceedings of the Nutrition Society, Vol. 62, No. 1, 2003, pp. 95-99. doi:10.1079/PNS2002213
- T. Wolever, P. Spadafora and H. Eshuis, “Interaction between Colonic Acetate and Propionate in Humans,” American Journal of Clinical Nutrition, Vol. 53, No. 3, 1991, pp. 681-687.
- C. J. Homko, P. Cheung and G. Boden, “Effects of Free Fatty Acids on Glucose Uptake and Utilization in Healthy Women,” Diabetes, Vol. 52, No. 2, 2003, pp. 487-491. doi:10.2337/diabetes.52.2.487
- J. W. Anderson and S. R. Bridges, “Short-Chain Fatty Acid Fermentation Products of Plant Fiber Affect Glucose Metabolism of Isolated Rat Hepatocytes,” Proceedings of the Society for Experimental Biology and Medicine, Vol. 177, No. 2, 1984, pp. 372-376. doi:10.3181/00379727-177-41958
- R. Reimer and M. I. McBurney, “Dietary Fiber Modulates Intestinal Glucagon Messenger Ribonucleic Acid and Postprandial Secretion of Glucagon-Like Peptide-1 and Insulin in Rats,” Endocrinology, Vol. 137, No. 9, 1996, pp. 3948-3956. doi:10.1210/en.137.9.3948
- G. T. Macfarlane, H. Steed and S. Macfarlane, “Bacterial Metabolism and Health-Related Effects of Galacto-Oligosaccharides and Other Prebiotics,” Journal of Applied Microbiology, Vol. 104, No. 2, 2008, pp. 305-344.
- FAO/WHO, “Report on Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Lactic Acid Bacteria,” 2001. www.who.int/foodsafety/publications/fs_management/en/probiotics.pdf
- S. Chen, Q. Zhao, L. R. Ferguson, Q. Shu, I. Weir and S. Garg, “Development of a Novel Probiotic Delivery System Based on Microencapsulation with Protectants,” Applied Microbiology and Biotechnology, Vol. 93, No. 4, 2012, pp. 1447-1457. doi:10.1007/s00253-011-3609-4
- F. Nazzaro, P. Orlando, F. Fratianni and R. Coppola, “Microencapsulation in Food Science and Biotechnology,” Current Opinion in Biotechnology, Vol. 23, No. 2, 2012, pp. 182-186. doi:10.1016/j.copbio.2011.10.001
- R. Tallon, S. Arias, P. Bressollier and M. C. Urdaci, “Strainand Matrix-Dependent Adhesion of Lactobacillus plantarum Is Mediated by Proteinaceous Bacterial Compounds,” Journal of Applied Microbiology, Vol. 102, No. 2, 2007, pp. 442-451. doi:10.1111/j.1365-2672.2006.03086.x
- M. de Vrese and J. Schrezenmeir, “Probiotics, Prebiotics, and Synbiotics,” Advances in Biochemical Engineering/ Biotechnology, Vol. 111, 2008, pp. 1-66. doi:10.1007/10_2008_097
- S. Kolida and G. R. Gibson, “Synbiotics in Health and Disease,” Annual Review of Food Science and Technology, Vol. 2, 2011, pp. 373-393. doi:10.1146/annurev-food-022510-133739
- P. D. Cani and N. M. Delzenne, “The Gut Microbiome as Therapeutic Target,” Pharmacology & Therapeutics, Vol. 30, No. 2, 2011, pp. 202-212. doi:10.1016/j.pharmthera.2011.01.012
- J. Salmerón, J. E. Manson, M. J. Stampfer, G. A. Colditz, A. L. Wing and W. C. Willett, “Dietary Fiber, Glycemic Load, and Risk of Non-Insulin-Dependent Diabetes Mellitus in Women,” JAMA, Vol. 77, No. 6, 1997, pp. 472- 477. doi:10.1001/jama.1997.03540300040031
- J. Salmerón, A. Ascherio, E. B. Rimm, G. A. Colditz, D. Spiegelman, D. J. Jenkins, M. J. Stampfer, A. L. Wing and W. C. Willett, “Dietary Fiber, Glycemic Load, and Risk of NIDDM in Men,” Diabetes Care, Vol. 20, No. 4, 1997, pp. 545-550. doi:10.2337/diacare.20.4.545
- S. Liu, W. C. Willett, M. J. Stampfer, F. B. Hu, M. Franz, L. Sampson, C. H. Hennekens and J. E. Manson, “A Prospective Study of Dietary Glycemic Load, Carbohydrate Intake, and Risk of Coronary Heart Disease in US Women,” The American Journal of Clinical Nutrition, Vol. 71, No. 6, 2000, pp. 1455-1461.
- G. Riccardi, G. Clemente and R. Giacco, “Glycemic Index of Local Foods and Diets: The Mediterranean Experience,” Nutrition Reviews, Vol. 61, No. 5, 2003, pp. S56-S60. doi:10.1301/nr.2003.may.S56-S60
- H. Mäkeläinen, H. Anttila, J. Sihvonen, R. M. Hietanen, R. Tahvonen, E. Salminen, M. Mikola and T. SontagStrohm, “The Effect of Beta-Glucan on the Glycemic and Insulin Index,” European Journal of Clinical Nutrition, Vol. 61, No. 6, 2007, pp. 779-785. doi:10.1038/sj.ejcn.1602561
- L. Tappy, E. Gügolz and P. Würsch, “Effects of Breakfast Cereals Containing Various Amounts of Beta-Glucan Fibers on Plasma Glucose and Insulin Responses in NIDDM Subjects,” Diabetes Care, Vol. 19, No. 8, 1996, pp. 831- 834. doi:10.2337/diacare.19.8.831
- K. L. Stanhope and P. J. Havel, “Fructose Consumption: Recent Results and Their Potential Implications,” Annals of the New York Academy of Sciences, Vol. 1190, 2010, pp. 15-24. doi:10.1111/j.1749-6632.2009.05266.x
- A. L. Jenkins, D. J. Jenkins, U. Zdravkovic, P. Würsch and V. Vuksan, “Depression of the Glycemic Index by High Levels of Beta-Glucan Fiber in Two Functional Foods Tested in Type 2 Diabetes,” European Journal of Clinical Nutrition, Vol. 56, No. 7, 2002, pp. 622-628. doi:10.1038/sj.ejcn.1601367
- H. N. Larsen, O. W. Rasmussen, P. H. Rasmussen, K. K. Alstrup, S. K. Biswas, I. H. Tetens, S. H. Thilsted and K. Hermansen, “Glycaemic Index of Parboiled Rice Depends on the Severity of Processing: Study in Type 2 Diabetic Subjects,” European Journal of Clinical Nutrition, Vol. 54, No. 5, 2000, pp. 380-385. doi:10.1038/sj.ejcn.1600969
- H. N. Larsen, C. Christensen, O. W. Rasmussen, I. H. Tetens, N. H. Choudhury, S. H. Thilsted and K. Hermansen, “Influence of Parboiling and Physico-Chemical Characteristics of Rice on the Glycaemic Index in Non-Insulin-Dependent Diabetic Subjects,” European Journal of Clinical Nutrition, Vol. 50, No. 1, 1996, pp. 22-27.
- M. Parillo, R. Giacco, G. Riccardi, D. Pacioni and A. A. Rivellese, “Different Glycaemic Responses to Pasta, Bread, and Potatoes in Diabetic Patients,” Diabetic Medicine, Vol. 2, No. 5, 1985, pp. 374-377. doi:10.1111/j.1464-5491.1985.tb00655.x
- R. Giacco, F. Brighenti, M. Parillo, M. Capuano, A. V. Ciardullo, A. Rivieccio, A. A. Rivellese and G. Riccardi, “Characteristics of Some Wheat-Based Foods of the Italian Diet in Relation to Their Influence on Postprandial Glucose Metabolism in Patients with Type 2 Diabetes,” British Journal of Nutrition, Vol. 85, No. 1, 2001, pp. 33-40. doi:10.1079/BJN2000218
- M. De Angelis, C. G. Rizzello, G. Alfonsi, P. Arnault, S. Cappelle, R. Di Cagno and M. Gobbetti, “Use of Sourdough Lactobacilli and Oat Fibre to Decrease the Glycaemic Index of White Wheat Bread,” British Journal of Nutrition, Vol. 98, No. 6, 2007, pp. 1196-1205. doi:10.1017/S0007114507772689
- US Food and Drug Administration, “FDA Final Rule for Federal Labelling: Health Claims: Oats and Coronary Heart Disease,” Federal Register, Vol. 62, No. 3, 1997, pp. 3584-3681.
- S. M. Haffner, S. Lehto, T. Rönnemaa, K. Pyörälä and M. Laakso, “Mortality from Coronary Heart Disease in Subjects with Type 2 Diabetes and in Nondiabetic Subjects with and without Prior Myocardial Infarction,” The New England Journal of Medicine, Vol. 339, No. 4, 1998, pp. 229-234. doi:10.1056/NEJM199807233390404
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults, “Executive Summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III),” JAMA, Vol. 285, No. 19, 2001, pp. 2486-2497. doi:10.1001/jama.285.19.2486
- D. J. Jenkins, K. Srichaikul, A. Mirrahimi, L. Chiavaroli and C. W. Kendall, “Functional Foods to Increase the Efficacy of Diet in Lowering Serum Cholesterol,” Canadian Journal of Cardiology, Vol. 27, No. 4, 2011, pp. 397-400. doi:10.1016/j.cjca.2011.03.008
- D. J. Jenkins, C. W. Kendall, A. Marchie, D. A. Faulkner, J. M. Wong, R. de Souza, A. Emam, T. L. Parker, E. Vidgen, K. G. Lapsley, E. A. Trautwein, R. G. Josse, L. A. Leiter and P. W. Connelly, “Effects of a Dietary Portfolio of Cholesterol-Lowering Foods vs Lovastatin on Serum Lipids and C-Reactive Protein,” JAMA, Vol. 290, No. 4, 2003, pp. 502-510. doi:10.1001/jama.290.4.502
- US Food and Drug Administration, “Food Labelling: Health Claims; Soluble Fiber from Certain Foods and Coronary Heart Disease,” Federal Register, Vol. 63, No. 68, 1998, pp. 17327-17329.
- D. J. Jenkins, C.W. Kendall, V. Vuksan, E. Vidgen, T. Parker, D. Faulkner, C. C. Mehling, M. Garsetti, G. Testolin, S. C. Cunnane, M. A. Ryan and P. N. Corey, “Soluble Fiber Intake at a Dose Approved by the US Food and Drug Administration for a Claim of Health Benefits: Serum Lipid Risk Factors for Cardiovascular Disease Assessed in a Randomized Controlled Crossover Trial,” The American Journal of Clinical Nutrition, Vol. 75, No. 5, 2002, pp. 834-839.
NOTES
*Corresponding author.

