American Journal of Plant Sciences
Vol.3 No.5(2012), Article ID:19516,4 pages DOI:10.4236/ajps.2012.35077
Kinetic Studies on β-Galactosidase Isolated from Apricots (Prunus armeniaca kaisa)
![]()
Department of Biochemistry, The University of Kashmir, Srinagar, India.
Email: *shajrul@rediffmail.com
Received January 23rd, 2012; revised February 23rd, 2012; accepted March 1st, 2012
Keywords: β-Galactosidase; Apricots; Chromatography; Enzyme Kinetics
ABSTRACT
β-galactosidase was extracted from apricots (Prunus armeniaca kaisa) and characterized biochemically. Three isoenzymes (β-gal I, β-gal II and β-gal III) were obtained by salt fractionation and ion-exchange and Sephadex G-100 column chromatography. β-galactosidase II showed a high ability to hydrolyze the substrate p-nitrophenyl β-D-galactopyranoside than that of β-galactosidase I and III. The individual peaks showed charge homogeneity as revealed by single band on polyacrylamide gel. The molecular weight of β-gal I, β-gal II and β-gal III as determined by gel filtration was found to be 44.15, 34.70 and 23.71 KDa respectively. The optimum pH for the activity different isozymes was found between 4 and 6. The isoenzymes were determined to be thermally stable up to 40˚C. The Km value for β-gal I was 1.85 mM which was higher than that of β-gal II (Km = 1.7), and β-gal III (Km = 1.19). The Vmax value for β-gal I, β-gal II and β-gal III was found to be 0.52, 0.70 and 0.38 µmole/min respectively.
1. Introduction
Fruits constitute a commercially important and nutritionally indispensable food commodity. Excessive ripening of fruits is a serious problem in the market and/or during storage. This is a major cause of quality loss and a significant problem for the food industry.
Ripening of fruit is usually accompanied by loss of firmness or softening, a process that changes the color and aroma of the fruit. The process may be mediated by the action of hydrolytic enzymes on the polysaccharides of the cell wall. Pectin modifying enzymes, polygalacturonase (PG) and pectin methylesterase (PME) are known to be involved in the solubilization of pectin during fruit softening processes. However, molecular evidence has suggested that these enzymes alone are not responsible for causing softening in tomato [1]. Further investigations demonstrated that pectin solubilization by β-galactosidase is actively involved in fruit softening [2-4]. Although some loss of galactosyl residues could result indirectly from the action of PG, β-galactosidase is the only enzyme identified in higher plants capable of directly cleaving β-(1, 3 and 4) galactan bonds, and probably plays a role in galactan side chain loss [5-7].
β-galactosidases are widely distributed in plant tissues e.g. leaves [8], seedlings [9], hypocotyls [10] and meristem zones of roots, cotyledons, vascular tissues, trichomes, and pollens [11,12]. Various studies have indicated remarkable increases in the activity of β-galactosidase during ripening of many fruits [13-18]. Increase in expression level of mRNA β-galactosidase during fruit ripening has also been reported in many fruits [19-24].
In this study, β-galactosidase from apricots (Prunus armeniaca kaisa) was isolated and purified to homogeneity. Three isoforms of the enzyme were identified and their physiochemical, functional and kinetic properties were studied.
2. Materials and Methods
2.1. Materials
p-nitrophenyl β-D-galactopyranoside (PNGP), Sephadex G-100 cytochrome c, ovalbumin and pepsin were obtained from Sigma Chemical Co. (USA). BSA and CMcellulose were obtained from SRL Chemicals, Mumbai, India. All other reagents and chemicals were of analytical grade. Ripe mature apricots (Prunus Armeniaca kaisa) were obtained from Sher-e-Kashmir University of Agriculture, Science and Technology, Srinagar, J&K, India.
2.2. Methods
2.2.1. Isolation of β-Galactosidase
Stored apricots were cut into pieces and seeds were separated apart. The cortical tissue was weighed (332 g) and homogenized for 10 min in ice cold extraction buffer (0.1 M sodium acetate pH 5.2) that contained 0.1% (v/v) β- mercaptoethanol, 2% (w/w) polyvinyl poly pyrroli done, 12% (w/v) polyethylene glycol and 2% (w/v) sodium bisulfate. The homogenate was filtered through 4 layers of cheese cloth & the suspension obtained was stirred on magnetic stirrer for about 4 hours at 4˚C and 1 M NaCl was added. The residue left after filtration was washed twice by homogenization in 300 ml ice cold extraction buffer, followed by filtration. The final residue was weighed and brought up to 300 g with 0.5 M NaCl. This was stirred for 1 h at 4˚C, and then filtered through Whatman No.1 filter paper. The filtrate was combined with the suspension and was centrifuged on Remi C 24 cooling at 13,000 rpm for 10 min at 4˚C. From the supernatant, the proteins were precipitated by ammonium sulphate at 85% saturation.The crude extract was further purified involving ion exchange chromatography on CMcellulose column using 0.1 M sodium acetate buffer, pH 5.2, using linear sodium chloride gradient from 0.1 to 0.5 M. The fractions eluted from ion-exchange column were chromatographed separately on Sephadex G 100 column in 0.1 M sodium acetate buffer, pH 5.2. The charge homogeneity of the fractions eluted from gel filtration column was checked by polyacrylamide gel electrophoresis.
2.2.2. Polyacrylamide Gel Electrophoresis
Polyacrylamide gel electrophoresis was carried out with a discontinuous buffer system at pH 8.3 on 10% gel with a discontinuous buffer system. The gels were stained with Coomassie Brilliant Blue R-250.
2.2.3. Protein Estimation
Protein concentration was determined by the method of Lowry et al. [25]. Bovine serum albumin (BSA) was used as standard.
2.2.4. Assay of β-Galactosidase
The activity of β-galactosidase was determined by using the method of Ali et al [16]. The β-galactosidase was assayed by following the release of p-nitrophenol from p-nitrophenyl β-D-galactopyranoside substrate. The reaction mixture consisted of 0.5 ml of 6 mM substrate, 1 ml of 0.1 M sodium acetate buffer (pH 5.2), 0.45 ml of 0.01 mg% BSA and 50 µl of diluted enzyme solution. The reaction was allowed to proceed for 1hour at 37˚C and was terminated by adding 1.0 ml of 1.0 M sodium carbonate. For substrate and the enzyme preparation separate blanks were used. The absorbance of liberated pnitrophenol was measured at 405 nm.
One unit of enzyme activity was defined as that amount of enzyme that catalyzes the liberation of 1 µmol of pnitrophenol/min at 37˚C. The enzyme activity was calculated according to following equation
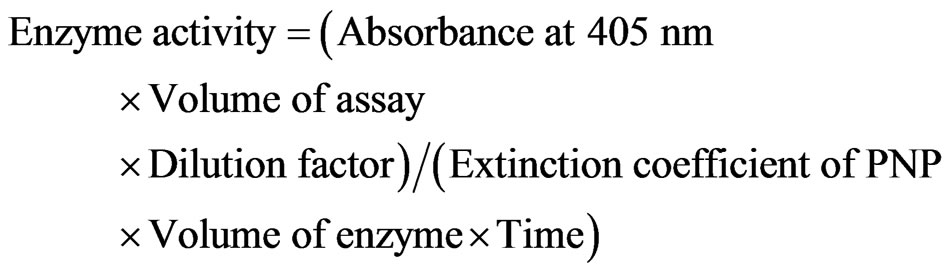
2.2.5. Molecular Weight Determination
Molecular weight was determined by Sephadex gel chromatography. The Sephadex G 100 column was pre equilibrated with standard proteins.
2.2.6. Effect of Substrate Concentrations on Enzyme Activity
In order to determine the Vmax and Km of β-galactosidases for PNGP as substrate, the effect of substrate concentration on enzyme activity was studied at pH 5.2 at 37˚C. Keeping the amount of enzyme constant in assay mixture, the concentration of PNGP was increased from 0.5 mM to 9 mM. For each substrate concentration and the enzyme preparation separate blanks were used. The enzyme activity was assayed by measuring the change in absorbance at 405 nm. Reciprocal plots were used to determine Vmax and Km.
2.2.7. Effect of pH on Enzyme Activity
The pH optima of apricot β-galactosidases was determined by incubating the enzyme at 30˚C in various pH adjusted buffers e.g. 0.1M glycine HCl buffer, pH 2.0 and 3.0, 0.1 M NaoAc buffer pH 4.2, 4.6, 5.0 and 5.2, 0.1 M phosphate buffer pH 6.0 and 7.0, 0.1 M Tris HCl buffer pH 8 and 9 0.1 M sodium carbonate buffer, pH 10. The enzyme assay was performed separately in each buffer system.
2.2.8. Effect of Temperature on Enzyme Activity
To determine the temperature optima the enzyme assay was performed by incubating reaction mixtures at various temperatures from 10˚C to 70˚C.
3. Results
3.1. Isolation and Purification of β-Galactosidase
The soluble form of β-galactosidase was isolated from apricot by homogenization. After the extraction of soluble enzyme the cell wall bound β-galactosidase was extracted successively with 2 M NaCl. The isolated enzyme which was subjected to 85% ammonium sulfate saturation showed 17.2 fold increase in purity of β-galactosidase. The crude preparation showed an increase in enzyme activity and specific activity over the homogenate. When applied on CM-cellulose column in 0.1 M sodium acetate buffer, pH 5.2 the protein eluted in two major peaks, I and II (Figure 1). The fractions under both peaks showed β-galactosidase activity. The fractions were pooled and chromatographed separately onto gel filtration column. Peak I was resolved into two enzymatically active protein peaks labeled as β-gal II & β-gal III, where as peak II was eluted as a single enzymatically active protein peak (β-gal I); the chromatographic profile is shown in Figure 2. The activity of β-gal I, II and III was found to be 0.39, 0.52 and 0.30 units respectively. At each purification step the specific activity of enzymes increased over the homogenate (Table 1).
3.2. Electrophoresis and Molecular Weight Determination
Purity of β-gal I, β-gal II and β-gal III was checked by
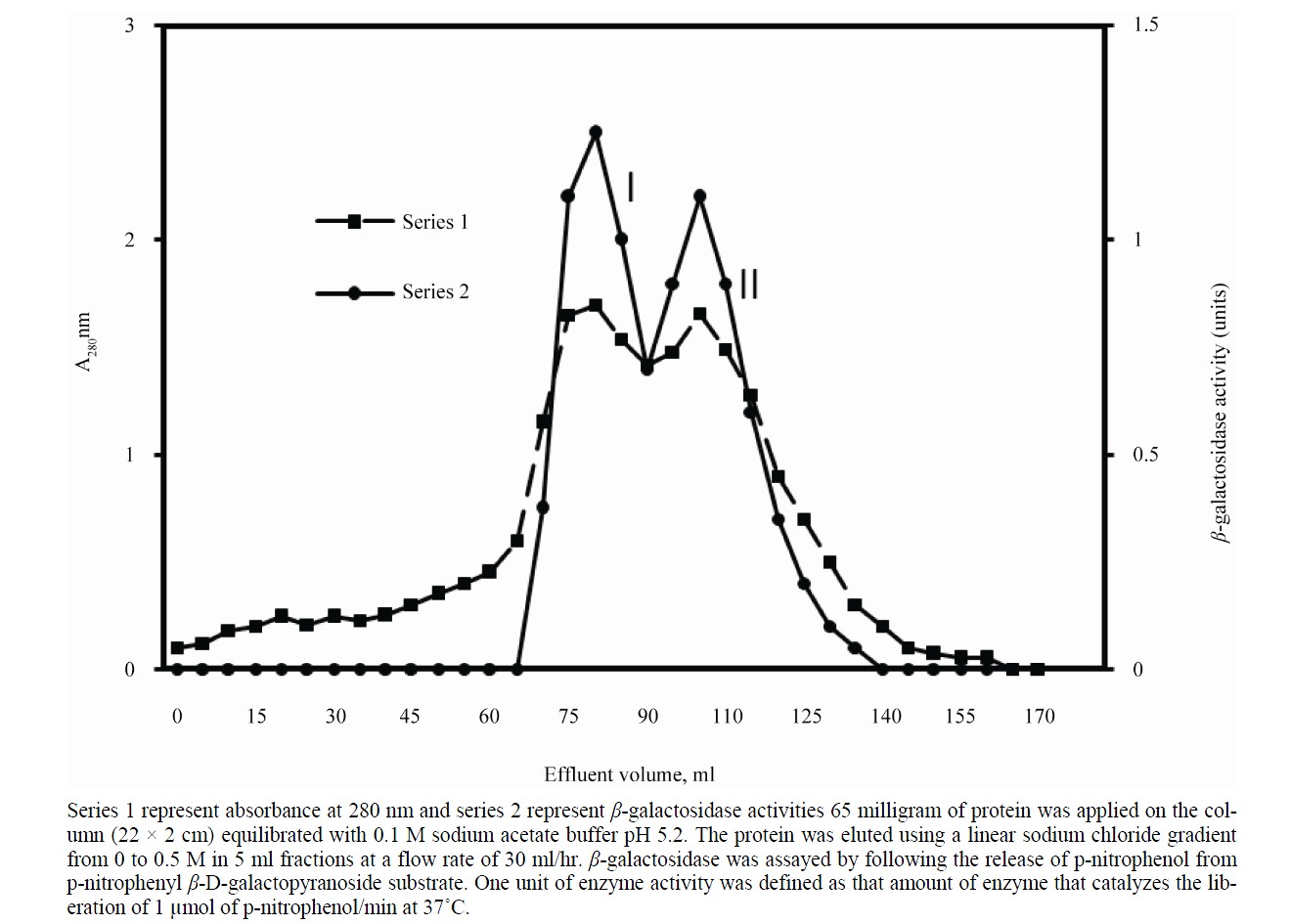
Figure 1. Elution profile of β-galactosidase extracted from apricot on CM cellulose column.
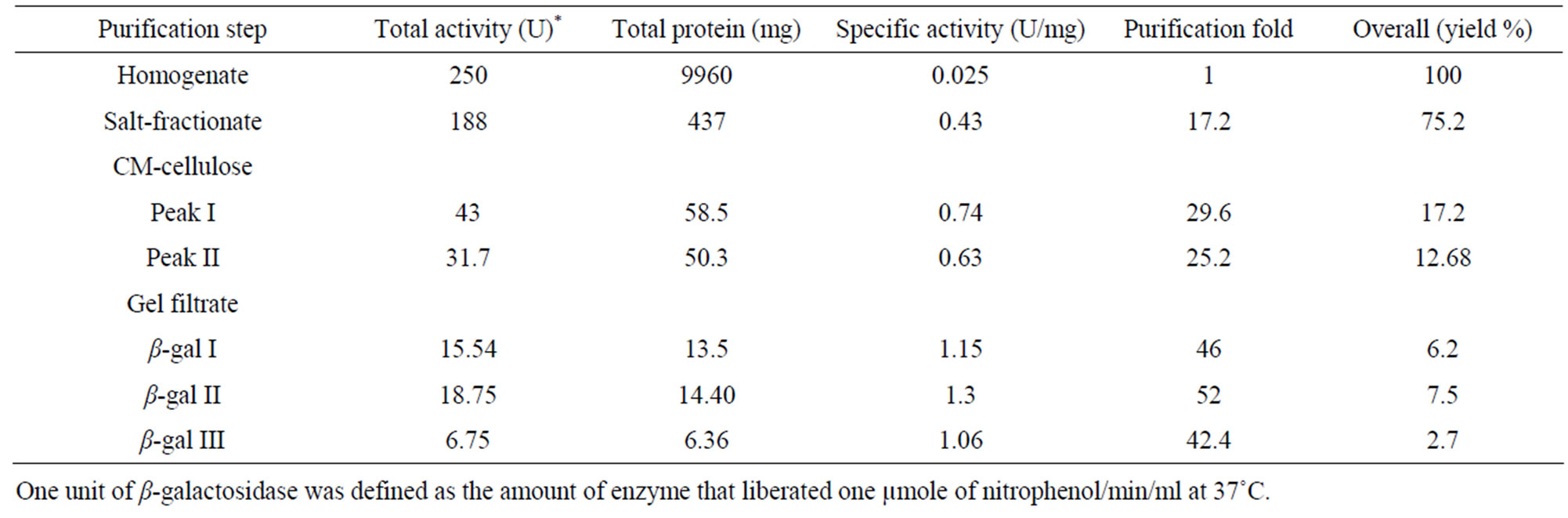
Table 1. Purification of β-galactosidase isoforms from apricot.
polyacrylamide gel electrophoresis. All the three isoenzymes gave single band indicating that the preparation was homogeneous with respect to charge (Figure 3). The molecular weight of isoforms of β-galactosidase was determined by gel filtration chromatography on Sephadex G 100 column. The column was pre equilibrated with standard proteins. The purified isoenzymes displayed a molecular weight of 44.15, 34.70 and 23.71 KDa for β-gal I, β-gal II and β-gal III respectively.
3.3. Effect of Substrate Concentrations
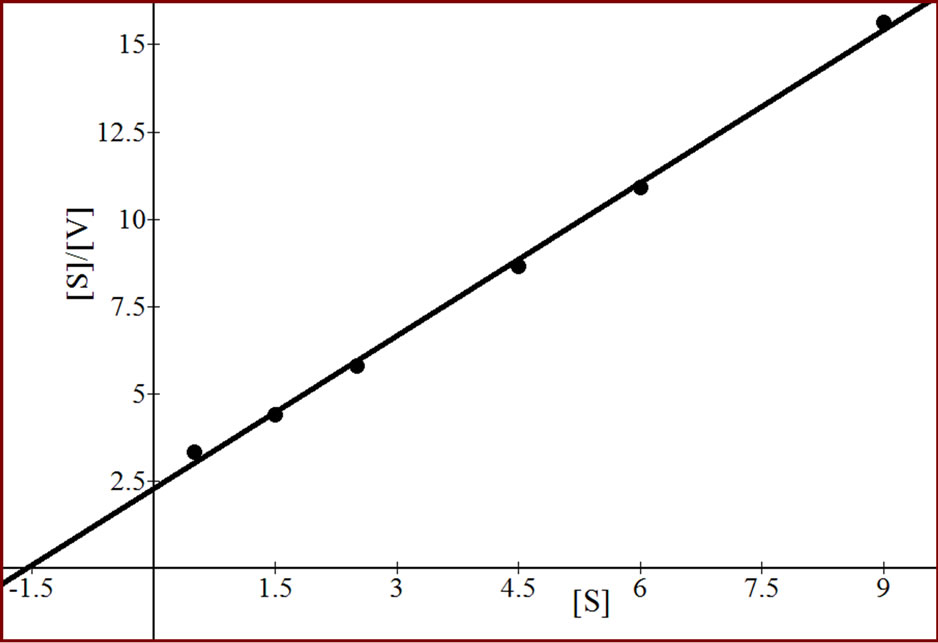
All the three isoforms of β-galactosidase showed the Michaelis-Menten kinetics (Figures 4, 6 and 8). The data
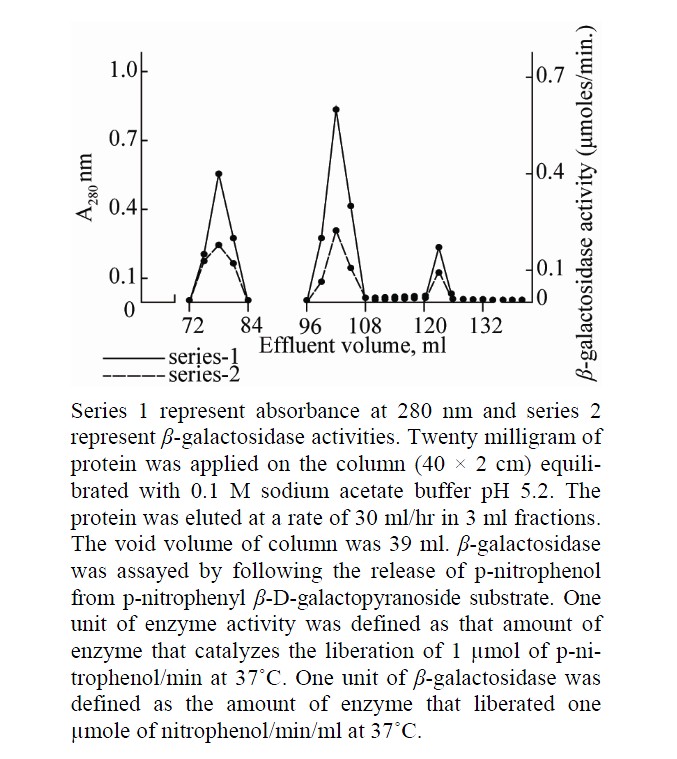
Figure 2. Elution profile of peak I and II obtained from cellulose column.

Figure 3. Polyacrylamide gel electrophoretic pattern of apricot β-galactosidases (Prunus armeniaca kaisa).
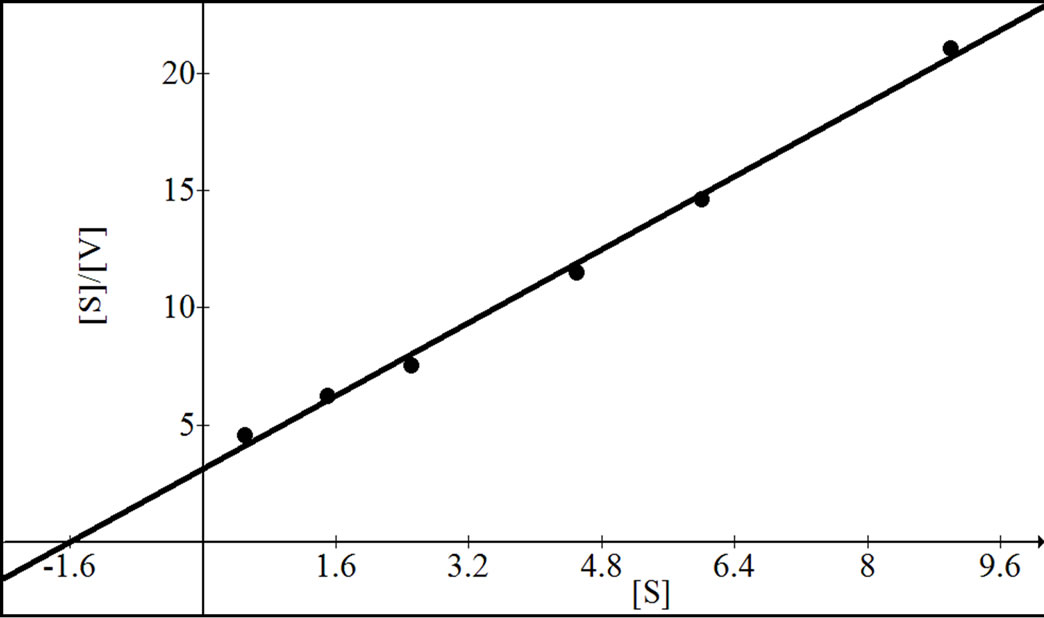
were analyzed according to Lineweaver Burk plot, WoolfAugustinsson-Hofstee plot, Hanes-Woolf plot and EadieScatchard plot (Figures 5, 7 and 9). The kinetic data of different isoforms obtained from the reciprocal plots is given in Tables 2, 3 and 4. The Km and Vmax value for each β-galactosidase isoform were taken as average of
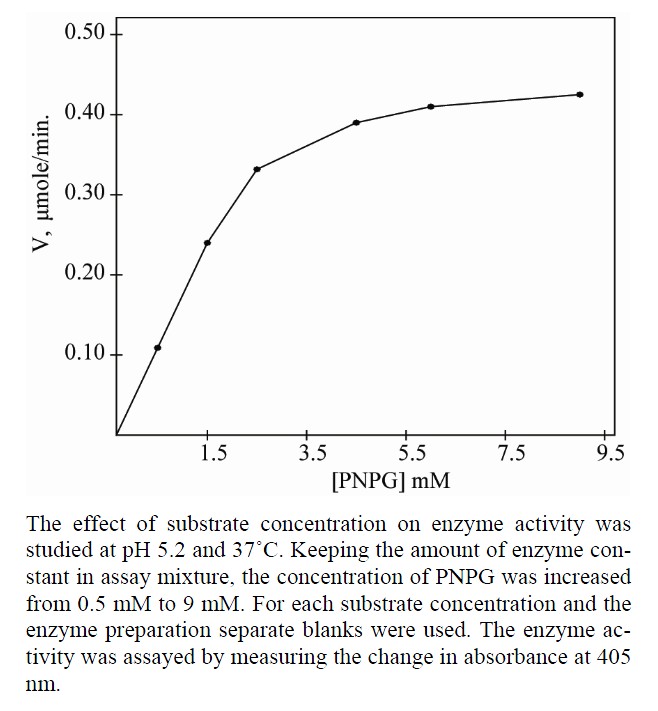
Figure 4. Effect of substrate concentration on the activity of apricot β-gal I.

Table 2. Kinetic data for the apricot β-gal I.
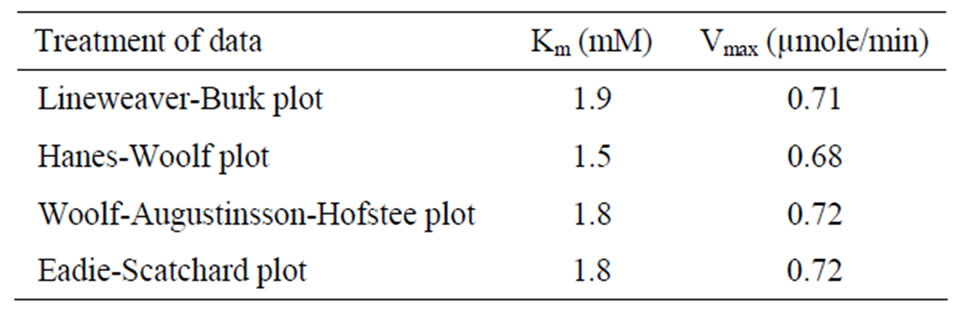
Table 3. Kinetic data for the apricot β-gal II.
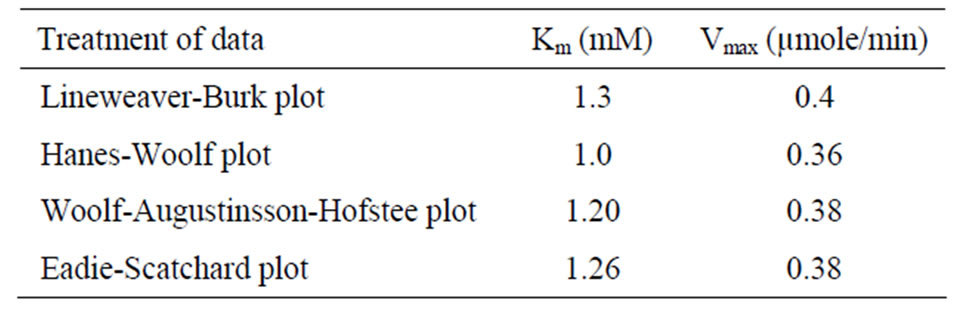
Table 4. Kinetic data for the apricot β-gal III.
 (a)
(a)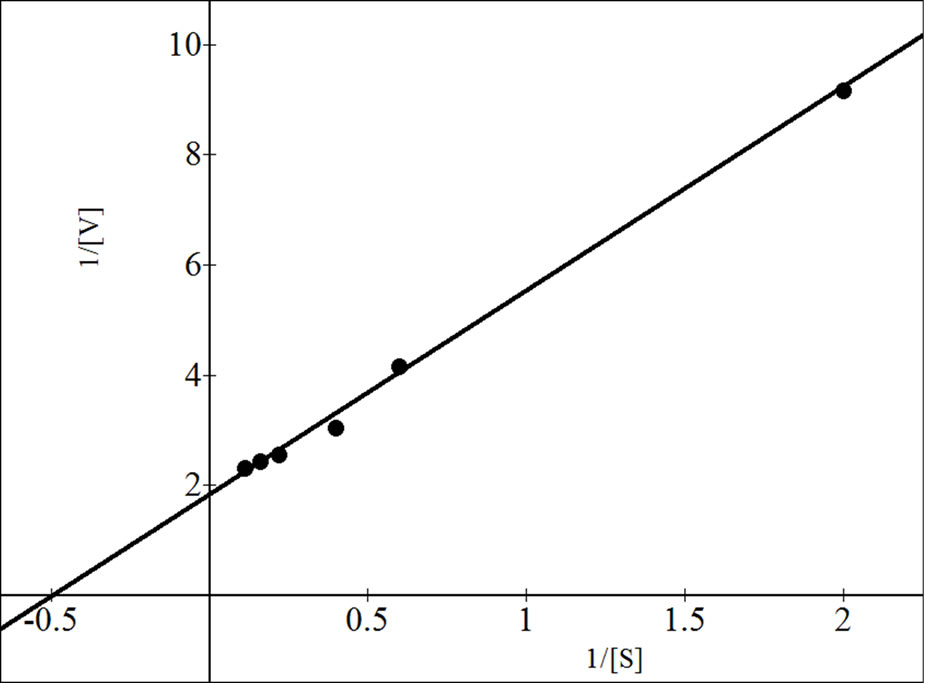 (b)
(b)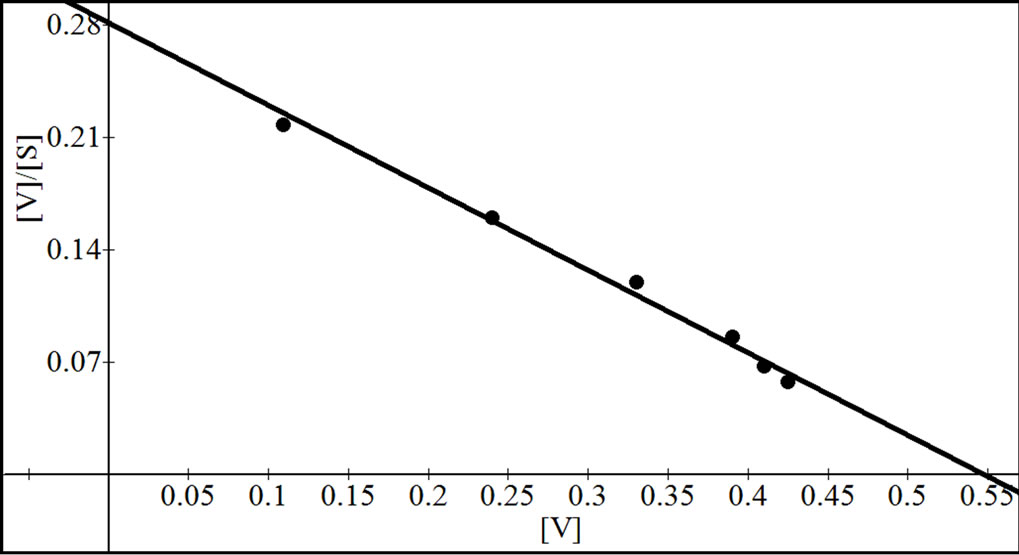 (c)
(c)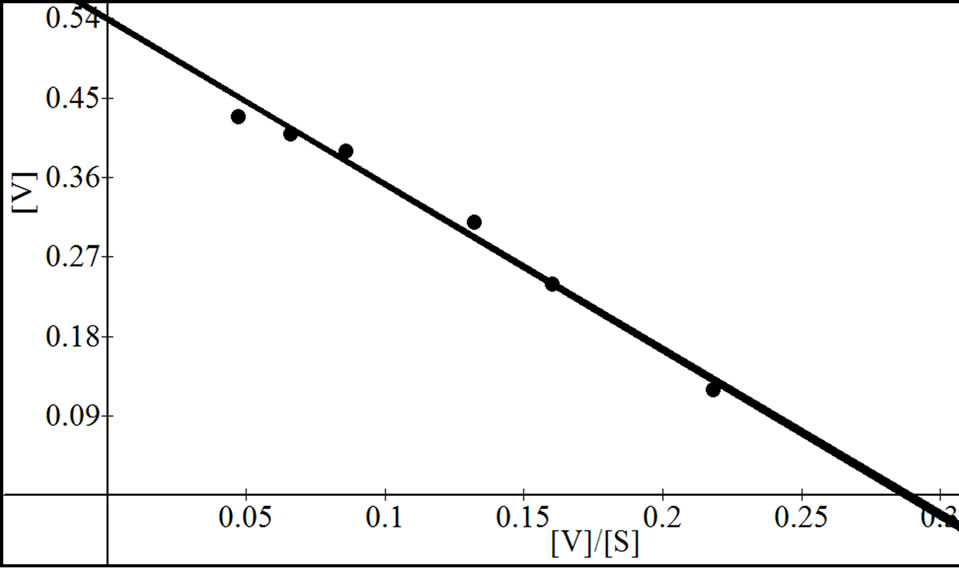 (d)
(d)
Figure 5. Reciprocal plots for β-gal I. Lineweaver Burk plot (a); Hanes-Woolf plot (b); Woolf-Augustinsson-Hofstee plot (c) and Eadie-Scatchard plot (d).
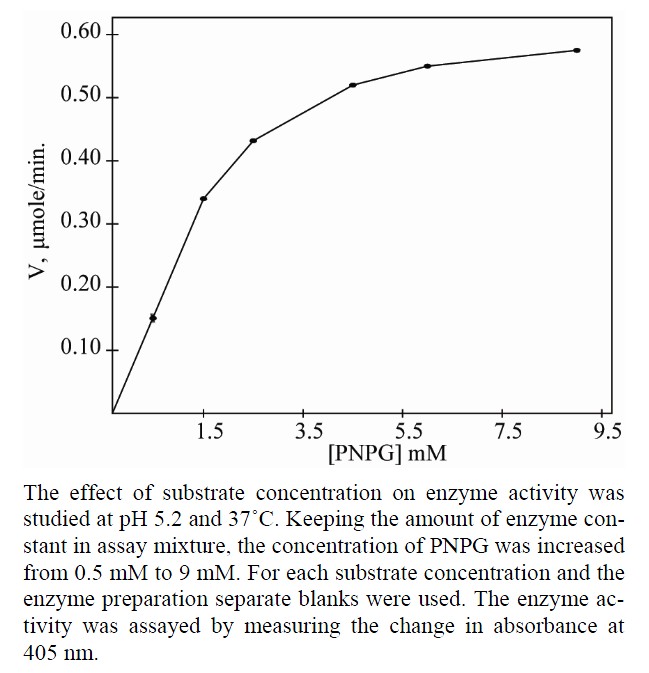
Figure 6. Effect of substrate concentration on the activity of apricot β-gal II.
four values obtained from reciprocal plots. The Km value for β-gal I was found to be 1.85 mM which was higher than that of β-gal II and β-gal III. The Vmax value for β-gal II (0.70 µmole·min–1) was found to be higher than that of β-gal I and β-gal III. The results are given in Table 5.
3.4. Effect of pH
The isozymes responded differently when exposed to different pH buffers. The optimum pH for the enzymes were between 4.2 to 6 for β-gal I and 4 to 6 for β-gal II and 4 to 5.2 for β-gal III. However β-gal I and β-gal II showed maximum activity at pH 5.2 and β-gal III at pH 4.2. A sharp decrease in activity of β-gal I, β-gal II and β-gal III was observed afterwards (Figure 10).
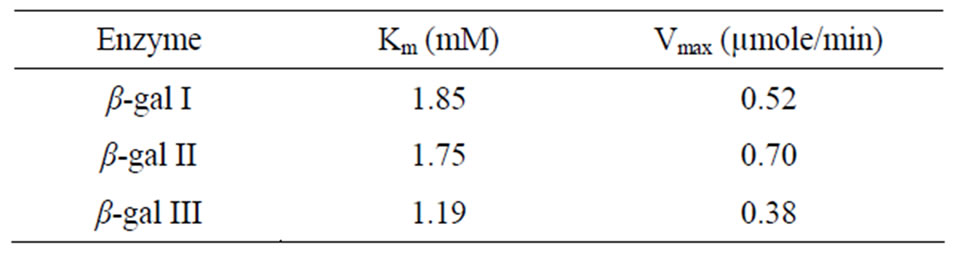
Table 5. Average Km and Vmax of β-gal I, II and III taken from Lineweaver Burk plot, Woolf-Augustinsson-Hofstee plot, Hanes-Woolf plot and Eadie-Scatchard plot.
 (a)
(a)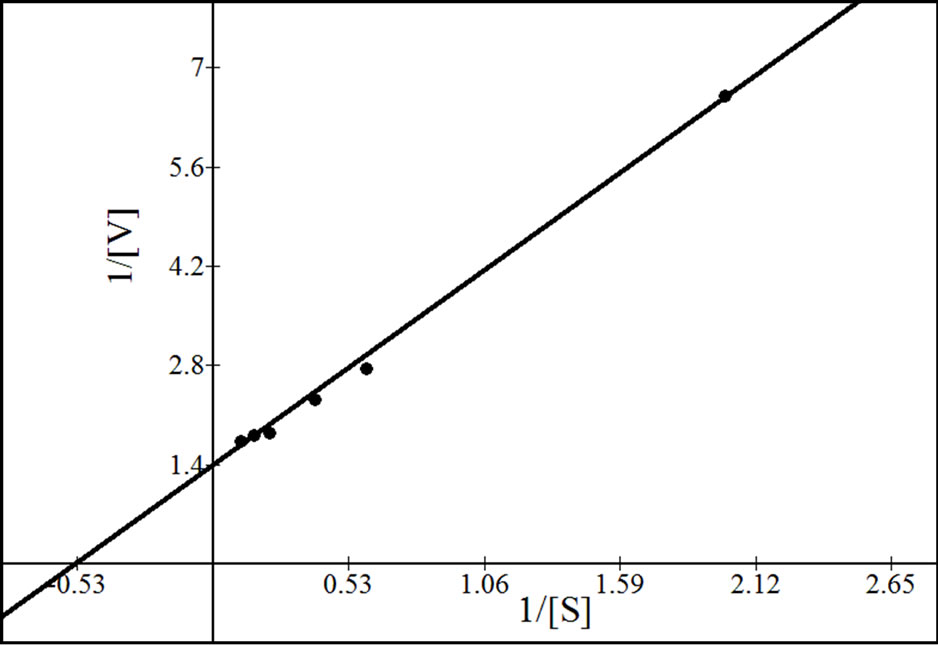 (b)
(b)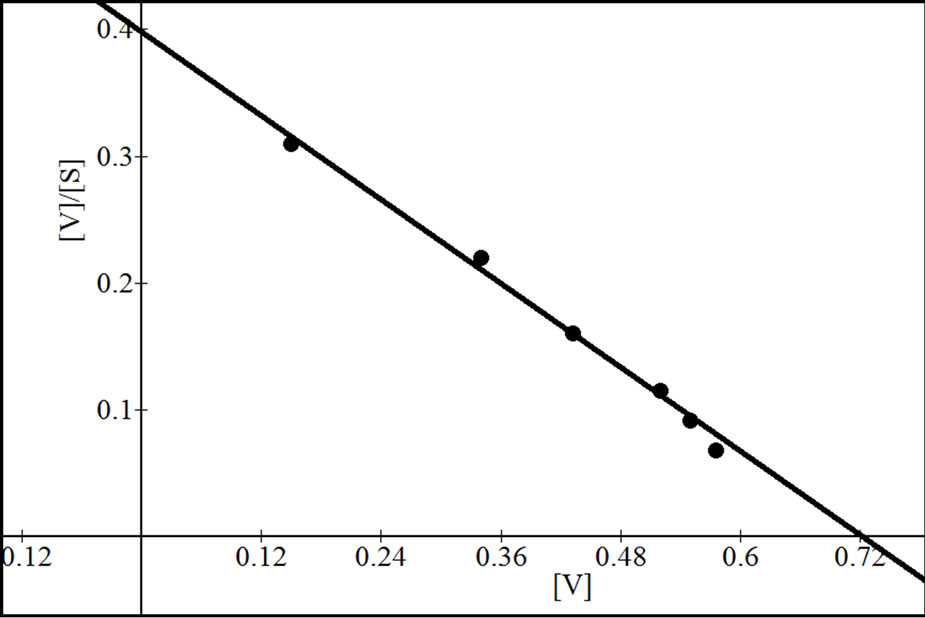 (c)
(c)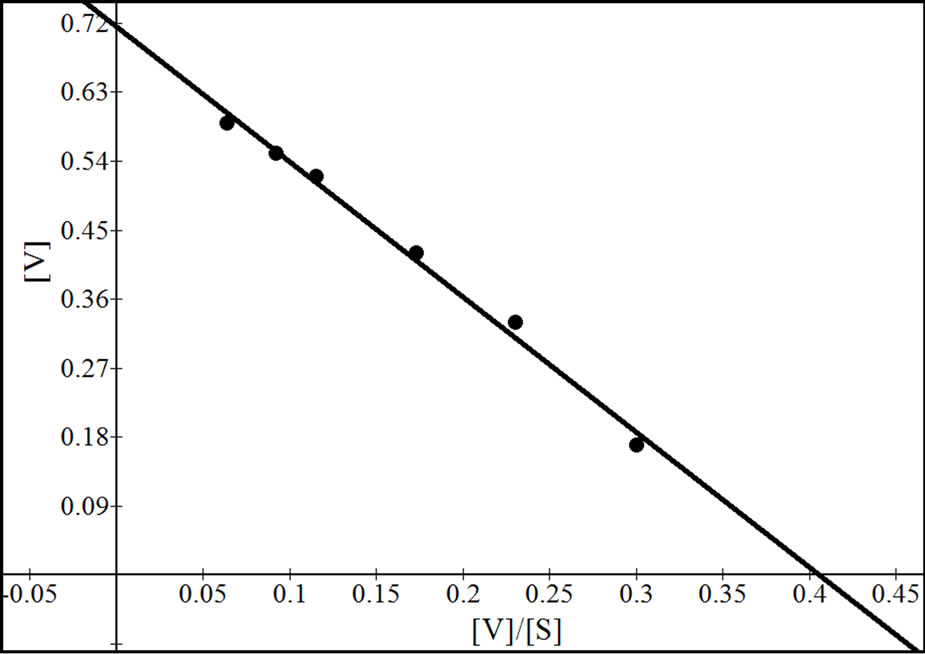 (d)
(d)
Figure 7. Reciprocal plots for β-gal II. Lineweaver Burk plot (a), Hanes-Woolf plot (b), Woolf-Augustinsson-Hofstee plot (c) and Eadie-Scatchard plot (d).
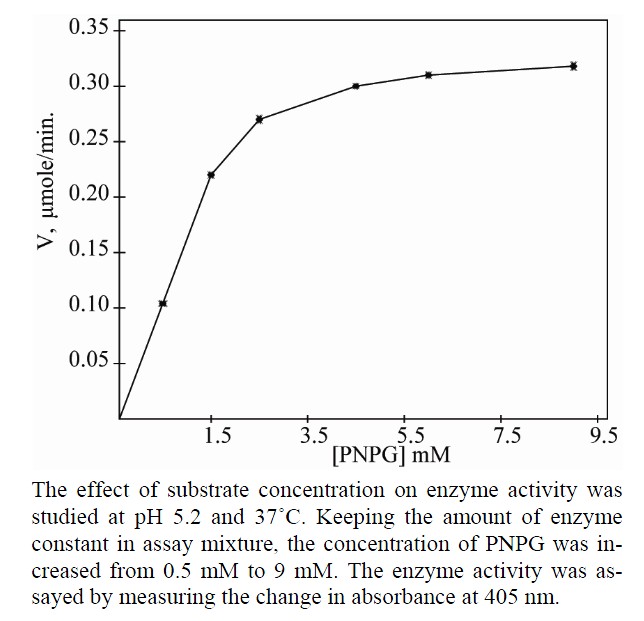
Figure 8. Effect of substrate concentration on the activity of apricot β-gal III.
3.5. Effect of Temperature
The thermal stability of β-galactosidase isozymes was determined by incubating reaction mixtures at temperatures ranging from 10˚C to 70˚C. The optimum temperature was found to be 40˚C for β-gal I, β-gal II and β-gal III (Figure 11). The enzymes showed a sharp decrease in activity afterward. At 60˚C there was complete loss of enzyme activity.
4. Discussion
β-galactosidase in fruits have been to exist in two forms: soluble and wall bound [2]. In the present study the soluble form of the enzyme was isolated by homogenization. After the extraction of soluble enzyme the wall bound enzyme was extracted successively with 2 M NaCl. The enzyme e separated into three peaks when subjected to ion exchange chromatography on CM-cellulose column suggesting that the β-galactosidase in apricot exists in three isoforms viz β-gal I, II and III. The results are comparable to those obtained from other fruit [17,18,20,21]. The molecular weight of β-galactosidase I, II and III as determined by Sephadex gel chromatography was found to be 44.157, 34.700 and 23.713 kDa respectively. In cortical tissue of ripe apples and persimmon fruit two polypeptides of 44 kDa and 32 kDa of β-galactosidase has been found [3,14]. In avocado 2 polypeptides of 21.5 kDa and 66.5 kDa were found [5].
β-galactosidase I, II and III showed different abilities to hydrolyze synthetic substrate p-nitrophenyl-β-galactoside. β-galactosidase II showed a high ability to hydrolyze the substrate PNGP than that of β-galactosidase I
 (a)
(a) (b)
(b)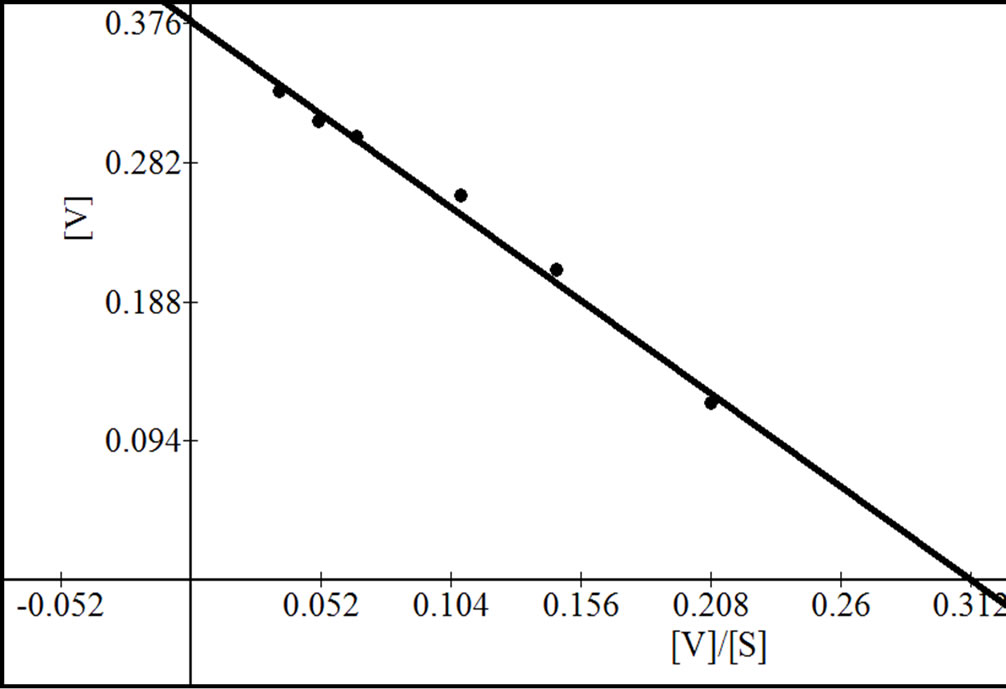 (c)
(c)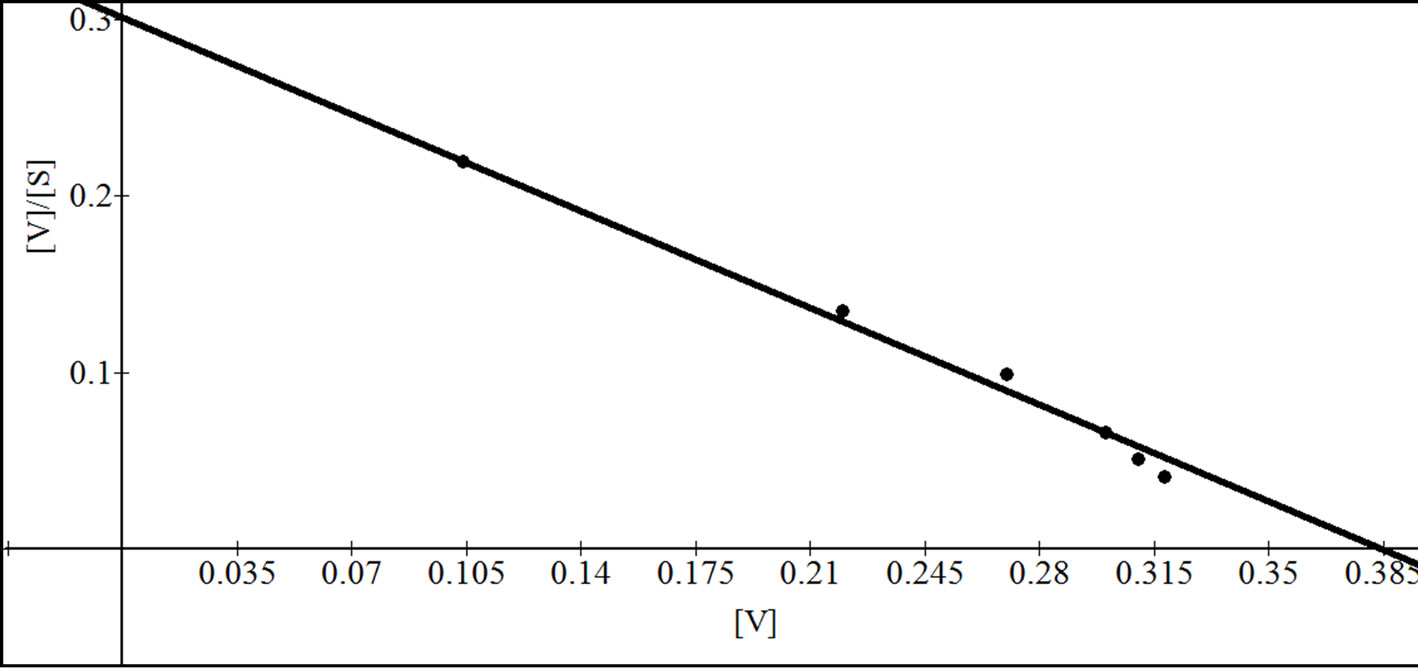 (d)
(d)
Figure 9. Reciprocal plots for β-gal III. Lineweaver Burk plot (a); Hanes-Woolf plot (b); Woolf-Augustinsson-Hofstee plot (c) and Eadie-Scatchard plot (d).
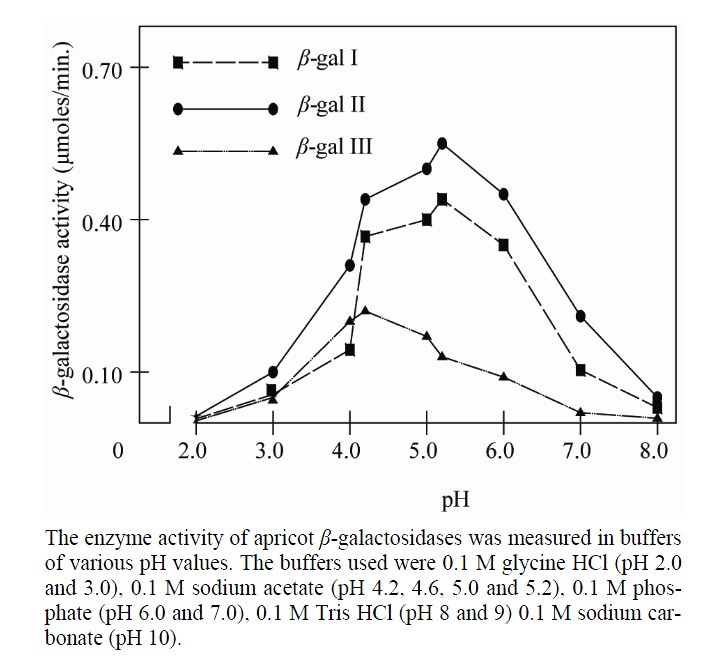
Figure 10. pH-activity profiles of different isoforms of apricot β-galactosidase.
that in turn showed a higher ability than that of β-galactosidase III suggesting that during apricot ripening the levels of the individual forms of β-galactosidase change markedly. The Km value for β-gal I was higher than that of β-gal II and β-gal III. However, the Km value of β-gal II was higher than reported earlier 0.17 mM for carrot [26] 1.25 mM for sweet cherry [15], 1.77 mM for tomato fruit [7] and 1.66 mM for musk melon [2] but was lower than that of other fruits such as 5.16 mM for peach β- galactosidase [17]. The Vmax value for β-gal I, β-gal II and β-gal III was found to be 0.52, 0.70 and 0.38 µmol/min respectively. The optimum pH of different isozymes was found between 4 and 6 which were consistent with earlier reports [3,6,7,15]. Moreover, β-gal I and β-gal II showed maximum activity at pH 5.2 where
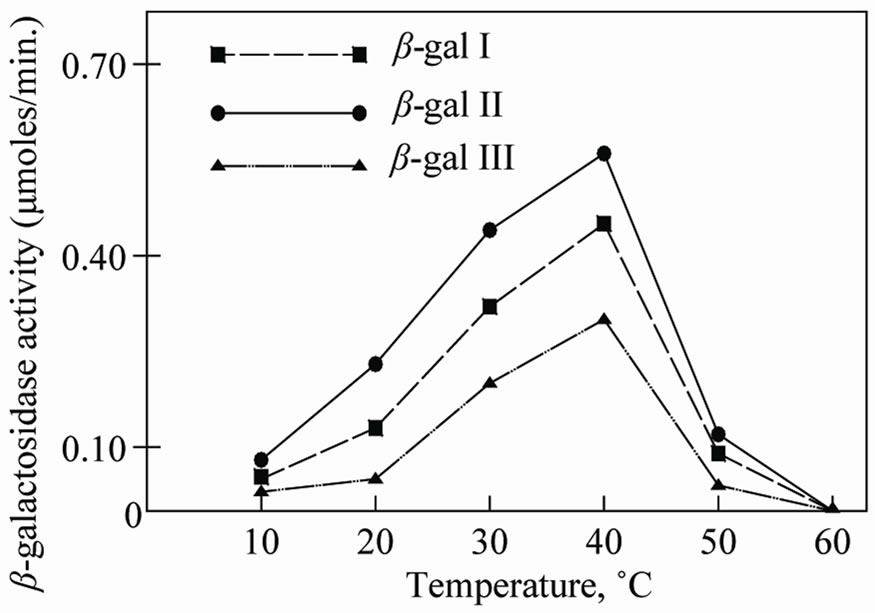
Figure 11. Temperature-activity profiles of different isoforms of β-galactosidase. The enzyme assays were performed by incubating reaction mixtures at various temperatures from 10˚C to 70˚C.
as β-gal III showed maximum activity at pH 4.2. These results indicate that the apricot β-galactosidases possesses a wide range of pH stability. The optimum temperature was found to be 40˚C. However the enzymes were active up to 50˚C but were completely inactivated at 60˚C. These results are in close agreement with that obtained from other fruits [16,17].
The above properties of β-galactosidase isoforms suggest that enzymes may be variably adaptable to temperature and pH fluctuations which are associated with physiological changes that are caused by the fruit softening. Earlier reports [16-18,21-23] and the results presented here indicate that the β-galactosidases may be involved in the modification of the cell wall structural polysaccharides of apricot fruits, suggesting its important role in softening. A detailed analysis of the cell wall structure of apricot will provide a better picture of the exact functions of different β-galactosidase isoforms in cell wall degradation of apricot.
REFERENCES
- J. Gray, S. Picton, J. Shabbeer, W. Schuch and D. Grierson, “Molecular Biology of Fruit Ripening and Its Manipulation with Antisense Genes,” Plant Molecualr Biology, Vol. 19, No. 1, 1992, pp. 69-87. doi:10.1007/BF00015607
- A. P. Ranwala, C. Suematsu and H. Masuda, “The Role of β-Galactosidases in the Modification of Cell Wall Components during Muskmelon Fruit Ripening,” Plant Physiology, Vol. 100, No. 3, 1992, pp. 1318-1325. doi:10.1104/pp.100.3.1318
- C. S. Ross, S. Cavin, T. Wegrzyn, E. A. MacRae and R. J. Redgwell, “Apple β-Galactosidase Activity against Cell Wall Polysaccharides and Characterization of a Related cDNA Clone,” Plant Physiology, Vol. 106, No. 2, 1994, pp. 521-528. doi:10.1104/pp.106.2.521
- D. A. Brummell and M. H. Harpster, “Cell Wall Metabolism in Fruit Softening and Quality and Its Manipulation in Transgenic Plants,” Plant Molecualr Biology, Vol. 47, No. 1-2, 2001, pp. 311-340. doi:10.1023/A:1010656104304
- E. J. DeVeau, K. C. Gross, D. J. Huber and A. E. Watada, “Degradation and Solubilization of Pectin by β-Galactosidases Purified from Avocado Mesocarp,” Physiologia Plantarum, Vol. 87, 1993, pp. 279-285
- A. T. Carey, K. Holt, S. Picard, R. Wilde, G. A. Tucker, C. R. Bird, W. Schuch and G. R. Seymour, “Tomato Exo- 1 → 4-β-D-Galactanase: Isolation, Changes during Ripening in Normal and Mutant Tomato Fruit and Characterization of a Related cDNA Clone,” Plant Physiology, Vol. 108, No. 3, 1995, pp. 1099-1107. doi:10.1104/pp.108.3.1099
- C. M. Carrington and R. Pressey, “β-Galactosidase II Activity in Relation to Changes in Cell Wall Galactosyl Composition during Tomato Ripening,” Journal of the American Society for Horticultural Science, Vol. 121, 1996, pp. 132-136.
- Y. Hirano, Y. Tsumuraya and Y. Hashimoto, “Characterization of Spinach leaf α-L-Arabinofuranosidases and β-Galactosidases and Their Synergistic Action on an Endogenous Arabinogalactan-Protein,” Physiologia Plantarum, Vol. 92, No. 2, 1994, pp. 286-296. doi:10.1111/j.1399-3054.1994.tb05339.x
- S. C. Li, J. W. Han, K. C. Chen and C. S. Chen, “Purification and Characterization of Isoforms of β-Galactosidases in Mung Bean Seedlings,” Phytochemistry, Vol. 57, No. 3, 2001, pp. 349-359. doi:10.1016/S0031-9422(01)00022-X
- T. Kotake, S. Dina, T. Konishi, S. Kaneko, K. Igarashi, et al., “Molecular Cloning of a β-Galactosidase from Radish That Specifically Hydrolyzes β-1 → 3- and β-1 → 6-Galactosyl Residues of Arabinogalactan Protein,” Plant Physiology, Vol. 138, No. 3, 2005, pp. 1563-1576. doi:10.1104/pp.105.062562
- P. Hrubá, D. Honys, D. Twell, V. Capková and J. Tupy, “Expression of β-Galactosidase and β-Xylosidase Genes during Microspore and Pollen Development,” Planta, Vol. 220, No. 6, 2005, pp. 931-940. doi:10.1007/s00425-004-1409-0
- A. Wu and J. Liu, “Isolation of the Promoter of a Cotton Beta-Galactosidase Gene GhGal1 and Its Expression in Transgenic Tobacco Plants,” Science in China Series C: Life Sciences, Vol. 49, No. 2, 2006, pp. 105-114. doi:10.1007/s11427-006-0105-7
- K. D. Golden, M. A. John and E. A. Kean, “β-Galactosidase from Coffea arabica and Its Role in Fruit Ripening,” Phytochemistry, Vol. 34, No. 2, 1993, pp. 355-360. doi:10.1016/0031-9422(93)80008-G
- I. K. Kang, S. G. Suh, K. C. Gross and J. K. Byun, “N-Terminal Amino Acid Sequence of Persimmon Fruit β-Galactosidase,” Plant Physiology, Vol. 105, No. 3, 1994, pp. 975-979. doi:10.1104/pp.105.3.975
- P. K. Andrews and S. Li, “Partial Purification and Characterization of β-D-Galactosidase from Sweet Cherry, a Non-Climacteric Fruit,” Journal of Agricultural and Food Chemistry, Vol. 42, No. 10, 1994, pp. 2177-2182. doi:10.1021/jf00046a019
- Z. M. Ali, S. Armugam and H. Lazan, “β-Galactosidase and Its Significance in Ripening Mango Fruit,” Phytochemistry, Vol. 38, No. 5, 1995, pp. 1109-1114. doi:10.1016/0031-9422(94)00804-3
- D. H. Lee, S.-G. Kang, S.-G. Suh and J. K. Byun, “Purification and Characterization of a β-Galactosidase from Peach Prunus persica,” Molecules and Cells, Vol. 15, 2003, pp. 68-74.
- H. Lazan, S. Y. Ng, L. Y. Goh And Z. M. Ali, “Papaya beta-Galactosidase/Galactanase Isoforms in Differential Cell Wall Hydrolysis and Fruit Softening during Ripening,” Plant Physiology and Biochemistry, Vol. 42, No. 11, 2004, pp. 847-853. doi:10.1016/j.plaphy.2004.10.007
- G. S. Ross, E. A. MacRae and R. J. Redgwell, “Isolation and Activity against Specific Fruit Cell-Wall Polysaccharides,” Planta, Vol. 189, No. 4, 1993, pp. 499-506. doi:10.1007/BF00198212
- D. L. Smith, D. A. Starrett and K. C. Gross, “A Gene Coding for Tomato Fruit β-Galactosidase II Is Expressed during Fruit Ripening Cloning, Characterization and Expression Pattern,” Plant Physiology, Vol. 117, No. 2, 1998, pp. 417-423. doi:10.1104/pp.117.2.417
- D. L. Smith and K. C. Gross, “A Family of at Least Seven Beta-Galactosidase Genes Is Expressed during Tomato Fruit Development,” Plant Physiology, Vol. 123, No. 3, 2000, pp. 1173-1183. doi:10.1104/pp.123.3.1173
- L. Trainotti, R. Spinello, A. Piovan, S. P. Spolaore and G. Casadoro, “β-Galactosidases with a Lectin-Like Domain Are Expressed in Strawberry,” Journal of Experimental Botany, Vol. 52, No. 361, 2001, pp. 1635-1645. doi:10.1093/jexbot/52.361.1635
- Z. Wu and J. K. Burns, “A β-Galactosidase Gene Is Expressed during Mature Fruit Abscission of ‘Valencia’ Orange Citrus sinensis,” Journal of Experimental Botany, Vol. 55, No. 402, 2004, pp. 1483-1490. doi:10.1093/jxb/erh163
- J. Zhuang, J. Su, X. Li and W. Chen, “Cloning and Expression Analysis of β-Galactosidase Gene Related to Softening of Banana Musa sp. Fruit,” Journal of Plant Physiology and Molecular Biology, Vol. 32, No. 4, 2006, pp. 411-419.
- O. H. Lowry, N. J. Rosebrough, A. L. Farr and R. J. Randall, “Protein Measurement with Folin Phenol Reagent,” Journal of Biological Chemistry, Vol. 193, 1951, pp. 265-275.
- R. Pressey, “β-Galactosidase in Ripening Tomatoes,” Plant Physiology, Vol. 71, No. 1, 1983, pp. 132-135. doi:10.1104/pp.71.1.132
NOTES
*Corresponding author.

