International Journal of Clinical Medicine
Vol.3 No.2(2012), Article ID:17882,4 pages DOI:10.4236/ijcm.2012.32026
Preoperative Prediction of Whether Intraoperative Fluorescence of Protoporphyrin IX Can Be Achieved by 5-Aminolevulinic Acid Administration
![]()
1Department of Neurosurgery, Kitasato University School of Medicine, Sagamihara, Japan; 2SBI ALApromo Co., Ltd., Tokyo, Japan.
Email: utsuki@med.kitasato-u.ac.jp
Received December 6th, 2011; revised January 20th, 2012; accepted February 16th, 2012
Keywords: 5-Aminolevulinic Acid; Intraoperative Fluorescence; Coproporphyrin; Protoporphyrin IX; Urinary Porphyrin
ABSTRACT
Protoporphyrin IX (PPIX) fluorescence-guided brain tumor resection, using 5-aminolevulinic acid (5-ALA), is among the most valuable tools for determining tumor removal area. However, PPIX fluorescence is not necessarily achieved during an operation visually even when 5-ALA is used, and we do not know until tumor exposure to the excitation light of the ultraviolet region whether PPIX fluorescence has been achieved. When a particular biopsy and frozen section diagnosis is made, the reason for lack of PPIX fluorescence in the tissue cannot be judged. We do not know whether the tumor fails to fluoresce or no fluorescence is seen because it is not the main body of the tumor. We investigated whether the presence or absence of tumor fluorescence could be predicted by examining urinary porphyrin before surgery, at the time of intraoperative fluorescence diagnosis using 5-ALA. The urine of brain tumor patients 2 hours after 5-ALA administration was irradiated with a 405 ± 1 nm laser light. The patients were divided into a fluorescent urine group and negative fluorescent urine group. Red fluorescence was observed in response to the 405 ± 1 nm laser beam for all tumors in the fluorescent urine group. Clear red fluorescence was not observed even with 405 ± 1 nm laser beam irradiation in any tumors in the negative fluorescent urine group. Preoperative prediction of the intraoperative fluorescence of PPIX can be achieved by observation of urine 2 hours after 5-ALA administration with exposure to a 405 ± 1 nm laser light.
1. Introduction
Protoporphyrin IX (PPIX) fluorescence-guided brain tumor resection using 5-aminolevulinic acid (5-ALA) is among the most useful tools for determining the removal area for infiltrating tumors like gliomas [1-4]. However, PPIX fluorescence is not observed in all tumors when 5-ALA is administered [3,4]. There is no method of ascertaining preoperatively whether PPIX fluorescence of the tumor tissue will be observed until exposure to an excitation light of the ultraviolet region during tumor resection. In a tumor showing strong PPIX fluorescence, the fluorescence around the tumor is reportedly vague [2]. Whether the resected region is the main body of the tumor or the peripheral zone intraoperatively can be judged before the operation if the tumor shows strong fluorescence, or not. The region is not the main body of the tumor which must be elsewhere when only weak PPIX fluorescence is obtained for a sample, in particular, a small tumor which was expected to show strong PPIX fluorescence. It has also been suggested that the biopsy region is not the main body of the tumor when only weakly fluorescent PPIX is observed in tumor biopsy samples. Coproporphyrin productions reportedly increase via the process generating PPIX within tumor cells after administration of 5-ALA [1,5]. We examined the possibility of predicting abundant PPIX production within tumor cells by a simple method allowing easy detection of urinary coproporphyrin.
2. Materials and Methods
This study was reviewed and approved by the Kitasato University Hospital Ethics Committee, and informed consent was obtained from all patients. Twenty-five patients with brain tumors, including 15 with glioblastomas, 6 with benign astrocytomas, and 4 with malignant lymphomawere surgically treated at the Department of Neurosurgery of Kitasato University Hospital. Two hours before induction of anesthesia, each patient was given a 1 g oral dose of 5-ALA. Two hours after administration of 5-ALA, 3 ml of new urine were obtained during anesthesia preparations via urethral catheters. One milliliter of this urine was exposed to a laser light with a peak wavelength of 405 ± 1 nm and an output of 120 mW by fiber-optic cable, using a semiconductor laser device (VLD-V1 version 2, M & M Co., Ltd., Tokyo, Japan). Urinary color was observed with an ultraviolet cut-off filter at the time and the colors were divided into two groups. One group was the fluorescent urine group which showed a clearly red color with laser beam irradiation (Figure 1). The other was the negative fluorescent urine group which showed an unclear red color with laser beam irradiation (Figure 2). Two milliliters of remaining urine were used for the coproporphyrin measurement by gas chromatography. The molar content of coproporphyrin was regulated with the creatinine concentration per gram (mg/gCr).
Also, once the tumor had been resected, the target region was exposed to a laser light with a peak wavelength

Figure 1. Photograph showing urine, from a glioblastoma patient, irradiated with a 405 nm laser beam delivered through an optic fiber 2 hours after 5-ALA administration. The urine showed slightly red fluorescence in response to the laser beam. When this tumor was irradiated with a 405 nm laser beam through the optic fiber during surgery, the tumor showed red fluorescence.

Figure 2. Photograph showing urine, from a benign astrocytoma patient, irradiated with a 405 nm laser beam delivered through an optic fiber 2 hours after 5-ALA administration. The urine showed no response to the laser beam, and the color was unchanged. When this tumor was irradiated with a 405 nm laser beam through the optic fiber during surgery, the tumor did not show red fluorescence.
of 405 ± 1 nm and an output of 120 mW using a semiconductor laser device. The color of the tumor in response to the laser light was observed. The tumors were divided into two groups, fluorescent and non-fluorescent. When the tumor showed no red fluorescence, it belonged to the non-fluorescent tumor group. The other tumors belonged to the fluorescent tumor group.
Significant differences were analyzed using Stat View 5.0 software. The p value was derived using a t-test.
3. Results
The fluorescent urine group included all of the glioblastomas, two of the four benign astrocytomas and three of the six metastatic brain tumors. The negative fluorescent urine group included two of the four benign astrocytomas and three of the six metastatic brain tumors. The molar contents of coproporphyrin ranged from 353 mg/gCr to 5200 mg/gCr. The molar contents of coproporphyrin in the fluorescent urine group ranged from 353 mg/gCr to 1172 mg/gCr. The molar contents of coproporphyrin in the negative fluorescent urine group ranged from 2015 mg/gCr to 5200 mg/gCr (Figure 3). The fluorescent urine group had a significantly higher level of urinary coproporphyrin than the negative group (p < 0.001). The numbers of cases in the fluorescent urine and negative fluorescent urine groups were 20 and 5, respectively. The fluorescence of tumors corresponded perfectly to that of urine fluorescence on a case by case basis. Similarly, all of the patients with non-fluorescent tumors belonged to the negative fluorescent urine group. In the non-fluorescent tumor group, all non-fluorescent tumor tissues represented the main body of the tumor histologically and corresponded to the diagnostic imaging results.
4. Discussion
Fluorescence-guided tumor resection using 5-ALA is based on this compound being metabolized in tumor cells and PPIX accumulating within the tumor. When 5-ALA is administered, the tumor cells take it up and synthesize heme actively. However, ferrochelatase, which is necessary for synthesis of heme from PPIX in the tumor cells, is relatively insufficient, and PPIX accumulates in tumor cells because, as a result, synthesis does not advance [3, 6]. This PPIX may be found in surrounding tissues where no tumor cells exist [7]. This phenomenon is thought to be due to PPIX produced in tumor cells leaking out of the cells [8]. Also, more highly-concentrated porphyrin, particularly coproporphyrin, is reportedly excreted in the urine of brain tumor patients than by volunteers without brain tumors given 5-ALA, since porphyrins including PPIX leak out of tumor cells [5].
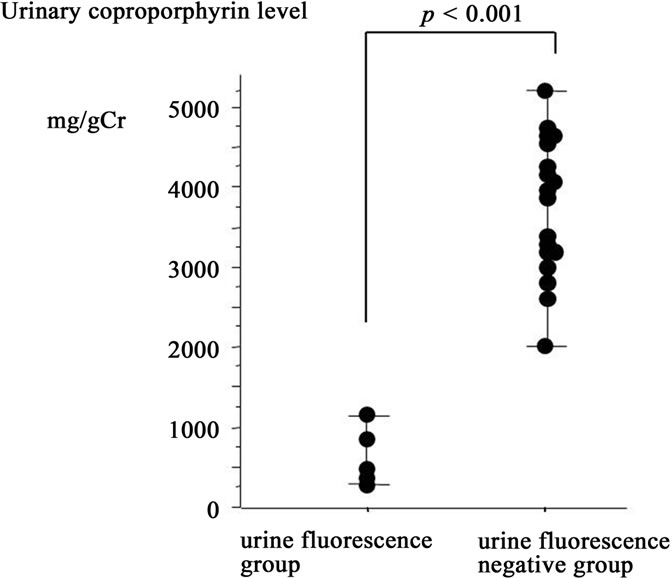
Figure 3. The graph shows values of urinary coproporphyrin 2 hours after 5-ALA administration in the fluorescent urine and negative fluorescent urine groups. There is a significant difference between the two groups in coproporphyrin, with the value being higher in the fluorescent urine group.
In this study, the fluorescent urine group had high urinary coproporphyrin levels and PPIX fluorescence was observed in tumors. In contrast, the negative fluorescent urine group had low urinary coproporphyrin levels and PPIX fluorescence was not observed in tumors. Therefore, neurosurgeons can estimate whether PPIX fluorescence will be present in a tumor by observing the color of urine exposed to a 405 nm laser light 2 hours after the 5-ALA administration. When no urinary fluorescence is observed 2 hours after 5-ALA administration, tumor fluorescence is highly unlikely. However, neurosurgeons should perform another biopsy in case the original biopsy site was inadequate, when tissue at the perioperative biopsy shows no PPIX fluorescence despite red fluorescence of urine 2 hours after 5-ALA administration.
The details underlying the mechanism of porphyrin leakage in metabolic processes from 5-ALA to heme remain unknown. ABCG2 and ABCB6, which are ATPbinding cassette (ABC) transporters, are known to be associated with transport of these porphyrins [9,10]. ABCG2 is overexpressed in cancer cells, and, porphyrins and anticancer drugs are actively transported outside of cells by this molecule [9]. ABCG2 was found to be over-expressed in brain tumors, and there is abundant expression in malignant tumors [11,12]. Furthermore, coproporphyrinogen is transported from the cytoplasm into mitochondria by ABCB6. t was found that the functions and the levels of expressions of these ABC transporters varied according to genetic polymorphisms [10]. A cumulative dose of PPIX in tumor tissues (the fluorescent strength of the tumor) and the amount of urinary porphyrin change with differences in the expression levels or the functions of these transporters [3,5]. The fluorescent strength of the tumor tissue (cumulative PPIX dose) and the amount of urinary porphyrin show a degree of correlation [5]. When PPIX fluorescence-guided brain tumor resection using 5-ALA is performed, expressions of ABC transporters rise and metabolism of porphyrin is accelerated intraoperatively in tumors with fluorescence such as glioblastomas. Therefore, an abundance of porphyrin will be excreted from urine when PPIX accumulates in tumors after 5-ALA administration.
5. Acknowledgements
This work was supported by a Grant-in-Aid for Scientific Research (C) (Nos. 23592139).
REFERENCES
- S. K. Bisland, E. A. Goebel, N. S. Hassanali, C. Johnson and B. C. Wilson, “Increased Expression of Mitochondrial Benzodiazepine Receptors Following Low-Level Light Treatment Facilitates Enhanced Protoporphyrin IX Production in Glioma-Derived Cells in Vitro,” Lasers in Surgery and Medicine, Vol. 39, No. 8, 2007, pp. 678-684. doi:10.1002/lsm.20544
- W. Stummer, A. Novotny, H. Stepp, C. Goetz, K. Bise and H. J. Reulen, “Fluorescence-Guided Resection of Glioblastoma Multiforme by Using 5-Aminolevulinic Acid-Induced Porphyrins: A Prospective Study in 52 Consecutive Patients,” Journal of Neurosurgery, Vol. 93, No. 6, 2000, pp. 1003-1013. doi:10.3171/jns.2000.93.6.1003
- S. Utsuki, H. Oka and K. Fujii, “Intraoperative Photodynamic Diagnosis of Brain Tumors Using 5-Aminolevulinic Acid,” In: A. L. Abujamra, Ed., Diagnostic Techniques and Surgical Management of Brain Tumors, InTech, Rijeka, 2011, pp. 227-244.
- S. Utsuki, H. Oka, S. Sato, S. Suzuki, S. Shimizu, S. Tanaka and K. Fujii, “Possibility of Using Laser Spectroscopy for the Intraoperative Detection of Nonfluorescing Brain Tumors and the Boundaries of Brain Tumor Infiltrates,” Journal of Neurosurgery, Vol. 104, No. 4, 2006, pp. 618-620. doi:10.3171/jns.2006.104.4.618
- S. Utsuki, H. Oka, K. Fujii, N. Miyoshi, M. Ishizuka, K Takahashi and K. Inoue, “Differential Metabolism of 5-ALA in Patients with Brain Tumors,” In: N. Miyoshi and R. H. Pottier, Ed., In Hope of Going over the Present Clinical PD and PDT, Sankeisha, Nagoya, 2011, pp. 160- 173.
- Q. Peng, K. Berg, J. Moan, M. Kongshaug and J. M. Nesland, “5-Aminolevulinic Acid-Based Photodynamic Therapy: Principles and Experimental Research,” Photochemistry and Photobiology, Vol. 65, No. 2, 1997, pp. 235-251. doi:10.1111/j.1751-1097.1997.tb08549.x
- S. Utsuki, H. Oka, S. Sato, S. Suzuki, S. Shimizu, Y. Tanizaki, K. Kondo, Y. Miyajima and K. Fujii, “Histological Examination of False Positive Tissue Resection Using 5-Aminolevulinic Acid-Induced Fluorescence Guidance,” Neurologia Medico-Chirurgica (Tokyo), Vol. 47, No. 5, 2007, pp. 210-214. doi:10.2176/nmc.47.210
- S. Utsuki, N. Miyoshi, H. Oka, Y. Miyajima, S. Shimizu, S. Suzuki and K. Fujii, “Fluorescence-Guided Resection of Metastatic Brain Tumors Using 5-Aminolevulinic AcidInduced Porphyrins—Pathological Study,” Brain Tumor Pathology, Vol. 24, No. 2, 2007, pp. 53-55. doi:10.1007/s10014-007-0223-3
- T. Ishikawa and H. Nakagawa, “Human ABC Transporter ABCG2 in Cancer Chemotherapy and Pharmacogenomics,” Journal of Experimental Therapeutics and Oncology, Vol. 8, No. 1, 2009, pp. 5-24.
- A. Tamura, Y. Onishi, R. An, S. Koshiba, K. Wakabayashi, K. Hoshijima, W. Priebe, T. Yoshida, S. Kometani, T. Matsubara, K. Mikuriya and T. Ishikawa, “In Vitro Evaluation of Photosensitivity Risk Related to Genetic Polymorphisms of Human ABC Transporter ABCG2 and Inhibition by Drugs,” Drug Metabolism and Pharmacokinetics, Vol. 22, No. 6, 2007, pp. 428-440. doi:10.2133/dmpk.22.428
- A. M. Bleau, J. T. Huse and E. C. Holland, “The ABCG2 Resistance Network of Glioblastoma,” Cell Cycle, Vol. 8, No. 18, 2009, pp. 2936-2944. doi:10.4161/cc.8.18.9504
- Y. Jin, Z. Q. Bin, H. Qiang, C. Liang, C. Hua, D. Jun, W. A. Dong and L. Qing, “ABCG2 Is Related with the Grade of Glioma and Resistance to Mitoxantone, a Chemotherapeutic Drug for Glioma,” Journal of Cancer Research and Clinical Oncology, Vol. 135, No. 10, 2009, pp. 1369-1376. doi:10.1007/s00432-009-0578-4

