American Journal of Plant Sciences
Vol.3 No.8(2012), Article ID:22186,4 pages DOI:10.4236/ajps.2012.38134
Mutual Information Flow between Beneficial Microorganisms and the Roots of Host Plants Determined the Bio-Functions of Biofertilizers
![]()
College of Life Sciences, South Central University for Nationalities, Wuhan, China.
Email: *yusheen@163.com
Received May 13th, 2012; revised June 10th, 2012; accepted June 18th, 2012
Keywords: Biofertilizers; Host Plant Roots; Plant Growth-Promoting Rhizobacteria (PGPR); Plant Growth-Promoting Fungi (PGPF); Signal Transduction
ABSTRACT
Modern agriculture is facing new challenges in which ecological and molecular approaches are being integrated to achieve higher crop yields while minimizing negative impacts on the environment. The application of biofertilizers could meet this requirement. Biofertilizer is a natural organic fertilizer that helps to provide all the nutrients required by the plants and helps to increase the quality of the soil with a natural microorganism environment. This paper reviewed the types of biofertilizers, the biological basic of biofertilizers in plant growth promotion. This paper also assayed the bidirectional information exchange between plant-microbes in rhizoshpere and the signal pathway of plant growthpromoting rhizobacteria (PGPR) and plant growth-promoting fungi (PGPF) in the course of plant infection. At last, the challenges of the application and the promising future of biofertilizers were also discussed.
1. Introduction (Agricultural Implications of Biofertilizer Application)
The increase of agricultural food production worldwide over the past four decades has been associated with a remarkable increase in the use of fertilizers [1]. Concomitantly, the continue use and overuse of petrochemical based fertilizers have triggered a detrimental effect on soils and water supplies and caused environmental concerns, such as the eutrophication of freshwater and marine ecosystems [2,3]. Moreover, chemical fertilizers increasingly displayed the ill-effects such as destroying micro-organisms and friendly insects, making the crop more susceptible to the attack of diseases, reducing the soil fertility and thus causing irreparable damage to the overall system. Exploring the alternative fertilizers to increase in crop yield and food production without further negative impacts on the global environment is necessary to support the growing world population. The exploit and application of biofertilizers will be essential to meet this purpose. It is also the requirement of the development of organic and ecological friendly agriculture.
Biofertilizers are substances which contains beneficial living microorganisms which, when applied to seed, plant surfaces, or soil, colonizes the rhizosphere or the interior of the plant and promotes growth by increasing the supply or availability of primary nutrients to the host plant and thus, have lower manufacturing costs especially regarding nitrogen and phosphorus use [4]. Comparison with chemical fertilizers, Biofertilizers are environmentally friendly not only in that they prevent damaging the natural source but also help to some extent cleanse the plant from precipitated chemical fertilizers and in turn help build up the health soil [4].
2. Types of Biofertilizers
The diversities of microorganisms provided prolific resources for biofertilizer exploit. Long known fact is that the symbiosis of plants with mycorrhiza and nitrogenfixing bacteria will promote plant growth and crop yields, and helps plants cope with some harmful insects [5]. This phenomenon enlightened scientist to develop the nitrogen fixer fertilizers (Figure 1). In addition to the beneficial bacteria and fungi, there are also many other microorganisms that can be used for the fertilizer exploring, such as blue green algae, water floating ferns and some species of moss (Figure 1). Some biofertilziers are exploited to address the barren of some mineral elements in the soil, such as phosphate, sulphur, zinc solubilizing
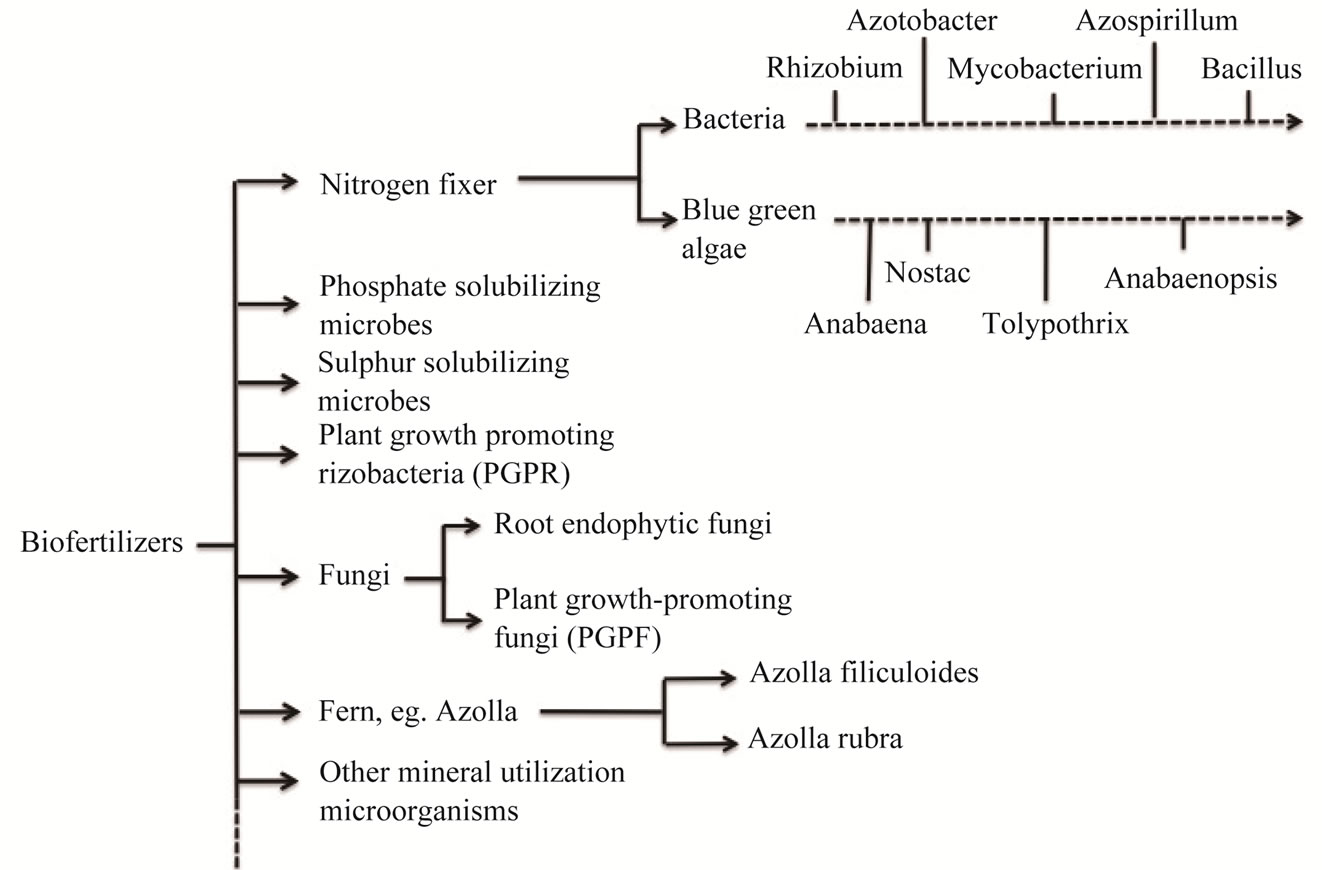
Figure 1. Microorganisms used for exploitation for biofertilizers up to date.
microbes and so on. The beneficial microorganisms in biofertilizers could mobilize the availability of nutrients in soil by their biological activity.
The microbe-mediated stimulation of plant growth can be explained by the improvement of plant nutrition and increase tolerance to biotic and abiotic stress. The beneficial effects of biofertilizers on plant growth have a high relevance for agricultural ecosystems because they reduce the need for fertilizers, leading to a decrease in pollution of agricultural soils and water [4,6]. Additionally, soil-borne microbes can enhance plant tolerance to abiotic stresses, such as drought, salt stress and the presence of heavy metals in the soil [7,8]. In addition to their plant growth-promoting effect, they induce systemic resistance against microbial pathogens and act as a biological control agent of such pathogens [9,10].
Some studies demonstrated that the mutual interaction between the benefical organisms and host plants could enhance flower visitation and seed set of mycorrhizal plants compared to control plants [11-13]. The underlying mechanisms reported include an increase in flower number, inflorescence size and nectar production [11,13], but whether plant volatiles are also involved remains to be uncovered.
Some microbes can also synthesize plant hormones that promote plant growth (e.g. indole-3-acetic acid, cytokinines, and gibberellins) [14,15] and can increase aboveground photosynthesis through the modulation of endogenous sugar and abscisic acid (ABA) signaling [16].
3. The Biological Basic of Biofertilizers to Promote Plant Growth and Crop Yields
From the evolution perspective, some conservation mechanism relevant with similar hormone signal components and signaling pathway lies at the heart of biological basic of biofertilizers. Data from large-scale genome sequencing projects such as those of Physcomitrella patens (moss), Selaginella (fern), Arabidopsis thaliana and Oriza sativa (rice) provided us some important insights into the conservation and evolution of hormone signaling pathways. For example, the moss genome (an ancient plant ancestor) encodes proteins that function in auxin, ABA and cytokinin signaling, whereas the genome of green algae does not, suggesting that these pathways emerged when plants were colonizing land [17,18]. In contrast, a comparison of the moss genome with more recently diverged plant genomes suggests that signaling mechanisms for gibberellin, ethylene and the brassinosteroids probably did not evolve until after the evolutionary split of moss and vascular plants [17,18]. Lots of evidences demonstrated that these plant hormones role in plant growth regulation and biomasses [19]. It is reasonable to explain the growth promotion of beneficial microorganisms on plants from the point view of the hormone providers. However, the diversities of plant hormones and their minute ratio of different hormones will trigger different plant reaction. For example, one kind of commercial biofertilizer, called Kelp (extracted from some kinds of marine Phaeophyta species) act as a soil conditioner by stimulating microbial activity in the soil which results in improved air-water relationships in soil, improved fertility and makes soil less prone to compaction and erosion. As biostimulants, Kelp are sold as powders or liquid extracts and their use is now well established for a variety of agricultural and horticultural crops. Cytokinins are considered as one of the active ingredients and many different cytokinins have been identified in some of these seaweed concentrates. Ten isoprenoid and seven aromatic cytokinins were identified in Kelpak® with a total concentration of 4.88 pmol·ml–1 [20]. This example showed that a key critical factor not ignored in assessing the function of different kinds of biofertilizers is that we should assay the contents and kinds of hormone, especially the cytokinins that the microbes produced.
4. Bidirectional Information Exchange between Plant-Microbes in Rhizoshpere
Densely colonized soil contains beneficial mycorrhizal fungi and rhizobia, which associate with roots and provide plants with mineral nutrients and fixed nitrogen, respectively, in exchange for carbon. By contrast, plants are constantly exposed to a range of fungal, bacterial and viral pathogens, and have evolved unique defense mechanisms to fight these infections. So the coevolution of plant and microbe make them to be friendly or hostile. Recent scientific discoveries demonstrated the complex signal pathway relationship between plant growth-promoting rhizobacteria (PGPR), root endophytic fungi and plant growth-promoting fungi (PGPF) and their host plants [5,21,22].
Much evidence uncovered the mechanism of PGPR and PGPF in the course of infection plants [21,23]. During the infection courses, the specific bidirectional information flow between microbes and host plants determined the specific immunity responses. Studies indicated that plant-derived signals from the root exudates could stimulate Arbuscularmycorrhiza (AM) fungal hyphae branching. The molecular identity of the “branching factors” was identified as strigolactones. This new kind of plant hormone was found to be responsible for the induction of branching and alterations in fungal physiology and mitochondrial activity [24,25]. Perception of AM fungal or rhizobia-derived signals triggers early signal transduction, which is mediated by at least seven shared components in host plant (Figure 2). The symbiosis receptor kinase SYMRK acts upstream of the Nod factor (from PGPR) and Myc factor (from PGPF) induced calcium signatures that occur in and around the nucleus. Perinuclear calcium spiking involves the release of calcium from a storage compartment (probably the nuclear envelope) through as-yet-unidentified calcium channels. The potassium-permeable channels CASTOR and POLLUX might compensate for the resulting charge imbalance. The nucleoporins NUP85 and NUP133 are required for calcium spiking, although their mode of involvement is currently unknown. The calcium-calmodulin-dependent protein kinase (CCaMK) forms a complex with CYCLOPS, a phosphorylation substrate, within the nucleus. Together with calmodulin, this complex might decode the symbiotic calcium signatures. Upstream of the common pathway, the Nod factor receptor kinases NFR1 and NFR5 are specifically required for Nod factor perception. It is possible that similar receptors are involved in Myc factor perception [21].
5. The Challenge of Biofertilizer Application
In the traditional agriculture, scientists are focusing on the study of the breeding of high yield crop through the crop modification. However, the breeding of high-yield crops varieties and the use of pesticides, fertilizers and more water [26]. But the leap in crop yield increasing was accompanied by a depletion of groundwater and bringing environmental problems such as polluted waterways. Biofertilizers are compounds that enrich the nutrient quality of soil by the use of microorganisms, which have a symbiotic relationship with the plants and the development of biofertilizers has promising bright future in the contributing to the modern sustainable and green agriculture. However, there are full of huge challenges in the course of the application of this technology. Some relevant reasons are assayed as follows.
Because roots have much potential genetic variation that has been neglected before, many scientists are starting to see roots as central to their efforts to produce crops with a better yield and turning their attention to roots to increase crop yields, and regarded roots as the key to a second green revolution and booster for food production [26,27]. However, this underground revolution is still at its infant stage. The complexity of the interaction between plants and beneficial microorganisms in biofertilizers make it difficult to investigate the function of biofertilizers. Plant-microbes interactions have the species specificity from the diversities of both plants and microbes. For example, some kinds of symbiosis arbuscular mycorrhiza (AM) cannot independently live after isolation from with its host plants [21]. In this situation, the study of the biofertilizer function is just like the “blackbox”.
Another future challenge will come from the understanding how multiple signaling pathways integrated to regulate plant growth. For example, Cytokinin plays a central role in the formation of nitrogen fixing root nodules following inoculation with rhizobia. Recent study demonstrated that that exogenous cytokinin induces formation of discrete and easily visible nodule primordia in Lotus japonicas root [28,29]. This study stressed the role of cytokinin in legume root endosymbiosis and provided a new strategy for the use of biofertilizers. However, root nodulation involves multiple plant hormone participation, such as auxin, gibberellin, ethylene and so on [28]. Although our knowledge of the molecular components
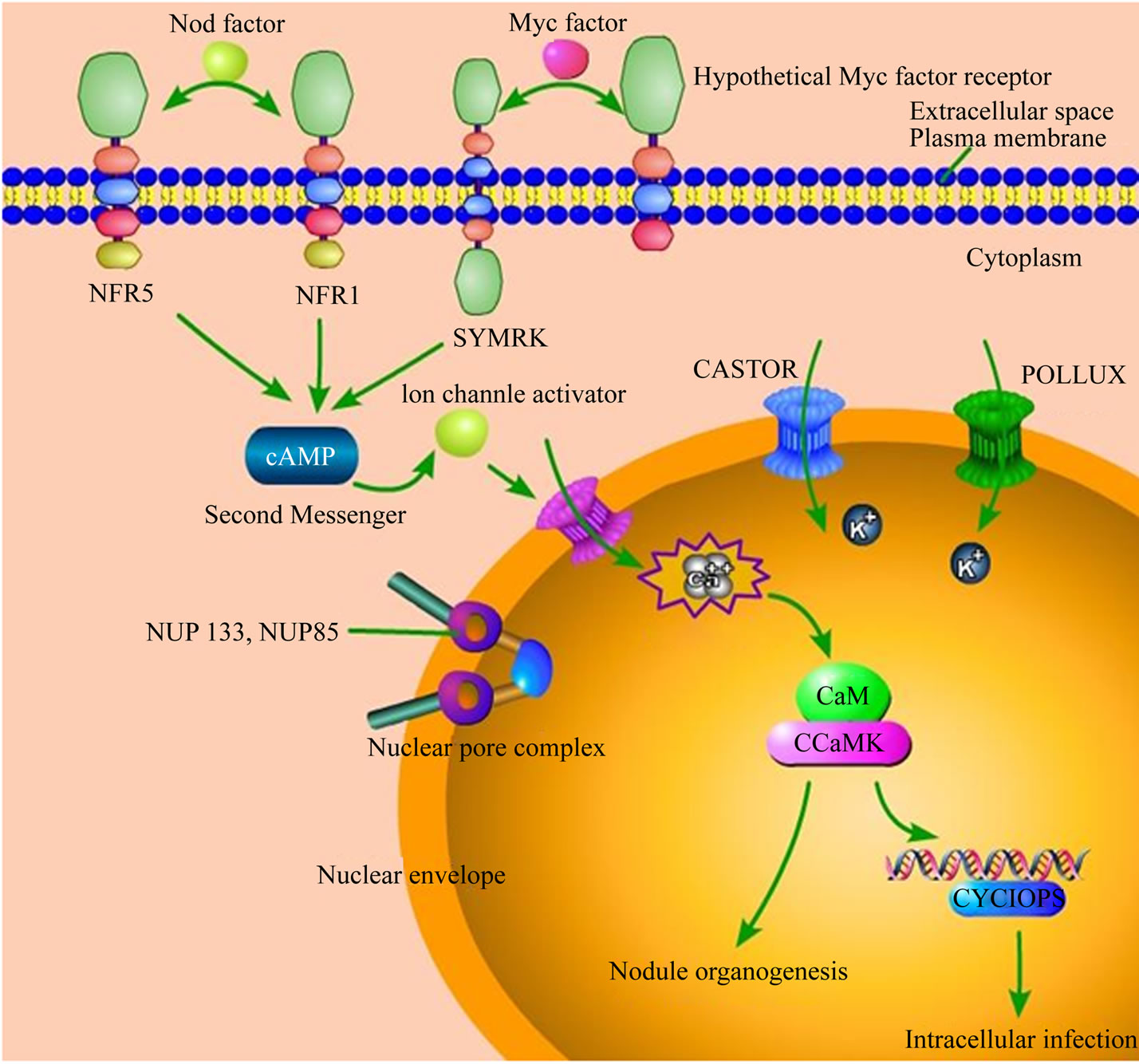
Figure 2. Signal components participated in arbuscular mycorrhiza (AM) and root-nodule symbiosis (Adapted from Parniske 2008, [21]).
and pathways that mediate hormone responses has improved enormously in recent years, the molecular mechanisms of hormone interaction remain poorly understood. Greater efforts are still needed to elucidate the crosstalk between different hormones interactions both derived from host plants and microbes.
In addition to the reasons mentioned above, other problems, such as the divergent function of cytokinins [30] added difficulty to the assay of the different kinds of cytokinins derived form microorganisms; the application itself of biofertilizers could also brought with some challenges. For example, the problem could come from the mix of different biofertilizers, the shelf-life of biofertilizers, their resistant ability to environmental stresses such as drought, extreme heat, early frost, pest and disease could also trigger the problems to affect the effect of biofertilizers.
6. Conclusion
Biofertilizers are the most advanced biotechnology necessary to support developing organic agriculture, sustainable agriculture, green agriculture and non-pollution agriculture. They contain a wide range of naturally chelated plant nutrients and trace elements, carbohydrates, amino acids and other growth promoting substances. Today, It has been widely used with excellent results in all kinds of plants and several countries. Consideration the fact that biofertilziers could enhance the fertility of soil, the conclusion that “human healthy of people depends upon healthy plants (for their food), healthy plants upon healthy soil, and healthy soil upon healthy biofertilizers” can be reached. At last, a famous saying in the Holy Bible (Mark 4:8) was citied to describe the function of biofertilizers in high crop yield production “Other seed fell into good soil and brought forth grain, growing up and increasing and yielding thirty, and sixty and a hundredfold”. This goal will be achieved in the foreseeable future in the development of modern sustainable agriculture.
7. Acknowledgements
Authors thank the foundation supports by the National Natural Science Foundation of China (30700427; 31270- 361) and Outstanding Young Talent Fund from Key Projects in Hubei Province Natural Science Foundation (2010CDA099).
REFERENCES
- B. Hirel, J. Le Gouis, B. Ney and A. Gallais, “The Challenge of Improving Nitrogen Use Efficiency in Crop Plants: Towards a More Central Role for Genetic Variability and Quantitative Genetics within Integrated Approaches,” Journal of Experimental Botany, Vol. 58, No. 9, 2007, pp. 2369-2387. doi:10.1093/jxb/erm097
- J. G. London, “Nitrogen Study Fertilizes Fears of Pollution,” Nature, Vol. 433, No. 7028, 2005, p. 791. doi:10.1038/433791a
- J. M. Beman, K. Arrigo and P. A. Matson, “Agricultural Runoff Fuels Large Phytoplankton Blooms in Vulnerable Areas of the Ocean,” Nature, Vol. 434, No. 7037, 2005, pp. 211-214. doi:10.1038/nature03370
- N. Weyens, D. van der Lelie, S. Taghavi, L. Newman and J. Vangronsveld, “Exploiting Plant-Microbe Partnerships to Improve Biomass Production and Remediation,” Trends in Biotechnology, Vol. 27, No. 10, 2009, pp. 591-598. doi:10.1016/j.tibtech.2009.07.006
- A. Pineda, S.-J. Zheng, J. J. A. van Loon, C. M. J. Pieterse and M. Dicke, “Helping Plants to Deal with Insects: The Role of Beneficial Soil-Borne Microbes,” Trends in Plant Science, Vol. 15, No. 9, 2010, pp. 507- 514. doi:10.1016/j.tplants.2010.05.007
- J. Yang, J. W. Kloepper and C.-M. Ryu, “Rhizosphere Bacteria Help Plants Tolerate Abiotic Stress,” Trends in Plant Science, Vol. 14, No. 1, 2008, pp. 1-4. doi:10.1016/j.tplants.2008.10.004
- H. Evelin, R. Kapoor and B. Giri, “Arbuscular Mycorrhizal Fungi in Alleviation of Salt Stress: A Review,” Annals of Botany, Vol. 104, No. 7, 2009, pp. 1263-1280. doi:10.1093/aob/mcp251
- H. Bae, R. C. Sicher, M. S. Kim, S.-H. Kim, M. D. Strem, R. L. Melnick and B. A. Bailey, “The Beneficial Endophyte Trichoderma hamatum Isolate DIS 219b Promotes Growth and Delays the Onset of the Drought Response in Theobroma cacao,” Journal of Experimental Botany, Vol. 60, No. 11, 2009, pp. 3279-3295. doi:10.1093/jxb/erp165
- M. I. Trillas and G. Segarra, “Interactions between Nonpathogenic Fungi and Plants,” Advances in Botanical Research, Vol. 51, 2009, pp. 321-359. doi:10.1016/S0065-2296(09)51008-7
- G. Segarra, S. Van der Ent, I. Trillas and C. M. J. Pieterse, “MYB72, a Node of Convergence in Induced Systemic Resistance Triggered by a Fungal and a Bacterial Beneficial Microbe,” Plant Biology, Vol. 11, No. 1, 2009, pp. 90-96. doi:10.1111/j.1438-8677.2008.00162.x
- A. C. Gange and A. K. Smith, “Arbuscular Mycorrhizal Fungi Influence Visitation Rates of Pollinating Insects,” Ecological Entomology, Vol. 30, No. 5, 2005, pp. 600- 606. doi:10.1111/j.0307-6946.2005.00732.x
- B. E. Wolfe, B. C. Husband and J. N. Klironomos, “Effects of a Belowground Mutualism on an Aboveground Mutualism,” Ecology Letters, Vol. 8, No. 2, 2005, pp. 218-223. doi:10.1111/j.1461-0248.2004.00716.x
- J. F. Cahill Jr., E. Elle, G. R. Smith and B. H. Shore, “Disruption of a Belowground Mutualism Alters Interactions between Plants and Their Floral Visitors,” Ecology, Vol. 89, No. 7, 2008, pp. 1791-1801. doi:10.1890/07-0719.1
- L. C. van Loon, “Plant Responses to Plant GrowthPromoting Rhizobacteria,” European Journal of Plant Pathology, Vol. 119, No. 3, 2007, pp. 243-254. doi:10.1007/s10658-007-9165-1
- H. A. Contreras-Cornejo, L. Macías-Rodríguez, C. Cortés-Penagos and J. López-Bucio, “Trichoderma virens, a Plant Beneficial Fungus, Enhances Biomass Production and Promotes Lateral Root Growth through an AuxinDependent Mechanism in Arabidopsis,” American Society of Plant Biologists, Vol. 149, No. 3, 2009, pp. 1579-1592. doi:10.1104/pp.108.130369
- H. M. Zhang, X. T. Xie, M.-S. Kim, D. A. Kornyeyev, S. Holaday and P. W. Paré, “Soil Bacteria Augment Arabidopsis Photosynthesis by Decreasing Glucose Sensing and Abscisic Acid Levels in Planta,” The Plant Journal, Vol. 56, No. 2, 2008, pp. 264-273. doi:10.1111/j.1365-313X.2008.03593.x
- S. A. Rensing, D. Lang, A. D. Zimmer, A. Terry, A. Salamov, H. Shapiro, et al., “The Physcomitrella Genome Reveals Evolutionary Insights into the Conquest of Land by Plants,” Science, Vol. 319, No. 5859, 2008, pp. 64-69. doi:10.1126/science.1150646
- F. Vandenbussche, A. C. Fierro, G. Wiedemann, R. Reski and D. Van Der Straeten, “Evolutionary Conservation of Plant Gibberellin Signalling Pathway Components,” BMC Plant Biology, Vol. 7, 2007, p. 65. doi:10.1186/1471-2229-7-65
- A. Santner and M. Estelle, “Recent Advances and Emerging Trends in Plant Hormone Signaling,” Nature, Vol. 459, No. 7250, 2009, pp. 1071-1078. doi:10.1038/nature08122
- W. A. Stirk and J. van Staden, “Flow of Cytokinins through the Environment,” Plant Growth Regulation, Vol. 62, No. 2, 2010, pp. 101-116. doi:10.1007/s10725-010-9481-x
- M. Parniske, “Arbuscular Mycorrhiza: The Mother of Plant Root Endosymbioses,” Nature Reviews Microbiology, Vol. 6, No. 10, 2008, pp. 763-775. doi:10.1038/nrmicro1987
- S. C. M. Van Wees, S. Van der Ent and C. M. J. Pieterse, “Plant Immune Responses Triggered by Beneficial Microbes,” Current Opinion in Plant Biology, Vol. 11, No. 4, 2008, pp. 443-448. doi:10.1016/j.pbi.2008.05.005
- P. Bonfante and A. Genre, “Mechanisms Underlying Beneficial Plant—Fungus Interactions in Mycorrhizal Symbiosis,” Nature Communications, Vol. 1, No. 4, 2010, pp. 1-11.
- K. Akiyama, K. Matsuzaki and H. Hayashi, “Plant Sesquiterpenes Induce Hyphal Branching in Arbuscular Mycorrhizal Fungi,” Nature, Vol. 435, No. 7043, 2005, pp. 824-827. doi:10.1038/nature03608
- A. Besserer, V. Puech-Pagès, P. Kiefer, V. GomezRoldan, A. Jauneau, S. Roy, J. C. Portais, C. Roux, G. Bécard and N. Séjalon-Delmas, “Strigolactones Stimulate Arbuscular Mycorrhizal Fungi by Activating Mitochondria,” Public Library of Science Biology, Vol. 4, No. 7, 2006, pp. 1239-1247. doi:10.1371/journal.pbio.0040226
- V. Gewin, “Food: An Underground Revolution,” Nature, Vol. 466, No. 7306, 2010, pp. 552-553. doi:10.1038/466552a
- G. Den Herder, G. Van Isterdael, T. Beeckman and I. De Smet, “The Roots of a New Green Revolution,” Trends in Plant Science, Vol. 15, No. 11, 2010, pp. 600-607. doi:10.1016/j.tplants.2010.08.009
- G. J. Desbrosses and J. Stougaard, “Root Nodulation: A Paradigm for How Plant-Microbe Symbiosis Influences Host Developmental Pathways,” Cell Host & Microbe, Vol. 10, No. 4, 2011, pp. 348-358. doi:10.1016/j.chom.2011.09.005
- A. B. Heckmann, N. Sandal, A. S. Bek, L. H. Madsen, A. Jurkiewicz, M. W. Nielsen, L. Tirichine and J. Stougaard, “Cytokinin Induction of Root Nodule Primordia in Lotus japonicus Is Regulated by a Mechanism Operating in the Root Cortex,” Molecular Plant-Microbe Interactions, Vol. 24, No. 11, 2011, pp. 1385-1395. doi:10.1094/MPMI-05-11-0142
- S. Perilli, L. Moubayidin and S. Sabatini, “The Molecular Basis of Cytokinin Function,” Current Opinion in Plant Biology, Vol. 13, No. 1, 2010, pp. 21-26. doi:10.1016/j.pbi.2009.09.018
NOTES
*Corresponding author.

