World Journal of Cardiovascular Diseases
Vol.3 No.1A(2013), Article ID:29341,8 pages DOI:10.4236/wjcd.2013.31A025
Comparative study of quality of life after hospital-at-home or in-patient admission for acute decompensation of chronic heart failure*
![]()
1Hospital-at-Home Service, Araba University Hospital, Vitoria, Spain
2Araba Research Unit, Araba University Hospital, Vitoria, Spain
3Department of Cardiology, Araba University Hospital, Vitoria, Spain
4Department of Internal Medicine, Nuestra Señora de Sonsoles Hospital, Ávila, Spain
5Emergency Department, Araba University Hospital, Vitoria, Spain
Email: #argayosil@hotmail.com
Received 18 January 2013; revised 15 February 2013; accepted 20 March 2013
Keywords: Heart Failure; Health-Related Quality of Life; Hospital at Home
ABSTRACT
AIMS: Hospitalisation at home (HaH) achieves comparable health outcomes to conventional in-patient care, in terms of cost-effectiveness and quality of life. Our objective was to assess the impact of episodes of acute decompensation of heart failure (ADCHF) on functional status and quality of life, in patients under HaH care compared with a matched group receiving conventional in-patient care at a Cardiology ward (CW). Methods: Randomised clinical trial in 71 patients with ADCHF attending the Emergency Department. Patients were either admitted to the hospital’s CW or to the HaH service. The functional status and health related quality of life (HRQOL) were assessed using the Barthel Index (BI) and the EQ-5D, Short Form-36 (SF-36) and Minnessota Living with Heart Failure (MLHFQ) questionnaires, administered at admission and, depending on the test, at discharge and 1, 3, 6 and 12 months after discharge. Results: MLHFQ and SF-36 experienced significant improvement in most domains with respect to the initial values at discharge and 6 months after only in the HaH group; though by 12-months there were no significant differences. Barthel Index and EQ-5D value scores showed improvement with respect to initial values in both arms. The improvement could be appreciated in more components and for longer among the HaH patients. Conclusions: Functional independence and quality of life improve at discharge compared to admission, after both home-based and in-patient hospitalisation, outcomes being better in various respects over the first year of follow-up after hospital-at-home care.
1. INTRODUCTION
Heart failure (HF) is one of the main health problems today in Western countries, given its high prevalence, with associated morbidity and mortality, increasing with age. In addition, it is one of the chronic diseases with the greatest impact on health-related quality of life (HRQOL) [1]. Research into HRQOL in patients with this disease is for this reason considered very important and health interventions focused on the management of HF, should include HRQOL in the outcome measures, in addition to clinical indicators such as mortality or frequency of decompensation. Indeed, many patients with advanced heart failure rate quality of life as more important than the length of survival [2].
The main objective of hospital at home (HaH) services is to reduce hospitalisations, by delivering in the home setting a care of a comparable intensity and complexity to that provided to inpatients while achieving equivalent clinical outcomes. Theoretically, this translates to reductions in healthcare costs, lower rates of hospital-related complications (nosocomial infections, thrombotic diseases and delirium) and increased comfort for patients and their families [3,4]. Several publications have reported the treatment of decompensated HF by HaH teams involving various types of interventions. The key results include: a reduction in the frequency of decompensation in the period following HaH care, compared to previous months [5]; comparable clinical effectiveness to conventional hospitalisation [6-9]; and lower direct healthcare costs [7-9]. Some authors have assessed HRQOL and found little or no difference between home-based and conventional hospitalisation [6-9].
On the other hand, two recent studies [10,11] have revealed how the perception of HRQOL changes over the 6 to 12 months following in-hospital admission for ADCHF. In both cases, there was an initial improvement between admission and the first month of follow-up, and the level then remained stable.
The objective of the present study was to analyse the impact of acute decompensation of chronic HF requiring hospitalisation on functional status and perceived HRQOL, comparing the effect in two groups of patients: those referred directly from the Emergency Department (ED) to the HaH and those transferred from the ED to the Cardiology Ward for conventional inpatient care, with a 12-month follow-up after the decompensation.
2. METHODS
2.1. Design
This was a randomised controlled trial conducted in Alava-Araba University Hospital, Vitoria-Gasteiz, Spain. The results of this trial related to the cardiovascular events have already been published [9].
2.2. Patients
From May 2006 to March 2007, we identified 80 patients of 65 years of age or older with a history of heart failure of at least one year, an aetiological diagnosis and known prognosis, NYHA Class II or III, attended in the Emergency Department and requiring admission for decompensation. Patients who met any of the following criteria were excluded: they had been admitted in the previous two months for decompensated HF or for acute coronary syndrome; showed indicators of severity such as sudden deterioration compared to baseline, or factors associated with a poor prognosis (haemodynamic instability, severe arrhythmias, baseline creatinine levels of over 2.5 mg/dl); failed to respond to the treatment provided in the Emergency Department; had active cancer or pulmonary tuberculosis, current alcohol abuse, acute psychiatric illness, severe dementia or any other disease in an advanced stage with a life expectancy of less than six months; or were psychogeriatric facilities residents as well as if there was no guarantee of sufficient support throughout the day or no telephone at home, or they lived more than 10 km from the hospital.
Patients were randomly assigned to care under the HaH service or to the Cardiology ward using a randomised sequence externally generated, researchers remaining blind to group allocation until an eligible patient had given written informed consent to participate in the study.
2.3. Intervention
HaH group: in the Emergency Department, the characteristics of the HaH service were explained to the patients assigned to this model of care, a patient information sheet being provided with the operating hours and contact telephone numbers. The following day, patients were visited at home by a doctor and a nurse from the HaH team. Subsequent visits, tests and other examinations were programmed and appropriate treatment indicated on a case-by-case basis. The HaH nurse visited patients every day and the HaH doctor every one to two days, depending on their progression. Besides clinical evaluation and treatment administration, the nurse uses the visits to educate the patient about their condition. The protocol stipulated that patients were to be transferred to hospital for ward admission in the event of worsening or failure to respond to treatment.
CW patients were managed in accordance with the guidelines of the Department of Cardiology in terms of treatment, indications for requesting tests and nursing care.
After the intervention, patients were followed-up in the usual way in the primary care setting, outside the control of the researchers.
2.4. Study Variables and Measurement/Assessment Instruments
Generic questionnaires were used for assessing functional status (Barthel Index, BI) and HRQOL (SF-36 and EQ-5D) while a HF specific questionnaire was used to assess HRQOL related to heart failure (the Minnesota Living with Heart Failure, MLHFQ). All the questionnaires were administered by one of two members of the research team.
An initial face-to-face interview was conducted covering the four questionnaires in the first 48 hours after admission, asking the patient and their family to consider the situation prior to the decompensation. At discharge, the BI and EQ-5D were re-administered, on this occasion considering the patient’s status at the time of the interview.
Subsequent assessments were made for all four instruments at 6 and 12 months after discharge, and additionally, the BI and EQ-5D at discharge and 1 and 3 months after. The questionnaires were sent to patients by mail and telephone support was offered to help with completing the forms.
The Barthel Index (BI) is a 10-item questionnaire with 2 or 3 response options with different scores. The maximum score of 100 corresponds to the highest level of independence [12].
The SF-36 is a structured QOL questionnaire with 36- items that assesses both positive and negative aspects of well-being, across 8 dimensions: physical functioning, social functioning, role limitation—physical, role limitation—emotional, mental health, vitality, bodily pain, and general health. For each of the dimensions, the item scores are coded, summed and transformed onto a scale from 0 (the worst state of health for the corresponding dimension) to 100 (best health) [1,10,13]. The questionnaire has not been designed to generate an overall score. On the other hand, two summary scores, for the physical and mental health components, can be calculated by combining the scores on the other dimensions. In the EQ-5D, patients rate their current state, first in terms of levels of severity on various dimensions (the descriptive system) and then on a more general visual analogue scale (VAS). The descriptive system has five dimensions (mobility, self-care, daily activities, pain/discomfort and anxiety/depression) and for each there are three levels of severity (no problems, some or moderate problems, extreme problems). The second part of the EQ-5D is a vertical 20-cm VAS (Visual Analogic Scale) from 0 (worst imaginable health state) to 100 (best imaginable health state) [1,14,15]. Lastly, the MLHFQ measures self-perceived quality of life in patients with heart failure. It is composed of 21 items, rated on a scale from 0 to 5, the highest score of 105 indicating the poorest quality of life [1,16-18].
We also recorded anthropometric data; Charlson Comorbidity Index; clinical data related to the episode leading to the admission; serum NT-proBNP concentrations at admission and discharge from the CW or the HaH Unit, as well as 12 months after discharge; mortality; and hospital admissions for further episodes of decompensation or other cardiovascular conditions (stroke, acute coronary syndrome or coronary revascularisation) in the 12-months following the intervention.
2.5. Ethical Considerations
The study was approved by the Clinical Research Ethics Committee of Araba University Hospital and all the patients gave written informed consent before inclusion in the study, in accordance with the principles of the 1975 Declaration of Helsinki.
2.6. Statistical Analysis
Changes in quality of life, as reflected in MLHFQ and SF-36 physical and mental health summary scores, were assessed in each group with the Student’s t-test for paired data, while differences these quality of life scores during the follow-up period (at 6 and 12 months) were compared between the two groups using ANCOVA, adjusting for baseline values.
Similarly, changes in functional status and quality of life over the follow-up period, in terms of the Barthel Index scores and EQ-5D values, were assessed in each group with the Student’s t-test for paired data and also with McNemar’s test for each component of the EQ-5D. For continuous variables ANCOVA was used for comparisons between the groups, adjusting for baseline values, and chi-square tests were used for each component of the EQ-5D.
The analysis was performed using SPSS for Windows (version 18), with a significance level of alpha = 0.05.
3. RESULTS
Of the 80 patients included in the study, 71 completed the follow-up (37 in the HaH group and 34 in the CCU group). The reasons for the withdrawals were: for personal reasons (3 patients) or by decision of the follow-up committee (another 3), or that they developed a nonheart-related medical condition that made follow-up impossible (another 3). Figure 1 illustrates the flow of patients through the study.
The mean age was 79 years and 41 (58%) of the 71 of participants were women. In 49 cases (69%) the Charlson Comorbidity Index score was greater than 2, while the NYHA functional class prior to the decompensation was II in 42 cases (59%) and III in the others (29 cases, 41%). The 24 patients with systolic dysfunction (34%) had a left ventricular ejection fraction of 0.2 to 0.45 (mean = 0.36). The mean NT-proBNP concentration at admission was 2912 pg/ml (1300 - 5138).
Patients assigned to HaH had a worse cardiovascular risk profile with higher percentage rated as NYHA Class III, systolic disfunction and atrial fibrillation and higher prevalence of COPD and renal failure (Table 1).
The initial admission lasted a mean of 7.9 days in the CW group compared to 10.9 days in HaH. There were no deaths during the initial period, nor were any patients
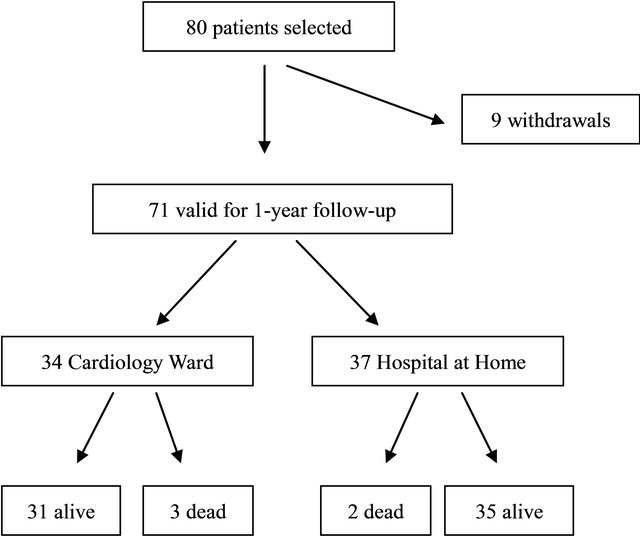
Figure 1. Flow chart outlining the study design.
Table 1. Demographic and baseline clinical characteristics.
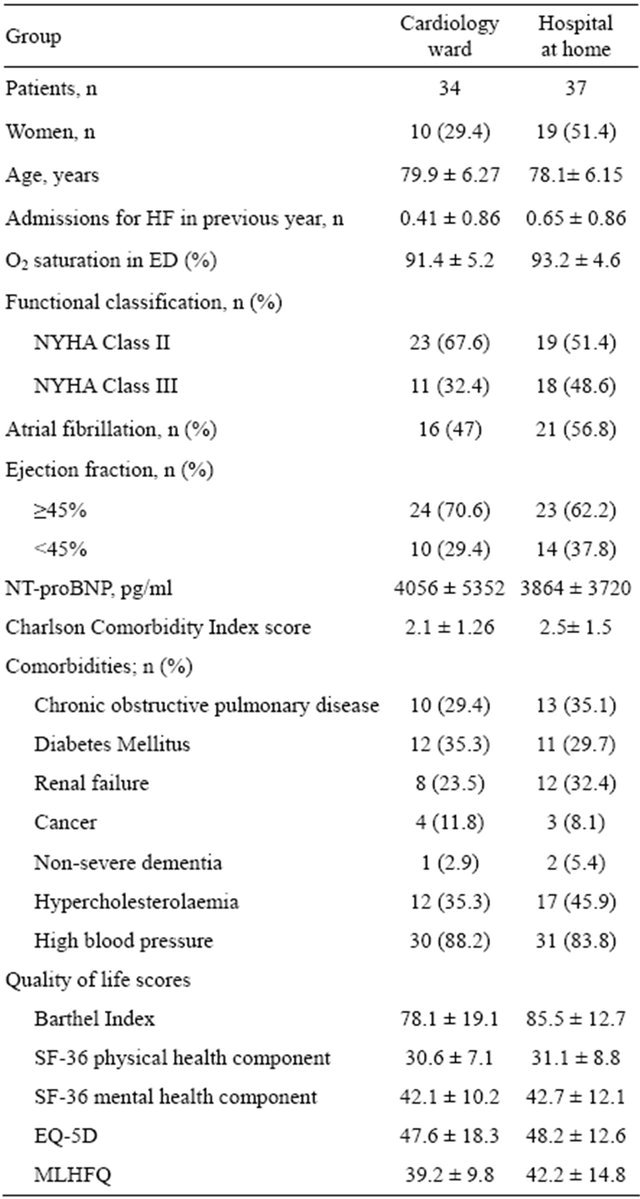
HF: heart failure; MLHFQ: Minnesota Living with Heart Failure Questionnaire.
transferred from the HaH to the CW. Later, during the follow-up year, 5 patients (7%) died: 3 (8.8%) and 2 (5.4%) from the CW and HaH groups respectively (p = 0.665). In total, over the 12-months of follow-up 19 of 34 (55.9%) of patients in the CW group died or were readmitted for HF or cardiovascular events, compared to 20 of 37 under HaH care (54.1%) (p = 0.877).
The questionnaires were completed by 71 patients at baseline, discharge and 1 month later. Given the death of 5 patients, the number of participants fell to 70 at 3 months, 69 at 6 months and 66 at 12 months. For comparing functional status and quality of life we only considered the 66 patients who completed the full year of follow-up.
3.1. MLHF and SF-36
Table 2 shows the scores on the MLHFQ and SF-36 physical and mental health summary measures at 6 and 12 months after discharge compared to the initial level in the two groups. As might be expected, the mean values for each of the eight components of the SF-36 were lower (worse quality of life) than the reference values for the Spanish population over 60 years of age [13].
Among patients who had received HaH care, the assessment at 6 months showed significant improvements in scores with respect to the initial values on the MLHFQ (10.3; p = 0.001) and the SF-36, both in the physical and mental health summary measures (5.4; p = 0.001 and 3.8; p = 0.053, respectively) and in 6 of the 8 dimensions explored: physical functioning, role-physical, general health, vitality, social functioning and role-emotional. In contrast, among patients treated as inpatients in the CW there were no significant changes in scores on either of these questionnaires, neither overall nor considering specific dimensions.
At 12 months, we observed differences with respect to the initial scores in only three dimensions of the SF-36 in the HaH group: role-physical, social functioning and roleemotional; and in one in the CW group: role-physical. All the other scores, for the SF-36 component dimensions and physical and mental health summary measures as well as for the MLHFQ, were similar to those in the initial measurements.
Comparison between groups of the changes in scores on each questionnaire at 6 and 12 months with respect to the initial values are reported in Table 3. The analysis indicates that at 6 months there were significantly greater improvements in the HaH group, compared to the CW group, in scores on the MLHFQ and the SF-36 physical component summary, as well as the physical functioning, role-physical, bodily pain and vitality dimensions. By 12 months, however, no significant differences were found between groups on either of these questionnaires.
3.2. Barthel Index and EQ-5D
Table 4 lists the scores for each of the two arms of the study at every point of the analysis. To simplify the presentation of the results, similar to the approach used in other studies [15], we present the percentage of patients who reported moderate or severe problems for every 5 dimensions of the EQ-5D. BI and EQ-5D-VAS are shown as mean and SD. In both groups there were significant improvements with respect to the initial measurements, these being more common considering the measurements at discharge, 1 and 3 months (16 and 20 of 21 being significant in the CW and HaH groups respectively) than those at 6 and 12 months (6 and 10 of 14 being significant in the CCU and HaH groups respectively).
Table 2. Changes in the mean MLHFQ and SF-36 scores by treatment group (n = 66).
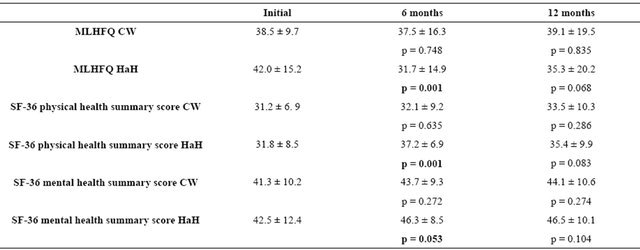
MLHFQ: Minnesota Living with Heart Failure Questionnaire; CW: Cardiology Ward group; HaH: hospital at home group; p value for comparisons with initial values; Comparisons within groups between initial scores and scores at 6 and 12 months with the Student’s t-test for paired data.
Table 3. Differences between the two groups in the magnitude of change in scores between the initial and 6 and 12 months measurements.
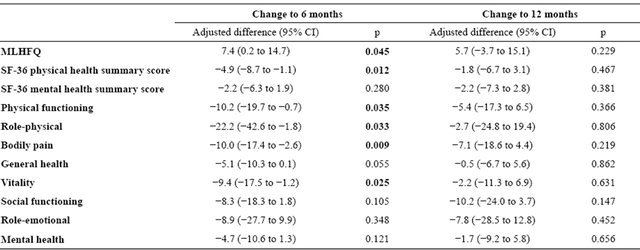
Comparison between CW and HaH groups for the magnitude of change in score with ANCOVA, adjusting for baseline values. Negative values for SF-36 scores and positive ones for MLHFQ scores represent differences in favour of the HaH group.
Comparing the changes in the scores over the followup period in the two arms of the study, we found differences in mobility and self-care dimensions (EQ-5D) and in the Barthel Index at 6 months (p = 0.033, p = 0.036, and p = 0.004 respectively), and in the pain/discomfort dimension and VAS (EQ-5D) at 1, 3 and 6 months (p = 0.043, p = 0.033, and p = 0.011), indicating greater improvement in the HaH group.
4. DISCUSSION
The study was designed to analyse differences in recovery of functional capacity and quality of life after an episode of decompensation of HF as a function of hospitalisation under the care of the Department of Cardiology or the HaH service. Quality of life was assessed with respect to the baseline condition prior to the decompensation. The starting hypothesis was that decompensation leads to deterioration with respect to baseline but that patients’ condition tends to recover in the days or months following discharge. This was expected to translate into better quality of life scores in the initial baseline measurement, followed by poorer scores in the measurement before discharge and gradual recovery to baseline status over the follow-up period. The pattern observed, however, was the poorest perceived quality of life in the first assessment, with improvements from discharge to a level at which it remained fairly steady for the rest of the
Table 4. Scores on the Barthel Index and EQ-5D over the follow-up period (n = 66).
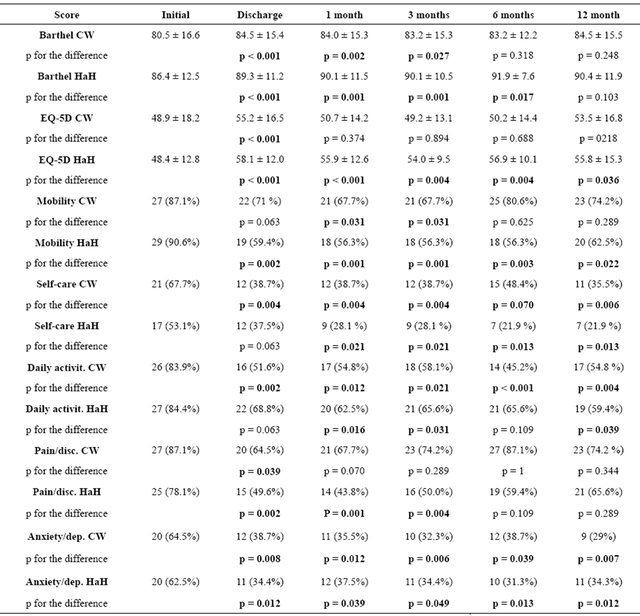
CW: Cardiology Ward group; HaH: Hospital at home group. Scores on Barthel (Barthel Index) and EQ-5D (visual analogue scale or overall from 0 to 100): reported as mean ± SD. Mobility, Self-care, Daily activities, Pain-Disc. (pain/discomfort) and Anxiety-dep. (anxiety/depression): reported as the total percentage of patients who reported “some or moderate” or “severe” problems (19,20). The p values refer to the comparison between the scores at the time point indicated and the initial assessment. Changes in functional status and quality of life (Barthel Index and EQ-5D scores respectively) during the follow-up period in each treatment group were assessed with the Student’s t-test for paired data and McNemar’s test for each component of the EQ-5D.
follow-up period. This suggests that the initial assessment, though theoretically referring back to the situation prior to the decompensation, was strongly influenced by the clinical condition of the patient at the time at which it was carried out (in the first 24 - 48 hours after admission). One finding in particular supports this interpretation. Patients classified as NYHA Class II prior to the decompensation had an mean MLHFQ score of 41.1, which is notably higher (indicating poorer quality of life) than the value reported by Lupón et al. [16] in patients over 65 years of age with stable HF in NYHA class II (20.7 ± 12.6).
Soriano et al. [10], studying 883 patients surviving an admission for HF with a one-year mortality rate of 23%, observed that SF-36 physical and mental health summary scores improved to some extent between admission (means of 34.1 and 40.1 respectively) and one month after discharge (36.5 and 43.4 respectively), and were then steady to the end of the 12-month follow-up. The MLHFQ score was 37.5 ± 21.3 a month after discharge and remained at 32 - 34 in subsequent measurements. Both the scores and the way they changed over time are similar to the findings of the present study. Further, though Moser et al. [11], analysing 433 patients admitted with severe systolic dysfunction (ejection fraction < 30%) with a 6-month mortality rate of 19%, found considerably higher MLHFQ scores (poorer quality of life) than in our study, the values evolved in a similar way: decreasing from 74.2 ± 17.4 during admission to 56.7 ± 22.7 a month after discharge, and remaining close to this level at 3 and 6 months.
With respect to comparisons between the groups managed by the HaH service and the Cardiology Department, improvements with respect to the initial measurement were more numerous and of bigger magnitude in the HaH group than in CW group, and, for the two groups, in assessments up to 6 months, than in those conducted at 6 and 12 months, a finding which could reasonably be attributed to progression of the disease. On the other hand, the improvements continued to be more numerous in the HaH group in these later assessments and this seems to indicate a greater and more sustained improvement than in the CW group. We believe that this may be related to patients feeling that they are more comfortable and have better functioning when treated in their everyday environment, rather than being transferred to an unfamiliar setting, namely the hospital ward.
A relatively small number of patients were included in the study and, given the selection criteria, the participants are not representative of the full spectrum of patients who present at the Emergency Department with acutely decompensated chronic HF. Further, this study has considered a particular HaH service. These factors limit the external validity of the conclusions. Patients under HaH care progressed very well, none requiring transfer to hospital for inpatient care, but the sample size was small. This could lead us to believe that there was a selection bias, given the application of very restrictive selection criteria, namely that we had only included cases that were very mild and might not even have needed hospitalisation. On the other hand, the baseline characteristics indicated that patients assigned to HaH care had a worse cardiovascular risk profile than those treated as inpatients. Further, this latter group, managed following the standard practice of the Cardiology Department, were hospitalised for a mean of almost 8 days, suggesting that their admission was fully justified.
Scores on the questionnaires could have been influenced by the educational level of patients or the exact time when questionnaires were completed and, as we did not take these factors into account, they could represent a bias in data collection. Similarly, the fact that some questionnaires were completed at home and returned by post means that the responses could have been from a relative rather than the patient themselves. These potential sources of bias, though, can be expected to have a similar effect in both treatment groups.
5. CONCLUSIONS
During acute decompensation of chronic heart failure patients probably have a distorted perception of the quality of life and level of functional independence they had during the preceding phase of stability.
Indicators of functional independence and health-related quality of life assessed after an episode of decompensated heart failure show improvements on discharge compared to admission, both in patients managed by the Hospital at Home service and those admitted to a Cardiology Ward (conventional hospitalisation). In the following 12 months, improvement is maintained in more indicators and for more longer among the patients who were treated by hospital-at-home.
Further research should be conducted, avoiding certain weaknesses of the present study, to confirm the findings.
![]()
![]()
REFERENCES
- Banegas, J.R. and Rodríguez-Artalejo, F. (2008) Insuficiencia cardiaca e instrumentos para medir la calidad de vida. Revista Española de Cardiología, 61, 233-235. doi:10.1157/13116649
- Lewis, E.F., Johnson, P.A., Johnson, W., Collins, C., Griffin, L. and Stevenson, L.W. (2001) Preferences for quality of life or survival expressed by patients with heart failure. The Journal of Heart and Lung Transplantation, 20, 1016-1024. doi:10.1016/S1053-2498(01)00298-4
- Shepperd, S., Doll, H., Angus, R.M., Clarke, M.J., Iliffe, S., Kalra, L., et al. (2009) Avoiding hospital admission through provision of hospital care at home: A systematic review and meta-analysis of individual patient data. CMAJ, 180, 175-182. doi:10.1503/cmaj.081491
- González-Ramallo, V.J. and Segado-Soriano, A. (2006) Veinticinco años de hospitalización a domicilio en Espa- ña. Medicina Clinica (Barcelona), 126, 332-333. doi:10.1157/13085746
- Mendoza Ruiz de Zuazu, H., Regalado de los Cobos, J., Altuna Basurto, E., Cia Ruiz, J.M., Aros Borau, F. and Lopetegui Eraso, P. (2003) Tratamiento de la insuficiencia cardiaca en régimen de hospitalización a domicilio: Estudio de 158 pacientes. Medicina Clinica (Barcelona), 120, 405-407. doi:10.1157/13045304
- Brotons, C., Falces, C., Alegre, J., Ballarin, E., Casanovas, J., Cata, T., et al. (2009) Ensayo clínico aleatorizado para evaluar la efectividad de una intervención domiciliaria en pacientes con insuficiencia cardiaca: Estudio IC-DOM. Revista Española de Cardiología, 62, 400-408. doi:10.1016/S0300-8932(09)70897-8
- Tibaldi, V., Isaia, G., Scarafiotti, C., Gariglio, F., Zanocchi, M., Bo, M., et al. (2009) Hospital at home for elderly patients with acute descompensation of chronic herat failure. Archives of Internal Medicine, 169, 1569-1575. doi:10.1001/archinternmed.2009.267
- Patel, H., Shafazand, M., Ekman, I., Höjgärd, S., Swedberg, K. and Schaufelberger, M. (2008) Home care as an option in worsening chronic heart failure. A pilot study to evaluate feasibility, quality adjusted life years and cost effectiveness. European Journal of Heart Failure, 10, 675-681. doi:10.1016/j.ejheart.2008.05.012
- Mendoza, H., Martin, M.J., Garcia, A., Aros, F., Aizpuru, F., Regalado De Los Cobos, J., et al. (2009) “Hospital at home” care model as an effective alternative in the management of decompensated chronic heart failure. European Journal of Heart Failure, 11, 1208-1213. doi:10.1093/eurjhf/hfp143
- Soriano, N., Ribera, A., Marsal, J.R., Brotons, C., Cascant, P. and Permanyer-Miralda, G. (2010) IC-QoL study investigators. Evolución de la calidad de vida relacionada con la salud en pacientes ingresados por insuficiencia cardiaca. Estudio IC-QoL. Revista Española de Cardiología, 63, 668-676. doi:10.1016/S0300-8932(10)70159-7
- Moser, D.K., Yamokoski, L., Sun, J.L., Conway, G.A., Hartman, K.A., Graziano, J.A., et al. (2009) For the ESCAPE investigators. Improvement in Health-related quality of life after hospitalization predicts event-free survival in patients with advanced heart failure. Journal of Cardiac Failure, 15, 763-769. doi:10.1016/j.cardfail.2009.05.003
- Mahoney, F.I. and Barthel, D.W. (1965) Functional evaluation: The Barthel index. A simple index of independence useful in scoring improvement in the rehabilitation of the chronically ill. Maryland State Medical Journal, 14, 61-65.
- López-García, E., Banegas, J.R., Graciani Pérez-Regadera, A., Gutiérrez-Fisac, J.L., Alonso, J. and Rodrí- guez-Artalejo, F. (2003) Valores de referencia de la versión española del cuestionario de salud SF-36 en la población adulta mayor. Medicina Clinica (Barcelona), 120, 568-573. doi:10.1157/13046436
- Brooks, R.G., Jendteg, S., Lindgren, B., Persson, U. and Bjork, S. (1991) Euroqol: Health related quality of life measurement. Results of the Swedish questionnaire exercise. Health Policy, 18, 37-48. doi:10.1016/0168-8510(91)90142-K
- Almenar-Pertejo, M., Almenar, L., Martínez-Dolz, L., Campos, J., Galán, J., Gironés, P., et al. (2006) Study on health-related quality of life in patients with advanced heart failure before and after transplantation. Transplantation Proceedings, 38, 2524-2526. doi:10.1016/j.transproceed.2006.08.017
- Lupón, J., Altimir, S., González, B., Parajón, T., Prats, M., Urrutia, A., et al. (2006) Valoración de la calidad de vida en pacientes ancianos con insuficiencia cardiaca mediante el cuestionario Minnesota Living with Heart Failure. Rev Esp Geriatr Gerontol, 41, 150-157. doi:10.1016/S0211-139X(06)72945-6
- Morcillo, C., Aguado, O., Delás, J. and Rosell, F. (2007) Utilidad del Minnesota Living with Heart Failure Questionnaire en la evaluación de la calidad de vida en enfermos con insuficiencia cardiaca. Revista Española de Cardiología, 60, 1093-1096. doi:10.1157/13111242
- Garin, O., Soriano, N., Ribera, A., Ferrer, M., Pont, A., Alonso, J., et al. (2008) Validación de la versión española del Minnesota Living with Heart Failure Questionnaire (MLHFQ). Revista Española de Cardiología, 61, 251- 259. doi:10.1157/13116652
NOTES
*The study was funded by the charitable foundation of the Caja Vital building society.
#Corresponding author.

