Advances in Bioscience and Biotechnology
Vol.4 No.4(2013), Article ID:30313,14 pages DOI:10.4236/abb.2013.44075
Tolerance and biosorption capacity of Zn2+, Pb2+, Ni3+ and Cu2+ by filamentous fungi (Trichoderma harzianum, T. aureoviride and T. virens)
![]()
1Biotechnology Research Institute, Universiti Malaysia Sabah, Jln UMS, Kota Kinabalu, Malaysia
2Borneo Marine Research Institute, Universiti Malaysia Sabah, Jln UMS, Kota Kinabalu, Malaysia
3Biology Department, Faculty of Science, Universiti Putra Malaysia, Jln UPM, Serdang, Malaysia
Email: *shafiqpab@ums.edu.my, *shafiq.siddiquee@gmail.com
Copyright © 2013 Shafiquzzaman Siddiquee et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received 19 January 2013; revised 28 February 2013; accepted 6 April 2013
Keywords: Bioaccumulation; Biosorption; Heavy Metals; Trichoderma Species; Bioremediation; Wastewater Treatment
ABSTRACT
Heavy metal pollution has become a serious environmental issue in the last few decades. There is a need to develop potential technology that can remove toxic heavy metals ions found in polluted environments. This study was undertaken to determine the resistance levels of different concentrations of heavy metals using filamentous fungi of Trichoderma aureoviride, T. harzianum, and T. virens. Based on the results, the T. virens strain T128 gave the highest tolerance ability for Ni3+ and Pb2+ in a 1200 mg/L concentration. The accumulation and uptake capacity was determined by the maximum removal of Pb2+, Cu2+, and Ni3+ by a T. harzianum in liquid medium when compared to other fungi. The metal removal occurred at a concentration of 500 mg/L and was 13.48 g/g for Pb2+, 3.1254 g/g for Cu2+ and 0.8351 g/g for Ni3+. For Zn2+, the highest tolerance and uptake capacity of metal was recorded at 3.1789 g/g by T. virens.
1. INTRODUCTION
Environmental degradation is a global phenomenon today. Nevertheless, it is significantly more deleterious in the developing countries that house some of the largest populations of human inhabitants over relatively small areas. With an increasing population, the demand for development, especially in the areas of agricultural and marine sectors, is required for economic growth and industrialization. The accelerated growth in the agricultural sector through the enhanced dependency on fertilizers, pesticides, and chemical uses as well as the development of industrialization has resulted in an overwhelming effect on the environment. This is because all the related biota and processes are not given enough time to recover for their optimal conditions.
Due to the recent development of industries, heavy metal pollution has become one of the most serious environmental concerns today. Heavy metals have drastically increased in the environment and are also found in nature and industrial wastewater. They naturally occur in varying concentrations as heavy metal elements in all ecosystems. Elements or compounds having different properties, such as Zn, Cu, Ni, Fe, and Mn are essential trace elements in living organisms too [1].
However, if these metals accumulate at high levels or are ingested in greater amounts than the required concentration, they can cause serious problems in living organisms, including human beings. Alleviating the concentrations of heavy metals in water is crucial to the quality of life of aquatic organisms. Besides, heavy metals can cause severe toxic effects in exposed plants, animals, and humans when present in excessive concentrations [1]. Due to their capability of binding with proteins and other bio-molecules, they act as potent enzyme inhibitors that hamper biochemical processes and compromise DNA and cell membrane integrity. With the rapid development of many industries, such as mining, surface finishing, energy and fuel production, fertilizers, pesticides, metal surface treating, electric appliance manufacture, and others activities, wastes containing metals are directly or indirectly discharged into the environment, producing serious environmental pollution and posing a significant threat to the health of humans, soil, and sediments [2,3].
Therefore, researchers have manipulated the microbemetals interactions, including reduction for anaerobic respiration, reduction for detoxification, biosorption, bioleaching, bioaccumulation, and biomineralization [3,4]. It is important for researchers to explore microbes from the ecological environment for use in metal biosorption. The biosorption process has been studied extensively using microbial biomass as a biosorbent for heavy metal removal. Biosorption is the process that involves the use of microbes to detoxify and control the environmental contaminants. Based on the interaction between living and non-living microorganisms, metallic ions in the system clean up the polluted sites. Consequently, the combined living and dead biomass can be entirely utilized in the biosorptive process as it often demonstrates clear tolerance toward metals and other adverse conditions, such as low pH [3,5].
Fungi are a versatile group as they can adapt and grow under various extreme conditions of pH, temperature, and nutrient availability as well as a high metal concentration [6,7]. Qazilbash [8] reported that fungi are one of the most suitable organisms used to run bioremediation. It is also able to tolerate and detoxify heavy metal ions from contaminated sites by several mechanisms, including valence transformation, extra and intracellular precipitation, and active uptake. Kapoor et al. [9] reported that living and dead cells of fungi are able to remove heavy metal ions from aqueous solutions and the uptake of heavy metal ions by fungal microorganisms may offer an alternative method for their removal from wastewater. The removal of heavy metal ions by the fungal biomass is much better compared to the removal of heavy metal pollutants by conventional adsorbent techniques including use of activated carbon, coal, or ion exchanges [8]. The increasing use of fungal biomass in numerous biotechnological processes is widely documented, especially in the fermentation and bioremediation industries, which include the production of antibiotics, enzymes, and industrial acids.
Investigations and studies carried out by some researchers have reported that the metal uptake mechanisms of free-living fungi involves three distinct processes that include the extra cellular uptake through an ion exchange process, intracellular accumulation, and the trapping of particulates rich in metal contents [7,10]. Researchers have found good agents for the biosorption of heavy metal ions using Trichoderma autroviride, T. harzianum and T. virens; these are also being used for cleaning polluted areas [2,3,7,11]. Comparative studies done by Filipovic et al. [12] report that the Aspergillus niger strain showed better biosorption capabilities of Cu2+, Zn2+ and Ni2+, at pH ranging from 4 to 6. Michael et al. [13] also found that A. niger is able to remove 70% of Zn2+ and 91% of Cu2+ from the wastewater.
The tolerance and ability to detoxify metals by several mechanisms, which include valence transformation, extra and intracellular precipitation, and active uptake are among the reasons that they considered as potential alternatives to synthetic resins for the remediation of dilute solutions of metals and solid wastes. They are a versatile biosorption group as they can grow and work under extreme conditions of pH, temperature, and nutrient availability as well as high metal concentrations. The resistance toward heavy metals can be defined as the ability of an organism to survive metals toxicity by means of mechanisms produced in direct response to the metal group concerned. Compared to other biosorption agents, fungi biomasses have a high percentage of cell wall materials that show excellent metal binding properties in which they can take considerable quantities of heavy metals even in the absence of any physiological activity.
Many definitions for heavy metals can be found but scientists define heavy metals according to three different criteria-their density, atomic number, or chemical properties. According to Radojevic et al. [14], heavy metals can be defined as metals having a density of more than 5 g/cm3, which is five times denser than water [15], and can be categorized as metals with an atomic number of higher than 20 [16], excluding alkaline metals, alkaline earth, lanthanides, and actinides [17].
The accumulation of different types of heavy metals, such as Pb2+, Cd2+, Cu2+, Ni3+, Zn2+, and Mn2+, in sea water not only contaminates the water but also the soil. As a result, it also affects the sources of drinking water and builds up a dangerous concentration of heavy metals in grains and vegetables. There are several cases that have involved heavy metals contamination, with the most popular case occurring in 1963 in Minamata Bay, Japan. Its tragedy is related to the high amount of mercury concentration in shellfish, which was consumed by locals near the Minamata Bay. The spread of diseases happened due to the chemical substances released and discharged without control by the chemical factory that operated near the bay [18]. High amount of mercury concentration was discharged into the sea as wastewater and affected the marine food chains, such as shellfish and other seafood that can build up high concentrations of mercury and become poisonous to consumers [18].
Heavy metals are one of the serious environment pollutants as they derive from both direct sources, such as industrial effluents and sludge dumping and indirectly through highway runoffs. As a result of these problems, a great interest in metal-microbe interactions has arisen in recent years from researchers as well as industrialists who wish to find suitable methods to remove as well as recover and stabilize the heavy metals in seawater, soil, and effluents [19]. In the present study, the Trichoderma species have been investigated for their ability to grow in the presence of four heavy metals, that is, Pb2+, Cu2+, Ni3+, and Zn2+, using different concentrations. Solid and liquid medium were used for the determination of the resistance levels of different concentrations of heavy metals. The maximum biosorption capacity based on dry weight was determined by varying the concentrations of four heavy metals ions in aqueous solution.
2. MATERIALS AND METHODS
2.1. Trichoderma Samples
Trichoderma samples were collected from the Biotechnology Research Institute (IPB), Universiti Malaysia Sabah (UMS), which were morphologically and genetically previously identified by Siddiquee et al. [20-22].
2.2. Fungal Growth Screen
The Cu2+, Pb2+, Zn2+, and Ni3+ stock solutions were prepared by dissolving copper sulphate pentahydrate (CuSO4·5H2O), lead nitrate (Pb(NO3)2), Zinc sulphate (ZnSO4·7H2O), and nickel oxide (Ni2O3) (analytical grade, Merck) in distilled water. The solid medium was prepared by pouring 4 mL of stock solution of Cu2+/Pb2+/ Zn2+/Ni3+, followed by 16 mL of sterilized potato dextrose agar (PDA) into a universal bottle to obtain the desired heavy metal concentrations of 100 mg/L, 300 mg/L, 500 mg/L, 700 mg/L, 900 mg/L, and 1200 mg/L, respectively. The universal bottle was then gently shaken to homogenize the solution. The PDA incorporated with the above-mentioned heavy metal solutions were then immediately poured into the petri dishes and swirled gently. The medium was inoculated with 3 mm diameter of agar disc cut from the edge side of an actively growing pure culture of Trichoderma species samples and were observed daily until mycelia growth covered the whole petri dish. Three replicate plates were done for each treatment, and the experiment was repeated thrice under room temperature conditions (28˚C ± 2˚C, 12 h daylight and 12 h darkness).
2.3 Toxicity Test of Selected Fungi Species
Potato dextrose broth (PDB) (Difco, USA) was prepared as per the manufacturer’s instructions (10 g of PDB media were decanted into individual 500 mL Erlehnmeyer flasks with cotton wool stoppers placed over the flask mouths and then autoclaved at 121˚C, 1.4 kg·cm−1 for 15 min). The used stock solution of either Cu2+ or Pb2+ or Zn2+ or Ni3+ (10 mL) was added to the medium in each 250 mL conical flask separately to reach the required concentrations (100, 200, 300, 400 and 500 mg/L) in a volume of 100 mL. The medium was later inoculated with six disks of pure culture of the selected Trichoderma samples taken from the edge side of an actively growing PDA culture [23]. The initial concentrations of each heavy metal in each conical flask were checked using an inductive coupled plasma spectrometer (ICPOES Model Optima 2000 DV, PerkinElmer, United States) before fungal inoculation [10]. Cultures were incubated under room temperature conditions (28˚C ± 2˚C, 12 h daylight and 12 h darkness). Three replicates were done for each heavy metal, and the experimental works were repeated three times.
2.4. Determination of Fungal Growth at Different Concentrations of Selected Heavy Metals
After seven days of incubation, flasks containing fungal biomass at different concentrations of heavy metals were harvested and filtered through Whatman No. 1 filter paper. Biomass samples were rinsed several times with distilled water and left in an oven at 50˚C until a constant weight was achieved and defined as dry biomass (g/L) [6].
2.5. Removal of the Heavy Metals at Different Concentrations by the Selected Fungi
According to the method proposed by Lopez and Vazquez [10], concentrations of heavy metals in liquid cultures were measured with ICP-OES before fungal inoculation. The amount of heavy metal uptake (Q, mg/g) was calculated by using the following equation [3,10,24]:
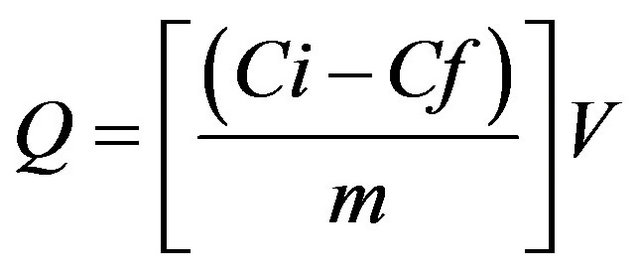
where Q (mg/g) = mg of metal ions uptake per gram biomass Ci (mg/L) = initial metals concentration Cf (mg/L) = final metals concentration m (g) = amount of dry biomass V (L) = volume of the medium
2.6. Data Analysis
Statistical analyses of data were performed by using the Statistical Package for the Social Science (SPSS), version 21.0. One way analysis of variance (ANOVA) was carried out to show the significance difference at p ≤ 0.05. Tukey test was used to compare the means.
3. RESULTS AND DISCUSSIONS
3.1. Screening for the Most Tolerant Fungi
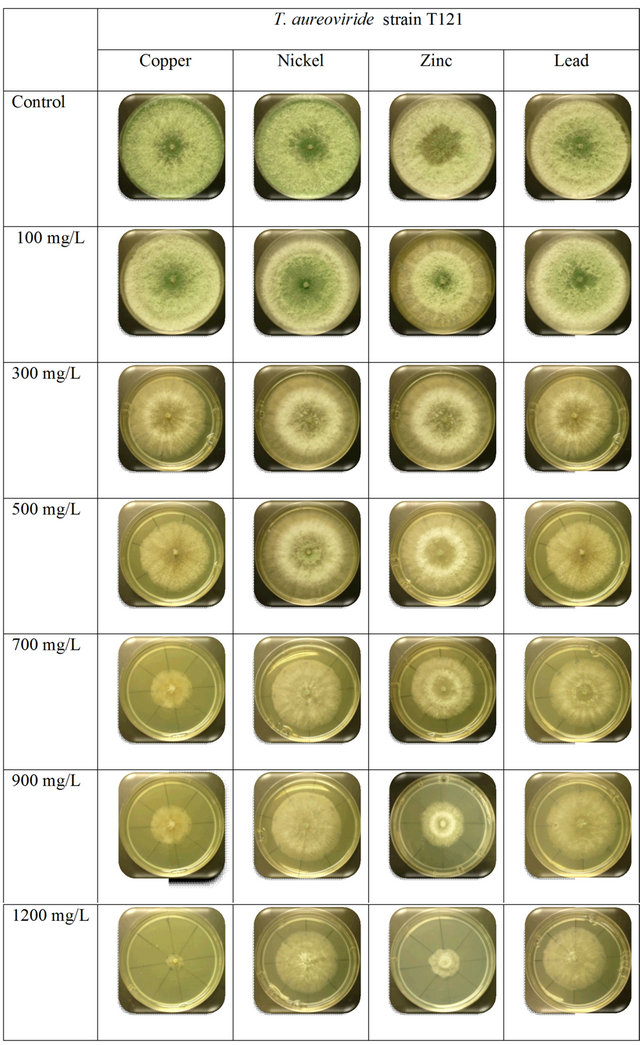
This research work was designed to determine the capability and absorption methods of the selected Trichoderma species for the different concentrations of heavy metals. According to Gadd [25], it has been reported that exposure of filamentous fungi to heavy metals can lead to physiological adaptation or the selection of mutants and such changes may be associated with increased metal absorption capacity. Filamentous fungi are able to grow in the presence of heavy metals even at higher concentrations. The selected fungi that were used belong to the genera Trichoderma which consisted of T. harzianum, T. aureoviride, and T. virens. In the present study, the selected fungi were observed at different concentrations of 100 mg/L, 300 mg/L, 500 mg/L, 700 mg/L, 900 mg/L, and 1200 mg/L in Cu2+, Zn2+, Pb2+, and Ni3+ heavy metals on PDA. From the analysis of Cu2+ ion in different concentrations, it was found that T. harzianum strain T32 obtained the highest mycelia growth values, followed by T. harzianum strain T30, T. virens strain T128, and T. aureoviride strain T121 within 3 days of incubation. At 100 mg/L-1200 mg/L concentration of Cu2+, the highest mycelia growth values were 7.5 cm for 100 mg/L, 7.2 cm for 300 mg/L, 6.2 cm for 500 mg/L, 5.4 cm for 700 mg/L, 2.9 cm for 900 mg/L, and 2.1 cm for 1200 mg/L, respectively, by using T. harzianum strain T32. When comparing with other fungi, T. harzianum strain FA30 recorded the lowest growth rate. Even though the same species of T. harzianum was used, a different strain exhibited different ability and biosorption capacity with different concentrations of Cu2+. This result suggests that different strains of even the same species do not exhibit the same tolerance capacity. After five days, all fungi fully covered the whole petri dish at concentration ranges of 100 mg/L - 700 mg/L. Concentrations of 900 mg/L and 1200 mg/L of Cu2+ showed completely grown mycelia within 20 days. Nur Liyana et al. [2] reported that Aspergillus niger has a high tolerance ability in the presence of Cu2+ than the P. simplicissimum species with a concentration of 1000 mg/L. Zapotoczny et al. [23] also reported that Acremonium pinkertoniae could tolerate up to 600 mg/L concentration of Cu2+ in malt agar. Anand et al. [6] reported that there is no growth at 300 mg/L of Cu2+ using T. viride species. Tsekova and Todorova [26] also reported similar level of tolerance to Cu2+ ions by A. niger B-77 strain where 300 mg/L of the Cu2+ ion was inhibitory to the growth of the organism. In comparison with previous research results, we first reported up to 1200 mg/L concentration of maximum tolerance on PDA.
By increasing the initial concentration of Zn2+ up to 1200 mg/L in the PDA culture medium, significantly different mycelia growth rate was found for those fungi. In Table 1, T. virens strain T128 showed the highest growth rate value compared to other fungi. Even though other fungi had a slow growth rate, they still showed tolerance with a high Zn2+ concentrations. Maximum concentrations (1200 mg/L) of Zn2+ with a value of 4.40 mg/L of T. harzianum strain FA30, 2.70 mg/L of T. harzianum strain T32, 4.70 mg/L of T. aureoviride, and 4.81 mg/L of T. virens strain 128 were found. In 100 mg/L 500 mg/L concentrations of Zn2+, the mycelia growth of T. harzianum strain T32 was higher than that of T. aureoviride strain T121; after that, with increasing concentrations of 700 mg/L - 1200 mg/L, the mycelia growth decreased. Based on the heavy metal study of Zn2+, the T. virens strain T128 showed the best selected filamentous fungi to tolerate and reduce the toxicity at low to high concentrations of metal solutions.
With increasing concentrations of Pb2+ from 0 mg/L - 1200 mg/L, the mycelia growth of the selected fungi in the PDA media decreased. The highest mycelia growth was recorded for the T. harzianum strain T32 at different concentrations for 3 days whereas compared to other fungi, almost the same growth rates were recorded. These fungi were determined as being the most tolerant fungi for Pb2+ when compared to other heavy metal ion concentrations. The maximum tolerance of Pb2+ for metal solution occurred at 1200 mg/L concentration with a value of 6.10 mg/L for T. harzianum strain FA30, 6.90 mg/L for T. harzianum strain T32, 6.71 mg/L for T. aureoviride, and 6.60 mg/L for T. virens, respectively.
There was fluctuation of the data as the fungal mycelia growth of T. harzianum decreased with the increasing concentration of Ni3+ when compared to T. virens. This result indicated that the T. virens is the most suitable fungus for biosorption and tolerance to Ni3+ pollutants because it can be adapted to such environments, followed by other fungal species.
According to Kapoor et al. [9], reported fungi are well-known to be tolerant and even able to accumulate Pb2+, Cu2+, Zn2+, Ni3+, and Cd2+ concentrations. Some species of fungi can accumulate a broad range of heavy metal contaminations, while others are specific for certain types of heavy metals. Different types of fungi, such as Trichoderma species that show high tolerance to toxic metals, may be useful in metal recovery systems.
This study showed that the T. virens strain T128 had the highest tolerance toward Ni3+ and Pb2+ concentrations when compared to other heavy metals, even at the maximum concentration of 1200 mg/L. For Cu2+ heavy metal ion solutions, T. harzianum recorded the highest average growth rate compared to other fungi. The colony growth rate observed in the T. aureoviride strain T121 after 5 day cultures on a solid medium containing Cu2+ solutions at low to high concentrations also supported fungal growth. According to Baldrian et al. [27], different ranges of heavy metals tolerance on solid medium may be caused by the metal binding property of various agar medium or the diverse ability of different fungi.
3.2. Comparison between the Growth Rates of Selected Fungi at Different Concentrations of Heavy Metals
T. harzianum strain FA30 showed the most tolerant and
Table 1. Growth rate of fungi on PDA at different concentrations of Cu2+, Zn2+, Pb2+ and Ni3+.
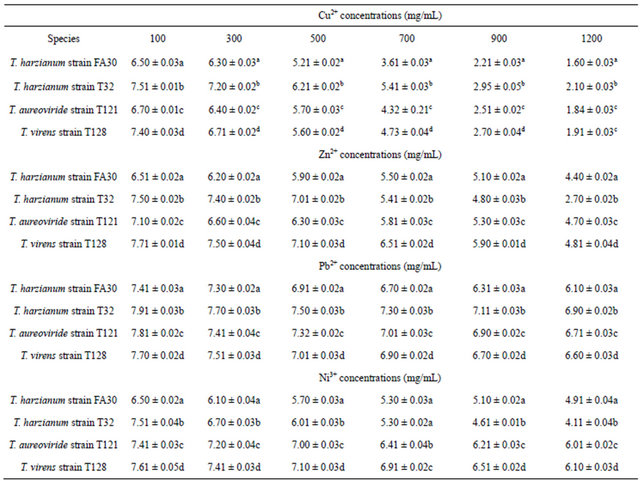
The growth rate of fungi were compared by using one-way ANOVA and Tukey test for different concentrations of Cu2+, Zn2+, Pb2+ and Ni3+. Means with different letters in each column of Cu2+, Zn2+, Pb2+ and Ni3+ concentrations were significantly different at p ≤ 0.05.
higher absorption skill with Ni3+ and Pb2+ concentrations, as shown in supplementary Figure S1. The increasing growth rate was found for Pb2+ and Ni3+ when compared to Cu2+ and Zn2+ concentrations, and no visible appearance changes occurred in the mycelium or sporulation. Subheading on screening for the most tolerant fungi results also indicated that T. harzianum strain T32 showed high ability and tolerance skills to absorb the toxicity of heavy metals when compared to T. harzianum strain FA30. T. harzianum strain T32 always recorded faster growth in the heavy metals consisting of Ni3+, Pb2+, and Zn2+ concentrations (supplementary Figure S2).
T. aureoviride strain T121 showed the highest tolerance toward the concentrations of Pb2+ and Ni3+, as shown in supplementary Figure S3. This species strain was fully grown at low concentrations of 100 mg/L - 500 mg/L, but no change occurred in physical appearances. Based on Figure 1, the T. virens strain T128 showed almost the same growth rates at low concentration when the concentration increased for Cu2+ and Zn2+, the growth rate of mycelia were decreased compared to Pb2+ and Ni3+ concentrations. The concentrations ranged from 300 mg/L to 1200 mg/L of Cu2+, the mycelia physical appearance changed as it turned from green to a darker colour. These similar changed results were also observed by Venkateswerlu et al. [28], who determined that there are blue colored particles in the presence of Cu2+ in Neurospora crassa and Cunninghamella blackesleeana and they proposed that this was caused by the binding of the Cu2+ ions to the protein in the cell wall of the mycelium. Anand et al. [6] tested in agar medium at concentration ranges of 1000 mg/L - 5000 mg/L of Cu2+; the mycelia turned blue both on the agar media and in broth culture at all concentrations of copper, which is due to the binding of Cu2+ to the fungal cell wall.
3.3. Metal Uptake Capacity by Selected Filamentous Fungi
Dry biomass of T. harzianum, T. virens, and T. aureo-
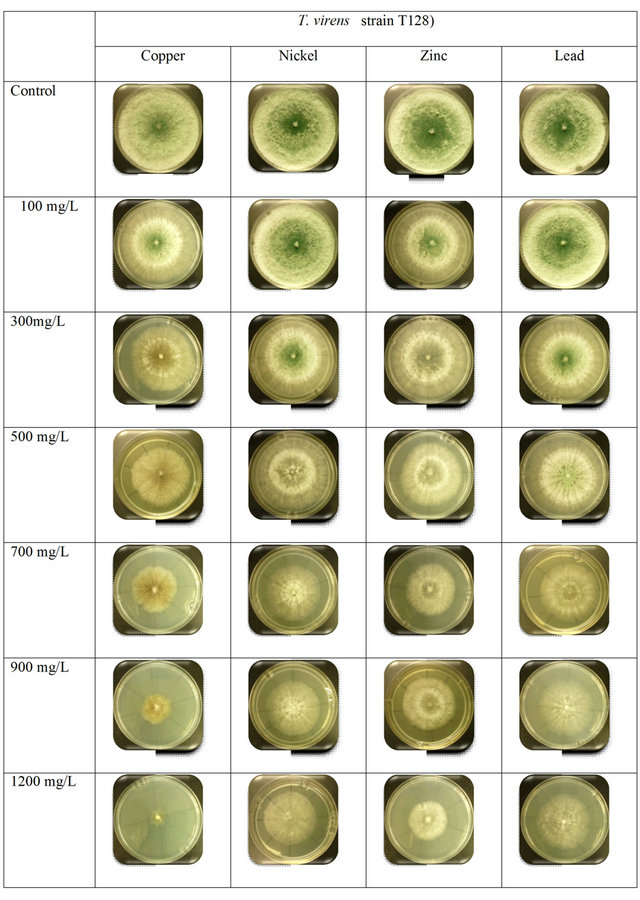
Figure 1. Comparison between the growth rates of T. virens strain T128 with different concentration of heavy metals.
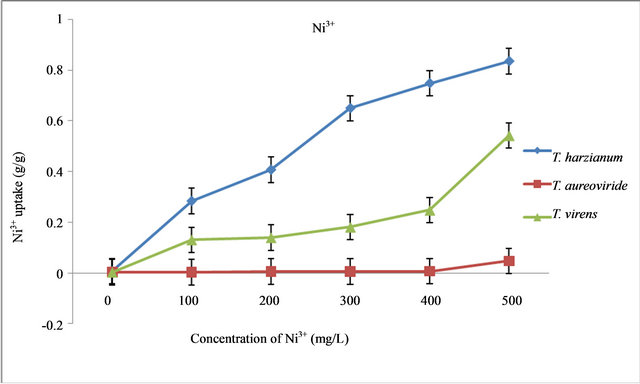
Figure 2. Uptake capacity of Ni3+ with different concentrations of Ni3+ in liquid medium by T. virens, T. aureoviride and T. harzianum.
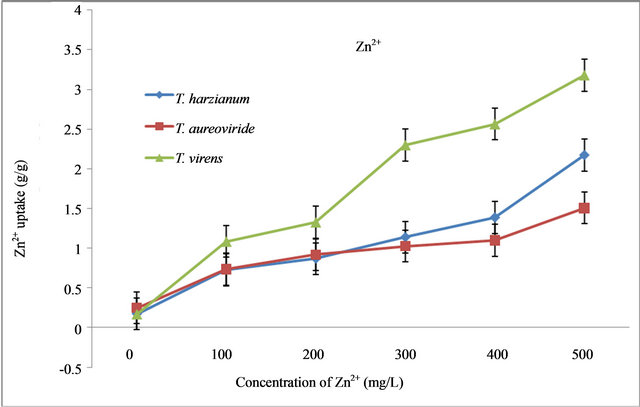
Figure 3. Uptake capacity of Zn2+ at different concentrations of Zn2+ in liquid medium by T. virens, T. aureoviride and T. harzianum.
viride decreased with increasing initial concentrations of heavy metals ( Ni3+, Zn2+, Pb2+, and Cu2+) for 7 days at room temperature conditions of 28˚C ± 2˚C. With increasing concentrations of Ni3+, the dry biomass values of those fungi decreased. The result indicated that the highest biomass values of those fungi were at the control concentration level while the lowest biomass values were recorded at 500 mg/L of Ni3+ (Table 2). It was found that the T. aureoviride strain T121 obtained the highest dry biomass values of 0.3518 g/L, followed by the T. virens strain T128 (0.1963 g/L) and T. harzianum strain T32 (0.137 g/L) at 500 mg/L concentration. The lowest dry biomass of T. harzianum strain T32 compared to the other fungi, however, gave the highest Ni3+ uptake capacity (Figure 2), even though the dry biomass of the T. aureoviride strain T121 recorded the highest values, but gave the lowest Ni3+ uptake capacity.
By increasing the initial concentration (0 mg/L) of Zn2+ up to 500 mg/L in the liquid culture medium using selected fungi, the level of dry biomasses decreased. A remarkable decrease of the biomass values was observed in the T. harzianum strain T32 while compared with the T. virens strain T128 and the T. aureoviride strain T121. From 0 mg/L - 500 mg/L concentration of Zn2+, the highest biomass values occurred between 0.5 g/L - 0.3 g/L by using T. aureoviride, followed by T. harzianum and T. virens (Table 2). With the increase of Zn2+, the metal uptake capacity by T. harzianum, T. virens, and T. aureoviride were always increasing. The maximum uptake of Zn2+ from the metal solution occurred at 500 mg/L concentration with a value of 3.1789 g/g for T. harzianum, 2.1719 g/g for T. virens, and 1.5065 g/g for T. aureoviride, as shown in Figure 3.
With different concentrations of Pb2+, dry biomass values of T. aureoviride were higher compared to T. virens and T. harzianum (Table 2). The dry biomass obtained for T. aureoviride was 0.1078 g/L and followed by 0.0981 g/L for T. virens and 0.0244 g/L for T. harzianum at 500 mg/L Pb2+ concentration. The noticeable decreased dry biomass value was found for T. harzianum at different concentrations of Pb2+ (0 mg/L - 500 mg/L). The growth and metal removal properties of the three fungi were highly affected by initial metal ion concentrations. The highest Pb2+ uptake (13.48 g/g) at 500 mg/L of the initial Pb2+ concentration was obtained for T. harzianum, whereas T. virens and T. aureoviride were only able to remove 7.9126 g/g and 4.6683 g/g of Pb2+, respectively (Figure 4).
Based on Table 2, dry biomass of T. virens, T. aureoviride, and T. harzianum decreased with increasing initial concentrations of Cu2+. The result indicated that the highest biomass values of those fungi were at the control concentration while the lowest biomass values was recorded at 500 mg/L of Cu2+. It was found that at 500 mg/L of Cu2+, the maximum Cu2+ uptake occurred and the highest dry biomass was obtained by T. aureoviride (0.3505 g/g), followed by T. harzianum (0.1678 g/g), and T. virens (0.1552 g/g) (Figure 5).
These three species of fungi are tested to determine the Minimum Inhibitory Concentration (MIC) for the different types of heavy metals. The following order of toxicity found Cu2+ > Zn2+ > Ni2+ > Pb2+ by the selected fungi. The MIC values suggest that the resistance level against individual metals was dependent on the fungi.
Table 2. Dry biomass values of fungi on PDB at different concentrations of Cu2+, Zn2+, Pb2+ and Ni3+.
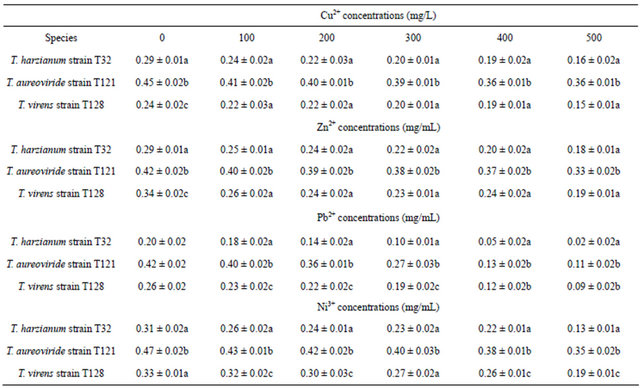
Dry biomass values of fungi were compared by using one-way ANOVA and Tukey test for different concentrations of Cu2+, Zn2+, Pb2+ and Ni3+. Means with different letters in each column of Cu2+, Zn2+, Pb2+ and Ni3+ concentrations were significantly different at p ≤ 0.05.
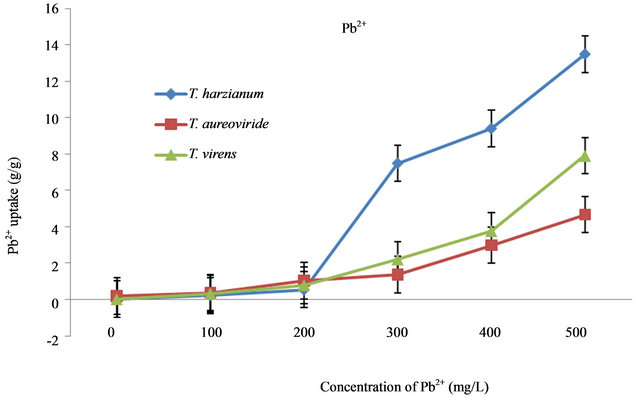
Figure 4. Uptake capacity of Pb2+ with different concentrations of Pb2+ in liquid medium by T. virens, T. aureoviride and T. harzianum.
The growth and metal removal properties of these fungi were highly affected by the initial concentration to increase different heavy metals concentrations. Based on the results, increasing Pb2+ concentrations may lead to the decrease of microbial growth biomass. At 500 mg/L, where the highest Pb2+ uptake occurred, the dry biomass obtained for T. aureoviride was 0.13 g/L and the uptake capacity of Pb2+ concentrations by T. aureoviride was the

Figure 5. Uptake capacity of Cu2+ at different concentrations of Cu2+ in liquid medium by T. virens, T. aureoviride and T. harzianum.
lowest at 4.7 g/g compared to other fungi.
According to Deshmokh and Rai [29], the increase of the uptake capacity of metals may be often associated with toxicity or the increasing permeability of cell membrane on account of further binding of the metal to exposed intracellular sites, causing the biomass to decrease with elevated metal exposure. Kapoor et al. [9] showed that the lower uptake at higher biomass concentrations can be attributed to the electrostatic interactions of the functional groups at the cell surfaces. The cells at higher concentrations in suspension attach to each other, thus lowering the cell surface area in contact with the solution.
Among the fungi, T. harzianum are able to absorb more amounts of Ni3+ metal concentrations, especially at lower concentrations between 300 mg/L - 500 mg/L. The differences may be due to the larger surface area of T. harzianum biomass for the adsorption processes. Zafar et al. [3] suggested that the main mechanism of heavy metal removal from aqueous solution was a redox reaction between the heavy metal ion concentrations and the fungal biomass. In addition to the removal of metals from solution during growth, the grown biomass cells can absorb metals from solution. The grown biomass cells can absorbs the metals at a high concentration especially where tolerance to metals during growth is fairly low. Kacprzak and Malina [11] reported that due to high growth, more ligands and negative charge are exposed, which in turn attract more positively charged metal ions from the aqueous solution.
Our result also proved that metal uptake occurred as a consequence of physical binding to cell surfaces rather than as an active process, since the mycelia appeared to be partially degraded and the fungal cell wall plays an important role in metal uptake (biosorption). The ability of the Trichoderma species is to tolerate and undergo the biosorption process of toxic metals based on ionic species associating with the cell surface or extra cellular polysaccharide, protein, and chitin. Usually, fungal cell walls contain chitin and chitosan. Chitin and chitosan contents of the fungal cell wall can change during the growth of mycelia and this can account for the variations in the metal-uptake capacity with the fungi [30].
The accumulation and uptake capacity of T. harzianum for Ni3+, Pb2+, and Cu2+ concentrations in liquid medium were found to be highest when compared to T. virens and T. aureoviride, while for Zn2+, the highest tolerance and uptake capacity of metals were recorded by T. virens. It was also showed that these fungi tested above had their own ability to absorb different types of heavy metals depending on the toxicity of each heavy metal. Based on this study, there is a possibility that by increasing the initial concentration, the removal of metals solution also increases due to the increase of metal uptake. These results are in agreement with those of Yalcin et al. [31], who reported that a higher initial concentration provided an important driving force to overcome all mass transfer resistances between the heavy metals solution and the fungi cell wall.
4. SUMMERY
The present study focused on recent evidence that identifies the potential selected filamentous fungi that are capable of tolerating and undergoing biosorption process toward different concentrations of heavy metals. Results support that the selected fungi have been successfully used as the absorbing agent for the removal of metals ion from different concentrations of heavy metals. These selected fungi are able to survive the maximum concentration of 1200 mg/L of heavy metals. Determination of the maximum absorption rate and the uptake capacity of various concentrations of heavy metals can be achieved in the following order: Ni3+, Cu2+, Pb2+, and Zn2+, according to T. harzianum, T. virens, and T. aureoviride. It was observed in the experiments that an increase in the initial metal concentrations results in an increase in the metal removal capacity and dry biomass of the biosorbent, which accumulates at very high metal concentration. The metal removal capacity of the fungi touches its peak at these higher metal concentrations and at low metal concentrations of heavy metals; the biosorption capacity of the biosorbent is not fully utilized.
The results in this work also indicated the possibilities that exist for the clean-up of the contamination or pollution of heavy metals with the use of natural resources. The contribution of the present study lies in the determination of the metal uptake by the biomass of naturally occurring microorganisms that are isolated from soil polluted with heavy metals ions. Although field studies are necessary, these results suggest that using these fungi may offer foundations to remediate and reclaim some contaminated or polluted areas.
5. ACKNOWLEDGEMENTS
The authors are grateful for the financial support provided by the Biotechnology Research Institute for this project.
REFERENCES
- Poli, A., Salerno, A., Laezza, G., et al. (2009) Heavy metal resistance of some thermophiles: Potential use of α- amylase from Anoxybacillus amylolyticusas a microbial enzymatic bioassay. Resource Microbiology, 160, 99- 106. doi:10.1016/j.resmic.2008.10.012
- Nur Liyana, I., Nur Ain Izzati, M.Z. and Tan, S.G. (2011) Tolerance and biosorption of copper (Cu) and lead (Pb) by filamentous fungi isolated from a freshwater ecosystem. Journal of Environmental Science, 23, 824-830. doi:10.1016/S1001-0742(10)60475-5
- Zafar, S., Aqil, F. and Ahmad, I. (2007) Metal tolerance and biosorption potential of filamentous fungi isolated from metal contaminated agricultural soil. Bioresource Technology, 98, 2557-2561. doi:10.1016/j.biortech.2006.09.051
- Lovely, D.R. and Philips, E.J.P. (1994) Reduction of chromate by Desulfovibrio vulgaris and its C3 cytochrome. Journal of Environmental Microbiology, 60, 726-728.
- Kapoor, A. and Viraraghavan, T. (1998) Biosorption of heavy metals on Aspergillus niger: Effect of pretreatment. Bioresource Technology, 63, 109-113. doi:10.1016/S0960-8524(97)00118-1
- Anand, P., Isar, J., Saran, S. and Saxena, R.K. (2006) Bioaccumulation of copper by Trichoderma viride. Bioresource Technology, 97, 1018-1025. doi:10.1016/j.biortech.2005.04.046
- Yazdani, M., Chee, K.Y., Faridah, A. and Tan, S.G. (2010) An in vitro study on the adsorption, absorption and uptake capacity of Zn by the bioremediator Trichoderma atroviride. Environmental Asia, 3, 53-59.
- Qazilbash, A.A. (2004) Isolation and characterization of heavy metal tolerant biota from industrially polluted soils and their role in bioremediation. Biological Science, 41, 210-256.
- Kapoor, A., Viraraghavan, T. and Cullimore, D.R. (1999) Removal of heavy metals using the fungus Aspergillus niger. Bioresource Technology, 70, 95-104. doi:10.1016/S0960-8524(98)00192-8
- Lopez, E.E. and Vazquez, C. (2003) Tolerance and uptake of heavy metals by Trichoderma atroviride isolated from sludge. Chemosphere, 50, 137-143. doi:10.1016/S0045-6535(02)00485-X
- Kacprzak, M. and Malina, G. (2005) The tolerance and Zn2+, Ba2+ and Fe2+ accumulation by Trichoderma atroviride and Mortierella exigua isolated from contaminated soil. Canadian Journal Soil Science, 85, 283-290. doi:10.4141/S04-018
- Filipovic, K.Z., Sipos, L. and Briski, F. (2000) Biosorption of chromium, copper, nickel and zinc ions onto fungal pellets of Aspergillus niger from aqueous solutions. Food Technology and Biotechnology, 38, 211-216.
- Michael, S.P., Classen, J.J. and Payne, G.A. (2001) Aspergillus niger absorbs copper and zinc from swine wastewater. Bioresource Technology, 77, 41-49.
- Radojevic, M., Abdullah, M.H. and Aris, A.Z. (2007) Analisis air. Scholar Press, Selangor. doi:10.1016/S0960-8524(00)00135-8
- Juliana, M.S. (2003) Kajian logam berat zink, plumbum, ferum, kadmium, chromium & kuprum dalam tumbuhan paku larat. disertasi sarjana sains. Universiti Malaysia Sabah, Kota Kinabalu.
- Yu, M.H. (2001) Environment toxicology: Impacts of environmental toxicants on living systems. CRS Press LLC.
- Thanapalasingam, V. (2005) Pollution status of the skudai river system through heavy metals. Master of Science Thesis, Universiti Teknologi Malaysia, Skudai.
- Blackmore, R. and Reddish, A. (1996) Global environmental issues. Hodder and Stoughton, London.
- Sani, R.K., Peyton, B.M. and Brown, L.T. (2001) Copperinduced inhibition of growth of Desulfovibrio deslfuricans G20: Assessment of its toxicity and correlation with those of zinc and lead. Environmental Microbial, 67, 4765-4772. doi:10.1128/AEM.67.10.4765-4772.2001
- Siddiquee, S., Abdullah, F., Tan, S.G. and Rohaza, E. (2007) Phylogenetic relationships of Trichoderma harzianum based on the sequence analysis of the internal transcribed spacer region-1 of the rDNA. Journal of Applied Science Research, 3, 896-903.
- Siddiquee, S., Tan, S.G., Umi Kalsom, Y., Nur Hasan, N.F. and Hasan, M.M. (2012) Characterization of Malaysian Trichoderma isolates using the Random Amplified Microsatellites (RAMS). Molecular Biology Reports, 29, 715-722. doi:10.1007/s11033-011-0790-6
- Siddiquee, S., Umi Kalsom, Y., Hossain, K. and Jahan, S. (2009) In vitro studies on the potential Trichoderma harzianum for antagonistic properties against Ganoderma boninense. International Journal of Food, Agriculture and Environmental, 7, 970-976.
- Zapotoczny, S., Jurkiewicz, A., Tylko, G., Anielska, T. and Turnau, K. (2006) Accumulation of copper by Acremonium pinkertoniae, a fungus isolated from industrial wastes. Microbiological Research, 26, 198-298.
- Fan, T., Liu, Y., Feng, B., Zeng, G., Yang, C. and Zhou, M. (2008) Biosorption of cadmium (II), zinc (II) and lead (II) by Penicillium simplicissimum: Isoterms, kinetics and thermodynamics. Journal of Hazardous Materials, 160, 655-661. doi:10.1016/j.jhazmat.2008.03.038
- Gadd, G.M. (1990) Fungi and yeast metal accumulation: Microbial mineral recovery. McGraw-Hill, New York.
- Tsekova, K. and Todorova, D. (2002) Copper (II) accumulation and superoxide dismutase activity during growth of Aspergillus niger B-77. Z. Naturforch, 57c, 319-322.
- Baldrian, P., Gabriel, J. and Nerud, F. (1996) Effect of cadmium on the ligninolytic activity of Stereum hirsutum and Phanerochaete chrysosporium. Folia Microbiology (Prague), 41, 363-367. doi:10.1007/BF02814716
- Venkateswerlu, G., Yoder, M.J. and Stotzky, G. (1989) Morphological, ultra structural and chemical changes induced in Cunninghamella blakesleeana. Applied Microbiology and Biotechnology, 31, 619-625. doi:10.1007/BF00270806
- Deshmokh, S.K. and Rai, M.K. (2005) Biodiversity of fungi; their role in human life. Science Publisher Inc., Enfield.
- Tobin, J.M., Cooper, D.G. and Neufeld, R.J. (1984) Uptake of metals ions by Rhizopus arrhizus biomass. Applied and Environmental Microbiology, 47, 821-824.
- Yalcin, E., Cavusoglu, K. and Kinalioglu, K. (2010) Biosorption of Cu2+ and Zn2+ by raw and autoclaved Rocella phycopsis. Journal of Environmental Science, 22, 367- 373. doi:10.1016/S1001-0742(09)60117-0
Supplementary Figures

Figure S1. Comparison between the growth rates of T. harzianum strain FA30 with different concentration of heavy metals.
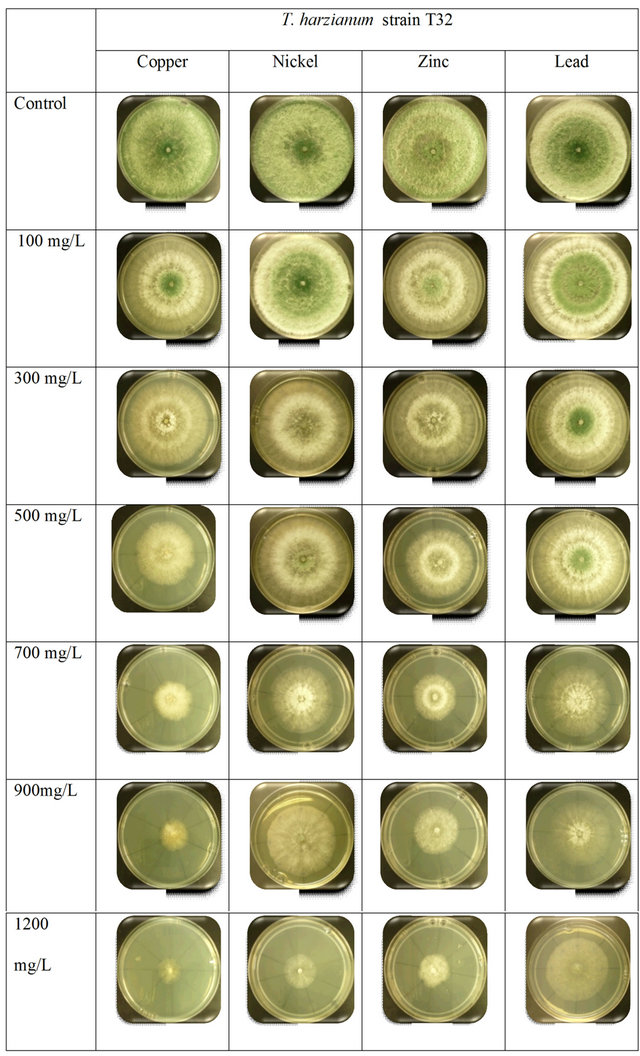
Figure S2. Comparison between the growth rates of T. harzianum strain T32 with different concentration of heavy metals.
NOTES
*Corresponding author.

