Advances in Bioscience and Biotechnology
Vol.2 No.5(2011), Article ID:7836,5 pages DOI:10.4236/abb.2011.25055
Effects of different colours in the visible region on Leishmania tropica
![]()
1Department of Physics, University of Balochistan, Quetta, Pakistan;
2Quaid-e-Azam University, Islamabad, Pakistan;
3Center for Advance Studies of Vaccinology and Bio-Technology (CASVAB), University of Balochistan, Quetta, Pakistan;
4Department of Chemistry, University of Balochistan, Quetta, Pakistan.
E-mail: *saminatazayyen@yahoo.com
Received 19 June 2011; revised 26 July 2011; accepted 10 August 2011.
Keywords: Leishmania tropica; Chromotherapy; Photo Stimulation
ABSTRACT
The aim of this study was to investigate the wavelength-dependency of chromotherapy effects on cutaneous Leishmaniasis parasite growth. Chromotherapy uses visible range radiations to improve healing; however, its effects on parasite are not well understood. Leishmania tropica was irradiated using seven (7) different wavelengths of visible region. Optical density was observed, which showed that red colour (644 nm) wavelength inhibited the growth of parasite while other colour wavelengths also affected the growth of parasite. It is, therefore, suggested that as red colour inhibits the growth of parasite so patients suffering from L. tropica can be treated with the application of red colour.
1. INTRODUCTION
Leishmania comprises a genus of flagellate protozoan parasites. The parasites are transmitted by the bite of infected female sand flies of the genera phlebotomus and Lutzomyia [1]. Leishmaniasis prevalence is estimated to be 12 million people, and, in developing countries, approximately 350 million people, mainly poor, are at risk of contracting the disease [2]. Nowadays, more and more nonimmune humans are entering areas where Leishmaniasis is endemic [3], and there is an evident increase of patients with human immunodeficiency virus type 1 being co-infected with Leishmania parasites [4].
The New World species cause manifestations of greater variety, of which mucocutaneous Leishmaniasis (MCL) is the most severe. MCL is more likely the result of the spreading of parasites from cutaneous lesions to the mucosal membranes, where they can progressively destroy connective tissues, such as cartilaginous structures, in the nasopharyngeal tract [5,6].
There are two common medicines containing antimony, meglumine antimoniate (Glucantim®) and sodium stibogluconate (Pentostam®), but is painful and expensive, too. It is not completely understood how these drugs act against the parasite; they may disrupt its energy production or trypanothione metabolism [4].
Most of the therapeutic systems induce vibrations indirectly but there are few in which the vibrations are used directly upon the body and Chromotherapy is one of them [7]. Chromotherapy is scientifically referred to as application of parts of visible electromagnetic spectrum for therapeutical purpose [8,9].
A case report has been presented recently in which Leishmaniasis lesion of a patient was treated using 644 nm (red colour) wavelengths [10].
2. MATERIALS AND METHODS
Samples were collected from 6 patients infected with cutaneous Leishmaniasis who didn’t receive any sort of treatment for this problem before. Leishmaniasis tropica promastigotes produced by F.N.A [11], taken from the skin lesion of patients with cutaneous Leishmaniasis in YP culture medium and incubated at 27˚C. YP culture medium consisted of the following ingredients: peptone, 1.00 g; yeast extract, 0.25 g; Na2HPO4, 0.75 g; NaCl, 0.80 g dissolved in 100 ml distilled water. Procedure of homogenization, and sterilization was adopted as given in [12]. The production was checked every other day, cultured twice and shifted to new medium for production. After two weeks promastigotes were counted with Neubauer chamber, 106/ml organisms were shifted into seven universal bottles containing 3 ml YP medium and wrapped in coloured cellophane filter sheets of specific wavelengths as given in Table 1. One bottle was taken as control without any filter sheet. The bottles were irradiated with 60 watt full spectrum incandescent bulb light from a distance of 0.32 meters after placing in the Herasus (Germany) incubator at 27˚C. Optical densities and absorbance of the samples were observed after 10 days of irradiation. The procedure was successfully repeated and observed the same behavior of parasites after irradiating them to different colours in the visible region. The change in samples was also photographed.
3. RESULTS
After initial incubation for production, out of six samples two were found positive and other four were contaminated with bacteria and fungus. Yellow (590 nm) and purple colour (464 nm) increased the size of the parasite (Figures 1 and 2). Red (644 nm), blue (483.5 nm) and violet (400 nm) decreased growth considerably; the size as well as the number of counts of the parasite, with red the most effective (Figures 3-5). Orange colour (610 nm) increased growth incredibly while in green colour (538 nm) the promastigotes appeared to change their size and shape and somewhat converted into rounded form (Figures 5 and 6). In the sample irradiated with red colour wavelength, the parasites were observed as lethargic with the Olympus IM 40-X (Japan) inverted microscope as well as least number of counts was observed both in the image and Optical Density.
4. DISCUSSIONS
It was discovered that Monochromatic Single-Wavelength Light Beams had an excellent therapeutic effect on afflicted cell tissue. This occurs through a process called “Photo-Stimulation.” The low intensity (non-coagulative) visible laser radiation has been successfully used in some areas of medicine (photodynamic therapy of tumors, therapy of infant hyperbilirubinemia, some dermatological diseases, etc.) [13]. Also the therapy with red (632.8 nm) laser light (stimulation of tissue regeneration) used for irradiation of the patients with trophic and indolent wounds has gained acceptance in the clinical practice [14]. The biomodulatory effect can have a positive effect on the repair of cutaneous wounds [15]. Various studies have been carried out that show the effect of monochromatic light on cells, but the research lacks empirical data regarding effects on parasite.
The data provided by this study actually gives first hand knowledge and invite other researchers, also. As the procedure was repeated successfully, so it confirms that this kind of study is reproducible which undoubtedly provides a room for further researches to be carried out on this topic.
We couldn’t perform the counting of Leishmania parasite due to unavailability of QUANTIMENT (Quantitative Microscopic Electron Tomography). This shortcoming of counting the parasites was however dealt by experimental observations of absorbance of visible range radiations (colors) in Leishmaniasis of different categories (Figure A). Figures 1-8 are self explanatory to reflect the image of optical density of Leishmaniasis. L. tropica when exposed to red light (644 nm), the decay in this case followed the Gaussian tail and finally to a ramp function. This is an indication that the cutaneous Leishmaniasis after decay just disappears because the parasite become dead. We developed an empirical relationship for the decay of all categories of Leishmania parasite i.e.
Absorbance 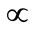
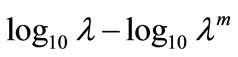
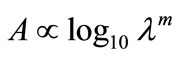
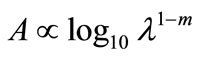
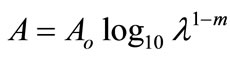
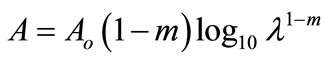
where m is the slope of the ramp function and Aο the extrapolated absorbance. The slope of the ramp function is negative. The Gaussian tail followed by a resonance peaks show that the parasites of cutaneous leishmaniasis reject absorption of energy initially and then attain maximum absorption of energy at 544 nm wavelength for orange and red (Figure A).
Although Low Level Laser Therapy (LLLT) has been used previously in most studies, coherence is not impor

Table 1. Dominant wavelength of monochromatic light measured by Hitachi U-2000 UV-Vis double beam spectrophotometer, spectral bandwith 0.1 nm and ordinates selected 10.

Figure 1. Yellow—parasites irradiated with 590 nm wavelength monochromatic light.

Figure 2. Purple—parasites irradiated with 464 nm wavelength monochromatic light.

Figure 3. Red—parasites irradiated with 644 nm wavelength monochromatic light.
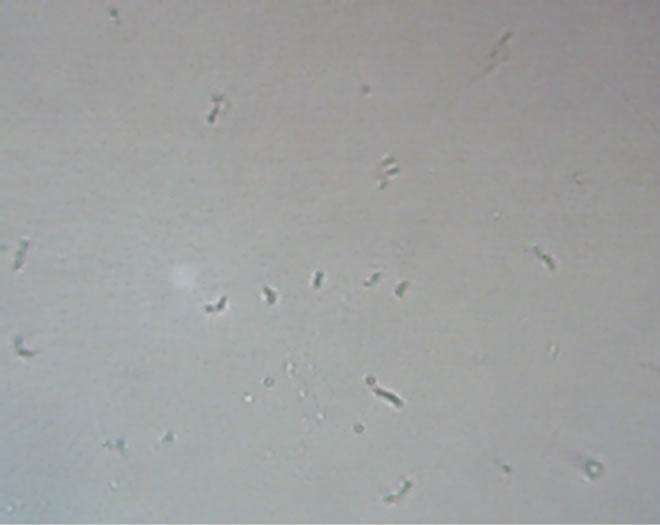
Figure 4. Blue—parasites irradiated with 453.5 nm wavelength monochromatic light.

Figure 5. Violet—parasites irradiated with 400 nm wavelength monochromatic light.
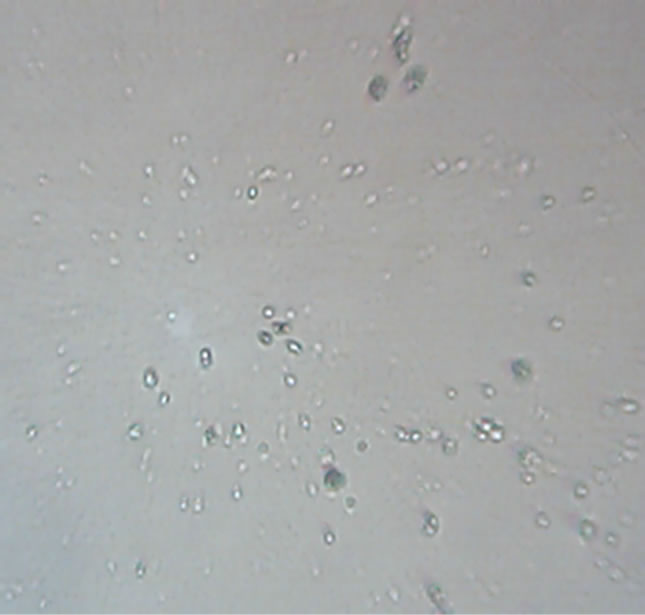
Figure 6. Orange—parasites irradiated with 610 nm wavelength monochromatic light.

Figure 7. Green—parasites irradiated with 538 nm wavelength monochromatic light.
Figure 8. Control—parasites untreated.
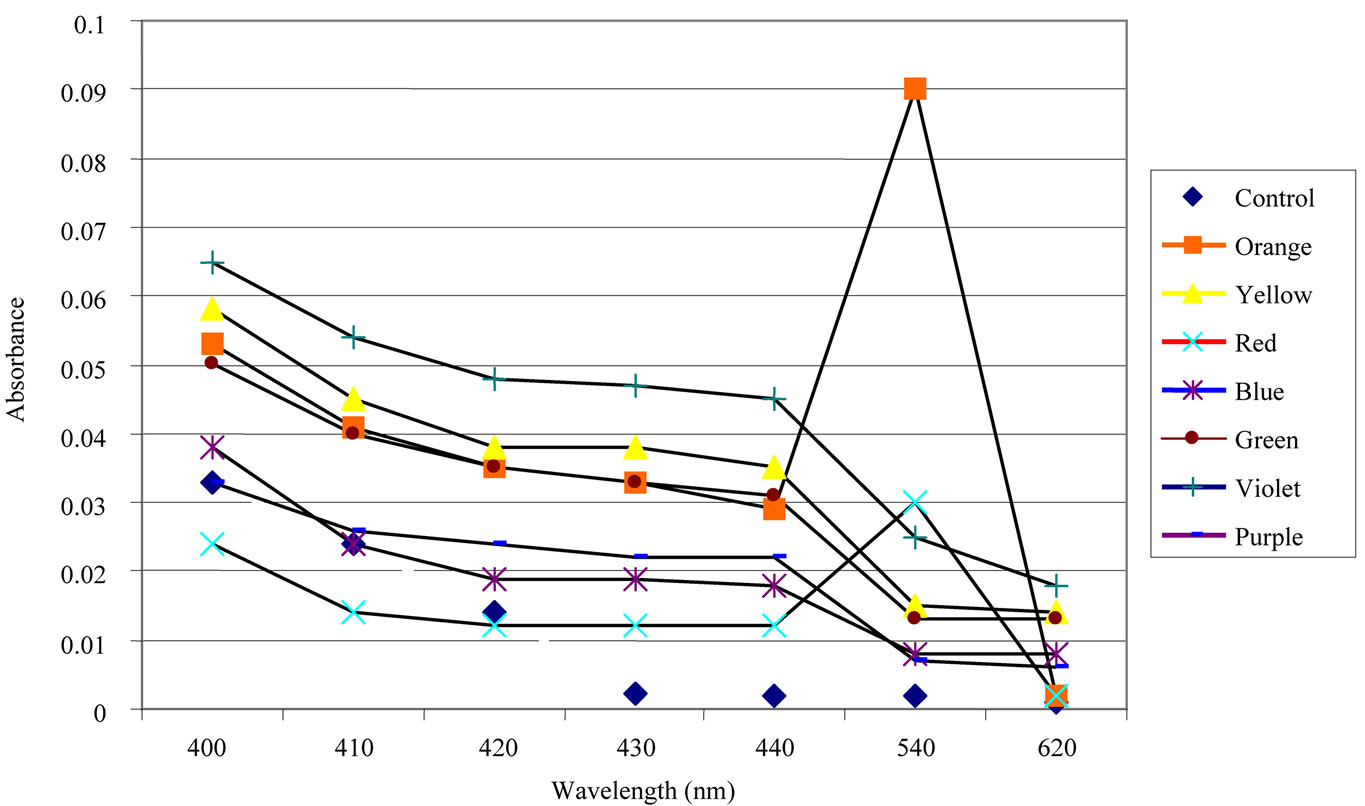
Figure A. Absorbance in samples after monochromatic light irradiation.
tant when photo-biological effects are expected because both coherent and non-coherent light have been shown to be effective [16]. In our study it is observed that red light showed a great change to inhibit the growth of Leishmania tropica organisms. From this study it is evident that cutaneous Leishmaniasis can be eliminated with higher wavelength, i.e., at 644 nm and with least energy (filtered light beams).
5. CONCLUSIONS
Red color (644 nm) inhibits the growth and become responsible for the decay of leishmania parasite while orange color (610 nm) increases the growth of parasite. Undoubtedly this makes the procedure of chromotherapy for treatment of leishmaniasis cost effective and easy approachable. The response of Leshmania parasite to each color is unique and this confirms Chromotherapy (with 644 nm wavelength), to be very easily manageable by the patient with no problems during the treatment. This kind of study opens new doors for research in biosciences and in bio technology.
REFERENCES
- Van der Meide, W.F., et al. (2005) Quan-titative nucleic acid sequence-based assay as a new molecular tool for detection and quan-tification of leishmania parasites in skin biopsy samples. Journal of Clinical Microbiology, 43, 5560-5566. doi:10.1128/JCM.43.11.5560-5566.2005
- World Health Organization (1990) The leish-maniasis. World Health Organization Technical Report Series, 753, 154.
- Berman, J.D. (2005) Recent developments in Leishmaniasis: Epidemiology, diagnosis, and treatment. Current Infectious Disease Reports, 7, 33-38. doi:10.1007/s11908-005-0021-1
- Alavr, J., Canavate, C., Gutierrez-Solar, B., Jimenez, M., Laguna, F., Lopezvelez, R., Molena, R. and Moreno, J. (1997) Leishmania and human immunodefi-ciency virus coinfection: The first 10 years. Clinical Microbiology Reviews, 10, 298-319.
- Tavares, C.A.P., Fernandes, A.P. and Melo, M.N. (2003) Molecular diagnosis of Leishmaniasis. Expert Review Molecular diagnosis of leishmaniasis, 3, 89-99. doi:10.1586/14737159.3.5.657
- Herwaldt, B.L. (1999) Leishmaniasis. Lancet, 354, 1191- 1199. doi:10.1016/S0140-6736(98)10178-2
- Shamsuddin, K., (1999) Colour therapy. Al-Kitab Publications, Karachi.
- Samina, A. and Raza, S.M. (2005) A critical analysis of chromotherapy and its scientific evolution. Evidence-Based Complementary and Alternative Medicine (ECAM), 2, 481-488. doi:10.1093/ecam/neh137
- Klotsche, C. (1999) Color medicine: The secrets of color/ vibrational healing. Light Technology Publishing, Flagstaff, 51-54.
- Samina, T., Mohsin, S. and Yasinzai, M. (2011) A case history of treatment of cutaneous leish maniasis by chromotherapy. Chinese Medicine, 2, 43-46. doi:10.4236/cm.2011.22008
- Kassi, M., Tareen, I. and Kasi, P.M. (2004) Fine needle aspiration cytology in the diagnosis of cutaneous leishmaniasis. Annals of Saudi Medicine, 24, 93-97.
- Moncu, M.E.N.L., Lu, C.B., Yereli, K., OZbel, Y. and Lgin, A.O.Z.˙(1997) A new experimental in vitro culture medium for cultivation of leishmania species. Journal of Clinical Microbiology, 35, 2430-2431.
- Pratesi, R. and Sacchi, C.A. (1980) Laser in pho-tomedicine and photobiology. Springer Series in Optical Sciences, 22, Springer-Verlag, Berlin.
- Karu, T.I., et al. (1984) Biostimulating action of lowintensity monochromatic visible light: Is it possible. Laser Chemistry, 5, 19-25. doi:10.1155/LC.5.19
- Mendez, T., Pinheiro, A., Pacheo, M., Nascimento, P. and Ramalho, L. (2004) Dose and wavelength of laser light have influence on the repair of cutaneous wounds. Journal of Clinical Laser Medicine & Surgery, 22, 19-25. doi:10.1089/104454704773660930
- Karu, T.I. (1987) Photobiological fundamentals of lowpower laser therapy. IEEE Journal of Quantum Electronics, 23, 1703-1717. doi:10.1109/JQE.1987.1073236

