Journal of Geoscience and Environment Protection
Vol.04 No.01(2016), Article ID:62725,7 pages
10.4236/gep.2016.41011
Microbial Community from MTBE-Contaminated Soil for Aerobic Biodegradation of MTBE
Bahareh Montazeri, Mohammad Hossein Sarrafzadeh*
School of Chemical Engineering, College of Engineering, University of Tehran, Tehran, Iran

Copyright © 2016 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 21 October 2014; accepted 10 January 2016; published 13 January 2016
ABSTRACT
This Methyl tert-butyl ether (MTBE) is one of the main additives in gasoline to increase octane rating and consequently reduce air pollution. The physico-chemical properties of this substance (high water solubility, low sorption in soil) result in high mobility and considerable concentrations in aquifers. In this survey, Isfehan Refinery that was encountered with MTBE contamination problem was selected as a case study and the MTBE degradation ability of this contaminated area by its indigenous microorganisms was investigated. In the first step of this survey, the influence of various factors on the aerobic degradation of MTBE such as mixed culture type, incubation time, microbial culture and optimal concentration of MTBE were investigated in shaking flasks and the most important factors were specified by means of fractional factorial design ½. In the second stage by using optimal values which obtained from the first stage, the effects of co-substare parameter and inoculum parameter were assayed by means of response surface method. The results of the experiments showed that the mixed culture type and initial concentration of MTBE were the most significant factors. The results of the experiments showed that the mixed indigenous culture acted better than activated sludge. The initial concentration of MTBE was also one of the most significant factors. At the best condition about 31 percent of MTBE was treated by co-substrating with n-hexane in a ratio of 0.2.
Keywords:
Bioremediation, MTBE Contamination, Groundwater, Cometabolism, Indigenous Microorganisms

1. Introduction
Methyl tert-butyl ether (MTBE), a synthetic compound produced from methanol and isobutylene, has been used as a gasoline additive to increase octane ratings instead of lead-based and aromatic compounds since the late 1970s. For the last 15 years, it has been added in higher concentrations (up to 15% (v/v)) to increase fuel combustion efficiency and to lower emissions of CO and other air pollutants [1] . The release of MTBE to the environment, mainly from damaged gasoline underground storage tanks or distribution systems spills, has provoked extended groundwater pollution [2] . Solubility of MTBE in water is approximately 50,000 mg/L at 25˚C and has a low organic carbon-based partitioning coefficient (KOC) of 11 cm3/g resulting in minimal sorption and retardation in natural aquifer [3] . MTBE has a higher vapor pressure than benzene, its Henry’s Law constant (0.022 at 25˚C) is 1/10th benzene. This results in minimal losses of MTBE from the aqueous phase to the atmosphere. Due to significant partitioning of MTBE into the aqueous phase and limited sorption to soil, MTBE plumes in polluted groundwater are generally longer and more stable than coexisting gasoline plumes [4] .
There are different technologies that have been applied to remediate groundwater polluted by MTBE and other gasoline compounds such as pump and treat, air sparging, multi-phase extraction, and in-situ chemical oxidation [5] - [7] . Traditional pump and treat technologies, including air stripping, used to eliminate gasoline components from groundwater have not been as successful for MTBE removal [4] . Bioremediation is an alternative to traditional technologies because it is considered to be cost-effective and energy efficient. Its main advantage is that it can completely mineralize MTBE to harmless products such as carbon dioxide, biomass, and water. Initial studies show that MTBE is highly recalcitrant (because of its structure containing both an ether link and a branched moiety) but recently some authors have reported that certain bacteria can degrade MTBE as their sole carbon and energy source. Depending on the microorganisms involved in the process, biodegradation is more frequently observed under aerobic conditions by pure or mixed cultures [5] . To date, the only bacterial strains that have been reported to completely consume MTBE as their sole source of carbon and energy are Methylibiumpetroleiphilum PM1, Hydrogenophagaflava ENV735, Mycobacterium austroafricanum IFP2012 and Aquincolatertiaricarbonis. A diverse group of bacterial strains such as Methylobacterium, Rhodococcus, Arthrobacter, Bacillus, Pseudomonas or an entrapped microbial consortium have been reported to partially degrade MTBE [8] -[11] .
Cometabolic MTBE biodegradation has received significant research attention. Cometabolism involves the use of an additional carbon source for growth and may have a role in natural attenuation. Some compounds present in gasoline have been reported for their ability to induce enzymes able to degrade MTBE [12] . Cometabolic MTBE degradation has been reported by C3-C7 n-alkanes [12] - [16] and generally, aromatic compounds do not support MTBE degradation [12] . Bacteria reported to degrade MTBE by cometabolism include Nocardia [14] , Mycobacterium [17] , Rhodococcus [18] and Pseudomonas [12] .
In this study, the aerobic MTBE degrading capacity of the indigenous microbial community in soil from gasoline contaminated sites in Isfehan Refinery, is examined in batch experiments simulating different in-situ conditions.
2. Material and Methods
2.1. Soil Sample and Enrichment Culture
Soil samples were collected at 5 - 20 cm depth from MTBE contaminated ground surface of Isfehan Refinery. These samples were used for reaching the indigenous microorganisms. Ten grams of soil sample were dissolved in 100 ml sterilized water and shaken at 150 rpm and 30˚C for 3 hrs and then left standing for 30 mins. The supernatant 10 ml was then incubated into 200 ml modified M9 as growth medium with the following formulation: Na2HPO4, 6.0 g/l; KH2PO4, 3.0 g/l; NaCl, 0.5 g/l; NH4Cl, 1.0 g/l; MgSO4.7H2O 1.0 M, 2.0 ml/l; CaCl2 1.0 M, 0.1 ml/l (all chemicals were purchased from Merck). MTBE as the sole carbon and energy source was added to a final concentration of 200 ppm and the mixture was shaken in a 500 ml flask for 24 h at 150 rpm and 30˚C. Subsequently, 20 ml of the inocula (supernatants of the above cultures) was transferred to another 500 ml flask containing 200 ml of modified fresh M9 medium supplemented with MTBE (300 ppm) and was incubated for another 24 hrs under the same conditions. This procedure was repeated over 10 days until the concentration of MTBE reached to 1000 ppm in the last fresh modified M9 medium. After the final transfer, 100 µl of the culture was spread on Nutrient agar medium plates and incubated for 24 hrs at 30˚C. Nutrient agar medium plates were maintained at 4˚C.
2.2. Batch Scale Study
In this part of study, feasibility of MTBE bioremediation was assessed by a combined microbial culture that was separated from MTBE contaminated soil. To evaluate this method’s applicability, a series of experiments based on design of experiments by Minitab software were done in which superficial MTBE contaminated water samples were used for preparing the mixed bacterial culture. Experiments were conducted to examine the feasibility of MTBE biodegradation under aerobic and aerobic-cometabolic condition. Experiments were conducted in two stages. At first stage, the effects of mixed culture type, initial concentration of MTBE, incubation time and culture medium type were investigated. All experiments were carried out in sealed 250-mL Erlenmeyer flasks. The flasks contained 90 mL of a culture medium type (M9 or nutrient broth) and MTBE. Based on design table (see Table 1) 10 ml of isolated microorganisms from contaminated soil with the optical density adjusted to 1 at 600 nm by sterile normal saline (0.89% NaCl) and municipal activated sludge with MLSS of 12 g/l with the optical density adjusted to 1 at 600 nm by sterile normal saline, were added to the flasks as inoculums. For each flask, one flask was prepared as abiotic control which every control contains 250 mg/l HgCl2 and 500 mg/l NaN3 [19] . Abiotic controls for each flask were prepared based on Table 1. Flasks were incubated at 25˚C and 150 rpm for 24 and 120 hours based on Table 1. The volume of the flask headspace was sufficient to prevent any limitation to O2 [20] . The pH of media was adjusted to 7.2 using 0.1 N NaOH.
At the second stage (after analyzing the results of stage 1) the effects of inoculum size and the presence of co substrate factors were investigated based on Table 2. Indigenous microorganisms of the region were used at this stage. Initial concentration of MTBE was 200 ppm at M9 salt broth. All the conditions of experiments were the same as stage 1. Incubation time in shaking flask was considered 48 hours.
2.3. Design of Experiments
Minitab 16 was used for designing of experiments and all of statistical analysis. The method for the first stage designing was fractional factorial ½ with resolution of ΙV and for second stage was response surface method (Central Composite Design). ANOVA was used to determine the significance of regression and factors. ANOVA was also used to assess differences between appropriates measured parameters. The first stage design table and the second stage design table are given in Table 1 and Table 2 respectively.
2.4. Analysis of MTBE
MTBE in groundwater are measured using headspace solvent micro extraction (HSME) method, with gas chromatography-flame ionization (GC-FID). HSME is a novel method of sample preparation for chromatography
Table 1. Design of experiments based on fractional factorial ½ method.
Table 2. Design of experiments based on response surface method.
analysis. This system involves a microdrop of a high boiling point organic solvent (n-decane in this study) extruded from the needle tip of gas chromatographic syringe, which is exposed to the headspace above a sample. Volatile organic compounds (MTBE in this study), are extracted and concentrated in the microdrop. Then, the microdrop is retracted in the microsyringe and injected directly into a chromatograph. The high volatility and low polarity of MTBE lead the compound to have a fast diffusion to headspace and an enhanced distribution into the microdrop, respectively. Theses characteristic leads to study the determination of MTBE in water samples by a HSME method using a GC-FID equipment [21] . The GC-FID analysis was performed using a Varian CP 300 gas chromatography equipped with a flame ionization detector. The injector and detector temperatures were 210˚C and 230˚C, respectively. A constant flow (1 ml/min) of Helium was used as carrier gas. The analysis was performed with an initial column temperature 35˚C held for 5 min followed by heating to 150˚C at 25˚C/min. MTBE eluted in 3.7 mins. All quantifications made in this study were based on the relative peak area of MTBE to the internal standard (1000 ppm chloroform in methanol) from the average of three replicate measurements.
2.5. MTBE Biodegradation Determination
The control samples were prepared for considering MTBE removal by abiotic processes. The percentages of biotic and abiotic removal of MTBE (R%) was obtained by the following equations:
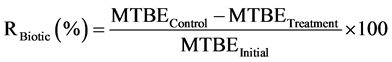 (1)
(1)
 (2)
(2)
3. Result and Discussion
Influences of Factors and Analysis of Variance
The results of stage one showed that indigenous microorganisms of the region could biodegrade MTBE effectively comparing to activated sludge. Based on the analysis of Minitab software, it was found that out of four main factors, two of them including culture type and initial concentration of MTBE had significant effects on biodegradation of MTBE with p-value < 0.05. In contrast, incubation time and culture medium factors had insignificant effect on MTBE degradation with p-value > 0.05. MTBE disappearance in the all controls was negligible.
Result showed that the indigenous microorganisms capability in MTBE degradation, as predicted, were higher compared to activated sludge. This phenomenon was probably interpreted in order of indigenous microorganisms acclimation with MTBE. According to data from experiments number 1 and 2 and also experiments number 3 and 4 and considering culture medium factor as an insignificant factor (p-value of media culture > 0.05), MTBE degradation percent is decreased when MTBE initial concentration is increased. Similar results are reported in [20] when the MTBE initial concentration is increased above 50 - 100 ppm, the MTBE degradation rate decreased.
Incubation time factor has no significant effect on MTBE degradation (p-value > 0.05). According to data from experiments number 1 and 3 and also 2 and 4 and considering culture medium factor as an insignificant factor, it is found that although MTBE degradation percent is increased with spreading time, this factor has insignificant effect on MTBE degradation percent practically. This could be ascribed to the presence of tert-butyl alcohol (TBA) as an intermediate in the most of MTBE-degrading culture and its inhibitory effect on MTBE degradation so that is suggested that a competition between two components occurred. It can be presumed that the same enzyme possibly responsible for MTBE and TBA metabolism and they may be competing for the same active site [20] . The TBA accumulation had a negative effect on the MTBE degradation rate in the beginning hour of experiment, the similar result was also reported about butane-oxidizing Arthrobacter (ATCC 27778) bacteria [15] .
According to the Minitab software, the medium culture has no significant effect on MTBE degradation. Although both M9 and NB can prepare Nitrogen and Phosphorous supply for microorganisms, MTBE degradation percent in M9, was higher comparison to NB. This can be intrepid by considering MTBE as the sole carbon source in M9.
Microorganism, MTBE initial concentration, incubation time and MTBE biological removal percent for similar studies are presented in Table 3.
According to results of stage two, the presence of cometabolite has significant effect on MTBE degradation (p-value < 0.05) and MTBE biological removal percent is increased. One potential reason is that some oxidizing enzymes during consumption of n-hexane are produced which promote MTBE removal efficiency. The result indicated that the highest MTBE degradation percent is achieved in MTBE to n-Hexane ratio (w/w) equal to 0.6.
The effect of inoculum size on MTBE biodegradation was tested. According to result of stage two, inoculum size factor had no significant effect on MTBE biodegradation (p-value > 0.05) and no difference was observed with the increasing of inoculum size. Although, inoculum size could play a role in MTBE removal soon after the inoculation, the degradation rate reaches a constant. This interpretation is consistent with findings of Zhang et al. [20] . Therefore, biological removal of MTBE could enhance with the decrease of MTBE to n-hexane ratio, while inoculum size had negligible impact on MTBE degradation.
4. Conclusion
The results of this study provide evidence of MTBE degradation by indigenous microorganisms. Aerobic microbial consortia is taken from the groundwater and soil of a contaminated site and enriched, to test whether they are capable of degrading MTBE as sole source of carbon. Current study shows that indigenous microbial populations are able to remove MTBE from aqueous samples. Furthermore, it emerges from the results of this study that when the initial concentration of MTBE is 200 ppm, biological removal of MTBE is enhanced. The results of design of experiments by response surface method show that using of n-hexane as the co metabolite provides a solution to overcome low biodegradation of MTBE.
Table 3. Aerobic MTBE degradation observed with different pure and mixed cultures of bacteria.
Acknowledgements
The authors would like to thank Mr. Damirchi and financial support of Isfahan Petroleum Refinery.
Cite this paper
BaharehMontazeri,Mohammad HosseinSarrafzadeh, (2016) Microbial Community from MTBE-Contaminated Soil for Aerobic Biodegradation of MTBE. Journal of Geoscience and Environment Protection,04,93-99. doi: 10.4236/gep.2016.41011
References
- 1. Eixarch, H. and Constanti, M. (2010) Biodegradation of MTBE by Achromobacter xylosoxidans MCM1/1 Induces Synthesis of Proteins That May Be Related to Cell Survival. Process Biochemistry, 45 794-798.
http://dx.doi.org/10.1016/j.procbio.2009.12.015 - 2. Nava, V., Morales, M. and Revah, S. (2007) Cometabolism of Methyl Tert-Butyl Ether (MTBE) with Alkanes. Reviews in Environmental Science and Biotechnology, 6, 339-352.
- 3. Martienssen, M., Fabritius, H., Kukla, S., Balcke, G.U., Hasselwander, E. and Schirmer, M. (2006) Determination of Naturally Occurring MTBE Biodegradation by Analyzing Metabolites and Biodegradation By-Products. Journal of Contaminant Hydrology, 87, 37-53.
http://dx.doi.org/10.1016/j.jconhyd.2006.04.007 - 4. Kern, E.A., Veeh, R.H., Langer, H.W., Macur, R.E. and Cunningham, A.B. (2002) Characterization of Methyl Tert-Butyl Ether-Degrading Bacteria from a Gasoline-Contaminated Aquifer. Bioremediation Journal, 6, 113-124.
http://dx.doi.org/10.1080/10588330208951208 - 5. Saponaro, S., Negri, M., Sezenna, E., Bonomo, L. and Sorlini, C. (2009) Groundwater Remediation by an in Situ Biobarrier: A Bench Scale Feasibility Test for Methyl Tert-Butyl Ether and Other Gasoline Compounds. Journal of Hazardous Materials, 167, 545-552.
http://dx.doi.org/10.1016/j.jhazmat.2009.01.026 - 6. Zargar, M., Sarrafzadeh, M., Taheri, B. and Keshavarz, A. (2014) Assessment of in-Situ Bioremediation of Oil Contaminated Soil and Groundwater in a Petroleum Refinery: A Laboratory Soil Column Study. Petroleum Science and Technology, 32, 1553-1561.
http://dx.doi.org/10.1080/10916466.2012.690482 - 7. Zargar, M., Sarrafzadeh, M. and Taheri, B. (2013) The Surveying of Soil and Groundwater Pollution in Petroleum Refinery and Potential of Bioremediation for Oil Decontamination. Petroleum Science and Technology, 31, 2585-2595.
http://dx.doi.org/10.1080/10916466.2011.559507 - 8. Nakatsu, C.H., Hristova, K., Hanada, S., Meng, X.Y., Hanson, J.R., Scow, K.M., et al. (2006) Methylibium petroleiphilum Gen. Nov., sp. Nov., a Novel Methyl Tert-Butyl Ether-Degrading Methylotroph of the Betaproteobacteria. International Journal of Systematic and Evolutionary Microbiology, 56, 983-989.
http://dx.doi.org/10.1099/ijs.0.63524-0 - 9. Streger, S.H., Vainberg, S., Dong, H.L. and Hatzinger, P.B. (2002) Enhancing Transport of Hydrogenophaga flava ENV735 for Bioaugmentation of Aquifers Contaminated with Methyl Tert-Butyl Ether. Applied and Environmental Microbiology, 68, 5571-5579.
- 10. Francois, A., Mathis, H., Godefroy, D., Piveteau, P., Fayolle, F. and Monot, F. (2002) Biodegradation of Methyl Tert-Butyl Ether and Other Fuel Oxygenates by a New Strain. Mycobacterium austroafricanum IFP 2012. Applied and Environmental Microbiology, 68, 2754-2762.
- 11. Muller, R.H., Rohwerder, T. and Harms, H. (2008) Degradation of Fuel Oxygenates and Their Main Intermediates by Aquincola tertiaricarbonis L108. Microbiology, 154, 1414-1421.
http://dx.doi.org/10.1099/mic.0.2007/014159-0 - 12. Morales, M., Nava, V., Velasquez, E., Razo-Flores, E. and Revah, S. (2009) Mineralization of Methyl tert-Butyl Ether and Other Gasoline Oxygenates by Pseudomonads Using Short n-Alkanes as Growth Source. Biodegradation, 20, 271-280.
http://dx.doi.org/10.1007/s10532-008-9219-x - 13. Hardison, L.K., Curry, S.S., Ciuffetti, L.M. and Hyman, M.R. (1997) Metabolism of Diethyl Ether and Cometabolism of Methyl tert-Butyl Ether by a Filamentous Fungus, a Graphium sp. Applied and Environmental Microbiology, 63, 3059-3067.
- 14. Steffan, R.J., McClay, K., Vainberg, S., Condee, C.W. and Zhang, D. (1997) Biodegradation of the Gasoline Oxygenates Methyl tert-Butyl Ether, Ethyl tert-Butyl Ether and tert-Amyl Methyl Ether by Propane-Oxidizing Bacteria. Applied and Environmental Microbiology, 63, 4216-4222.
- 15. Liu, C.Y., Speitel, G.E.J. and Georgiou, G. (2001) Kinetics of Methyl t-Butyl Ether Cometabolism at Low Concentrations by Pure Cultures of Butane-Degrading Bacteria. Applied and Environmental Microbiology, 67, 2197-2201.
http://dx.doi.org/10.1128/AEM.67.5.2197-2201.2001 - 16. Garnier, P., Auria, R., Augur, C. and Revah, S. (1999) Cometabolic Biodegradation of Methyl t-Butyl Ether by Pseudomonas aeruginosa Grown on Pentane. Applied Microbiology and Biotechnology, 51, 498-503.
http://dx.doi.org/10.1007/s002530051423 - 17. Smith, C.A., O’Reilly, K.T. and Hyman, M.R. (2003) Characterization of the Initial Reactions during the Cometabolic Degradation of Methyl tert-Butyl Ether (MTBE) by Propane-Grown Mycobacterium vaccae JOB5. Applied and Environmental Microbiology, 69, 796-804.
- 18. Haase, K., Wendlandt, K.D., Graber, A. and Stottmeister, U. (2006) Cometabolic Degradation of MTBE Using Methane-Propane- and Butane-Utilizing Enrichment Cultures and Rhodococcus sp. BU3. Engineering in Life Sciences, 6, 508-513.
http://dx.doi.org/10.1002/elsc.200520144 - 19. Chen, K.F., Kao, C.M., Chen, T.Y., Weng, C.H. and Tasi, C.T. (2006) Intrinsic Bioremediation of MTBE Contaminated Groundwater at Petroleum-Hydrocarbon Spill Site. Environmental Geology, 50, 439-445.
http://dx.doi.org/10.1007/s00254-006-0227-z - 20. Zhang, R.L., Huang, G.Q., Lian, J.Y. and Li, X.G. (2007) Degradation of MTBE and TBA by a New Isolate from MTBE-Contaminated Soil. Journal of Environmental Science, 19, 1120-1124.
- 21. Bahramifar, N., yamini, Y., Shariati, S. and Shamsipur, M. (2004) Trace Analysis of Methyl tert-Butyl Ether in Water Samples Using Headspace Solvent Microextraction and Gas Chromatography-Flame Ionization Detection. Journal of Chromatography A, 1024, 211-217.
http://dx.doi.org/10.1016/j.chroma.2004.05.033 - 22. Munoz-Castellanos, L.N., Torres-Munoz, J.V., Keer-Rendon, A., Manzanares-Papayanopoulos, L.I. and Nevarez-Moorillon, G.V. (2006) Aerobic Biodegradation of Methyl tert-Butyl Ether (MTBE) by Pure Bacteria Culture Isolated from Contaminated Soil. World Journal of Microbiology and Biotechnology, 22, 851-855.
http://dx.doi.org/10.1007/s11274-005-9114-0
NOTES

*Corresponding author.


