Food and Nutrition Sciences
Vol. 2 No. 7 (2011) , Article ID: 7237 , 8 pages DOI:10.4236/fns.2011.27103
Occurrence of Fungi and Mycotoxins in Some Commercial Baby Foods in North Africa
![]()
1School of Biological and Biomedical Sciences, Glasgow Caledonian University, Glasgow, UK; 2Department of Food Technology, Sebha University, Faculty of Engineering and Technology, Brack Ashati, Libya; 3Present address: Renewable Energies and Water Desalination Research, Center, Tajoura, Libya.
Email: *K.Aidoo@gcu.ac.uk
Received February 14th, 2011; revised July 21st, 2011; accepted July 28th, 2011.
Keywords: Mycotoxins, Aflatoxins, HPLC, Infant Food, North Africa
ABSTRACT
Aflatoxins, are one of over 200 known mycotoxins produced by filamentous fungi. They are toxic, carcinogenic, and mutagenic and may be present in many raw and processed food commodities including cereals and milk used as ingredients in infant food products. Consumption of these commodities may pose a potential risk to the health of infants. The mycoflora of 84 samples of baby food commercially available in North Africa was determined. The feeds were screened for mycotoxins using thin layer chromatography (TLC) and further analyses were carried for the total aflatoxins by high performance liquid chromatography (HPLC) using a Kobra cell to derivatise the aflatoxins (B1, G1, B2 and G2). Twenty-four fungal isolates were recovered from the samples consisting of 5 Aspergillus. spp, 13 Penicillum. spp, 5 Mucor. spp and an unidentified species Of these, 45.8% of the isolates were found to be mycotoxigenic however only 2.4% of the samples contained aflatoxins (19 to 70 µg·kg–1) and the remaining contained undetectable levels of the toxin. Storage at elevated relative humidity for 7 days showed a slight increase in the fungal counts but the toxin levels were unaffected. The results show the importance of periodic testing regime of ingredients used infant food formula for the presence of fungal contamination.
1. Introduction
Powdered infant formula and feed products have been associated with serious illness and death in infants due to microbial infections, in particular pathogenic bacteria. Cereals, dairy products, fruits and nuts are some of the materials used in the formulation of baby food. Baby food and feed formula can be contaminated with microorganisms through inadequate manufacturing process. During the preparation of infant formula, inappropriate handling practices can exacerbate the problem. The FAO/WHO, European Commission and Food and Drug Administration have set permissible levels for total bacterial counts in infant food and formula, however there is very information on levels of fungi in such products. Agricultural products may become contaminated with fungi during drying and storage. The FAO estimates that mycotoxins contaminate 25 percent of agricultural crops worldwide [1].
Mycotoxins are produced by various strains of fungi, particularly Penicillium, Aspergillus and Fusarium. Mycotoxins are well known as a worldwide health problem particularly in countries with high ambient temperature and relative humidity typical of tropical countries [1-3]. A great deal of attention has been given to aflatoxigenic fungi such as Aspergillus flavus and A. parasiticus. Of the aflatoxins, aflatoxin B1 (AFB1) is considered by the International Agency for Research on Cancer [4] to be the most carcinogenic. About 250,000 hepatocellular carcinoma related deaths are reported to occur annually in Africa due to aflatoxin ingestion where 80% of cases and deaths of liver cancer occur in Western and Central Africa [5,6]. The carcinogens may be present in many raw and processed foods and feeds. Among these products, cereals and milk are very susceptible to mycotoxin contamination and are often used as ingredients in many infant food products [7-10]. Consumption of foods produced from these commodities may pose a potential risk to the health of the infants since the toxins are resistant to various food processes, including heat treatment [11-14]. Regulations to control the mycotoxin content of foods have been established in about one hundred countries, out of which fifteen are African, to protect the consumer [15-17]. The European Community and Codex Alimentarius have set a maximum level of total aflatoxin (AF) exposure in infant foods, AFB1 and aflatoxin M1 (AFM1) at level 0.04 µg·kg–1, 0.02 µg·kg–1 and 0.025 µg·kg–1 respectively [18,19]. Most African countries have a climate characterised by high humidity and high temperature which favours the growth of fungi in food products, and therefore the potential for the production of mycotoxins. Hence, there is a need to investigate the occurrence of aflatoxins in infant foods since some of the ingredients used in the foods are usually prone to fungal contamination.
The objective of the present work was to determine the mycoflora of a range of commercial baby foods, evaluate toxigenic capability of the isolates and the effect of storage conditions on the samples. Natural occurrence of aflatoxins in the samples was also determined. It is anticipated that the results from this study would help to understand the general mycoflora and potential mycotoxins which may be present in infant food and feed formulae retailed in North Africa.
2. Materials and Methods
2.1. Mycological Examination of Infant Food Samples
Eighty-four samples of baby food were collected from several local sources including retailers, factories and stores in Libya. The ingredients of the samples comprised of rice flour, wheat flour, mixed grain cereal, wheat, rice, barley, and oat flour, skimmed milk powder or whole milk powder and in various combinations. The samples were examined for mycological profile before storage at 22˚C and 80% relative humidity in humidity chamber (Cryotechnics, 2000 Series, Edinburgh, UK) for 7 days. Standard methods were used for isolation, enumeration and identification of fungi [20]. Samples were reconstituted in maximum recovery dilutent (MRD, Oxoid, Basingstoke, UK) and plated out on malt extract agar (MEA, VWR International, Lutterworth, UK) and potato dextrose agar (PDA, Oxoid CM 139). The plates were then incubated at 25˚C for 5 days.
2.2. Thin Layer Chromatography (TLC) Assay
Twenty five fungal strains isolates were grown on malt extract agar (MEA, VWR International, Lutterworth, UK) and potato dextrose agar (PDA, Oxoid CM 139), the culture plates were incubated at 25˚C for 7 days and examined for mycotoxins using the TLC technique as reported by others [20,21]. Agar plugs were prepared by cutting the fungal colony to a diameter of 5 mm. The plugs were immersed in 2 mL of methanol for extraction of toxin from fungi and culture medium centrifuged (Stuart 2500 RMP) at 2500 rpm for 30 s. The extracts (10 μL) were spotted on TLC plates (20 ´ 20 cm, Merck) coated with 0.25 mm thin layer of silica gel. Subsequently, 10 μL of mycotoxin standard solutions total aflatoxins, ochratoxin A (Biopharm, Glasgow, UK) and citrinin (Sigma-Aldrich, Dorset, UK) were used as reference standards (40 μg·mL–1) and spotted along with the fungal samples extract. The plates were developed in a solvent tank containing toluene—ethyl acetate—formic acid (5:4:1) for 1h and viewed under long wave UV light (365 nm). Sample extracts were compared with reference standards spots.
2.3. Detection of Aflatoxins in the Samples by High Performance Liquid Chromatography Technique (HPLC)
2.3.1. Sample Extraction and Preparation
Food samples (50 g) were extracted using 250 mL of methanol/water (60/40, v/v) containing 4 g NaCl and homogenised for 1min using a high speed homogeniser. Distilled water (250 mL) was added and this extract was filtered through a Whatman No 4 filter paper (55mm) and the filtrate collected. The filtrate was then analysed by passing 10 mL through an immunoaffinity column followed by 10 mL distilled water (twice) to remove any interfering material. Aflatoxins were eluted by passing 1 mL of acetonitrile through the column. The stock solution and standards were prepared and determined as described by the manufacturer and Candlish et al. [22]. Quantitative analysis was conducted on all samples by injection of 100 µL extract through the HPLC system and using total aflatoxins (AFs) as standards (Biopharm Rhone, Glasgow, UK).
2.3.2. Preparation of Artificially Contaminated Sample
Infant food samples free of aflatoxins (AFs) were artificially spiked with 10 ng of AFs/g (in triplicate) and extracted as above and then passed a known volume of sample through an immunoaffinity column. Recovery of aflatoxins (AFs) from immunoaffinity columns was expressed as a percentage of aflatoxins detected by HPLC compared to the known concentration of aflatoxins added to the 10 ml sample filtrate.
2.3.3. Chemicals, Reagents and HPLC Conditions
Acetonitrile and methanol were purchased from Fisher Scientific (Loughborough, UK). Aflatoxin standard solutions and immunoaffinity columns (Aflaprep®) were purchased from R-Biopharm Rhone Ltd (Glasgow, Scotland). The HPLC system consisted of a pump (Wellchrom K 1001 Knauer) and a fluorescence detector (Waters 470, UK) combined with a Kobra cell® (R-Biopharm Rhone Ltd, Glasgow) as an electrochemical reaction cell. The mobile phase consisted of water-acetonitrile-methanol (60: 20:20, v/v/v) with 350 µL of 4 M nitric acid and 119 mg of potassium bromide per litre and flow rate of 0.8 mL/min for aflatoxins detection in both the standard and the extracts from the baby food samples. Aflatoxins were derivatised in the Kobra cell and later detected at λex = 366 nm, λem = 465 nm. The aflatoxins B1, B2, G1 and G2 were detected by HPLC and the four peaks quantified.
2.4. Statistical Analysis
All tests were conducted in triplicate. Colonies were counted and expressed logarithmically (log10 CFUg–1). Mean and standard deviation (SD) were calculated using Microsoft Office Excel 2003 software (Microsoft Corporation, Redmont, USA). The toxin values obtained for samples were acquired and analyzed using SPSS version 15 and averages results are expressed with standard deviation mean (SD). The t-test was used to compare the differences in recoveries and detection limits.
3. Results and Discussion
3.1. Mycological Profile of Infant Food Products
A total of 84 baby food samples were tested for the presence of fungi and metabolites produced by these isolates. Table 1 shows the fungal counts in the infant products as received and after storage of samples at 25˚C and 80% relative humidity for 7 days. For products which were positive for presence of fungi, the count was between 1.1 and 4.7 log10CFUg–1. Two samples (2.4%) showed the highest fungal counts of ≥4.0 log10CFUg–1. Fungi were not detected in 50 of the samples (17.9%) on any of the 2 media even after incubation of products at 25˚C and 80% humidity The genera Aspergillus spp., Penicillum spp. and Mucor were isolated from the samples but no further morphological studies were conducted to classify the isolates to species level. The twenty four isolated strains consisted of five Aspergillus spp., thirteen Peniciillum spp., five Mucor and one unknown. There was a slight increase in the count of fungi in most of samples after 7 days incubation at 25˚C and 80% relative humidity, although one sample showed an increase in the count from 3.43 log10CFUg–1 to 4.48 log10CFUg–1.
3.2. Occurrence of Mycotoxigenic Fungi and Mycotoxins in Infant Food Products.
The mycotoxins produced by the isolated strains on two mycological media are shown in Table 2. Both media showed similar results. The TLC analysis indicated that all stains of Aspergillus spp isolates were able to produce aflatoxins, seven out of thirteen strains of Penicillium spp were mycotoxin positive and most strains were able to produce citrinin; five strains produced unidentified fluorescent bands, which are thought to be either unidentified mycotoxins or pigments produced by fungi. The results also indicated that there was a slight increase ≤ 1.0 log10CFUg–1 in the total number of fungi in some samples after storage of the samples at 80% relative humidity (Table 1).
The percentage recoveries of AFs in the products spiked with 10 ng of AFs·g–1 were 98% for AFB1, 95% for AFB2, 90% for AFG1 and 94% for AFG2. In this study, aflatoxins were not quantifiable in the majority of samples (97.6%), while 2.4% of the samples were found to contain the toxins. The amounts of aflatoxins (AFs) of the two samples were 19 and 70 µg·kg–1 as shown in Figures 1(c) and (d). In view of the high recovery of aflatoxins from artificially contaminated samples this indicates that these samples indeed contained significant levels of aflatoxin (Table 3). These amounts were higher than the maximum tolerance limit accepted by European Union as guideline levels for cereal-based feeds (5 µg·kg–1 for AFB1 and 10 µg·kg–1 for total aflatoxin in cereal based feeds) [19]. However, common aflatoxin producing fungi such as A. flavus and A. parasticus were not isolated from positive samples. The production of mycotoxins does not correlate directly with the growth of the fungi and it is possible that the mycotoxins may have been within the raw materials before processing.
Since mycotoxin limits have not been established in Libya, limits for the EC Regulations were used as a guide for this study. Many studies on the presence and quantity of mycotoxins in food and feed products have been conducted worldwide [23-25] but only a few studies for the presence of mycotoxins in baby food [7,26,27]. This is in spite of the fact that cereals and dried fruits are used as ingredient in infant food in combination with milk. In a Korean study, 85% infant formulae samples were found to be contaminated with AFM1 with a mean concentration of 46 ng·kg–1 [28].
Araguas et al. [29] examined twenty samples of cereals-based baby foods in Spain (Navarra) for OTA contamination. Ochratoxin A was detected in fourteen of the samples (limit of detection 0.035 µg·kg–1). In another survey Razzazi-Fazeli et al. [30] examined twelve baby foods and found no aflatoxins in the products. Sewram et al. [31] investigated corn-based infant food from markets in Brazil. They found that AFB1 was present in seven of eight samples ranging from 30 to 6127 µg·kg–1 and two of eight samples were positive for AFB2 ranging from 53 to 1738 µg·kg–1. A study of mycotoxins in infant foods including cereal-based infant foods—soy, wheat, oat,



Table 1. Total count of fungi before and after storage of samples at 25˚C and 80% relative humidity (RH) for 7 days.

Table 2. Screening of fungi isolates for their ability to produce mycotoxins using thin layer chromatography (TLC).
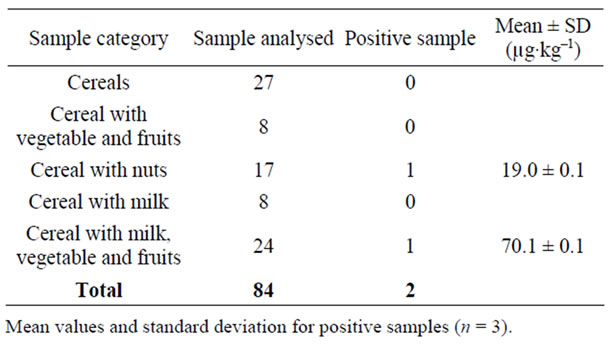
Table 3. Aflatoxins in infant cereal-based foods
barley, rice was conducted in Canada between 1997 and 1999 showed that 74% of 363 samples contained multiple mycotoxins [26]. In another survey carried out in UK [27], 199 food samples including breakfast/rusk products, baby rice, savoury products and dessert/cereal bar/biscuits were analysed for total aflatoxins, OTA and patulin. It was found that aflatoxins were not quantifiable in the majority of samples (99.5%) with only one sample containing detectable levels of AFB1, The OTA was not detected in 5.5% of the samples, while patulin was not detected in any of the samples tested [27].
Tam et al. [32] analysed 349 breakfast and mixed-grain infant cereals samples and should that 50% of both breakfast and infant cereals contained (limit of detection = 0.002 ng·g–1) AFB1. Only 4% of the breakfast cereals and 1% of the infant cereals had AFB1 concentrations exceeding 0.1 ng·g–1, which is the European Union maximum limit for aflatoxin B1 in baby foods and processed cereal-based foods for infants and young children. Marit-Aralt [33] analysed milk samples including organic cow's milk, conventional cow’s milk, and cow’s milk-based infant formulas for the occurrence of OTA. The toxin was detected in six out of forty conventional cow's milk samples with a range of 11 - 58 ng·L–1, and in five out of forty seven organic milk samples with a range of 15 - 28 ng·L–1. No OTA was detected in any of the twenty infant formula samples. Beretta et al. [34] studied 338 samples of baby food for OTA and indicated that 16.8% contained detectable quantities of OTA and four of these (3.4% of the total) contained OTA above 0.5 µg·kg–1.
3.3. Effect of Mycotoxins on Health and Its Occurrence in Human Milk
The presence of aflatoxins in the human diet, and especially in the diet of infants, is a major concern as cereal foods are a significant source of nutrition for infants and are among the first solid foods offered to babies. Studies indicate that susceptibility to aflatoxin is greatest in the young children [35,36]. Aflatoxin exposure has been associated with growth faltering and immune suppression in young children. Early life exposures could be a contributing factor towards the early onset of hepatocellular carcinoma [37]. Due to high toxicity and carcinogenic properties of AFB1, its presence in dried food is a concern. Aflatoxins are resistant to thermal inactivation, pasteurisation, autoclaving and other varieties of food processing procedures [13,14]. So to produce high qual-
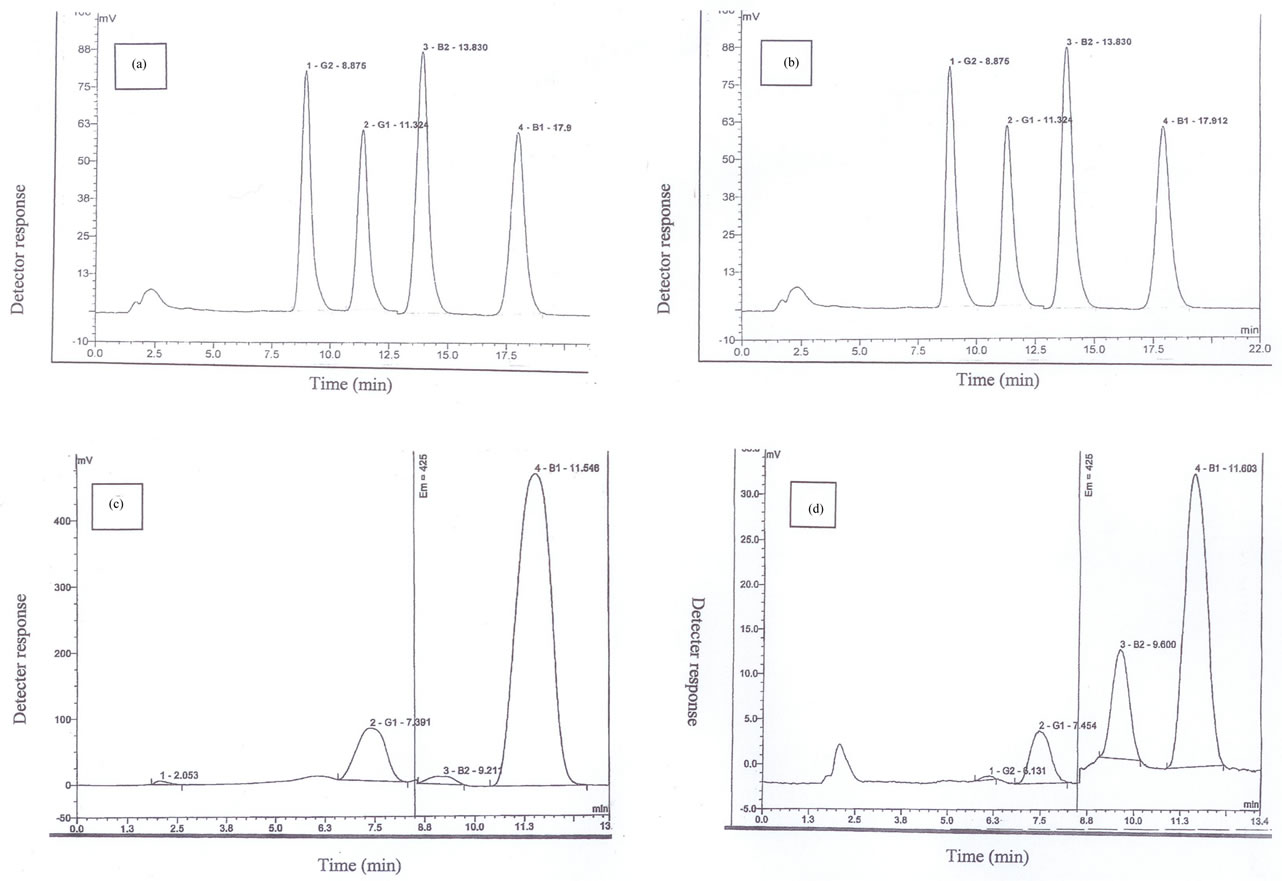
Figure 1. Chromatograms of aflatoxins (AFs) standard at 40 ng·mL–1 (a) AFs recovery sample; (b) positive AFs samples; (c) and (d).
ity milk, it is essential to keep raw materials from contamination. Mycotoxin concentrations in raw materials can be reduced by good manufacturing practice (GMP) and good storage practices. In addition, infant formula manufacturers must obtain substantial reductions in contaminants through careful selection of raw materials and/ or avoidance of contamination during processing.
Human breastfeeding is now being encouraged for mothers and their infants and some studies show that breast milk may be contaminated with fungal metabolites. The presence of AFM1 in human breast milk is clearly of concern. Aflatoxin M1 has been reported in breast milk particularly in developing countries. For example in Egypt, AFM1 was detected in breast milk at level 13.5 pg·mL–1 [38], in The United Arab Emirates in the range of 5 to 3400 pg·mL–1 [39] and from 5 - 64 pg·mL–1 in the Sudan [40]. In Gambia AFM1 was detected in one hundred samples of breast milk at ≥1.4 pg·mL–1 [41]. However in Libya, there is little or no published information on occurrence of AFM1 in human milk. A preliminary study by Elgerbi [42] showed that 66.7% of 27 human breast milk samples from Libya were contaminated with aflatoxin M1 ranging from 0.015 to 0.343 pg·mL–1. This again is of major concern to the health of mothers and infants.
4. Conclusions
The present work demonstrates that periodical testing of infant food products especially cereal products for mycotoxins is important since these toxins cannot be eliminated completely from the grain supply and very difficult to destroy by heating. All efforts should be made to ensure that the toxins are below permissible limits in baby foods. Such effort should involve agriculture practices, food importers, food processors, dairy industry, infant food manufacturers, food safety enforcement officers and the public on food storage conditions that are likely to promote and/or encourage microbial growth. Periodic testing of baby food and their ingredients for mycotoxins is important to control occurrence of the toxin in the food chain destined for infants.
5. Acknowledgments
The authors gratefully acknowledge financial support from the Libyan government for research studentship. The authors are also grateful to Martin Gallagher (RBiopharm Rhone Ltd, Glasgow) for technical assistance with aspects of the aflatoxin assay.
REFERENCES
- World Health Organization (WHO), “Mycotoxins in African Foods: Implications to Food Safety and Health,” AFRO Food Safety Newsletter, World Health Organisation Food Safety (FOS), July 2006.
- P. Sean and P. D. Abbott, “Mycotoxins and Indoor Moulds,” Indoor Environment Connections, Vol. 3, No. 4, 2002, pp. 14-24.
- J. M. Wagacha and J. W Muthomi, “Mycotoxin Problem in Africa: Current Status, Implications to Food Safety and Health and Possible Management Strategies,” International Journal of Food Microbiology, Vol. 124, No. 1, 2008, pp. 1-12. doi:10.1016/j.ijfoodmicro.2008.01.008
- International Agency for Research on Cancer (IARC), “Some Naturally Occurring Substances: Food Items and Constituents, Heterocyclic Aromatic Amines and Mycotoxins,” IARC Monograph on Evaluation of Carcinogens and Risks in Humans, Vol. 56, 1993, pp. 1-599.
- S. Dow, “The Politics of Breast Cancer,” The Australia, Vol. 35, 1994, pp. 6-17.
- H. Nagy, A. Loutfy and A. Moussa, “Influence of Gamma-Radiation on Mycotoxin Producing Moulds and Mycotoxins in Fruits,” Food Control, Vol. 13, No. 4-5, 2001, pp. 281-288.
- R. Shipra, D. D. Premendra, K. K. Subhash and D. Mukul, “Detection of Aflatoxin M1 Contamination in Milk and Infant Milk Products from Indian Markets by ELISA,” Food Control, Vol. 15, No. 4, 2004, pp. 287-290. doi:10.1016/S0956-7135(03)00078-1
- C. Tarik-Haluk, S. Belgin and K. Ozlem, “Aflatoxin M1 Contamination in Pasteurized Milk,” Veterinary Arski Arhives, Vol. 75, No. 1, 2005, pp. 57-65.
- O. Mohammad-Reza, J. Behrooz, S. Naficeh, H. Mannan and N. Azadeh, “Presence of Aflatoxin M1 in Milk and Infant Milk Products in Tehran, Iran,” Food Control, Vol. 18, No. 10, 2007, pp. 1216-1218. doi:10.1016/j.foodcont.2006.07.021
- H. Huseyin-Oruc, C. Recep, E. Engin and G. Ertan. “Fate of Aflatoxin M1 in Kashar Cheese,” Journal of Food Safety, Vol. 27, No. 1, 2007, pp. 82-90.
- A. Pittet, “Natural Occurrence of Mycotoxins in Foods and Feeds,” Medical Veterinary, Vol. 149, 1998, pp. 479-499.
- E. E. Creppy, “Update of Survey, Regulation and Toxic Effects of Mycotoxins in Europe,” Toxicology Letters, Vol. 127, 2002, pp. 667-743. doi:10.1016/S0378-4274(01)00479-9
- S. S. Deshpande, “Fungal Toxins. In: Handbook of Food Toxicology,” Marcel Decker, New York, 2002, pp. 387- 456. doi:10.1201/9780203908969
- D. L. Park, “Effect of Processing on Aflatoxin,” Advances in Experimental Medical Biology, Vol. 504, 2002 pp. 173-179. doi:10.1007/978-1-4615-0629-4_17
- H. P. van Egmond, “Worldwide Regulations for Mycotoxins,” Advances in Experimental Medical Biology, Vol. 504, 2002, pp. 257-269. doi:10.1007/978-1-4615-0629-4_27
- D. Barug, H. P. van Egmond, R. Lopez-Garcia, T. van Osenbruggen and A. Visconti, “Meeting the Mycotoxins Menace,” Wageningen Academic, The Netherlands, 2003.
- A. Fellinger, “Worldwide Mycotoxin Regulations and Analytical Challenges,” World Grain Summit: Foods and Beverages, San Francisco, California, September 2006.
- EC (Codex Alimentarius Commissions), “Discussion and Possible Opinion on Draft Commissions Regulation Amending Regulation (EC) NO. 466/2001 as Regards Aflatoxins and Ochratoxin A in Foods for Infant and Young Children (SANCO/0983/2002 REV. 6),” 2004.
- European Commission (EC), “The Commission Decision, 2006/504/EC,” Official Journal of the European Union L199, 2006, pp. 21-32.
- R. Samson, E. Hoekstra, J. Frisvad and O. Filtenborg, “Introduction to Food Borne Fungi,” 6th Edition, Centraalbureau voor Schimmelcultures, Utrecht, The Netherlands, 2002.
- P. V. Hans and H. P. Walter, “Determination of Mycotoxins,” Pure and Applied Chemistry, Vol. 58, No. 2, 1986, pp. 315-326. doi:10.1351/pac198658020315
- A. A. G. Candlish, C. A. Haynes and W. H. Simson, “Detection and Determination of Aflatoxins Using Affinity Chromatography,” International Journal of Food Science and Technology, Vol. 23, No. 5, 1988, pp. 479-485. doi:10.1111/j.1365-2621.1988.tb00604.x
- H. Bacha, R. Hadidane, E. Creppy, C. Regnault, F. Ellouze and G. Dirheimer, “Monitoring and Identification of Fungal Toxins in Food Products, Animal Feed and Cereals in Tunisia,” Journal of Stored Products Research, Vol. 24, 1988, pp. 199-206. doi:10.1016/0022-474X(88)90019-7
- J. Marika, C. S. Maria, K. Merja, V. Pirojo, R. Aldo, R. Alberto and P. Kimmo, “Levels of Mycotoxins and Sample Cytotoxicity of Selected Organic and Conventional Grain-Based Products Purchased from Finnish and Italian Markets,” Molecular and Nutritional Food Research, Vol. 48, No. 4, 2004, pp. 299-307. doi:10.1002/mnfr.200400026
- B. Terken, B. E. Ayse, G. Gözde, A. Sevtap and S. Gönul. “Aflatoxin and Ochratoxin in Various Types of Commonly Consumed Retail Ground Samples in Ankara, Turkey,” Annals of Agricultural and Environmental Medicine, Vol. 12, No. 2, 2005, pp. 193-197.
- Health Canada Scientists, “Federal Study Finds Toxins in Baby Food,” 2003. http://www.breakingtheviciouscycle.info/news/baby_food_study.htm
- FSIS 68/2004, “Food Survey Information Sheets Survey of Baby Foods for Mycotoxins,” 2004. http://www.food.gov.uk
- E. K. Kim, D. H Shon, D. Ryu, J. W. Park, H. J. Hwand and Y. B. Kim, “Occurrence of Aflatoxin M1 in Korean Dairy Products Determined by ELISA and HPLC,” Food Additives and Contaminants, Vol. 17, No. 1, 2000, pp. 59-64. doi:10.1080/026520300283595
- C. Araguas, E. Gonza’lez-Penas and C. de Lopez, “Study on Ochratoxin A in Cereal Derived Products from Spain,” Food Chemistry, Vol. 92, No. 3, 2005, pp. 459-464. doi:10.1016/j.foodchem.2004.08.012
- E. Razzazi-Fazeli, C. Noviandi, A. Porasuphatana and J. Bohm, “A Survey of Aflatoxins B1 and Total Aflatoxins Contamination in Baby Food, Peanut and Products Sold at Retail in Indonesia Analyzed by ELISA and HPLC,” Mycotoxin Research, Vol. 20, No. 2, 2004, pp. 51-58. doi:10.1007/BF02946735
- V. Sewram, G. S. Shephard, W. F. O. Marasas and M. F. Castro, “Improving Extraction of Fumonisin Mycotoxins from Brazilian Corn-Based Infant Foods,” Journal of Food Protection, Vol. 66, No. 5, 2003, pp. 854-859.
- J. Tam, M. Mankotia, M. Mably, P. Pantazopoulos, R. J. Neil, P. Calway and P. Scott, “Survey of Breakfast and Infant Cereals for Aflatoxins B1, B2, G1 and G2,” Food Additives and Contaminants, Vol. 23, No. 7, 2006, pp. 693-699. doi:10.1080/02652030600627230
- S. Marit-Aralt, “Analysis of Norwegian Milk and Infant Formulas for Ochratoxin A,” Food Additives and Contaminants, Vol. 16, No. 2, 1999, pp. 75-78. doi:10.1080/026520399284235
- B. Beretta, R. de Domenico, A. Gaiaschi, C. Ballabio, C. L. Galli, C. Gigliotti and P. Restani, “Ochratoxin A in Cereal-Based Baby Foods: Occurrence and Safety Evaluation,” Food Additives and Contaminants, Vol. 19, No. 1, 2002, pp. 70-75. doi:10.1080/02652030110070021
- H. Mykkänen, H. Zhu, E. Salminen, R. O. Juvonen, W. Ling, J. Ma, N. Polychronaki, H. Kemiläinen, O. Mykkänen, S. Salminen and H. El-Nezami, “Faecal and Urinary Excretion of Aflatoxin B1 Metabolites (AFQ1, AFM1 and AFB1-N7-Guanine) in Young Chinese Males,” International Journal of Cancer, Vol. 115, No. 6, 2005, pp. 879-884. doi:10.1002/ijc.20951
- N. Polychronaki, R. M. West, P. C. Turner, H. Amra, M. Abdel-Wahhab, H. Mykkänen and H. El-Nezami, “A Longitudinal Assessment of Aflatoxin M1 Excretion in Breast Milk of Selected Egyptian Mothers,” Food Chemistry and Toxicology, Vol. 45, No. 7, 2007, pp. 1210- 1215. doi:10.1016/j.fct.2007.01.001
- Y. Y. Gong, S. Egal, S. Hounsa, A. J. Hall, K. F. Cardwell and C. P. Wild, “Determinants of Aflatoxin Exposure in Young Children from Benin and Togo, West Africa: The Critical Role of Weaning,” International Journal of Epidemiology, Vol. 32, No. 4, 2003, pp. 556-662. doi:10.1093/ije/dyg109
- N. Polychronakiab, P. C. Turner, H. Mykkänen, Y. Gong, H. Amra, M. Abdel-Wahhab and H. El-Nezami, “Determinants of Aflatoxin M1 in Breast Milk in a Selected Group of Egyptian Mothers,” Food Additives and Contaminants, Vol. 23, No. 7, 2006, pp. 700-708. doi:10.1080/02652030600627222
- Y. M. Abdulrazzaq, N. Osman, Z. M. Yousif and S. Al-Falahi, “Aflatoxin M1 in Breast-Milk of United Arab Emirates Women,” Annals of Tropical Paediatrics, Vol. 23, No. 3, 2003, pp. 173-179. doi:10.1179/027249303322296484
- J. B. Coulter, S. M. Lamplugh, G. I. Suliman, M. I. Omer and R. G. Hendrickse, “Aflatoxins in Human Breast Milk,” Annals of Tropical Paediatrics, Vol. 4, 1984, pp. 61-66.
- A. Zarba, C. P. Wild, A. J. Hall, R. Montesano, G. J. Hudson and J. D. Groopman, “Aflatoxin M1 in Human Breast Milk from the Gambia, West Africa, Quantified by Combined Monoclonal Antibody Immunoaffinity Chromatography and HPLC,” Carcinogenesis, Vol. 13, No. 5, 1992, pp. 891-894. doi:10.1093/carcin/13.5.891
- A. M. Elgerbi, “Studies on Occurrence of Aflatoxins M1 in Milk Products and Effect of Lactobacilli and Related Genera on Toxin,” Ph.D. Thesis, Glasgow Caledonian University, Glasgow, 2005.

