Food and Nutrition Sciences
Vol. 4 No. 9A1 (2013) , Article ID: 36130 , 4 pages DOI:10.4236/fns.2013.49A1010
Fatty Acid Compositional Studies of Lesser Known Acacia tortilis Seed Oil
![]()
1Phytochemistry Division CSIR-National Botanical Research Institute, Lucknow, India; 2Dayanand Dinanath College, Institute of Pharmacy, Kanpur, India.
Email: ms_sks2005@yahoo.co.in
Copyright © 2013 Manjoosha Srivastava, Shailesh Kumar. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received May 15th, 2013; revised June 15th, 2013; accepted June 22nd, 2013
Keywords: A. tortilis; Seed Oil; Fatty Acid; Cholesterol; Fat
ABSTRACT
A. tortilis is commonly known as “Israeli babool”. The species is exposed to the world for its pods, gum and other aerial parts as fodder &feed for cattles. The plant was used as antidiabetic, antidiarrhoeal, antiasthmatic and also had several other medicinal benefits but the seed oil was not explored to the extent of its real potential. Hence the study was carried out to characterize the seed for oil and analysis of fatty acid composition through GC for quality control and maintenance. Commercial indices of oil were also estimated. Extracted oil is rich in Linolenic acid (50.43%) and Linoleic acid (36.74%). Beside this palmitic, stearic and oleic acids are other fatty acids identified. MUFA, PUFA, SFA and omega-6/ omega-3 ratio were also found to be promising for its potentiality in edible and clinical uses. The investigation thus scopes the utility of oil and prospects the lesser known plant seed as alternative oil source of nutritional and therapeutic importance.
1. Introduction
In recent times, change in eating and dietary habits leads to emergence of several lifestyle disorders like hypertension, obesity, diabetes and other cardio-vascular diseases. However, more than diseased conditions they are disorders as in most of the patients underlying cause is idiopathic. These disorders however could be fatal as one being the cause of other and hence forming a vicious circle. In general sense “fats” are the metabolites which are being cursed for the above conditions, although clinically there are “good fats” and “bad fats” too. Currently, there is great interest in dietary omega-3 fatty acids to promote health and there is stress for improving the ratio by eating less omega-6 and more omega-3 fat in food and other dietary sources, as diet is the only source of the same and body cannot make it [1]. These symptomatic disorders can be cured/controlled by synthetics but long term ingestion may result in immediate or delay adverse effect. Hence the need of safe and effective counterpart is always a point of concern in recent times and the plant based on therapeutics can be the best alternatives.
Acacia is a genus of shrubs and trees which includes about 135 species today, most of them are widely spread through out the arid and semi-arid tropics either as pure stands or in mixtures with allied woody species [2]. A. tortilis, commonly known as umbrella thorn belonging to family fabaceae (sub family Mimosoideae) is a medium umbrella-shaped tree. Pods are flat, glabrous, coiled into a spring-like array. It is reported that pods contain close to 19% protein, 2.5% fats, 46.5% carbohydrates, 5.1% minerals and 20.1% crude fiber [3]. Species provide high quality animal fodder, fuel wood, charcoal, gums and other products as well as contribute to soil stabilization and improvement through nitrogen fixation. Their particular value in arid zones lies in their extreme resistance to heat, drought, salinity and alkalinity, drifting sand, grazing and repeated cutting [4]. Apart from this, timber is also used for fence posts, firewood, furniture and wagon wheels. The prolific pods made good fodder for desert grazers and the foliage is also palatable. Traditionally acacia is used as anthelmintic, antidiarrhoeal, anti asthmaticand in pulmonary diseases [5].
As evident from literature, aerial part of species is widely used but the seeds which are discarded as waste could also be used efficiently for food and health. Hence the study includes extraction and characterization of A. tortilis seed oil to evaluate its oil potential and explore newer source of fatty acid for nutritional purposes.
2. Material and Methods
2.1. Plant Material
Acacia tortilis seeds were procured from market, its specimen was prepared and matched with authenticated sample deposited in institute’s herbarium (LWG No. NBRE-B4) Seeds were shade dried to constant weight, grinded and passed through sieves to obtain powder of 40 mesh.
2.2. Reagents
The fatty acid methyl esters (FAMES) references viz. palmetic, oleic, steric, linoleic and linolenic acid (99% pure) and all the reagents (AR grade) were procured from sigma aldrich.
2.3. Extraction of Oil
For oil extraction, 5.0 g of air dried powdered seeds were extracted with petroleum ether (40˚C - 60˚C) in a soxhlet apparatus and dried over anhydrous sodium sulphate in vacuum at 40˚C for removal of solvents and percentage yield was calculated [6].
2.4. Methyl Estrification
The oil was hydrolyzed with 0.5 N KOH in ethanol (15 ml) for 4 - 6 h. Fatty acids were separated from unsaponified part of the oil after hydrolysis. Thehydrolysate was extracted with di-ethyl etherand allowed to stand for few minutes. The upper layer containing unsaponified part was separated from the lower aqueous layer containing fatty acids. HCl:H2O (1:2) was added to make the aqueous layer acidic and finally extracted with di-ethyl ether for complete separation of fatty acids.
The fatty acids were derivatized into their methyl esters after complete evaporation of di-ethyl ether adding 25 ml of 2% methanolic H2SO4 (both methanol and H2SO4, AR grade) which was refluxed on a water bath for 4 h. Washing of the methyl esters was carried out with 1% aqueous potassium hydroxide and distilled water 3 - 4 times, finally transferred to ependorff tube after removing moisture through anhydrous sodium sulphate (if any) for injection in gas chromatograph.
2.5. Fatty Acid Analysis
Gas chromatography was performed to determine the fatty acid composition of oil by Hewlett Packard Gas Chromatograph Model 5890 Series II using a stainless steel column coated with diethylene glycol succinate (DEGS) on chromosorb W (HP) (180 × 0.3 cm), equipped with flame ionization detector attached to Pentium III Computer. The column, injector and detector temperatures were maintained at 170˚C, 230˚C and 250˚C respectively using nitrogen as carrier gas [7].
2.6. Measurement of Commercial Indices
Different commercial indices were established to calculate monounsaturated fatty acids (MUFA), poly unsaturated fatty acid (PUFA), total unstauration (TU = PUFA + MUFA), total saturation (SFA), polyunsaturated to saturated ratio (PUFA/SFA) and omega-6/omega-3 ratio (n-6/n-3) [8,9].
3. Result & Discussion
The oil extracted was viscous and cream to light yellow in colour with the total oil content of 3.8%. The quality of oil is mainly governed by the fatty acid composition, hence the standardization of oil on the basis of fatty acid composition is mandatory. Table 1 shows that seed oil consists mainly of essential saturated and unsaturated fatty acid. In plant tissues, the most abundant saturated fatty acids are palmitic and stearic acids and the most common unsaturated fatty acids are oleic, linoleic and linolenic [10]. In A. tortilis seed oil linolenic acid (50.43%) was found to be maximum, followed by linoleic acid (36.75%), Palmitic acid (6.41%), Oleic acid (3.96%) and stearic acid (0.63%). Higher content of Linoleic acid and Linolenic acid i.e. 36% and 50% respectively in analyzed oil is noteworthy. Content of linoleic acid in seed i.e. 36.75% is similar to soya bean, sunflower and safflower vegetable oils having specified codex standard range of 48% - 55%, 48% - 74% and 67.6% - 73.2% respectively. Thus such linoleic acid rich oils as grape seed oil, sunflower oil and sufflower oil including A. tortilis oil can play a significant role in reducing blood cholesterol levels when consumed regularly as part of the diet [11-13]. Linolenic acid was found to be significantly high i.e. 50.43%, which is comparable (Table 2) to various commercially available edible oils [1]. In addition to this omega-3 fatty acid lowers the blood cholesterol, reduces the symptoms in of depression, dementia, anxiety and possess anti inflammatory action. It is also vital for infants’ health, pregnant women, seems to improve rheumatoid arthritis, diabetes and improves the life span of patients with auto immune diseases [1]. The high content of linoleic and linolenic acid of the oil analysed may have a significant role in clinical use [14] and contribute significantly in cholesterol lowering effect also [15,16]. Moreover, the higher content of saturated fatty acids like Palmitic acid (6.41%) and stearic acid (0.63%) support the fact that this genus belonging to the family Fabaceae
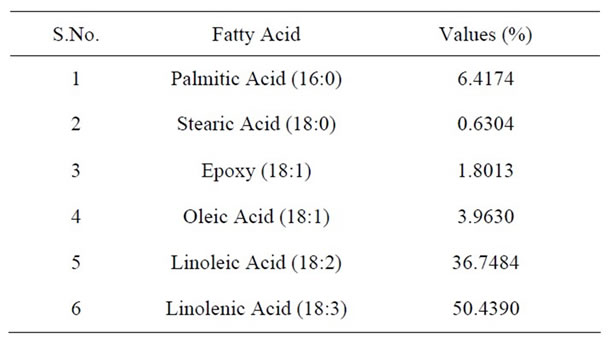
Table 1. Fatty acid composition of A. tortilis seed oil.

Table 2. Comparison of linoleic and linolenic content in A. tortilis seed oil with some commercial oils.
and being a legume species A. tortilis have low seed oil content but significant saturated and unsaturated fatty acid.
Other commercial indices essential for accessing the nutrition value of oil were also estimated (Figure 1). Higher index of mono unsaturated fatty acid and poly unsaturated fatty acid (PUFA) espically omega-3 fatty acid as linolenic acid and omega-6 fatty acid as linoleic acid play important role in human and animal health [11-13]. In recent years there has been interest in MUFA as suitable replacement of saturated fatty acid. Their net effect on serum lipids & protein is not much different from PUFA but they are not susceptible to oxidation which may play role in artherogenesis [17]. Poly unsaturated fatty acids (PUFA) and total unsaturation of the oil were also found to be promising viz. 87.17% and 92.94% respectively. PUFA/SFA was 12.36 and is significantly
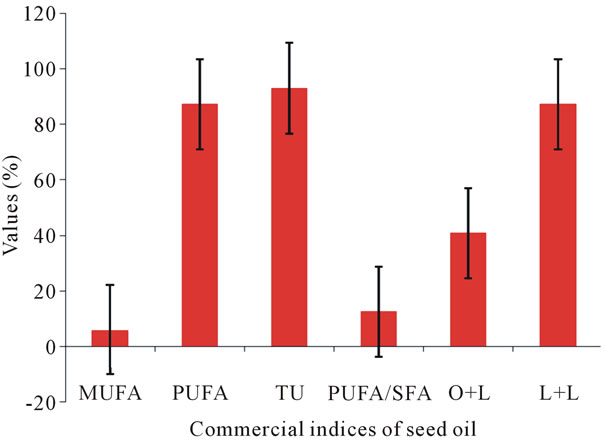
Figure 1. Commercial indices of A. tortilis seed oil.
higher than the standards laid for nominal dietary intake. n-6/n-3 was found to be 0.72, which is quite close to the WHO codex and imbalance in the ratio is linked to the serious health conditions such as heart attack, obesity, strokes etc. [18].
4. Conclusion
The present investigation reveals the newer source of oil and contains beneficial saturated and unsaturated fatty acid of significant value. Oil is rich in linoleic and linolenic acid content which is beneficial for lowering body cholesterol if taken on daily basis for dietary suppliments. Beside this presence of palmitic, oleic and stearic acid increases the nutritional value and adds to the overall health benefits of oil. Commercial indices viz. MUFA, PUFA, TU, SFA, PUFA/SFA and n-6/n-3 established were also found to be very promising. Oil and fatty acid values analysed meets the set standards and is significant in terms of nutritional value. This bioprospects the use of seed oil in dietary intake and thus could be a promising alternative to other edible oils especially in specific locations where there is higher abundance of species to explore the real potential of the source.
5. Acknowledgements
The authors are thankful to the Director NBRI for providing facilities and encouragement throughout the work. Thanks to the technical staff for their support.
REFERENCES
- R. Banerji, P. Shukla, A. Bajpai and B. S. Dixit, “Review Article: Omega-3 Fatty Acid,” Applied Botany Abstracts, Vol. 27, No. 4, 2007, pp. 315-333.
- A. R. Pellew, “The Production and Consumption of Acacia Browse and It’s Potential for Animal Protein Production,” In: H. N. Le Houerou, Ed., Browse in Africa, International Livestock Centre for Agriculture, Addis Ababa, 1980.
- http://www.worldagroforestry.org/
- H. N. Le Houerou, “Browse in Northern Africa,” In: Browse in Africa. The Current State of Knowledge, International Livestock Centre for Agriculture, Addis Ababa, 1980, pp. 55-82.
- Anonymous, “A Guide to Medicinal Plants in North Africa: Database on Medicinal Plants in North Africa: Database on Medicinal Plants in North Africa,” IUCN, 2005.
- R. Banerji, R. C. Gupta and B. S. Dixit, “Comparitive Study of Fats and Fatty Acids in Mangifer Indica Varieties,” The Journal of Oil Technologists’ Association of India, Vol. 35, No. 1, 2003, pp. 9-10.
- B. E. Phillips, C. R. Smith and J. W. Hangemann, “Glyceride Structure of Erlangatomentosa Seed Oil, a New Source of Vernolic Acid,” Lipids, Vol. 4, No. 6, 1969, pp. 473-477. doi:10.1007/BF02531027
- Y. Maralidharu and Virupakshappa, “Extent of Variation in Oil, Protein and Fatty Acid Composition of High Oleic Hybrids, Wild Species and Some Cultivars of Sunflower,” JOTAI, Vol. 30, 1998, p. 4.
- H. L. Shearer, D. H. Urpin and D. T. Dennis, “Characterization of NADP Dependent Malic Enzyme from Developing Castor Oil Endosperm,” Archives of Biochemistry and Biophysics, Vol. 42, 2004, pp. 134-144.
- J. Murphy, “Structure, Function and Biogenesis of Storage Lipid Bodies and Oleo Resin in Plants,” Progress in Lipid Research, Vol. 32, No. 3, 1993, pp. 247-280. doi:10.1016/0163-7827(93)90009-L
- S. V. Isay and N. G Busarova, “Study of Fatty Acid Composition of Marine Organism-Unsaturated Fatty Acid of Japan Sea Invertibrates,” Compendium of Biochemistry and Physiology, Vol. 77, 1984, p. 803.
- B. E. Phillipson, et al., “Reduction of Plasma Lipids, Lipoproteins and Apoproteins by Dietary Fish Oil in Patients with Hyper Triglyceridemia,” The New England Journal of Medicine,” Vol. 312, 1985, p. 1210. doi:10.1056/NEJM198505093121902
- Y. H. Hui, “Baleey’s Industrial Oil and Fat Products,” 5th Edition, John Wiley and Sons, Inc., New York, 1996, pp. 163-383, 394.
- J. X. Kang and A. Leaf, “Protective Effects of Free Poly Un-Saturated Fatty Acids on Arrhythmias Induced by Lysophosphatidylcholine or Palmitoylcarnitine in Neonatal Rat Cardiac Myocytes,” European Journal of Pharmacology, Vol. 297, No. 1-2, 1996, pp. 97-106. doi:10.1016/0014-2999(95)00701-6
- J. K. Chan, “Dietary Alpha Linolenic Acid Is as Effective as Oleic Acid and Linoleic Acid in Lowering Blood Cholesterol; in Normolipidemic Men,” American Journal of Nature, Vol. 53, 1991, pp. 1230-1234.
- R. David, “Combined Fatty Acid Composition for Lowering Blood Cholesterol and Triglyceride Levels,” Canadian Patent CA 1239587, 1985.
- R. M. Krauss, R. H. Eckel, B. Howard, L. J. Appel, S. R. Daniels, et al., “AHA Dietary Guidelines: Revision 2000: A Statement for Healthcare Professionals from the Nutrition Committee of the American Heart Association,” Circular, Vol. 108, No. 18, 2000, pp. 102-2284.
- WHO, “Diet, Nutrition and the Prevention of Chronic Disease,” WHO Technical Report, Series 916, 2003.

