Surgical Science
Vol.4 No.2(2013), Article ID:27882,10 pages DOI:10.4236/ss.2013.42032
Clinical Evaluation of TruFUSE® Lumbar Facet Fusion System
Department of Neurosurgery, University of Pittsburgh Medical Center, Pittsburgh, USA
Email: maroonjc@upmc.edu, *bostj@upmc.edu
Received December 12, 2012; revised January 14, 2013; accepted January 23, 2013
Keywords: Laminectomy; Pedicle Screw Fusion; Spondylolisthesis; TruFUSE®
ABSTRACT
Introduction: The TruFUSE® lumbar facet fusion system is a unique allograft milled bone dowel used to fuse facet joints. We evaluated subjects undergoing TruFUSE® fusion for stable grade I spondylolisthesis and stenosis comparing operative time, length of stay, blood loss and outcome to a similar literature-based cohort of patients undergoing pedicle screw fusion (PSF). Methods: From 2009 to 2011, 41 subjects (17 M, 24 F, aver. age 69.5 yr) underwent TruFUSE® facet fusion along with transverse process bone fusion and laminectomy. Length of stay, operative time, blood loss and outcomes were compared to eight literature-based cohort that analyzed similar parameters following pedicle screw fusion. Results: The 41 subjects’ mean operative time for laminectomy, transverse process fusion and TruFUSE® facet fusion was 106 min, with a mean blood loss of 145 cm3, and a mean hospital stay of 1.7 days (77% one day). A follow-up at average six months, 33 (80%) subjects reported subjective outcomes of “excellent” or “somewhat improved”, four (10%) “unchanged” and four (10%) “worse”. Flexion and extension radiographs showed 39 of the 41 patients (95%) had spinal stability at an average six months post-op and all (100%) had signs of early fusion. Discussion: TruFUSE® subjects had significantly (p < 0.0001) shorter surgeries (106 min compared to the literature data range of 185 - 240 min); significantly (p < 0.0001) shorter hospitalization (1.7 days compared to 4 - 19 days range). Mean estimated blood loss (EBL) was significantly lower (p < 0.001) (145 cm3 compared to 321 cm3 and 1082 cm3 range for PSF). Subjective outcome and radiographic stability were comparable between groups. Conclusion: This comparison using the TruFUSE® lumbar facet fusion system demonstrates improvements in length of stay, surgical blood loss, and operative time in our selected patient population compared to several published lumbar pedicle screw fusion systems outcomes. There may be potential economic benefits as a result of these improvements.
1. Introduction
In general, there is no consensus among spine surgeons as to the need for surgical fusion of the lumbar spine, of any type, following a laminectomy surgery for spinal decompression. Most surgeons, however, will agree that multiple laminectomy levels, the extent of facet disruption, underlying spinal degeneration or prior discectomy, can increase the chances of post-operative spinal instability in some patients. There is additional agreement that underlying pre-existing fixed degenerative spondylolisthesis can be a risk factor for post-operative spinal instability following lumbar laminectomy, and spinal fusion should be considered.
The decision to then fuse the associated motion segment in this subset of patients, as a means of preventing post-operative instability, must include the consideration of additional surgical risks associated with spinal fusion surgery compared to the benefit of preventing the possibility of future instability and its associated sequela. Risks associated with traditional pedicle screw fusion (PSF) with transverse process bone fusion continue to evolve. But in aged populations, especially with significant underlying co-morbidities, PSF can add significant morbidity and mortality risks [1].
Moller and Hedlund (2000) found that PSF increased surgical time and blood loss but did not improve clinical outcomes compared to non-instrumented fusion [2]. Many other studies have found significant long term outcome benefits following PSF but most concede placement of PSF instrumentation increases surgical time, blood loss, and length of hospital stay compared to laminectomy with or without transverse process fusion [3,4]. These parameters alone have been shown to add significant mortality and morbidity following any type of spine surgery with PSF [5,6].
Considering these factors, the authors undertook an evaluation of a newly introduced allograft bone dowel (TruFUSE®, minSURG, Clearwater, Fl, USA) designed to fuse the articular surfaces of the facet joint. The patient population evaluated included patients who had underlying pre-existing fixed degenerative spondylolisthesis with symptomatic lumbar stenosis, who were believed to benefit from this less invasive fusion procedure. In this article we will compare our results using TruFUSE® to a literature-based cohort of patients undergoing lumbar pedicle screw fusion that had collected similar categories of data for comparison. Significant differences in length of hospital stay, operative time, blood loss and, objective and subjective outcome measures were compared and will be discussed, along with economic data and a discussion of facet fusion using TruFUSE® technology. In addition, this article includes a detailed description of the insertion process used for the TruFUSE® implant.
2. Methods
From 2009 to 2011, 41 consecutive patients (17 M, 24 F, aver. age 69.5 yr) requiring laminectomy and fusion underwent a TruFUSE® facet fusion, performed by a single surgeon, along with transverse process bone fusion and laminectomy. Data from these patient records were collected and analyzed retrospectively. The indications for surgical intervention included a diagnosis of grade I degenerative spondylolisthesis and stenosis as determined radiographically, patient pain or other neurological deficiencies associated with this diagnosis, and minimal to no movement on flexion and extension radiographs. Preoperatively, all patients were assessed for leg or back and pain and neurological exams were performed. All patients had either pre-operative lumbar spine CT or MRI scans, along with flexion and extension spine films to assess spinal pathology and mobility. All patients had undergone routine medical evaluations and clearance prior to surgery.
2.1. Device Placement
Following the laminectomy procedure the facet joints were identified. By design, the TruFUSE® facet fusion system procedure eliminates the need to drill through the pedicle into the vertebral body. Instead, the facet joint is accessed between the superior and inferior facet articular surfaces at the most posterior point of the joint; first with guidance instrumentation (guide wire) and then a spatula, which is placed over the guide wire and seated between the facet joints (Figure 1(a)). Next, a drill guide is placed over the spatula into the mid-portion of the facet joint and two small teeth on the end of the guide engage the superior and inferior articular processes equally (Figure 1(b)). The drill guide is lightly tapped to engage the drill guide’s teeth into the joint. Once secure, the spatula and guide wire are removed and a Morse taper reamer is used to drill to a depth of 13 mm. An inserter with the TruFUSE® allograft is placed into the drill guide (Figure 1(c)) and an impactor is used to countersink the TruFUSE® allograft into the pre-drilled hole (Figure 1(d)). The TruFUSE® procedure was performed bilaterally. The standard transverse process fusions were done bilaterally with a mixture of autograft and allograft bone.
2.2. Post-Operative Assessment
Post-operative follow-up assessments continued in all patients for an average of six months (<6 months n = 26, <12 months n = 3, >12 months n = 12). During follow-up, radiographic assessment with lumbar flexion and extension, anterior-posterior, and lateral radiographs were obtained starting at three months post-op. Films were reviewed for spinal stability, facet bone dowel placement, and evidence of both facet and transverse process fusion. In certain cases, MRI with and without contrast was employed for additional visualization and assessment of post-operative status. Successful stability was defined as no movement at the affected vertebral fusion segment(s) during flexion and extension positions. The presence of early bone fusion in the transverse process area was defined as increased opacity in the area of the transverse process bone fusion. Additionally the facet joints at the level(s) of the TruFUSE® implant were evaluated for any evidence of migration, early fusion, or dislodgement from the joint space.
Post-operative assessments included ambulation status, pain symptoms and neurological changes compared to pre-operative exams. Wound healing was also assessed along with any occurrence of post-operative complications. Medical records from their latest follow-up visit were compared to the patients’ pre-operative examination. The extent to which the patients’ subjective preoperative complaints improved or worsened were then graded as excellent, somewhat improved, unchanged, somewhat worse or significantly worse compared to preoperative statements.
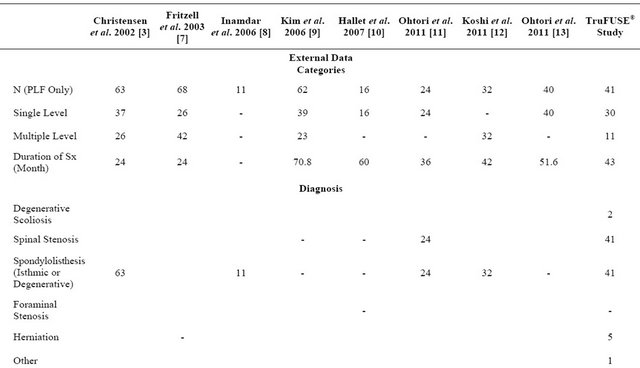
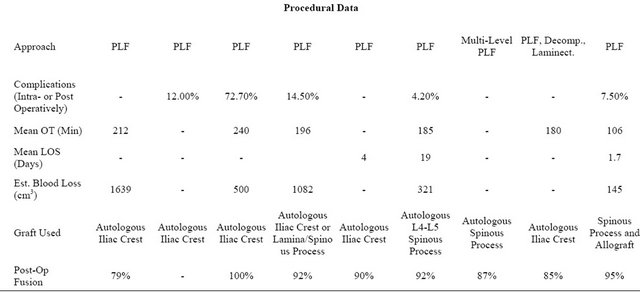
Surgical data was recorded from the patients’ records for length of hospital stay, operative time, and blood loss. These results were compared to eight studies published in the literature (Table 1) that analyzed pedicle screw fusion to the augment process fusion and laminectomy procedures that included results on the parameters listed above.
2.3. Literature Search
The PubMed database was searched for key terms until
 (a)
(a) (b)
(b)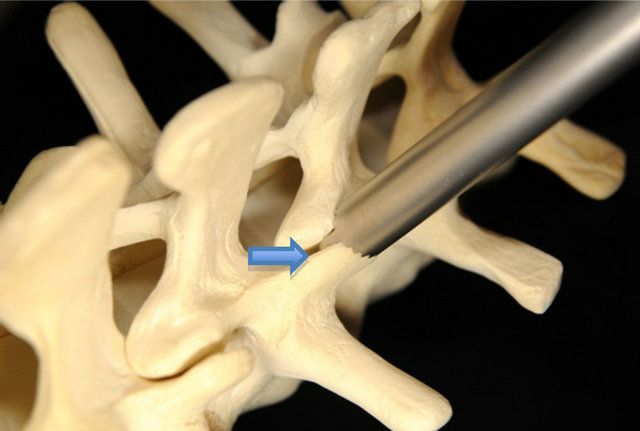 (c)
(c)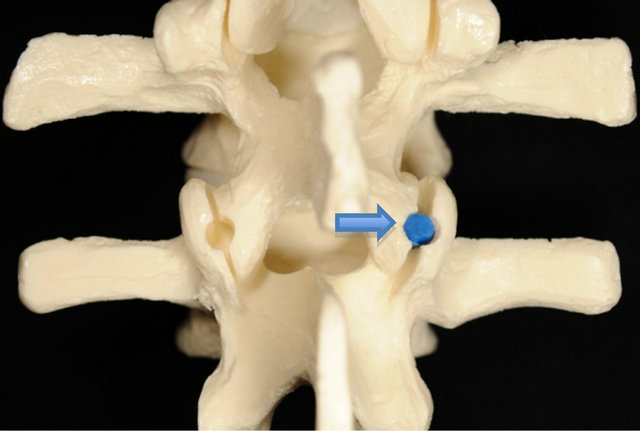 (d)
(d)
Figure 1. After the facet joint has been properly located with the guide wire, a metal spatula is inserted into the facet joint (Figure 1(a), upper left); the drill guide is then inserted over the spatula (Figure 1(b), upper right); and a Morse taper reamer is used to drill to a depth of 13 mm within the facet joint. At that time, the TruFUSE® allograft is inserted through the drill guide (Figure 1(c), bottom left); and countersunk into the facet joint (Figure 1(d), bottom right).
June 27, 2011 (last date searched) to find appropriate articles for comparison: pedicle screw fusion, lumbar fusion, outcome for pedicle screw fusion, blood loss pedicle screw fusion, length of hospital stay pedicle screw fusion, and operative time pedicle screw fusion. Of the articles found, selection criteria for comparison included: 1) the use of a cohort of at least 10 patients having received decompression and conventional instrumented PSF with posterolateral lumbar fusion; 2) the use of local or iliac crest bone grafting techniques; 3) recording of at least one of the desired parameters for comparison for the given cohort (mean operating time, mean length of stay, mean estimated blood loss); and 4) published no earlier than 2000. Studies that focused exclusively on non-instrumented fusion, alternative or minimally invasive fixation methods, anterior surgical approaches, or non-conventional grafting techniques were excluded. Studies were not excluded on the basis of patient diagnosis, demographics or length of follow-up in order to capture the performance of conventional instrumented PSF with posterolateral instrumented fusion across a variety of patient populations and practice settings. Our search revealed eight peer-reviewed articles of which met these inclusion criteria and were used to make a statistical comparison to our data collection [3,7-13].
3. Results
3.1. Treatment Related Parameters
Forty-one consecutive patients received TruFUSE® implantation and follow-up treatment over a 24-month period (24 female and 17 male). Mean age among the surgical population was 69.5 years (52 - 84 years). Patient body composition varied among the population, with a mean height of 65.5 inches (48 - 75 inches), weight of 173.7 lbs (120 - 250 lbs.), and an average BMI of 28.6 (19.9 - 49.4). Each patient had a pre-op lumbar MRI and flexion and extension radiographs demonstrating a fixed grade I degenerative spondylolisthesis and spinal stenosis. Additional radiographic findings included disk herniation (12%) and degenerative scoliosis (2.4%). All individuals presented with initial complaints of focal lower back and radicular lower extremity pain. In addition to these primary symptoms, 66% exhibited lower extremity motor
Table 1. Comparison of TruFUSE® procedural outcomes to eight selected published studies using pedicle screw fusion instrumentation.
deficits, 44% suffered from lower extremity reflex abnormalities, and 20% presented with lower extremity sensory deficiencies associated with their radicular pain. They all subsequently underwent laminectomy, hemilaminectomy and/or discectomy with transverse process fusion at affected levels.
TruFUSE® facet fusion was done at either one level (n = 30, 73%) or two levels (n = 11, 27%) for a total of 52 TruFUSE® implantations in 41 patients. The majority of implantations occurred at the level of L4-L5 (75%), with the L3-L4 and L5-S1 levels accounting for 21% and 2.0% of implantations, respectively. The mean operative time for a laminectomy with transverse process fusion and TruFUSE® facet fusion was 106 ± 23 min with a mean blood loss intra-operatively of 145 ± 66.8 cm3. Patients were hospitalized for an average of 1.7 ± 1.6 days post-operatively.
3.2. Evaluation of Post-Op Spine Stability
There was no evidence of facet fracture or TruFUSE® bone dowel dislodgement on any post-operative radiographs including flexion and extension radiographs. Flexion and extension radiographs showed 39 of the 41 patients (95%) had spinal stability at an average six months post-op and all (100%) had signs of early fusion. Additionally, evidence of bone fusion along the transverse processes was noted by increased opacity and early ossification was found in all subjects.
3.3. Patient Outcomes
In 33 subjects subjective outcome was determined to be either excellent or somewhat improved (80%) compared to pre-operative statements. Of the remaining patients, four were unchanged (10%), and four patients were worse (10%) compared to pre-operative symptom comments (Table 2).
3.4. Complications
No surgical site infections, excessive intra-operative blood loss requiring transfusions, or obvious nerve root damage occurred during the surgical procedures. Two patients reported urinary incontinence following their operations. Both cases resolved with medication. A single patient developed unexplainable painful polyneuropathy in the lower extremities that was believed to be unrelated to the surgical intervention. The patient was started on gabapentin and improved. One patient had post-operative pulmonary emboli that required shortterm re-hospitalization and long-term anti-coagulation. One additional patient lost sight in his right eye approximately one year after his operation and was referred for further neurosurgical care secondary to a suspected glioblastoma brain tumor, a complication unrelated to surgery.
3.5. Intra-Operative and Length of Stay Parameters (Table 1)
3.5.1. Operative Time
The mean OT for the TruFUSE® fusion surgery was 106 ± 23 min. This value was lower than all four of the reported literature values and statistically significant (p < 0.0001) for the two data sets where complete statistical information was available (means = 185 min, 196 min) [9,13]. The other two studies, used only for comparison without statistical confirmation, had mean OTs of 212 min and 240 min [3,8].
3.5.2. Blood Loss (cm3)
The mean estimated blood loss (EBL) for the TruFUSE® fusion surgery was 145.0 ± 66.9 cm3. This value was lower than those for the four data sets that recorded this information, and statistically significant (p < 0.0001) against the two data sets with complete information (means = 321 cm3, 1082 cm3) [9,13]. The other two studies were used only as comparison without statistical confirmation had EBL means of 500 cm3 and 1639 cm3 [3,8].
3.5.3. Length of Stay
The mean LOS for the TruFUSE® fusion surgery was 1.71 ± 1.63 days. This value was lower than those for the two data sets that reported this information (means = 4 days, 19 days) and statistically significant (p < 0.0001) [10,11]. Seventy-seven percent of the TruFUSE® patients spent one day or fewer in the hospital following surgery.
4. Discussion
The use of pedicle screw fusion (PSF) for the lumbar spine continues to grow both in the number of procedures
Table 2. Definitions of the rating scales used to categorize patient outcomes and result summaries.

and the number of spine surgeons being trained in their use. PSF outcome measures have certainly improved over the years, especially with reduced hardware failures and improved post-operative spinal stability when compared to the previous standard of transverse process or lamina bone fusion without instrumentation [3,14,15]. Additionally, PSF has allowed for early ambulation and mobility that has no doubt led to fewer post-operative complications such as pulmonary emboli and pneumonia [6,16,17]. But now as the pendulum has swung almost completely to the universal use of pedicle screw instrumentation as the preferred choice for the lumbar spine fusion, potentially less traumatic lumbar fusion systems are now being employed which can be viable alternatives in certain patient populations.
As the surgical patient continues to be older with an increasing number of co-morbidities, the potential for surgical complications continues to grow. Alternative, less invasive fusion procedures, such as facet fusion, may help to reduce complications associated with potentially more invasive, traumatic and time-consuming PSF procedures.
The concept of fusing facet joints with bone packed in and around facet joints for lumbar spondylolisthesis as part of a non-instrumented lateral mass bone fusion has been described for decades [18]. Historically this was done with either allograft or autograft bone chips, and generally included bone placement both in and around the lamina and transverse processes. With the introducetion of spinal instrumentation and eventually PSF, the motion segments are now instrumented with interlocking screws and rods to maintain stability. Eventually bone fusion occurs within the now static facet joint as over time bridging osteophytes, along with added layered bone chips and other allograft materials surround and fuse the joints and motion segment.
In 2010, Pateder D.B., and Benzel E., published their retrospective series of 62 elderly subjects for which they preformed lumbar laminectomy and facet bone fusion for both stable (n = 39) and unstable spondylolisthesis (n = 23). Their results showed that 64% of those patients in the stable pre-operative group maintained post-operative stability on flexion and extension films. Of this group, 96% reported “much improved” post-operative outcomes. Conversely, of the 36% that were unstable post-operatively, only 50% reported clinical improvement [19].
This study demonstrates that pre-operative spinal stability is a significant advantage for a successful outcome when considering facet fusion alone. Furthermore, it highlights that traditional facet fusion techniques using bone chips, following laminectomy, may not result in successful stabilization in some patients.
Recent facet fusion advances in allograft bone milling, like that employed by the TruFUSE® bone dowel, are able to produce uniformed implants in various shapes and sizes which biomechanically have been shown to resist motion within the facet joint following implantation [20,21]. Biomechanical in vitro studies on cadaveric specimens conducted by Johnson found a 14% increase in spinal range of motion (ROM) and 18% decrease in stiffness following laminectomy without fusion. When augmented with the TruFUSE® facet fusion device, 18% spinal stiffness was returned (3% above baseline) with an 18% decrease in range of motion (4% below baseline) [20]. The TruFUSE® milled dowel is designed to lock the facet joints in place after insertion. By preserving most of the facet joint capsule during surgical dissection, a ligament and joint capsule tension band is preserved around the joint that, with the dowel in place, can the lock the joint and stop motion segment movement.
TruFUSE® facet fusion allograft implant (minSURG, Clearwater, Fl, USA) was used, along with transverse process fusion, in this group of patients following lumbar laminectomy or hemi-laminectomy with discectomy, to preserve spinal stability in a population known to be at increased risk for post-operative instability. In this group of subjects, TruFUSE® facet fusion, with transverse process fusion, provided a less tissue traumatic and timeconsuming fusion alternative to traditional PSF. For most cases in this series, placement of bilateral bone dowels was completed in 10 - 15 minutes and without the need for additional radiograph or neuro-monitoring. Additionally, blood loss was generally less than 200 cm3.
4.1. Fusion Stability
Assessing bone fusion solidity by any radiographic method is difficult, but the stabilizing effects of fusion can be demonstrated with sufficient accuracy by measuring radiographic flexion and extension intervertebral movements [22]. Post-operative flexion and extension radiographs using TruFUSE® facet fusion with transverse process bone fusion, demonstrated spinal stability in all but two patients. Additionally, no additional surgery has been required and these patients remain symptom free at their last assessment.
Studies evaluating patients undergoing lumbar laminectomies have shown that up to 30% will experience post-operative instability at the surgical site, while 5% of patients undergoing laminectomies with pedicle screw instrumented posterolateral fusion require follow-up surgery due to progressive spondylolisthesis at the same level [23,24]. Additionally, Pateder D.B., and Benzel E., found that 36% of their patients were unstable following laminectomy with facet bone chip fusion [19]. Our limited follow-up study suggests that TruFUSE® facet fusion and transverse process fusion following lumbar laminectomy can be a viable alternative method for spinal fusion in select groups of patients with stable grade I spondylolisthesis.
As noted, post-operative flexion and extension radiographs showed that 39 of the 41 patients (95%) had spine stability at an average six months post-op and all (100%) had early signs of fusion. The two patients who had post-operative movement at the level fused were assessed at 12.9 and 13.3 months post-op and both were asymptomatic. Future long-term, prospective, randomized studies with direct patient outcome measures will be needed in order to confirm these early fusion results.
Subjective post-operative improvements reveal a majority of subjects (80%) improved (Table 2). This result was similar to the results reported by Pateder DB and Benzel E. (2010), who observed improvements in their pre-operative group that did not show preor post-operative instability following facet fusion with transverse process fusion [19].
4.2. Operative Time (OT), Blood Loss (BL) and Length of Stay (LOS)
Using the TruFUSE® facet fusion with transverseprocess bone fusion, our limited evaluation demonstrated a clear advantage in surgical time, length of stay, and intra-operative blood loss compared with literature results of similar patients using pedicle screw instrumented fusion with transverse process bone fusion. Our patients had significantly shorter surgical times compared with PSF by an average of 85 minutes, a 45% reduction in operating time. This dramatic time difference between the TruFUSE® procedure and standard PSF studies obtained for this comparison is likely due to the greater technical requirements and increased tissue dissection needed to place the screw and rod instrumentation that accompany the latter [7]. The need for additional time for radiographic or fluoroscopic guidance for screw placement and not for TruFUSE® dowels may also play a role [25,26].
Additional operative time was saved by utilizing morselized bone from the spinous processes and lamina, along with allograft bone, at affected levels to achieve transverse process fusion. In contrast, three of the four published data sets for which operating time was recorded used iliac crest to achieve transverse process fusion [3,8,9]. Adjusting the TruFUSE® OTs for the time that would have been required to harvest donor bone, TruFUSE® implantation operations remained 42% shorter on average, or about 77 mins (p < 0.0001) by comparison.
Differences in surgical technique may have also accounted for the 655.5 cm3 average reduction of blood loss in the TruFUSE® population relative to those receiving conventional PSF fixation. The TruFUSE® approach, which required 13 mm of drilling into the articular surfaces of the facet joint, spares the removal of a significant amount of highly vascularized trabecular bone volume from the pedicle and vertebral body. Additionally, because of the dorsal placement of TruFUSE® facet fusion system, there is less potential for major vascular or nerve root injury.
Differences in pre-operative diagnoses and extent of spinal decompression between our TruFUSE® surgical population and those included in the published data will certainly contribute to some of the differences in blood loss and operative time between the two groups. Of the 41 patients receiving the TruFUSE® facet fusion, 11 (27%) required fusion at two levels. Among the five data sets that included blood loss information for pedicle screw fixation, the two studies reporting the largest blood loss among surgical patients examined populations in which 41% and 37% of surgical candidates required fusion at multiple levels [3,9]. All other studies included a proportion of patients receiving fixation at multiple levels that was higher than that of the TruFUSE® population with the exception of that conducted by Ohtori et al. (2011), which excluded patients requiring fixation at multiple levels. Even compared to this study, the TruFUSE® population exhibited a 63.6% reduction in blood loss relative to those receiving pedicle screw fixation (p < 0.0001) [11].
In addition to observing shorter operative times with reduced blood loss compared to pedicle screw fixation, the TruFUSE® procedure also exhibited significantly shorter hospital stays in the studied population compared to two studies of pedicle screw fixation in which complete data was available (p < 0.0001) [10,11]. In this study, 77% of patients receiving TruFUSE® implantation spent one day or less in the hospital.
4.3. Economic Consideration
The economic implications associated with using the TruFUSE® facet fusion system demonstrates potentially significant savings compared to PSF. Candrilli (2006), estimated the average cost of a hospital stay across all conditions to be $1237/day [27]. Using this conservative figure, it can be suggested that the TruFUSE® procedure could result in hospital admission savings of $2800 - $21,000 when considering decreased LOS alone (TruFUSE® LOS cost = $2115 compared with $4948, LOS = 4 days and $23,503, LOS = 19 days). It should be noted that a LOS of 19 days is unnaturally high for this or similar procedures, and any cost reduction due to the use of the TruFUSE® system is only an estimation (Table 3).
Reduction in blood loss and OT found with TruFUSE® procedures would also reduce the overall hospital cost of
Table 3. Review of savings based upon length of stay (LOS) and operating time (OT).
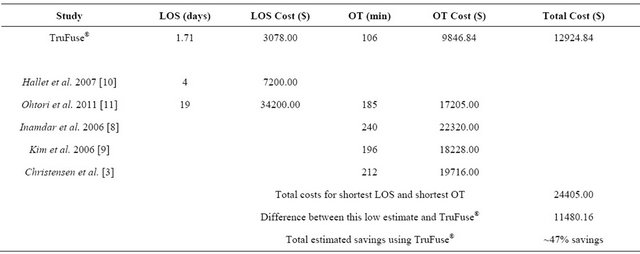
Note: Calculated savings do not include savings due to cost of implant, additional equipment, or neuromonitoring.
lumbar fixation by reducing the likelihood of a transfusion and reducing the time requirements for operative instruments and personnel. Cost benefits associated with TruFUSE® implantation will certainly vary with the geography, payer mix, patient population and extent of surgical decompression.
4.4. Limitations of Study
This retrospective evaluation has inherent flaws that must be considered. No standardized tool was used to assess preand post-operative subjective improvements and the literature-based cohort of studies used for comparison may not actually represent the outcomes for all PSF surgeries and therefore could lead to inflated comparisons. The severity of pre-operative diagnosis and extent of fusion surgeries used to compare the TruFUSE® results to PSF may be significantly different and again lead to errors in comparison. The limited follow-up radiographic and subjective outcomes on average of six months may be too early to predict overall success. And finally, the results presented here by one surgeon with a relatively small population will need to be confirmed in larger numbers with multiple surgeons and for a longer period of follow-up to confirm the extremely positive results reported here.
5. Conclusions
This retrospective analysis demonstrates the successful implementation of the TruFUSE® facet fusion system to augment laminectomy and transverse process fusion procedures for the treatment of stable grade I degenerative spondylolisthesis in a cohort of 41 patients. Flexion and extension radiographs showed 39 of the 41 patients (95%) had spinal stability at an average six months post-op and all (100%) had signs of early fusion. 88% of patients presenting no neurological deficits on post-operative physical examination and 80% reporting perceived improvement or relief of pre-operative symptoms.
Additionally, patients receiving TruFUSE® facet fusion had a significant reduction in operative time of 85 minutes (45% reduction, p < 0.0001) on average when compared to a literature-based group of similar subjects receiving pedicle screw fixation. TruFUSE® fusion patients also benefited from significantly (p < 0.0001) shorter hospital stays (1.7 days compared to 4 - 19 days in literature), and reduced mean estimated intra-operative blood loss (145 cm3 compared to 321 - 1082 cm3 in literature).
These initial findings suggest that one or two level TruFUSE® facet fusion maybe a viable alternative to pedicle screw fixation for spinal stabilization following lumbar laminectomy and transverse process fusion for stable grade I degenerative spondylolisthesis with symptomatic lumbar stenosis. Furthermore, observed reductions in operative time, blood loss and length of stay may provide reduced operative complications and economic advantages to patients and physicians over traditional fusion methods. Additional long-term, prospective studies are needed to confirm these results.
6. Funding & Conflicts of Interest
Neuroscience Research Foundation-Research contributions for the Neuroscience Research Foundation include numerous spine-related manufacturers, including minSURG (Clearwater, Fl, USA), and individual foundations, including the Heindl Foundation and the Nelson Peltz Foundation.
There are no conflicts of interest reported by the authors with regard to stock ownership, consultant work, or employment related to minSURG.
Photographs provided by minSURG (Clearwater, Fl, USA) for the purpose of this article (Permission release will be provided under a separate correspondence from the company).
REFERENCES
- R. A. Deyo, D. C. Cherkin, J. D. Loeser, et al., “Morbidity and Mortality in Association with Operations on the Lumbar Spine: The influence of Age, Diagnosis, and Procedure,” The Journal of Bone and Joint Surgery (American Volume), Vol. 74, No. 4, 1992, pp. 536-543.
- H. Moller and R. Hedlund, “Instrumented and Noninstrumented Postereolateral Fusion in Adult Spondylolisthesis,” Spine, Vol. 25, No. 13, 2000, pp. 1716-1721. doi:10.1097/00007632-200007010-00017
- F. B. Christensen, E. S. Hansen, M. Laursen, et al., “LongTerm Functional Outcome of Pedicle Screw Instrumentation as a Support for Postereolateral Spinal Fusion: Randomized Clinical Study with a 5-Year Follow-Up,” Spine, Vol. 27, No. 12, 2002, pp. 1269-1277. doi:10.1097/00007632-200206150-00006
- D. Dindo, N. Demartines and P. A. Clavien, “Classification of Surgical Complications: New Proposal with Evaluation in a Cohort of 6336 Patients and Results of Survey,” Annals of Surgery, Vol. 240, No. 2, 2004, pp. 205- 213. doi:10.1097/01.sla.0000133083.54934.ae
- R. A. Deyo, S. K. Mirza, B. I. Martin, et al., “Trends, Major Medical Complications and Charges Associated with Surgery for Lumbar Spinal Stenosis in Older Adults,” The Journal of the American Medical Association, Vol. 303, No. 13, 2010, pp. 1259-1265. doi:10.1001/jama.2010.338
- R. Johnsson, P. Axelsson and B. Stromqvist, “Posterolateral Lumbar Fusion Using Facet Joint Fixation with Biodegradable Rods: A Pilot Study,” European Spine Journal, Vol. 6, No. 2, 1997, pp. 144-148. doi:10.1007/BF01358748
- P. Fritzell, O. Hagg and A. Nordwall, “Complications in Lumbar Fusion Surgery for Chronic Low Back Pain: Comparison of Three Surgical Techniques Used in a Prospective Randomized Study. A Report from the Swedish Lumbar Spine Study Group,” European Spine Journal, Vol. 12, No. 2, 2003, pp. 178-189.
- D. N. Inamdar, M. Alagappan, L. Shyam, S. Devadoss and A. Devadoss, “Posterior Lumbar Interbody Fusion versus Intertransverse Fusion in the Treatment of Lumbar Spondylolisthesis,” The Journal of the Orthopaedic Surgery, Vol. 14, No. 1, 2006, pp. 21-26.
- K. T. Kim, S. H. Lee, Y. H. Lee, S. C. Bae and K. S. Suk, “Clinical Outcomes of 3 Fusion Methods through the Posterior Approach in the Lumbar Spine,” Spine, Vol. 31, No. 12, 2006, pp. 1351-1357. doi:10.1097/01.brs.0000218635.14571.55
- A. Hallet, J. S. Huntley and A. Gibson, “Foraminal Stenosis and Single-Level Degenerative Disc Disease,” Spine, Vol. 32, No. 13, 2007, pp. 1375-1380. doi:10.1097/BRS.0b013e318064520f
- S. Ohtori, T. Koshi, M. Yamashita, et al., “Single-Level Instrumented Posterolateral Fusion versus Non-Instrumented Anterior Interbody Fusion for Lumbar Spondylolisthesis: A Prospective Study with a 2-Year Follow-Up,” Journal of Orthopaedic Science, Vol. 16, No. 4, 2011, pp. 352-358. doi:10.1007/s00776-011-0088-5
- T. Koshi, S. Ohtori, M. Suzuki, et al., “Uniand Bi-Lateral Instrumented Posterolateral Fusion of the Lumbar Spine with Local Bone Grafting: A Prospective Study with a 2-Year Follow-Up,” Spine, Vol. 36, No. 26, 2011, pp. E1744-E1748.
- S. Ohtori, M. Suzuki, M. Takaso, et al., “Single-Level Instrumented Posterolateral Fusion of the Lumbar Spine with a Local Bone Graft versus an Iliac Crest Bone Graft: A Prospective, Randomized Study with a 2-Year Follow-Up,” European Spine Journal, Vol. 20, No. 4, 2011, pp. 635-639. doi:10.1007/s00586-010-1656-7
- S. M. Mardjetko, P. J. Connolly and S. Shott, “Degenerative Lumbar Spondylolisthesis: A Meta-Analysis of Literature 1979-1993,” Spine, Vol. 19, No. 20, 2004, pp. 2256-2265. doi:10.1097/00007632-199410151-00002
- G. W. Wood, R. J. Boyd, T. A. Carothers, et al., “The Effect of Pedicle Screw/Plate Fixation on Lumbar/Lumbosacral Autogenous Bone Graft Fusions in Patients with Degenerative Disc Disease,” Spine, Vol. 20, No. 7, 1995, pp. 819-830. doi:10.1097/00007632-199504000-00017
- H. G. Deen, D. S. Fenton and T. J. Lamer, “Minimally Invasive Procedures for Disorders of the Lumbar Spine,” Mayo Clinic Proceedings, Vol. 78, No. 10, 2003, pp. 1249- 1256. doi:10.4065/78.10.1249
- R. I. Riesenburger and C. A. David, “Lumbar Microdiscectomy and Microendoscopic Discectomy,” Minimally Invasive Therapy & Allied Technologies, Vol. 15, No. 5, 2006, pp. 267-270. doi:10.1080/13645700600958432
- C. K. Lee and N. A. Langrana, “Review of Spinal Fusion for Degenerative Disc Disease: Need for an Alternative Treatment Approach of Disc Arthroplasty,” The Spine Journal, Vol. 4, No. 6, 2004, pp. 173-176. doi:10.1016/j.spinee.2004.07.002
- D. B. Pateder and E. Benzel, “Noninstrumented Facet Fusion in Patients Undergoing Lumbar Laminectomy for Degenerative Spondylolisthesis,” Journal of Surgical Orthopadic Advances, Vol. 19, No. 3, 2010, pp. 153-158.
- W. M. Johnson, “The Use of the TruFUSE Structural Facet Joint Fusion Allograft in Conjunction with a Lumbar Laminectomy,” 2011.
- W. M. Johnson, “Understanding the Morphology of Lumbar Facets and Its Correlation to the TruFUSE® Facet Joint Fusion Procedure,” 2011.
- A. E. Brodsky, E. S. Kovalsky and M. A. Khalil, “Correlation of Radiologic Assessment of Lumbar Spine Fusions with Surgical Exploration,” Spine, Vol. 16, No. 6S, 1991, pp. 261-265. doi:10.1097/00007632-199106001-00017
- A. Abla, H. El Kadi, J. Bost, J. C. Maroon and M. Wert, “Evaluation of Spondylolisthesis after Lumbar Laminectomy,” Contemporary Neurosurgery, Vol. 26, 2004, pp. 1-6.
- W. A. Abdu, J. D. Lurie, K. F. Spratt, et al., “Degenerative Spondylolisthesis: Does Fusion Method Influence Outcome? Four-Year Results of the Spine Patient Outcomes Research Trial,” Spine, Vol. 34, No. 21, 2009, pp. 2351-2360. doi:10.1097/BRS.0b013e3181b8a829
- O. Schwarzenbach, U. Beremann, B. Jost, et al., “Accuracy of Computer-Assisted Pedicle Screw Placement: An in Vivo Computed Tomography Analysis,” Spine, Vol. 22, No. 4, 1997, pp. 452-458. doi:10.1097/00007632-199702150-00020
- F. Asgarzadie and L. T. Khoo, “Minimally Invasive Operative Management for Lumbar Spinal Stenosis: Overview of Early and Long-Term Outcomes,” Orthopedic Clinics of North America, Vol. 38, No. 3, 2007, pp. 387-399. doi:10.1016/j.ocl.2007.02.006
- S. D. Candrilli and J. A. Mauskopf, “How Much Does a Hospital Day Cost?” The 11th Annual International Meeting of the International Society for Pharmacoeconomics and Outcomes Research, Philadelphia, 20-24 May 2006.
Abbreviations
PSF: pedicle screw fusion;
EBL: estimated blood loss;
OT: operation time;
LOS: length of stay;
ROM: range of motion.
NOTES
*Corresponding author.

