American Journal of Plant Sciences
Vol.08 No.03(2017), Article ID:74016,20 pages
10.4236/ajps.2017.83031
Expression Profiles of psbA, ALS, EPSPS, and Other Chloroplastic Genes in Response to PSII-, ALS-, and EPSPS-Inhibitor Treatments in Kochia scoparia
Vijay K. Varanasi1*, Shahniyar Bayramov2*, P. V. Vara Prasad1, Mithila Jugulam1#
1Department of Agronomy, Kansas State University, Manhattan, USA
2Institute of Botany, Azerbaijan National Academy of Sciences, Baku, Azerbaijan

Copyright © 2017 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: December 4, 2016; Accepted: February 6, 2017; Published: February 9, 2017
ABSTRACT
Kochia (Kochia scoparia L. Schrad.), also known as tumbleweed, is an economically important annual C4 broadleaf weed found throughout the US Great Plains. Several herbicides with different modes of action are used in the management of kochia. The effect of commonly used herbicides on the expression of their target site(s) and photosynthetic/chloroplastic genes is poorly understood in weed species, including kochia. The objective of this research was to characterize the expression profiles of herbicide target-site genes, KspsbA, KsALS, and KsEPSPS upon treatment with PSII- (e.g. atrazine), ALS- (e.g. chlorsulfuron), and EPSPS- (e.g. glyphosate)-inhibitors, respectively, in kochia. Furthermore, the expression of genes involved in photosynthesis (e.g. KsRubisco, KsCAB, and KsPPDK) was also determined in response to these herbicide treatments. KspsbA was strongly upregulated (>200-fold) 24 h after atrazine treatment. Transcript levels of the KsALS or KsEPSPS genes were 7 and 3-fold higher 24 h after chlorsulfuron or glyphosate treatment, respectively. KsRubisco, a Calvin cycle gene important for CO2 fixation, was upregulated 7 and 2.6-fold 8 and 24 h after glyphosate and chlorsulfuron treatments, whereas it downregulated 8 and 24 h after atrazine treatment. The transcript levels of KsPPDK remained unchanged after glyphosate treatment but increased 1.8-fold and decreased 2-fold at 24 h after chlorsulfuron and atrazine treatments, respectively. KsCAB remained unchanged after chlorsulfuron treatment, but was downregulated after glyphosate and atrazine treatments. The results show that herbicide treatments not only affect the respective target-site gene expression, but also influence the genes involved in the critical photosynthetic pathway.
Keywords:
Kochia scoparia L. Schrad., C4, Herbicide Stress, Gene Expression, psbA, ALS, EPSPS, Photosynthesis, Rubisco, PPDK, CAB

1. Introduction
Kochia scoparia L. Schrad. (a Chenopodiaceae member) is an important C4 annual broadleaf weed distributed throughout North American Great Plains [1] . Kochia can germinate and emerge early in the spring season when temperatures are low, and can also tolerate high temperatures of summer. Kochia also tolerates drought stress, low soil water potentials, and saline conditions. These characteristics make kochia a highly competitive weed causing extensive yield losses in several mid-western US cropping systems [1] .
Ribulose-1,5-bisphosphate (Rubisco) is the key enzyme in C3 or C4 plants for carbon dioxide (CO2) fixation. In C3 plants, atmospheric CO2 is fixed through the Calvin cycle, catalyzed by Rubisco inside the chloroplast of mesophyll cells [2] . On the other hand, in C4 plants such as kochia, photosynthetic activities are partitioned anatomically and biochemically in mesophyll and bundle sheath cells [2] . This partitioning of photosynthetic activities into distinct regions (Kranz anatomy) in C4 plants helps to reduce photorespiration and increase the photosynthetic efficiency [3] . In addition to Rubisco, certain other enzymes also play an important role in the photosynthetic activity and are therefore critical for plant survival. Pyruvate-orthophosphate dikinase (PPDK) catalyzes the conversion of pyruvate to phosphoenol pyruvate (PEP) in the mesophyll cells, which is further carboxylated by PEPC to oxaloacetate (OAA) in the C4 cycle [4] . The chlorophyll a/b binding proteins (CAB) are the light-harvesting apoproteins, serving as the antenna complex for the photosystem (PS) II pathway [5] [6] .
Even though the C3 and C4 photosynthetic pathways are well established and the properties of individual enzymes well characterized, there remain several areas which remain poorly understood, especially related to herbicide and weed physiology. Many herbicides target a specific enzyme in the chloroplast resulting in depletion of CO2 fixation, thereby leading to plant death. In response to herbicide treatments, the expression patterns of herbicide target and other critical genes involved in PS II, Calvin, and C4 cycles are yet to be studied in detail. An understanding of the expression of such genes which play an important role in plant survival helps to better elucidate the mode of action of (MOA) of commonly used herbicides for weed control.
Herbicides that inhibit PSII (e.g. atrazine), acetolactate synthase-(ALS; e.g. chlorsulfuron), or 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS; e.g. glyphosate) are commonly used to manage kochia in cropping systems. PSII-in- hibitors inhibit photosynthesis by binding to the secondary quinone acceptor QB within the D1 protein encoded by the psbA gene and block transport of electrons to the plastoquinone [7] . Blockage of the electron transport chain by these herbicides results in depletion of ATP and NADPH synthesis, and thereby CO2 fixation, leading to cellular damage by oxidative stress [7] [8] . Acetolactate synthase-inhibitors are one of the most commonly used type of herbicides for controlling wide spectrum of weeds in agronomic crops [9] . These herbicides inhibit ALS, which catalyzes the biosynthesis of branched-chain amino acids leucine, valine, and isoleucine [10] . The most commonly used non-selective herbicide in agriculture, glyphosate, inhibits EPSPS, the key enzyme of the shikimate pathway catalyzing the biosynthesis of aromatic amino acids [11] [12] .
In this study, using kochia as a model weed species, we investigated the expression profiles of herbicide target genes upon treatment with atrazine (KspsbA), chlorsulfuron (KsALS) or glyphosate (KsEPSPS) and also examined the expression of photosynthetic genes (KsRubisco, KsCAB, and KsPPDK) in response to treatment with these herbicides. Additionally, the physiological changes related to the photosynthetic apparatus, i.e. leaf chlorophyll index and chlorophyll fluorescence (Fv/Fm) were also determined. The objective of the current study was therefore to investigate if both target as well as non-target site genes (affecting photosynthetic activity) are altered by herbicide treatments in kochia.
2. Materials and Methods
2.1. Plant Material and Growth Conditions
Seeds of kochia (susceptible to atrazine, chlorsulfuron, and glyphosate), were collected from a field in Manhattan, Kansas, and germinated in plastic trays filled with Miracle-Gro potting mix (Scotts Miracle-Gro Company, Marysville, OH, USA) in the greenhouse (29/25 C temperature; 15/9 h light day/night, supplemented with 1200 µmol illumination using sodium vapor lamps). Individual seedlings (~4 cm tall) were transplanted to 6.5 cm × 6.5 cm × 6.5 cm pots in the greenhouse for herbicide treatments.
2.2. Herbicide Treatments and Tissue Collection
Nine kochia seedling (10 - 12 cm) split into replicates of three seedlings each (three seedlings for each time-point) were separately treated with field rates (1×) of three sites of action (SOA) herbicides including recommended adjuvants: atrazine, a PSII-inhibitor (Aatrex 4L® @ 2240 g ai ha−1; 1% v/v crop oil concentrate (COC)), chlorsulfuron, an ALS-inhibitor (Glean® XP @ 18 g ai ha−1; 0.25% v/v nonionic surfactant (NIS)), and glyphosate, an EPSPS inhibitor (Roundup Weathermax® @ 840 g ae ha−1; 2% w/v ammonium sulfate (AMS)). Additionally, three seedlings (controls) were treated only with adjuvants and three seedlings were left untreated (no adjuvant and no herbicide treatment). The field rate (1×) was chosen for herbicide treatments based on our preliminary studies that indicated susceptibility of kochia population to above three SOA (data not shown).
Young leaf tissue from three treated plants (biological replicates) was separately collected at 8, 24 h, and 7 d after treatment, and flash frozen in liquid nitrogen (−196 C). Additionally, tissue was also collected at 48 h to study KspsbA gene expression after atrazine treatment. Tissue from control plants was separately collected at 7 d after adjuvant treatments and used for relative comparisons with other treated time points. The untreated leaf tissue with no adjuvant or herbicide treatments was collected at 8 h time point. All the harvested tissue was stored at −80 C until RNA isolation.
2.3. Total RNA Isolation
The frozen leaf tissue was homogenized in liquid nitrogen using a pre-chilled mortar and pestle to prevent thawing. The powdered tissue was transferred to a 1.5 mL microcentrifuge tube and the total RNA was isolated using a TRIazol® reagent (Thermo Fisher Scientific, Waltham, MA, USA) following manufacturer’s instructions with modifications. Ribonucleic acid was treated with DNase 1 enzyme (Thermo Fisher Scientific, Waltham, MA, USA) to remove genomic DNA contamination. The isolated RNA was stored at −80 C. The quantity and quality (integrity) of total RNA was determined using a spectrophotometer (NanoDrop 1000, Thermo Fisher Scientific) and agarose gel (1%) (Agarose Pro- ducts, MS, USA) electrophoresis.
2.4. cDNA Synthesis
Complementary DNA (cDNA) was synthesized from 1 μg of the total RNA using RevertAid First Strand cDNA Synthesis Kit (Thermo Fisher Scientific). The cDNA was then diluted with molecular grade water in a 1:5 ratio and used in a quantitative PCR (qPCR) reaction to study the expression of herbicide target-site and photosynthetic genes in kochia.
2.5. Gene-Specific qPCR Primer Designing
Quantitative Real-Time PCR primers (Table 1) were designed using OligoAnalyzer 3.1 [https://www.idtdna.com/calc/analyzer] and by the alignment of the nucleotide sequences from species representing several plant families available in GenBank (National Center for Biotechnology Information, Bethesda, MD). Nucleotide sequence alignment was done using MultAlin software [13] . Conserved K. scoparia psbA (KspsbA) primers were designed by aligning the nucleotide sequences of K. scoparia (accession no. AY251266.1) and Poa annua (accession no. AF131887.1). K. scoparia ALS (KsALS) primers were designed based on the sequence (accession no. EU517499.1) available at GenBank. Primers for K. scoparia Rubisco (large subunit) (KsRubisco) were designed by aligning nucleotide sequences from Bassia dasyphylla (Fisch. & C. A. Mey.) Kuntze (accession no. AY270150.1), Atriplex covillei (Standl.) J.F. Macbr. (accession no. HM587609.1), Kochia americana (S. Watson) A.J. Scott (accession no. AY270103.1), Salsola genistoides Poir. (Accession no. AY270128.1), and Suaeda linifolia Pall. (accession no. HM630106.1) respectively. Previously reported primers were used for amplifying the EPSPS, CAB and PPDK genes from K. scoparia [14] [15] . In this study, we tested previously reported reference genes such as SAND (SAND family protein), UBC (Ubiquitin-Conjugating Enzyme), ARF2 (Auxin Response Factor 2) from leafy spurge (Euphorbia esula L.) [16] and CAC (Clathrin Adaptor Complex) from mustard (Brassica juncea L.) [17] based on comparative delta CT method (data not shown). However, we found that most of these top performing reference genes in other plant systems were not stably expressed under the different herbicide stress conditions in kochia, except β- tubulin.
2.6. Quantitative Real-Time PCR
Quantitative PCR reaction was performed using a StepOnePlusTM real-time detection system (Thermo Fisher Scientific) to determine the expression of herbicide target-site and photosynthetic genes from K. scoparia. The qPCR reaction mix (14 µL) consisted of 8 µL of PowerUp SYBR Green master mix (Applied Biosystems, Waltham, MA, USA), 2 µL each of forward and reverse primers (5 µM), and 2 µL of cDNA. The primers for herbicide target-site and photosynthetic genes were used in the qPCR reactions (Table 1). The qPCR conditions were 95 C for 15 min and 40 cycles of 95 C for 30 s and an annealing at 59/60 C for 1 min (Table 1). A melt curve profile was included following the thermal cycling protocol to determine the specificity of the qPCR products. Single curves were obtained for the herbicide target-site and tubulin primers.
2.7. Leaf Chlorophyll Index
A non-destructive method for determining leaf chlorophyll index was adopted using a chlorophyll meter SPAD-502 plus (Konica Minolta Optics Inc., Japan) [18] . The SPAD meter readings are proportional to the amount of chlorophyll
Table 1. qPCR primer sequences used for studying the expression of herbicide target-site and photosynthetic genes in Kochia scoparia. LSU refers to the large subunit of KsRubisco.
present in the leaf samples [19] . Leaf chlorophyll index in kochia was measured at 6 h, 1, 2, 3, 4, 5, 6, and 7 d after treatment with different herbicide SOA.
2.8. Chlorophyll Fluorescence (Fv/Fm) Measurement
A chlorophyll fluorometer OS30p+ (Opti-Sciences Inc., Hudson, NH, USA) was used to measure the efficiency of photosynthetic light reaction (PSII) as indicated by chlorophyll fluorescence, which is highly sensitive to stress [20] . Chlorophyll fluorescence in kochia was measured at 6 h, 1, 2, 3, 4, 5, 6, and 7 d after treatment with different herbicide SOA.
2.9. Statistical Analysis
Fold-change in gene expression (as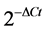 ) was calculated by the comparative Ct method [21] , relative to the adjuvant treated control samples, where △Ct = [Ct target gene − Ct reference gene]. β-tubulin was used as an endogenous reference gene (housekeeping gene) for normalizing the gene expression data [22] . A calibrator sample (untreated-no adjuvant) was also included to represent 1x expression of gene of interest. Gene expression data was analyzed using one-way ANOVA in SigmaPlot (version 12.3). Means were separated using Fisher’s LSD (p < 0.05) and standard deviations (SD) were calculated based upon three biological replicates (n = 3). Chlorophyll index and fluorescence were measured on four plants (four biological replicates, n = 4) for each herbicide treatment at each time point and the data was analyzed using one-way ANOVA in SigmaPlot (version 12.3). Means of herbicide treatments at each time point were compared with control (untreated means) using Holm-Sidak’s test (p < 0.05).
) was calculated by the comparative Ct method [21] , relative to the adjuvant treated control samples, where △Ct = [Ct target gene − Ct reference gene]. β-tubulin was used as an endogenous reference gene (housekeeping gene) for normalizing the gene expression data [22] . A calibrator sample (untreated-no adjuvant) was also included to represent 1x expression of gene of interest. Gene expression data was analyzed using one-way ANOVA in SigmaPlot (version 12.3). Means were separated using Fisher’s LSD (p < 0.05) and standard deviations (SD) were calculated based upon three biological replicates (n = 3). Chlorophyll index and fluorescence were measured on four plants (four biological replicates, n = 4) for each herbicide treatment at each time point and the data was analyzed using one-way ANOVA in SigmaPlot (version 12.3). Means of herbicide treatments at each time point were compared with control (untreated means) using Holm-Sidak’s test (p < 0.05).
3. Results
3.1. Gene Expression in Response to Atrazine Treatment
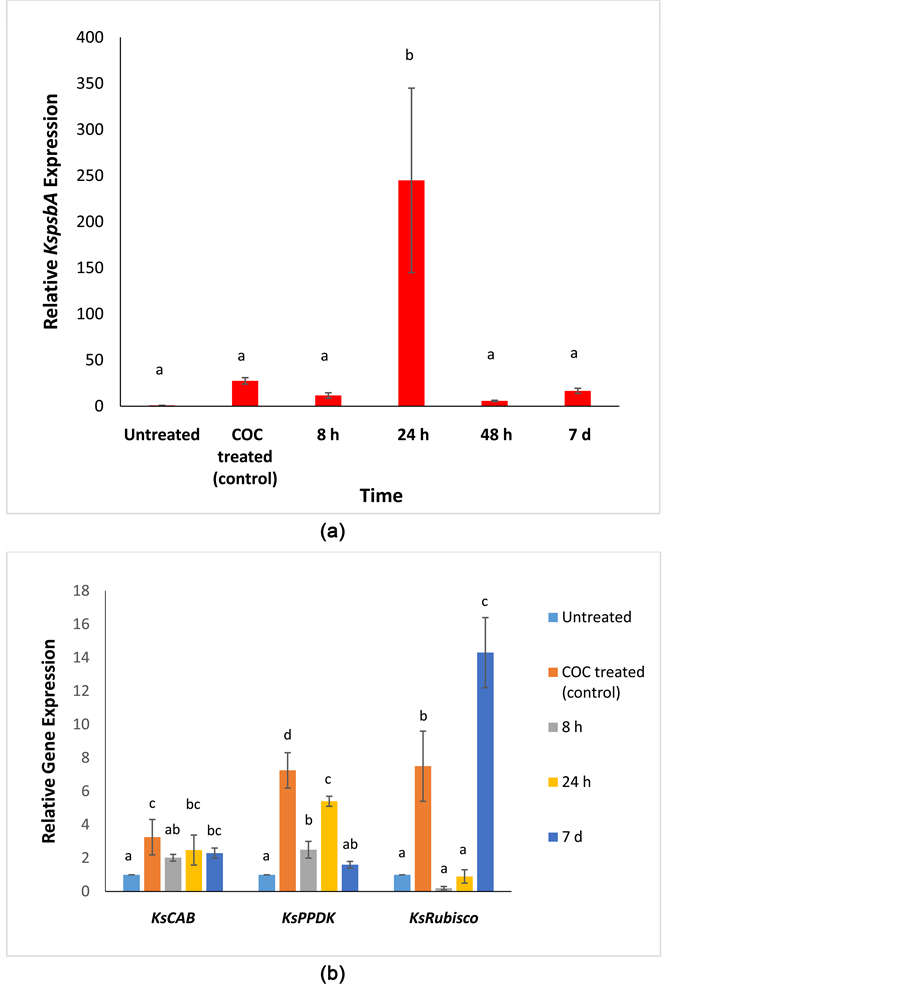
There was a >200-fold increase (p ≤ 0.001) in KspsbA gene expression at 24 h after treatment (HAT) with atrazine (Figure 1(a)). Levels of KspsbA transcript returned to the levels found in untreated plants 48 h after atrazine treatment. In contrast, transcript levels of KsCAB, KsPPDK, and KsRubisco were downregulated (p ≤ 0.020) at 8 and 24 h after atrazine treatment (Figure 1(b)). KsRubisco levels reached a peak (7-fold relative to control, p ≤ 0.001) at 7 d after atrazine treatment (Figure 1(b)).
3.2. Gene Expression in Response to Chlorsulfuron Treatment
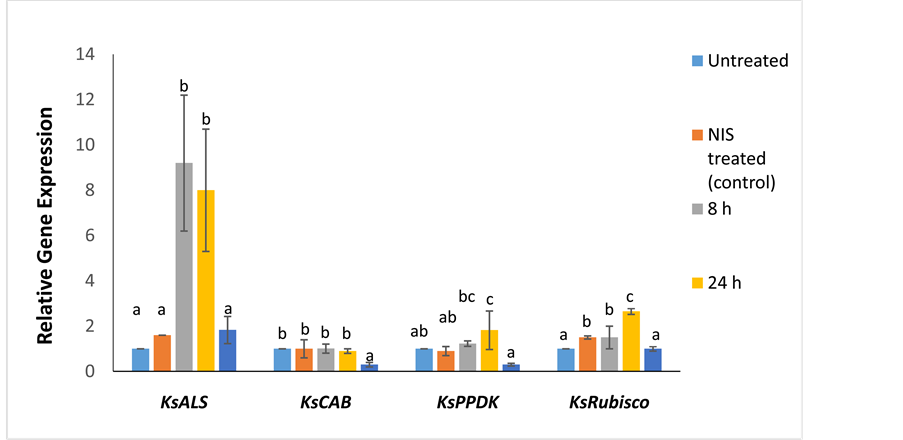
There was a significant upregulation of the KsALS gene (~9-fold relative to control, p ≤ 0.001) immediately after chlorsulfuron treatment, followed by a 7-fold increase at 24 HAT (Figure 2). KsALS transcript levels returned to near control levels at 7 d after treatment with chlorsulfuron. The expression of KsCAB remained unaffected at 8 and 24 HAT (p ≤ 0.008), whereas KsPPDK was upregulated at 24 HAT (p ≤ 0.011) (Figure 2). KsRubisco was upregulated 2.6-fold relative to untreated control at 24 HAT with chlorsulfuron (p ≤ 0.001) (Figure 2). At 7 d after chlorsulfuron treatment, the transcript levels of KsCAB, KsPPDK,
Figure 1. (a) Relative fold expression of KspsbA gene in Kochia scoparia after atrazine treatment at different time points (8 h, 24 h, 48 h, and 7 d). Different letters indicate significant differences (p < 0.05) among the means of each gene. The expression of the KspsbA gene was measured relative to a COC treated control. Untreated samples (not treated with either adjuvant or herbicide) were used as the calibrator. Error bars represent ± standard deviation from the mean (n = 3). The qPCR data was normalized using β-tubulin as the reference gene. COC refers to treatment with crop oil concentrate. (b) Relative fold expression of the photosynthetic genes (KsCAB, KsPPDK, and KsRubisco) in Kochia scoparia after atrazine treatment at different time points (8 h, 24 h, and 7 d). Different letters indicate significant differences (p < 0.05) among the means of each gene. The expression of the photosynthetic genes was measured relative to a COC treated control. Untreated samples (not treated with either adjuvant or herbicide) were used as the calibrator. Error bars represent ± standard deviation from the mean (n = 3). The qPCR data was normalized using β-tubulin as the reference gene. COC refers to treatment with crop oil concentrate.
Figure 2. Relative fold expression of the target-site (KsALS) and photosynthetic genes (KsCAB, KsPPDK, and KsRubisco) in Kochia scoparia at different time points (8 h, 24 h, and 7 d) after chlorsulfuron treatment. Different letters indicate significant differences (p < 0.05) among the means of each gene. The expression of the target-site and photosynthetic genes was measured relative to a NIS treated control. Untreated samples (not treated with either adjuvant or herbicide) were used as the calibrator. Error bars represent ± standard deviation from the mean (n = 3). Mean values were based upon three biological and nine technical replicates. The qPCR data was normalized using β-tubulin as the reference gene. NIS refers to treatment with non-ionic surfactant.
and KsRubisco were found to be lower compared to their controls.
3.3. Gene Expression in Response to Glyphosate Treatment
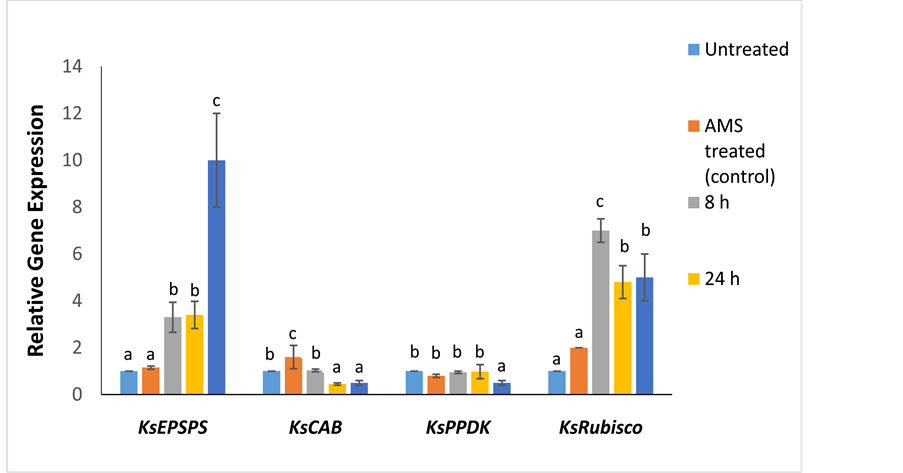
KsEPSPS gene was upregulated (3.3-fold, p ≤ 0.001) at 8 HAT with glyphosate, maintaining similar transcript levels (3.4-fold) at 24 HAT as well (Figure 3). There was a 10-fold increase in KsEPSPS levels 7 d after glyphosate treatment (Figure 3). Compared to other herbicide treatments, KsRubisco showed the maximum expression levels after glyphosate treatment, reaching a peak at 8 h (7-fold relative to control, p ≤ 0.001) (Figure 3). A high KsRubisco level (~5- fold) was maintained even 7 d after glyphosate treatment. In contrast, KsCAB and KsPPDK genes were relatively downregulated (~0.5-fold, p ≤ 0.001, p ≤ 0.01) 7 d after glyphosate treatment (Figure 3).
3.4. Chlorophyll Index and Fluorescence (Fv/Fm)
In general, upon treatment with either atrazine, chlorsulfuron, or glyphosate, there was no significant change (except at few time points) in chlorophyll index in kochia compared to untreated plants (control) during the seven-day period after herbicide treatments (Figure 4(a)). Chlorophyll fluorescence, on the other hand varied, especially after atrazine treatment (Figure 4(b)). There was a significant decline in chlorophyll fluorescence at 6 h (indicated by 0 on the x-axis), and the decreasing trend continued for the next 7 d after atrazine treatment (p ≤ 0.001) (Figure 4(b)). However, after glyphosate and chlorsulfuron treatments, the chlorophyll fluorescence remained unchanged compared to untreated kochia (Figure 4(b)).
4. Discussion
Inhibition of a specific biosynthetic pathway in plants can not only affect the expression of genes in that pathway, but also result in corresponding changes in the gene expression in other related pathways [23] . For example, interaction between different components of photosynthetic machinery, as well as with those in pathways associated with amino acid synthesis, stress response, hormonal regulation etc. has been reported [24] [25] [26] . The biochemical pathways that have target sites of most of the commonly used herbicides share a close relation [27] . For example, chorismate, one of the major intermediates of the shikimate pathway involving EPSPS, serves as a branch point for synthesis of various branched chain aromatic amino acids [28] . The study of the effect of herbicides on their target-sites and other related gene networks helps in deciphering the cross-talk between various biochemical pathways and the events leading to plant death [29] [30] .
4.1. Expression of Target-Site Genes upon Herbicide Treatments
There was a strong upregulation (~200 fold) of the KspsbA gene in kochia at 24 HAT with atrazine (Figure 1(a)). The KspsbA expression returned to the same level as in control by 48 h and continued to stay at that level 7 d after atrazine
Figure 3. Relative fold expression of the target-site (KsEPSPS) and photosynthetic genes (KsCAB, KsPPDK, and KsRubisco) in Kochia scoparia at different time points (8 h, 24 h, and 7 d) after glyphosate treatment. Different letters indicate significant differences (p < 0.05) among the means of each gene. The expression of the target-site and photosynthetic genes was measured relative to an AMS treated control. Untreated samples (not treated with either adjuvant or herbicide) were used as the calibrator. Error bars represent ± standard deviation from the mean (n = 3). The qPCR data was normalized using β-tubulin as the reference gene. AMS refers to treatment with ammonium sulphate.
Figure 4. (a) Chlorophyll index in Kochia scoparia during the 7 d period after treatment with herbicides. Error bars represent ± standard deviation from the mean (n = 4). Six h time point is indicated as 0 d. Significance between herbicide treatments and control at each time point is indicated by the colored asterisks corresponding to each herbicide treatment (*p ≤ 0.05, **p ≤ 0.01). (b) Chlorophyll fluorescence in Kochia scoparia during the 7 d period after treatment with herbicides. Error bars represent ± standard deviation from the mean (n = 4). Six h time point is indicated as 0 d. Significance between herbicide treatments and control at each time point is indicated by the colored asterisks corresponding to each herbicide treatment (*p ≤ 0.05, ***p ≤ 0.001).
treatment. D1 protein, encoded by the psbA gene is the target-site of PSII-inhi- bitors [31] [32] . A high Dl protein degradation and turnover rates have been reported after photoinhibition [33] . These high Dl protein turnover rates are essential for repair of the photoinhibitory damage caused by PSII-inhibitors. The high D1 protein turnover is also accompanied by corresponding increase in psbA gene transcription [34] . In this study, the high KspsbA gene expression in kochia after atrazine application can be explained by the rapid turnover of the D1 protein. The photoinhibitory damage caused by atrazine also leads to production of reactive oxygen species (ROS) such as singlet oxygen (1O2) [35] . The singlet oxygen and the other free radicals produced after the blockage of electron transfer by triazines result in an irreversible damage to photosynthetic machinery contributing to plant death [36] .
ALS-inhibitor herbicides suppress the biosynthesis of branched-chain amino acids (e.g. valine, leucine, and isoleucine) through the inhibition of acetolactate synthase. Transcript levels of the KsALS were approximately 9 and 7-fold higher in K. scoparia at 8 and 24 HAT (Figure 2). Several studies have determined the expression of protein after the application of ALS-inhibitors, but there is insufficient literature regarding the expression of ALS gene. A three- to four-fold variation in ALS gene expression has been reported in several tobacco organs such as leaves, seedlings, and flowers [37] . An increase in the free amino acid pool after the application of ALS-inhibitors has been reported [38] [39] [40] . This increased protein pool was attributed to an increase in protein turnover rates in response to treatment with ALS-inhibitors [41] . The higher expression levels of KsALS gene observed in this study, may lead to increased protein turnover and free amino acid pools after application of ALS-inhibitors. The increased protein turnover is due to increased protein degradation and reduced protein synthesis [42] .
Gaines et al. [43] found no difference in EPSPS transcript levels in Palmer amaranth (Amaranthus palmeri S. Wats.) 8 h after glyphosate application. Similarly, there was no induction of EPSPS transcripts in horseweed (Conyza canadensis L. Cronq.) at 21 d after treatment with glyphosate [44] . In contrast, Gao et al. [45] recently reported upregulation (two to three-fold) of EPSPS gene in annual wild soybean (Glycine soja) at 6 h after glyphosate treatment. In the current study, we observed a similar three-fold increase in KsEPSPS expression at 8 and 24 h followed by a ten-fold increase at 7 d after glyphosate treatment (Figure 3). Although both inhibit different pathways, glyphosate induces physiological responses similar to ALS-inhibitors in plants, resulting in an increased free amino acid content (as discussed above) and an impaired carbon metabolism leading to plant death [39] [42] [46] . Similar to ALS-inhibitors, glyphosate application also increases EPSPS transcript levels (Figure 3), possibly resulting in higher protein turnover and degradation.
4.2. Expression of Photosynthetic Genes upon Herbicide Treatments
The expression of Rubisco is dependent on various factors such as light [47] [48] , cell and tissue type [49] [50] , efficiency of photosynthetic machinery [51] , phytohormone and nutrient levels [52] [53] , and plant developmental stage [54] . Rubisco consists of two subunits (large and small), encoded by two separate genes (chloroplastic large subunit, LSU and nuclear small subunit, SSU) [55] . In this investigation the expression of KsRubisco (LSU) was determined in response to herbicides with different SOA.
KsRubisco levels varied with different herbicide treatments. KsRubisco was upregulated (6-fold) at 8 h and its transcript levels remained high (4-fold) in kochia even at 7 d after treatment with glyphosate (Figure 3). Glyphosate is known to induce physiological changes leading to increased photosynthetic rates and growth stimulations [56] . An increase in shikimate is the most commonly observed biochemical change in response to glyphosate application. Both shikimate and photosynthetic pathways are known to be closely related, as majority of carbon that is synthesized during photosynthesis feeds the shikimic acid pathway [57] . Increased photosynthetic rate observed in barley after glyphosate treatment was attributed to increased carbon fixation resulting from higher Rubisco expression and RuBP (ribulose-biphosphate) turnover rates [56] [58] . In the current study, the strong upregulation of KsRubisco at 8 HAT with glyphosate (Figure 3) supports previous reports. However, both chlorophyll index and fluorescence (Fv/Fm) remained unchanged (Figure 4(a) and Figure 4(b)) during the 7 d period after glyphosate treatment. There was a 2.6-fold increase in KsRubisco expression in kochia after chlorsulfuron treatment (Figure 2). The upregulation of Rubisco after chlorsulfuron treatment is likely to have caused as a result of the effect of ALS-inhibitors on carbon metabolism. Amino acid synthesis inhibitors such as glyphosate and chlorsulfuron, are known to induce such physiological responses in plants [42] .
In the current study, KsRubisco was downregulated (at 8 and 24 HAT with atrazine) when compared to the control, however maximum expression of KsRubisco (~7-fold relative to control) was seen 7 d after atrazine treatment (Figure 1). Previous reports suggest that the specific activity of Rubisco was significantly reduced in maize (Zea mays L.) during the first two days after atrazine treatment followed by a recovery phase after one week [59] . The high expression of KsRubisco observed in this study most likely resulted due to the initiation of recovery phase 7 d after atrazine treatment.
Similar to Rubisco, photosynthetic efficiency in plants is highly dependent on PPDK activity [60] . A reduction in the activity of PPDK would cause less production of PEP for PEPC action, leading to reduced CO2 fixation by Rubisco in the bundle sheath cells [59] . Nemat Alla et al. [59] observed a reduction in maize PPDK level within the first 2 d after treatment with ALS-inhibitors such as rimsulfuron and imazethapyr, followed by an increase in PPDK levels for the next 10 d. In our study, KsPPDK transcript levels were increased 1.8-fold at 24 h and then decreased (0.3-fold) 7 d after treatment with chlorsulfuron relative to the control (Figure 2).
In the current study, KsPPDK was found to be downregulated relative to the control at 8, 24 h, and 7 d after atrazine treatment (Figure 1(b)). Similarly, Nemat Alla et al. [59] reported a decreased PPDK activity in maize in the first two days after atrazine application and the decreasing trend continued for the next 10 d. PSII-inhibitors such as atrazine block the electron transport chain and the ATP production in the mesophyll cells, causing severe imbalance in the C4 photosynthetic pathway.
The transcript levels of KsPPDK were relatively unaffected by glyphosate treatment (Figure 3). The regulation of carbon flux is dependent on the availability of precursors E4P (D-erythrose 4-phosphate) and PEP for the enzyme 3-Deoxy-D-arabino-heptulosonate7-phosphate (DAHP) synthase, which catalyzes the first committed step of the shikimate pathway [61] . The conversion of pyruvate to PEP by plastidic PPDK is one of the several pathways that maintain a constant supply of PEP for the shikimate pathway to function [62] [63] . The requirement for PEP should be minimum when shikimate pathway is inhibited by glyphosate, and as a result PPDK activity may not be crucial.
KsCAB showed downregulation relative to control at 8 h after atrazine treatment (Figure 1(b)). Similarly, expression of CAB gene in soybean was reported to be downregulated at 1, 2, 4, and 8 HAT with atrazine [64] . Expression of CAB is influenced by several abiotic stress factors such as salinity, metal ions, and ultraviolet radiation [65] [66] , and is known to be active during the production ROS in chloroplasts [67] [68] .
KsCAB gene expression remained unchanged at 8 and 24 h, and downregulated (0.3-fold relative to control) at 7 d after treatment with chlorsulfuron (Figure 2). The cause for the downregulation of KsCAB at 7 d after chlorsulfuron treatment is unknown. Similar downregulation of KsCAB was observed at 24 h and maintained until 7 d after glyphosate treatment (Figure 3). Jiang et al. [27] reported a downregulation of CAB genes in soybean at 24 and 72 HAT with glyphosate. The observed downregulation of KsCAB after glyphosate application may be due to inhibition of glutamine synthetase 2 (GS2), a key enzyme for the synthesis of precursor glutamine for chlorophyll biosynthesis via the C5 pathway [27] [69] [70] .
4.3. Chlorophyll Index and Fluorescence (Fv/Fm)
Leaf chlorophyll index is an important parameter for evaluating general plant health [19] . In the current study, we observed no change (compared to untreated control) in the leaf chlorophyll index of kochia during the one week after treatment with three SOA herbicides (Figure 4(a)). Previous studies in common lambsquarters (Chenopodium album L.) and in some other weed species have shown no change in the leaf chlorophyll index in the first week after herbicide treatment [71] [72] .
Chlorophyll fluorescence (Fv/Fm) indicates the photosynthetic capability and the quantum efficiency of the PSII, which is highly sensitive to stress [20] . In this study, significant decrease in the chlorophyll fluorescence in kochia was observed after atrazine treatment (Figure 4(b)). A decrease in Fv/Fm values suggests an increase in dissipation of energy as heat and inhibition of photosynthetic machinery [73] . Atrazine, being a PSII inhibitor, has a more direct effect on the efficiency of PSII compared to other SOA herbicides. The blockage of the electron transport chain by the atrazine treatment would eventually affect the functioning of the PSII with corresponding decrease in Fv/Fm values.
4.4. Conclusions and Future Directions
In summary, this study clearly demonstrates differential expression of target-site and photosynthetic genes in response to herbicide treatments in kochia. Herbicide target-site genes (KsALS, KspsbA, and KsEPSPS) were readily upregulated after herbicide treatment. It is important to realize that herbicide application, directly or indirectly, not only influences the targeted pathway, but also other critical pathways such as Calvin and C4 cycles leading to tissue or plant death. In future, a more elaborate gene expression study (high-throughput) using RNA sequencing tools is essential in order to elucidate the expression levels of not only photosynthetic genes after herbicide application, but also genes related to plant defense and stress response.
Acknowledgements
The authors acknowledge the grant support provided by the Fulbright Fellowship Program (Dr. Shaniyar Bayramov), and the Kansas State University Research and Extension. The authors would also like to acknowledge Dr. David Horvath, Research Plant Physiologist (USDA-ARS Sunflower Research Unit, Fargo, USA) for critically reviewing this manuscript. This manuscript is approved for publication as Kansas Agricultural Experiment Station Contribution No. 16-370-J.
Cite this paper
Varanasi, V.K., Bayramov, S., Prasad, P.V.V. and Jugulam, M. (2017) Expression Profiles of psbA, ALS, EPSPS, and Other Chloroplastic Genes in Response to PSII-, ALS-, and EPSPS-Inhi- bitor Treatments in Kochia scoparia. Ame- rican Journal of Plant Sciences, 8, 451-470. https://doi.org/10.4236/ajps.2017.83031
References
- 1. Friesen, L.F., Beckie, H.J., Warwick, S.I. and Van Acker R.C. (2009) The Biology of Canadian Weeds. 138. Kochia scoparia (L.) Schrad. Canadian Journal of Plant Science, 89, 141-167.
https://doi.org/10.4141/CJPS08057 - 2. Wang, C., Guo, L., Li, Y. and Wang, Z. (2012) Systematic Comparison of C3 and C4 Plants Based on Metabolic Network Analysis. BMC Systems Biology, 6, S9.
https://doi.org/10.1186/1752-0509-6-s2-s9 - 3. Majeran, W., Friso, G., Ponnala, L., Connolly, B., Huang, M., Reidel, E., Zhang, C., Asakura, Y., Bhuiyan, N.H., Sun, Q., Turgeon, R. and van Wijk, K.J. (2001) Structural and Metabolic Transitions of C4 Leaf Development and Differentiation Defined by Microscopy and Quantitative Proteomics in Maize. Plant Cell, 22, 3509-3542.
https://doi.org/10.1105/tpc.110.079764 - 4. Tovar-Mendez, A. and Munoz-Clares, R. (2001) Kinetics of Phosphoenolpyruvate Carboxylase from Zea mays Leaves at High Concentration of Substrates. Biochimica et Biophysica Acta, 1546, 242-252.
https://doi.org/10.1016/S0167-4838(01)00148-0 - 5. Jansson, S. (1994) The Light-Harvesting Chlorophyll a/b-Binding Proteins Biochimica Biophysica Acta, 1184, 1-19.
- 6. Jansson, S. (1999) A Guide to the Lhc Genes and Their Relatives in Arabidopsis. Trends in Plant Science, 4, 236-240.
https://doi.org/10.1016/S1360-1385(99)01419-3 - 7. Hess, F.D. (2000) Light-Dependent Herbicides: An Overview. Weed Science, 48, 160-170.
https://doi.org/10.1614/0043-1745(2000)048[0160:LDHAO]2.0.CO;2 - 8. Steinback, K.E., McIntosh, L., Bogorad, L. and Arntzen, C.J. (1981) Identification of the Triazine Receptor Protein as a Chloroplast Gene Product. Proceedings of National Academy of Sciences, USA, 78, 7463-7467.
https://doi.org/10.1073/pnas.78.12.7463 - 9. Lamego, F.P, Charlson, D., Delatorre, C.A., Burgos, N.R. and Vidal, R.A. (2009) Molecular Basis of Resistance to ALS-Inhibitor Herbicides in Greater Beggarticks. Weed Science, 57, 474-481.
https://doi.org/10.1614/WS-09-056.1 - 10. Devine, M.D. and Eberlein, C.V. (1997) Physiological, Biochemical and Molecular Aspects of Herbicide Resistance Based on Altered Target Sites. In: Roe, R.M., Burton, J.D. and Kuhr, R.J., Eds., Herbicide Activity: Toxicology, Biochemistry and Molecular Biology, IOS, Amsterdam, 159-185.
- 11. Steinrücken, H.C. and Amrhein, N. (1980) The Herbicide Glyphosate Is a Potent Inhibitor of 5-Enolpyruvylshikimic Acid-3-Phosphate Synthase. Biochemical and Biophysical Research Communications, 94, 1207-1212.
https://doi.org/10.1016/0006-291X(80)90547-1 - 12. Schonbrunn, E., Eschenburg, S., Shuttleworth, W.A., Schloss, J.V., Amrhein, N., Evans, J.N. and Kabsch, W. (2001) Interaction of the Herbicide Glyphosate with Its Target Enzyme 5-Enolpyruvylshikimate 3-Phosphate Synthase in Atomic Detail. Proceedings of National Academy of Sciences, USA, 98, 1376-1380.
https://doi.org/10.1073/pnas.98.4.1376 - 13. Corpet, F. (1988) Multiple Sequence Alignment with Hierarchical Clustering. Nucleic Acids Research, 16, 10881-10890.
https://doi.org/10.1093/nar/16.22.10881 - 14. Lara, M.V., Offermann, S., Smith, M., Okita, T.W., Andreo, C.S. and Edwards, G.E. (2008) Leaf Development in the Single-Cell C4 System in Bienertia sinuspersici: Expression of Genes and Peptide Levels for C4 Metabolism in Relation to Chlorenchyma Structure Under Different Light Conditions. Plant Physiology, 148, 593-610.
https://doi.org/10.1104/pp.108.124008 - 15. Wiersma, A.T, Gaines, T.A., Preston, C., Hamilton, J.P., Giacomini, D., Buell, C.R., Leach, J.E. and Westra, P. (2015) Gene Amplification of 5-Enol-Pyruvylshikimate-3-Phosphate Synthase in Glyphosate-Resistant Kochia scoparia. Planta, 241, 463-474.
https://doi.org/10.1007/s00425-014-2197-9 - 16. Chao, W.S., Dogramaci, M., Foley, M.E., Horvath, D.P. and Anderson, J.V. (2012) Selection and Validation of Endogenous Reference Genes for qRT-PCR Analysis in Leafy Spurge (Euphorbia esula). PLoS ONE, 7, e42839.
https://doi.org/10.1371/journal.pone.0042839 - 17. Chandna, R., Augustine, R. and Bisht, N.C. (2012) Evaluation of Candidate Reference Genes for Gene Expression Normalization in Brassica juncea Using Real Time Quantitative RT-PCR. PLoS ONE, 7, e36918.
https://doi.org/10.1371/journal.pone.0036918 - 18. Ling, Q., Huang, W. and Jarvis, P. (2011) Use of a SPAD-502 Meter to Measure Leaf Chlorophyll Concentration in Arabidopsis thaliana. Photosynthesis Research, 107, 209-214.
https://doi.org/10.1007/s11120-010-9606-0 - 19. Uddling, J., Gelang-Alfredsson, J., Piikki, K. and Pleijel, H. (2007) Evaluating the Relationship between Leaf Chlorophyll Concentration and SPAD-502 Chlorophyll Meter Readings. Photosynthesis Research, 91, 37-46.
https://doi.org/10.1007/s11120-006-9077-5 - 20. Krause, G.H. and Weis, E. (1991) Chlorophyll Fluorescence and Photosynthesis: The Basics. Annual Review of Plant Physiology and Plant Molecular Biology, 42, 313-349.
https://doi.org/10.1146/annurev.pp.42.060191.001525 - 21. Schmittgen, T.D. and Livak, K.J. (2008) Analyzing Real-Time PCR Data by the Comparative CT Method. Nature Protocols, 3, 1101-1108.
https://doi.org/10.1038/nprot.2008.73 - 22. Godar, A.S., Varanasi, V.K., Nakka, S., Vara Prasad, P.V., Thompson, C.R. and Mithila, J. (2015) Physiological and Molecular Mechanisms of Differential Sensitivity of Palmer amaranth (Amaranthus palmeri) to Mesotrione at Varying Growth Temperatures. PLoS ONE, 10, e0126731.
https://doi.org/10.1371/journal.pone.0126731 - 23. Guyer, D., Patton, D. and Ward, E. (1995) Evidence for Cross-Pathway Regulation of Metabolic Gene Expression in Plants. Proceedings of National Academy of Sciences of the USA, 92, 4997-5000.
https://doi.org/10.1073/pnas.92.11.4997 - 24. Atkinson, N.J. and Urwin, P.E. (2012) The Interaction of Plant Biotic and Abiotic Stresses: From Genes to the Field. Journal of Experimental Botany, 63, 3523-3543.
https://doi.org/10.1093/jxb/ers100 - 25. Kerchev, P.I., Fenton, B., Foyer, C.H. and Hancock, R.D. (2012) Plant Responses to Insect Herbivory: Interactions between Photosynthesis, Reactive Oxygen Species and Hormonal Signalling Pathways. Plant, Cell and Environment, 35, 441-453.
https://doi.org/10.1111/j.1365-3040.2011.02399.x - 26. Yu, X., Zheng, G., Shan, L., Meng, G., Vingron, M., Liu, Q. and Zhu, X.G. (2014) Reconstruction of Gene Regulatory Network Related to Photosynthesis in Arabidopsis thaliana. Frontiers in Plant Science, 5, 273.
https://doi.org/10.3389/fpls.2014.00273 - 27. Jiang, L.X., Jin, L.G., Guo, Y., Tao, B. and Qiu, L.J. (2013) Glyphosate Effects on the Gene Expression of the Apical Bud in Soybean (Glycine max). Biochemical and Biophysical Research Communications, 437, 544-549.
https://doi.org/10.1016/j.bbrc.2013.06.112 - 28. Tzin, V. and Galili, G. (2010) The Biosynthetic Pathways for Shikimate and Aromatic Amino Acids in Arabidopsis thaliana. The Arabidopsis Book, 8, e0132.
- 29. Duke, S.O., Bajsa, J. and Pan, Z. (2013) Omics Methods for Probing the Mode of Action of Natural and Synthetic Phytotoxins. Journal of Chemical Ecology, 39, 333-347.
https://doi.org/10.1007/s10886-013-0240-0 - 30. Dayan, F.E. and Duke, S.O. (2014) Natural Compounds as Next-Generation Herbicides. Plant Physiology, 166, 1090-1105.
https://doi.org/10.1104/pp.114.239061 - 31. Zurawski, G., Bohnert, H.J., Whitfeld, P.R. and Bottomley, W. (1982) Nucleotide Sequence of the Gene for the M(r) 32,000 Thylakoid Membrane Protein from Spinacia oleracea and Nicotiana debneyi Predicts a Totally Conserved Primary Translation Product of M(r) 38,950. Proceedings of National Academy of Sciences, USA, 79, 7699-7703.
https://doi.org/10.1073/pnas.79.24.7699 - 32. Gronwald, J.W. (1994) Resistance to Photosystem II Inhibiting Herbicides. In: Powles, S.B. and Holtum, J.A.M., Eds., Herbicide Resistance in Plants: Biology and Biochemistry, Lewis Publishers, Boca Raton, 27-60.
- 33. Sundby, C., McCaffery, S. and Anderson, J.M. (1993) Turnover of the Photosystem II Dl Protein in Higher Plants under Photoinhibitory and Nonphotoinhibitory Irradiance. The Journal of Biological Chemistry, 268, 25476-25482.
- 34. Mulo, P., Sakurai, I. and Aro, E.M. (2012) Strategies for psbA Gene Expression in Cyanobacteria, Green Algae and Higher Plants: From Transcription to PSII Repair. Biochimica et Biophysica Acta, 1817, 247-257.
https://doi.org/10.1016/j.bbabio.2011.04.011 - 35. Boulahia, K., Carol, P., Planchais, S. and Abrous-Belbachir, O. (2016) Phaseolus vulgaris L. Seedlings Exposed to Prometryn Herbicide Contaminated Soil Trigger an Oxidative Stress Response. Journal of Agricultural and Food Chemistry, 64, 3150-3160.
https://doi.org/10.1021/acs.jafc.6b00328 - 36. Rutherford, A.W. and Krieger-Liszkay, A. (2001) Herbicide-Induced Oxidative Stress in Photosystem II. Trends in Biochemical Sciences, 26, 648-653.
https://doi.org/10.1016/S0968-0004(01)01953-3 - 37. Keeler, S.J., Sanders, P., Smith, J.K. and Mazur, B.J. (1993) Regulation of Tobacco Acetolactate Synthase Gene Expression. Plant Physiology, 102, 1009-1018.
https://doi.org/10.1104/pp.102.3.1009 - 38. Anderson, P. and Hibberd, K. (1985) Evidence for the Interaction of an Imidazolinone Herbicide with Leucine, Valine, and Isoleucine Metabolism. Weed Science, 33, 479-483. http://www.jstor.org/stable/4044133
- 39. Shaner, D.L. and Reider, M.L. (1986) Physiological Responses of Corn (Zea mays) to AC 243,997 in Combination with Valine, Leucine, and Isoleucine. Pesticide Biochemistry and Physiology, 25, 248-257.
https://doi.org/10.1016/0048-3575(86)90051-9 - 40. Orcaray, L., Igal, M., Marino, D., Zabalza, A. and Royuela, M. (2010) The Possible Role of Quinate in the Mode of Action of Glyphosate and Acetolactate Synthase Inhibitors. Pest Management Science, 66, 262-269.
https://doi.org/10.1002/ps.1868 - 41. Rhodes, D., Hogan, A.L., Deal, L., Jamieson, G.C. and Haworth, P. (1987) Amino Acid Metabolism of Lemna minor L. II. Responses to Chlorsulfuron. Plant Physiology, 84, 775-780.
https://doi.org/10.1104/pp.84.3.775 - 42. Zulet, A., Gil-Monreal, M., Villamor, J.G., Zabalza, A., van der Hoorn, R.A. and Royuela, M. (2013) Proteolytic Pathways Induced by Herbicides that Inhibit Amino Acid Biosynthesis. PLoS ONE, 8, e73847.
https://doi.org/10.1371/journal.pone.0073847 - 43. Gaines, T.A., Zhang, W., Wang, D., Bukun, B., Chisholm, S.T., Shaner, D.L., Nissen, S.J., Patzoldt, W.L., Tranel, P.J., Culpepper, A.S., Grey, T.L., Webster, T.M., Vencill, W.K., Sammons, R.D., Jiang, J., Preston, C., Leach, J.E. and Westra, P. (2010) Gene Amplification Confers Glyphosate Resistance in Amaranthus palmeri. Proceedings of National Academy of Sciences, USA, 107, 1029-1034.
https://doi.org/10.1073/pnas.0906649107 - 44. Dinelli, G., Marotti, I., Bonetti, A., Minelli, M., Catizone, P. and Barnes, J. (2006) Physiological and Molecular Insight on the Mechanisms of Resistance to Glyphosate in Conyza canadensis (L.) Cronq. Biotypes. Pesticide Biochemistry and Physiology, 86, 30-41.
https://doi.org/10.1016/j.pestbp.2006.01.004 - 45. Gao, Y., Tao, B., Qiu, L., Jin, L. and Wu, J. (2014) Role of Physiological Mechanisms and EPSPS Gene Expression in Glyphosate Resistance in Wild Soybeans (Glycine soja). Pesticide Biochemistry and Physiology, 109, 6-11.
https://doi.org/10.1016/j.pestbp.2013.12.005 - 46. Wang, C. (2001) Effect of Glyphosate on Aromatic Amino Acid Metabolism in Purple Nutsedge (Cyperus rotundus). Weed Technology, 15, 628-635. http://www.jstor.org/stable/3988541
https://doi.org/10.1614/0890-037X(2001)015[0628:EOGOAA]2.0.CO;2 - 47. Wang, J.L., Long, J.J., Hotchkiss, T. and Berry, J.O. (1993) C4 Photosynthetic Gene Expression in Light- and Dark-Grown Amaranth Cotyledons. Plant Physiology, 102, 1085-1093.
https://doi.org/10.1104/pp.102.4.1085 - 48. Sheen, J. (1999) C4 Gene Expression. Annual Review of Plant Physiology and Plant Molecular Biology, 50, 187-217.
https://doi.org/10.1146/annurev.arplant.50.1.187 - 49. Berry, J.O., McCormac, D.J., Long, J.J., Boinski, J.J. and Corey, A.C. (1997) Photosynthetic Gene Expression in Amaranth, an NAD-ME Type C4 Dicot. Australian Journal of Plant Physiology, 24, 423-428.
https://doi.org/10.1071/PP97001 - 50. Patel, M., Corey, A.C., Yin, L.P., Ali, S., Taylor, W.C. and Berry, J.O. (2004) Untranslated Regions from C4 Amaranth AhRbcS1 mRNAs Confer Translational Enhancement and Preferential Bundle Sheath Cell Expression in Transgenic C4 Flaveria bidentis. Plant Physiology, 136, 3550-3561.
https://doi.org/10.1104/pp.104.051508 - 51. Krapp, A., Hofmann, B., Schafer, C. and Stitt, M. (1993) Regulation of the Expression of rbcS and other Photosynthetic Genes by Carbohydrates: A Mechanism for the “Sink Regulation” of Photosynthesis? Plant Journal, 3, 817-828.
https://doi.org/10.1111/j.1365-313X.1993.00817.x - 52. Ookawa, T., Naruoka, Y., Sayama, A. and Hirasawa, T. (2004) Cytokinin Effects on Ribulose-1,5-Bisphosphate Carboxylase/Oxygenase and Nitrogen Partitioning in Rice during Ripening. Crop Science, 44, 2107-2115.
https://doi.org/10.2135/cropsci2004.2107 - 53. Imai, K., Suzuki, Y., Makino, A. and Mae, T. (2005) Effects of Nitrogen Nutrition on the Relationships between the Levels of rbcS and rbcL mRNAs and the Amount of Ribulose 1,5-Bisphosphate Carboxylase/Oxygenase Synthesized in the Eighth Leaves of Rice from Emergence through Senescence. Plant Cell and Environment, 28, 1589-1600.
https://doi.org/10.1111/j.1365-3040.2005.01438.x - 54. Hensel, L., Grbic, V., Baumgarten, D.A. and Bleecker, A.B. (1993) Developmental and Age-Related Processes That Influence the Longevity and Senescence of Photosynthetic Tissues in Arabidopsis. Plant Cell, 5, 553-564.
https://doi.org/10.1105/tpc.5.5.553 - 55. Patel, M. and Berry, J.O. (2008) Rubisco Gene Expression in C4 Plants. Journal of Experimental Botany, 59, 1625-1634.
https://doi.org/10.1093/jxb/erm368 - 56. Cedergreen, N. and Olesen, C.F. (2010) Can Glyphosate Stimulate Photosynthesis? Pesticide Biochemistry and Physiology, 96, 140-148.
https://doi.org/10.1016/j.pestbp.2009.11.002 - 57. Cobb, A.H. (1992) Auxin-Type Herbicides. In: Cobb, A.H., Ed., Herbicides and Plant Physiology, Chapman and Hall, London, 82-125.
- 58. Sharkey, T.D., Bernacchi, C.J., Farquhar, G.D. and Singsaas, E.L. (2007) Fitting Photosynthetic Carbon Dioxide Response Curves for C-3 Leaves. Plant Cell and Environment, 30, 1035-1040.
https://doi.org/10.1111/j.1365-3040.2007.01710.x - 59. Nemat Alla, M.M., Hassan, N.M. and El-Bastawisy, Z.M. (2007) Differential Influence of Herbicide Treatments on Activity and Kinetic Parameters of C4 Photosynthetic Enzymes from Zea mays. Pesticide Biochemistry and Physiology, 89, 198-205.
https://doi.org/10.1016/j.pestbp.2007.06.005 - 60. Naidu, S.L., Moose, S.P., AL-Shoaibi, A.K., Raines, C.A. and Long, S.P. (2003) Cold Tolerance of C4 Photosynthesis in Miscanthus x giganteus: Adaptation in Amounts and Sequence of C4 Photosynthetic Enzymes. Plant Physiology, 132, 1688-1697.
https://doi.org/10.1104/pp.103.021790 - 61. Maeda, H. and Dudareva, N. (2012) The Shikimate Pathway and Aromatic Amino Acid Biosynthesis in Plants. Annual Review of Plant Biology, 63, 73-105.
https://doi.org/10.1146/annurev-arplant-042811-105439 - 62. Fischer, K., Kammerer, B., Gutensohn, M., Arbinger, B., Weber, A., Hausler, R.E. and Flügge, U.I. (1997) A New Class of Plastidic Phosphate Translocators: A Putative Link between Primary and Secondary Metabolism by the Phosphoenolpyruvate/Phosphate Antiporter. Plant Cell, 9, 453-462.
https://doi.org/10.1105/tpc.9.3.453 - 63. Flugge, U.I., Hausler, R.E., Ludewig, F. and Gierth, M. (2011) The Role of Transporters in Supplying Energy to Plant Plastids. Journal of Experimental Botany, 62, 2381-2392.
https://doi.org/10.1093/jxb/erq361 - 64. Zhu, J., Patzoldt, W.L., Radwan, O., Tranel, P.J. and Clough, S.J. (2009) Effects of Photosystem-II Interfering Herbicides Atrazine and Bentazon on the Soybean Transcriptome. Plant Genome, 2, 191-205.
https://doi.org/10.3835/plantgenome2009.02.0010 - 65. Capel, J., Jarillo, J.A., Madueno, F., Jorquera, M.J., Martínez-Zapater, J.M. and Salinas, J. (1998) Low Temperature Regulates Arabidopsis Lhcb Gene Expression in a Light-Independent Manner. Plant Journal, 13, 411-418.
https://doi.org/10.1046/j.1365-313X.1998.00039.x - 66. Staneloni, R.J., Rodriguez-Batiller, M.J. and Casal, J.J. (2008) Abscisic Acid, High-Light, and Oxidative Stress Down-Regulate a Photosynthetic Gene via a Promoter Motif Not Involved in Phytochrome-Mediated Transcriptional Regulation. Molecular Plant, 1, 75-83.
https://doi.org/10.1093/mp/ssm007 - 67. Galvez-Valdivieso, G. and Mullineaux, P.M. (2010) The Role of Reactive Oxygen Species in Signaling from Chloroplasts to the Nucleus. Physiologia Plantarum, 138, 430-439.
https://doi.org/10.1111/j.1399-3054.2009.01331.x - 68. Xu, Y.H., Liu, R., Yan, L., Liu, Z.Q., Jiang, S.C., Shen, Y.Y., Wang, X.F. and Zhang, D.P. (2012) Light-Harvesting Chlorophyll a/b-Binding Proteins Are Required for Stomatal Response to Abscisic Acid in Arabidopsis. Journal of Experimental Botany, 63, 1095-1106.
https://doi.org/10.1093/jxb/err315 - 69. Von Wettstein, D., Gough, S. and Kannangara, C.G. (1995) Chlorophyll Biosynthesis. Plant Cell, 7, 1039-1057.
https://doi.org/10.1105/tpc.7.7.1039 - 70. Chatterjee, A. and Kundu, S. (2015) Revisiting the Chlorophyll Biosynthesis Pathway Using Genome Scale Metabolic Model of Oryza sativa japonica. Scientific Reports, 5, Article No. 14975.
https://doi.org/10.1038/srep14975 - 71. Ketel, D.H. (1996) Effect of Low Doses of Metamitron and Glyphosate on Growth and Chlorophyll Content of Common Lambsquarters (Chenopodium album). Weed Science, 44, 1-6.
http://www.jstor.org/stable/4045774 - 72. Sea, W.B., Sykes, N. and Downey, P.O. (2012) A New Method for Assessing Herbicide Damage in Weeds. 18th Australasian Weeds Conference, Melbourne, 8-11 October 2012, 108-111.
- 73. Song, Y., Chen, Q., Ci, D., Shao, X. and Zhang, D. (2014) Effects of High Temperature on Photosynthesis and Related Gene Expression in Poplar. BMC Plant Biology, 14, 111.
https://doi.org/10.1186/1471-2229-14-111
Abbreviations
ALS, Acetolactate synthase;
EPSPS, 5-enolpyruvylshikimate-3-phosphate synthase;
Rubisco, Ribulose-1,5-bisphosphate carboxylase;
LSU, Large subunit;
CAB, Chlorophyll a/b binding protein;
PPDK, Pyruvate, orthophosphate dikinase;
HAT, Hours after treatment;
DAT, Days after treatment;
COC, Crop oil concentrate;
NIS, Nonionic surfactant;
AMS, Ammonium sulfate;
MOA, Modes of action;
SOA, Sites of action;
PSII, Photosystem II;
ai, Active ingredient;
ae, Acid equivalent.

Submit or recommend next manuscript to SCIRP and we will provide best service for you:
Accepting pre-submission inquiries through Email, Facebook, LinkedIn, Twitter, etc.
A wide selection of journals (inclusive of 9 subjects, more than 200 journals)
Providing 24-hour high-quality service
User-friendly online submission system
Fair and swift peer-review system
Efficient typesetting and proofreading procedure
Display of the result of downloads and visits, as well as the number of cited articles
Maximum dissemination of your research work
Submit your manuscript at: http://papersubmission.scirp.org/
Or contact ajps@scirp.org
NOTES

*These authors contributed equally to the work.