Open Journal of Anesthesiology
Vol.2 No.5(2012), Article ID:24823,3 pages DOI:10.4236/ojanes.2012.25055
Late Thrombolysis in Massive Pulmonary Thromboembolism
![]()
Unidad Anestesia y Reanimación, Hospital Universitario Fundación Alcorcón, Alcorcon, Spain.
Email: crmolina@fhalcorcon.es
Received September 16th, 2012; revised October 18th, 2012; accepted October 27th, 2012
Keywords: Pulmonary Thromboembolism; Thrombolysis; Pulmonary Hypertension
ABSTRACT
Pulmonary thromboembolism (PTE) remains a diagnostic and therapeutic challenge for physicians. Anticoagulation with heparin remains as the cornerstone in its management, reserving thrombolysis for cases with hemodynamic impairment. The later has been associated with haemorrhagic complications and has proved beneficial when initiated within the first 48 hours of PTE; but there is little evidence supporting its use passed this time. We present a case of an 84 year old female admitted in our unit and treated successfully with thrombolysis for massive PTE at the fifth day of diagnosis. The patient improved towards complete recovery and was discharged from the hospital 22 days after admission without complications.
1. Introduction
Thrombolytic therapy is usually reserved for patients with clinically serious or massive pulmonary thromboembolism (PTE) [1]. However, evidence supporting thrombolysis introduction beyond 48 hours of massive PTE is scarce.
2. Case Report
An 84 year old female was admitted to the emergency room with an abrupt onset of dyspnoea, three days after a six hours bus ride. Her shortness of breath had worsened in the last 48 hours. She suffered from hypertension, asthma, moderate mitral incompetence and was heterozygous for prothrombin gene 2021A which was diagnosed after a PTE seven years earlier. She was anticoagulated for six months after that event. Neither the patient nor her file provided data of the development of pulmonary hypertension during or after this episode of PTE. The physical examination disclosed no signs of cyanosis or use of accessory respiratory muscles with 24 breaths per minute and oxygen saturation of 93%. Pulmonary auscultation revealed global hypoventilation and rales on the right pulmonary base. There were no signs of deep venous thrombosis. The chest x-rays pro-vided no positive information and the electrocardiogram revealed normal sinus rhythm of 90 beats per minute and a previously known right bundle branch block. The study of arterial blood gases without supplemental oxygen showed pH 7.45, PCO2 37.4 mmHg, PO2 48.3 mmHg, Bicarbonate 25.6 mmol/L and the levels of troponin I were 0.19 ng/ml and D-Dimer 9894 ng/ml. Other laboratory values were within normal limits.
A Computed Tomography Pulmonary Angiogram (CTPA) showed multiple repletion defects in the main pulmonary arteries and segmentary branches of all lobes (Figure 1). With the diagnosis of massive PTE, Enoxaparin (60 mg/12 hours) was initiated.
She complained of acute pain and burning sensation in her lower limbs one day after admission. An abdominal AngioCT showed massive embolic occlusion of both iliac arteries.
To rule out a paradoxical embolism, a transthoracic echocardiogram was performed revealing left ventricular hypertrophy with good systolic function, ejection fraction of 66%, and a dilated right ventricle with severe systolic dysfunction. The inter-ventricular septum was displaced to the left due to pressure overload in the right chambers. TAPSE was estimated in 15 mm. There was evidence of mild mitral and tricuspid insufficiency and severe Pulmonary Hypertension (PHT), with an estimated Pulmonary Artery Pressure of 86 mmHg. There were no findings of cardiac thrombi or inter-auricular communication.
A successful revascularization was accomplished through bilateral femoral embolectomy under general anaesthesia. Successful extubation was accomplished and

Figure 1. Multiple repletion defects in principal pulmonary arteries and the segmentary branches of all the lobes, confirming Massive PTE.
the patient remained stable without the need of respiratory or Vasoactive Drug support. Anticoagulation was achieved with Non Fractioned Heparin (NFH) infusion with a target of 3 times the Activated Throboplastin Time (APTT).
The patient remained stable in the immediate postoperative period, until her fifth day of admission, when she presented with hypotension refractory to fluid therapy and Oxygen saturation below 85% despite high inspired oxygen concrentration. Orotracheal intubation for mechanical ventilation and infusion of dobutamine (10.5 µg/kg/min) and norepinephrine (0.14 µg/kg/min) were needed. A new CTPA and echocardiogram revealed no changes. Due to hemodynamic instability and the life threatening condition, thrombolysis with 100mg of Recombinant Tissue Plasminogen Activator (rTPA) was administered together with the continuous infusion of NFH. Over the next 48 hours respiratory function and hemodynamic state improved, allowing lower doses of vasoactive drugs. There were no hemorrhagic complications secondary to thrombolysis. Nine days after admission successful weaning was accomplished.
On the twelfth day of admission notable clinical, respiratory and hemodynamic improvement was accomplished and oral tolerance was initiated. A CTPA (Figure 2) showed a significant improvement as compared with the previous ones. She was discharged home in good clinical condition after 22 days of admission.
3. Discussion
The initial clinical presentation was a massive PTE with no hemodynamic repercussion and echocardiographic evidence of right ventricular dysfunction, PHT (which we assumed as new onset as we had no evidence of development in the previous episode of PTE or chronicity) and elevation of Troponin I. In this stage most guidelines encourage the prompt initiation of antico-
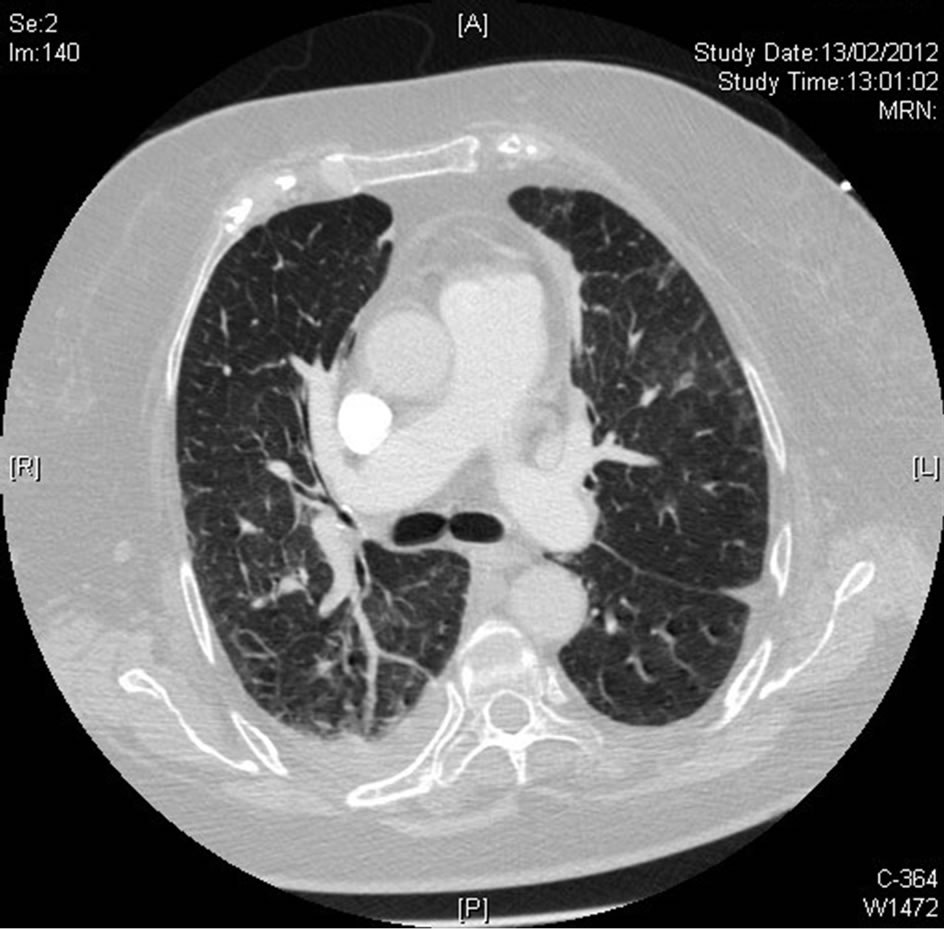
Figure 2. Significant improvement and revascularization compared with previous ACTP.
agulation with heparin and discourage (as a Grade 1C recommendation) the use of thrombolysis unless hemodynamic impairment, making little or no difference with the treatment of PTE with no PHT and right ventricular dysfunction [1,2]. Despite early thrombolysis might have been an option, the lack of hemodynamic compromise was the main criteria followed in our unit not to introduce thrombolysis at this stage. Although we are certain this conduct might be controversial, strict application of the guidelines do discourage thrombolysis at this point and leaves its application to the entire discretion of the practitioner only if two conditions are present: vital risk and low haemorrhagic risk. This final statement considered as a Grade 2C recommendation in the last guidelines published by the American College of Chest Physicians [1-3].
As the case reached day five, hemodynamic compromise was added to our prior scenario. Guidelines do favour thrombolysis under these circumstances. However, the use of thrombolysis could be also considered controversial, not only due to the haemorrhagic risks in a perioperative scenario, but also because of evidence supporting its use only during the first 48 hours [1,2]. Given the extreme vital risk in our patient and available evidence recommending not delaying thrombolysis if cardiogenic shock is present [1,3], the decision to administer rTPA was taken.
There is scarce available clinical evidence supporting that the use of thrombolysis in PTE beyond 48 hours can be beneficial in the clinical state and survival rates of these patients [3,4]. This might be explained by the difficulty of building appropriate clinical trials due to the high mortality in patients with hemodynamic compromise, associated haemorrhagic risks and the ethical implications of holding the treatment beyond 48 hours in a hypothetical control group.
This case also raises some queries about the hypothetical benefit of starting thrombolysis when having similar echocardiographic findings (PHT, right ventricular dysfunction) and before the onset of hemodynamic compromise. Although we do not pretend to encourage its use before any sign of cardiogenic shock, our case do serves as an example of the natural history of PTE and that hemodynamic alterations might occur even under appropriate anticoagulation therapy.
4. Conclusion
The use of thrombolysis before, and to some practitioners even during, hemodynamic impairment in PTE is still controversial, as there are not enough randomized controlled trials to pronounce on its ultimate contribution compared with anticoagulation alone [3]. We therefore believe that it might be considered as a therapeutic measure with not such a restrictive fashion and under appropriate risk stratification of our patients. We are also aware that we cannot generalize any of our conclusions, but according to the guidelines [1], thrombolysis should not be delayed in the presence of shock; so it should be considered as an alternative given high vital risk, and after comparing potential risks and benefits in PTE with more than 48 hours of evolution. Based on this, our final statement would be that although judicious with its use, potential benefits of thrombolysis must be always kept in mind in the presence of massive PTE.
REFERENCES
- C. Kearon, E. A. Akl, A. J. Comerota, et al., “Antithrombotic Therapy for Vte Disease. Antithrombotic Therapy and Prevention of Thrombosis, 9th Edition, American College of Chest Physicians Evidence-Based Clinical Practice Guidelines,” Chest, Vol. 141, Suppl. 2, 2012, pp. e419S-e494S.
- K. E. Wood, “Major Pulmonary Embolism. Review of a Pathophysiologic Approach to the Golden Hour on Hemodynamically Significant Pulmonary Embolism,” Chest, Vol. 121, No. 3, 2002, pp. 877-905. doi:10.1378/chest.121.3.877
- J. L. Todd and V. F. Tapson, “Thrombolytic Therapy for Acute Pulmonary Embolism: A Critical Appraisal,” Chest, Vol. 135, No. 5, 2009, pp. 1321-1329. doi:10.1378/chest.08-2125
- L. Daniels, A. Parker, S. R. Patel, F. Grodstein and S. E. Goldhaber, “Relation of Duration of Symptoms with Response to Thrombolytic Therapy in Pulmonary Embolism,” American Journal of Cardiology, Vol. 80, No. 2, 1997, pp. 184-188. doi:10.1016/S0002-9149(97)00315-9

