Paper Menu >>
Journal Menu >>
 Vol.1, No.3, 176-180 (2009) doi:10.4236/ns.2009.13022 SciRes Copyright © 2009 Openly accessible at http://www.scirp.org/journal/NS/ Natural Science Fabrication of two-dimensional periodic TiO2 pillar arrays by multi-beam laser interference lithography Hiroyo Segawa1, Hiroaki Misawa2 1Exploratory Nanomaterials Research Laboratory, National Institute for Materials Science, Tsukuba, Japan 2Research Institute for Electronic Science, Hokkaido University, Sapporo, Japan Received 19 August 2009; revised 21 September 2009; accepted 22 September 2009. ABSTRACT Two-dimensional (2D) periodic TiO2 pillar arrays, applicable to photonic crystals and micro- channels, were fabricated by direct patterning of a TiO2-organic hybrid material by multi-beam laser interference lithography and calcination of hybrid patterns. 2D periodic pillars of a TiO2– organic hybrid material were prepared by irra- diation with the interference pattern of femto- second laser beams and removal of the non- irradiated portions. Two types of periodic pillar arrays, standing pillars and top-gathering pillars (four pillars gathered at the top), were obtained, depending on laser irradiation conditions. After calcination of TiO2–organic hybrid pillars, TiO2 pillar arrays were obtained without collapse. Keywords: Lithography, Capillary Force, Assembly 1. INTRODUCTION Two-dimensional (2D) periodic arrays of dielectric ma- terials with submicrometer to micrometer repetitions have great potential in various applications, such as dif- fraction gratings [1], photonic crystals (PCs) [2,3], and molecular separation in a microchannel [4,5]. Most of these arrays are fabricated by lithography, in which di- electric materials are coated with photoresists, the photo- resist patterns are fabricated by beams such as electron beams, lasers, and beams of UV light, and the patterns of the dielectric material are chemically etched in the area not covered by the photoresist. The patterns of the di- electric materials do not feature a high aspect ratio (pat- tern height to pitch), and complex patterns, e.g.,three- dimensional (3D) patterns, cannot be obtained easily, because dielectric materials are generally stable for chemical etchants. However, patterns such as pillars [6,7], waveguides [8, 9,10], and diffraction gratings [11], can be fabricated in organic–inorganic hybrid materials by direct lithography. Organic–inorganic hybrid materials prepared by the sol–gel method are composed of inorganic networks modified with photosensitive organic molecules such as unsaturated hydrocarbons or β-diketonato ligands. When the hybrid films are exposed to UV light or a laser, pho- tosensitive organic groups react, and the solubility of the exposed parts in alcohol decreases. Removing the unex- posed portions produces the patterns of hybrid materials. Hybrid materials are generally more flexible than in- organic materials due to the characteristics of the organic groups; therefore, they have been used to fabricate many types of thick films with thickness greater than 1 μm. Most thick films are prepared by the sol–gel method, using organosiloxanes, in which one or two alkoxyl groups of the silicone alkoxide change to functional or- ganic groups. However, it is known to be difficult to fabricate thick films without using organosiloxane. In our previous work, different types of photosensitive TiO2–organic hybrid films were prepared by the sol–gel method [12,13,14]. TiO2 hybrid films were prepared from a Ti alkoxide, and modified with a β-diketone and/ or an unsaturated hydrocarbon. Two types of TiO2 hy- brid dot arrays, with a pitch spacing of 1.3 μm and a height of about 250–300 nm, were directly fabricated by multi-beam laser interference (MLI) lithography. In this method, photosensitive materials are exposed to the in- terference light pattern and developed in an appropriate solvent [13,14]. One type was fabricated by multi-pho- ton absorption (MPA) of a femtosecond laser at 800 nm, while another was fabricated by a nanosecond laser at 351 nm. Hybrid films with a thickness of about 1 μm were used for the lithography; however, the light for photopolymerization was absorbed in the film, and moreover, photopolymerization did not occur over the entire film. Both types of TiO2–organic hybrid dot arrays were calcined, and TiO2 arrays were fabricated without collapse or exfoliation; however, patterns with a high aspect ratio could not be produced. MPA is a well-known method of fabricating complex patterns by photopolymerization of organic compounds [15,16]. If MPA was used for producing patterns in  H. Segawa et al. / Natural Science 1 (2009) 176-180 SciRes Copyright © 2009 Openly accessible at http://www.scirp.org/journal/NS/ 177 Figure 1. Flowchart of experimental procedure. thick organic-inorganic hybrid films by lithography, fabrication of 3D patterns with a high aspect ratio would be possible. The hybrid materials could be easily chang- ed to ceramics by calcination, resulting in complex cera- mic patterns. In this study, 2D periodic pillar arrays of TiO2 with a high aspect ratio were fabricated directly from TiO2– organic hybrid films with a thickness greater than 1 μm, using the MLI technique and by calcination of the or- ganic groups. 2. EXPERIMENTAL PROCEDURE A flowchart of the experimental procedure is shown in Figure 1. A TiO2–organic hybrid gel film with a thickness greater than 1 μm was prepared by the sol–gel method [14]. Titanium tetra-n-butoxide (Ti(O-nBu)4, Kishida Chemical Co.) was reacted with 2-(methacryloyloxy)ethyl acetoacetate (MEAcAc, Acros Organics); the molar ratio between Ti(O-nBu)4 and MEAcAc was 1:1. After stirring the solution for 15 min at room temperature, H2O was poured into the solution; the molar ratio between H2O and Ti(O-nBu)4 was 2:1. The solution was stirred for 3 hat room temperature. The sol was then spin-coated at 1000 rpm for 20 sec on glass substrates. The hybrid film was baked at 80°C for 10 min. The film was then exposed to the interference pattern of femtosecond laser pulses. The optical setup has been described previously [17]. In brief, a diffractive beam splitter (G1023A, MEMS Optical Inc.) divided the input laser beam into four beams, which were then collected on the film using two lenses. The femtosecond pulses were produced by a Ti:sapphire regenerative amplifier (800-nm wavelength, 150-fs pulse duration, 1-kHz repe- tition rate). The single-pulse energy (sum of all beams) was 20–35 μJ. After laser irradiation for 1–10 min, the films were developed in 2-ethoxyethanol for 1 min to remove the non-irradiated portions. After dryingthe 2-ethoxyethanol, the TiO2–organic hybrid arrays re- Figure 2. UV-VIS spectrum of TiO2–organic hybrid film. mained on the substrates. The hybrid arrays were cal- cined at 450 °C for 1 h, finally producing TiO2 arrays. The periodic arrays before and after calcination were imaged by ascanning electron microscope (SEM; JSM- 6700FT, JEOL). A UV-VIS spectrum of the prepared hybrid film was measured by a spectrometer (JASCO, V-570). The hy- brid film was calcined at 120–750 °C for 1h, and refrac- tive indices of the film after calcination were measured at 632.8 nm with a Metricon 2010 prism coupler. 3. RESULTS & DISCUSSIONS Figure 2 shows the UV-VIS spectrum of the TiO2–or- ganic hybrid film. In the spectrum, the increase in ab- sorption around 400 nm might be assigned to the dike- tonate chelate between MEAcAc and Ti(OBu)4 [14]. As seen in Figure 2, the laser light at 800 nm was not ab- sorbed by the hybrid film. Typical SEM images of the TiO2–organic hybrid pil- lar arrays are shown in Figure 3. In Figure 3a, 3b, and 3c, the pillar arrays were obtained by irradiation with a 30-μJ single-pulse for 100, 140, and 300 sec, respec- tively. In the pillar arrays of Figure 3a, all pillars are gathered at the top, and top-gathering units composed of four (2 × 2) pillars are arranged periodically. In Figure 3b, all pillars are standing vertically on the substrate, and are arranged tetragonal, which corresponds to the light distribution. In Figure 3c, many pillars are gath- ered without any empty space between pillars, and have adhered to each other. The pillar diameter increased with increased laser irradiation time. When the laser irradia- tion time increases, the photopolymerization of the hy- brid film proceeds, leaving a wider area on the substrate after development, resulting in thicker pillars. In Figure 3, the pillar arrays were fabricated on 1.4-μm thick films, and the height of the pillars was 1.4 μm. The height of pillars were improved from previous ones [14] and as- pect ratio of the pillars became high. This represents that 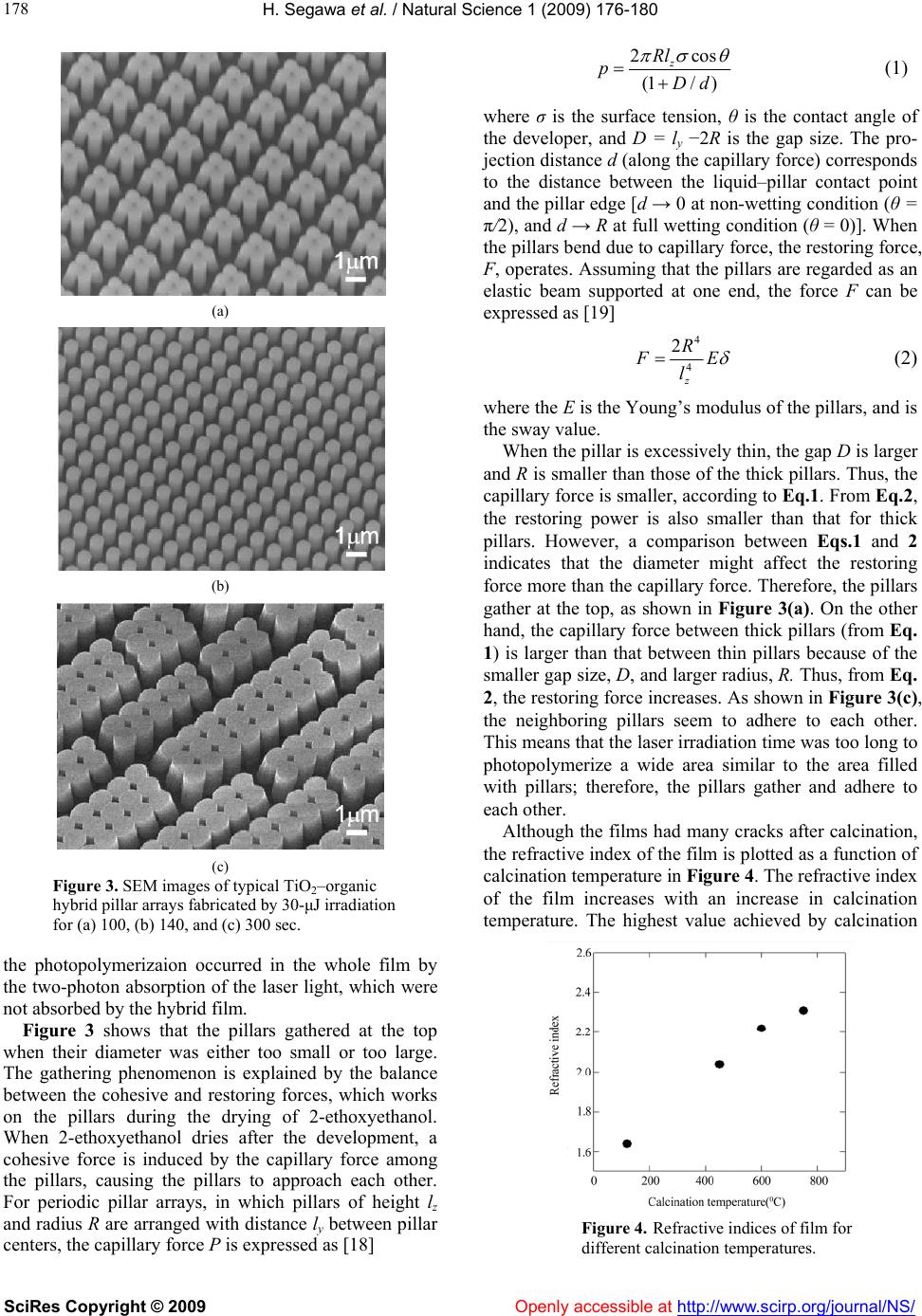 H. Segawa et al. / Natural Science 1 (2009) 176-180 SciRes Copyright © 2009 Openly accessible at http://www.scirp.org/journal/NS/ 178 (a) (b) (c) Figure 3. SEM images of typical TiO2–organic hybrid pillar arrays fabricated by 30-μJ irradiation for (a) 100, (b) 140, and (c) 300 sec. the photopolymerizaion occurred in the whole film by the two-photon absorption of the laser light, which were not absorbed by the hybrid film. Figure 3 shows that the pillars gathered at the top when their diameter was either too small or too large. The gathering phenomenon is explained by the balance between the cohesive and restoring forces, which works on the pillars during the drying of 2-ethoxyethanol. When 2-ethoxyethanol dries after the development, a cohesive force is induced by the capillary force among the pillars, causing the pillars to approach each other. For periodic pillar arrays, in which pillars of height lz and radius R are arranged with distance ly between pillar centers, the capillary force P is expressed as [18] 2cos (1/ ) z Rl pDd (1) where σ is the surface tension, θ is the contact angle of the developer, and D = ly −2R is the gap size. The pro- jection distance d (along the capillary force) corresponds to the distance between the liquid–pillar contact point and the pillar edge [d → 0 at non-wetting condition (θ = π/2), and d → R at full wetting condition (θ = 0)]. When the pillars bend due to capillary force, the restoring force, F, operates. Assuming that the pillars are regarded as an elastic beam supported at one end, the force F can be expressed as [19] 4 4 2 z R F E l (2) where the E is the Young’s modulus of the pillars, and is the sway value. When the pillar is excessively thin, the gap D is larger and R is smaller than those of the thick pillars. Thus, the capillary force is smaller, according to Eq.1. From Eq.2, the restoring power is also smaller than that for thick pillars. However, a comparison between Eqs.1 and 2 indicates that the diameter might affect the restoring force more than the capillary force. Therefore, the pillars gather at the top, as shown in Figure 3(a). On the other hand, the capillary force between thick pillars (from Eq. 1) is larger than that between thin pillars because of the smaller gap size, D, and larger radius, R. Thus, from Eq. 2, the restoring force increases. As shown in Figure 3(c), the neighboring pillars seem to adhere to each other. This means that the laser irradiation time was too long to photopolymerize a wide area similar to the area filled with pillars; therefore, the pillars gather and adhere to each other. Although the films had many cracks after calcination, the refractive index of the film is plotted as a function of calcination temperature in Figure 4. The refractive index of the film increases with an increase in calcination temperature. The highest value achieved by calcination Figure 4. Refractive indices of film for different calcination temperatures.  H. Segawa et al. / Natural Science 1 (2009) 176-180 SciRes Copyright © 2009 Openly accessible at http://www.scirp.org/journal/NS/ 179 (a) (b) Figure 5. SEM images of TiO2 pillar arrays fabricated by irradiation at 30 μJ for (a) 100 and (b) 140 sec followed by calcination at 450 °C for 1h. at 750 °C was 2.31-smaller than the refractive index of anatase (2.55) [20]. Since the films had cracks, the re- fractive indices might be assumed to be smaller than those of a dense TiO2 film. To investigate changes in morphology, SEM images of the periodic pillars calcined at 450 °C are shown in Figure 5. Figure 5(a) and (b) were calcined at 450 °C the TiO2–organic hybrid arrays which were shown in Figure 3(a) and (b), respectively.The top-gathering pil- lars in Figure 5(a) were calcined, after which they re- tained the top-gathering structure, although the size be- came smaller. Figure 5(b) shows that the pillar size de- creased after calcination. The several kinds of organic compounds were remained in the hybrid pillars before calcination, and those were decomposed by the heating and removed. Thus, the pillars shrunk after the calcina- tion. As shown in Figure 5, the pillars did not collapse. Generally, films without patterns were restricted by the substrate and subjected to large stresses during the cal- cination, resulting in the many cracks. In this case, the pillars were small not to restrict by the substrate and could shrink without stress during calcination. XRD patterns of the periodic array after calcination at 450 °C are shown in Figure 6. All the peaks can be as- signed to anatase. Figure 4 shows that the refractive index of the film calcined at 450 °C was 2.04, which is smaller than the reported value of anatase (2.55) [20]. However, the refractive index of the pillars might be Figure 6. XRD pattern of TiO2 pillar arrays. larger than that of the film, since the pillars appear dense. These results indicate that patterning of photosensitive hybrid materials by lithography and calcination of hy- brid materials are excellent methods of creating fine patterns with high aspect ratios in ceramics. Generally, the preparation of the photosensitive hybrid films with chelate compounds are useful to obtain patterns of such as ZrO2 [21] and PbZr0.52Ti0.48O3 (PZT) [22]. If thick organic-inorganic hybrid films are obtained, complex ceramic patterns can be easily produced by direct li- thography of hybrid films and calcination of hybrid pat- terns. 4. CONCLUSIONS We produced TiO2 pillar arrays with a high aspect ratio by MLI lithography and calcination. Pillars with a high aspect ratio were successfully fabricated by the mul- tiphoton absorption of a femtosecond laser. TiO2–or- ganic hybrid pillar arrays were fabricated by MLI, and two types of patterns, a periodic pillar array and a top- gathering pillar array, were obtained, depending on the laser irradiation time. The laser irradiation time affected the balance between the cohesive and restoring forces during the drying of the developer in lithography. After calcination of the hybrid patterns, TiO2 patterns were obtained without collapse. This suggests that direct li- thography of organic–inorganic hybrid materials and calcination can be applied to fabrication of complex pat- erns in ceramics. t REFERENCES [1] H. P. Herzig (1997), Micro-optics. Taylor & Francis, Ltd., London, UK. [2] J. D. Joannapoulos, R. D. Meade, and J. N. Winn, Photonic crystals.Princeton University Press, Princeton, NJ, 1995. [3] E. Yablonovitch, (1993) Photonic band-gap structures. J.  H. Segawa et al. / Natural Science 1 (2009) 176-180 SciRes Copyright © 2009 http://www.scirp.org/journal/NS/Openly accessible at 180 Opt. Soc. Am. B, 10283-295. [4] W. D. Volkmuth and R. H. Austin, (1992) DNA electro- phoresis in microlithographic arrays. Nature, 358, 600- 602. [5] N. Kaji, Y. Tezuka, Y. Takamura, M. Ueda, T. Nishi- moto, H. Nakanishi, Y. Horiike, and Y. Baba, (2004) Separation of long DNA molecules by quartz nanopillar chips under a direct current electric field. Anal. Chem., 76, 15-22. [6] H. Segawa, Y. Yamazaki, T. Yano, and S. Shibata, (2006) Top-gathering periodic array derived from self-organi- zation of hybrid organic-inorganic pillars. Jpn. J. Ceram. Soc., 114, 120-124. [7] H. Segawa, S. Yamaguchi, Y. Yamazaki, T. Yano, S. Shibata, and H. Misawa, (2006) Top-gathering pillar ar- ray of hybrid organic-inorganic material by means of self-organization. Appl. Phys. A, 83, 447-451. [8] H. Krug, F. Tiefence, P. W. Oliveira, and H. Schmidt, (1992) Organic-inorganic composite materials: Optical properties of laser-patterned and protective-coated wave- guides. SPIE, 1758, 448-455. [9] H. Schmidt, J. Krug, R. Kasemann, and F. Tiefensee, (1991) Devel opment of optical wave guides by sol-gel techniques for laser patterning. SPIE, 1590, 36-43. [10] S. I. Najafi, T. Touam, R. Sara, M. P. Andrews, and M. A. Fardad, (1998) Sol-gel glass waveguide and grating on silicon. J. Lightwave Tech., 16, 1640-1646. [11] H. J. Jiang, X. C. Yuan, Y. Zhou, Y. C. Chan, and Y. L. Lam, (2000) Single-step fabrication of diffraction grat- ings on hybrid sol-gel glass using holographic interfer- ence lithography. Opt. Commun., 185 19-24. [12] H. Segawa, S. Adachi, Y. Arai, and K. Yoshida, (2003) Fine patterning of hybrid titania films by ultraviolet irra- diation. J. Am. Ceram. Soc., 86, 761-64. [13] H. Segawa, H. Misawa, and S. Matsuo, (2004) Fabrica- tion of fine-pitch TiO2-organic hybrid dot arrays using multiphoton absorption of femstosecond pulses. Appl. Phys. A, 79, 407-409. [14] H. Segawa, N. Abrams, T. E. Mallouk, I. Divliansky, and T. S. Mayer, (2006) Fabrication of TiO2-Organic hybrid dot arrays using nanosecond laser interference lithogra- phy. J. Am. Ceram Soc., 89, 3507-3510. [15] T. Kondo, S. Juodkazis, V. Vygantas, and H. Misawa, (2006) Holographic lithography of periodic two- and three- dimensional microstructures in photoresist SU-8. Opt. Exp., 14, 7943-7953. [16] J. Xing, X. Dong, W. Chen, X. Duan, N. Takeyasu, T. Tanaka, and S. Kawata, (2007) Improving spatial resolu- tion of two-photo microfabrication by using photoinitia- tor. Appl. Phys. Lett., 80, 131106. [17] T. Kondo, S. Matsuo, S. Juodkazis, and H. Misawa, (2001) Femtosecond laser interference technique with diffractive beam splitter for fabrication of threedimen- sional photonic crystals. Appl. Phys. Lett., 79, 725. [18] J. N. Israelachvili, (1992) Intermolecular and surface forces. Academic Press Ltd., London, 2nd ed.. [19] (1984) JSME mechanical engineers’ handbook, A4: Strength of materials. Japan Society of Mechanical En- gineers, Tokyo. [20] D. R. Lide ed., (1998) CRC handbook of chemistry and physics, 82th Ed., CRC Press Inc., Boca Raton, Florida. [21] K. Kintaka, J. Nishii, and N. Tohge, (2000) Diffraction gratings of photosensitive ZrO2 gel films fabricated with the two ultraviolet beam interference method, Appl.Opt., 39, 489-493. [22] N. Tohge and Y. Takama, (1999) Direct fine-patterning of PZT thin films using photosensitive gel films derived from chemically modified metal alkoxides. J. Mater. Sci.: Mater. Electron., 10, 273-277. |

