Advances in Materials Physics and Chemistry
Vol.3 No.2(2013), Article ID:33313,6 pages DOI:10.4236/ampc.2013.32023
Study of Oxidation Kinetics in Air of Zircaloy-4 by in Situ X-Ray Diffraction
Nuclear Research Center of Birine, Birine, Algeria
Email: selm2002nor@yahoo.fr, sariali63@yahoo.fr
Copyright © 2013 Noureddine Selmi, Ali Sari. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received April 2, 2013; revised May 9, 2013; accepted May 20, 2013
Keywords: Zircaloy-4; High Temperature Oxidation; Zirconium Oxides; In Situ X-Ray Diffraction
ABSTRACT
The zircaloy-4 is an alloy of zirconium, which has a very weak thermal neutron absorption, satisfactory mechanical properties and good corrosion resistance at high temperature. For these reasons, zircaloy-4 is used as a material of cladding fuel rod of nuclear reactors. In this environment, it is submitted to different severe conditions of temperature and pressure. The objective of this work is to study the oxidation kinetics of zircaloy-4 in air by the X-ray diffraction technique. The experiments were realized in a “HTK1200” furnace installed as a sample holder in the diffractometer at different temperatures; 25˚C, 350˚C, 500˚C, 830˚C and 1000˚C. The results show that the monoclinic and the tetragonal phases are formed at 350˚C temperature. The volume fraction of these phases increased with the temperature until 1000˚C where the α phase disappears completely. For simulating the case of loss-of-coolant-accident (LOCA), we have done x-ray diffraction of Zry-4 samples water quenched at 1050˚C with different ageing times at this temperature. At 10 seconds and more, there is an important evolution of monoclinic and tetragonal zirconias, which leads to the degradation of zircaloy-4 properties.
1. Introduction
The fuel rod cladding represents the first barrier of the radioactive products confinement. The use of zirconium alloys for the cladding fuel rod of the pressurized water of the nuclear reactors (P.W.R.) is related to their very weak absorption of the thermal neutrons, good water corrosion resistance, good mechanical properties at high temperature [1], compatibility with the fuel and sufficient resistance to the irradiation effects (embrittlement, swelling …) (Figure 1). This material is submitted to the extreme conditions of the use, and various phenomena which degrade their characteristics. Among these phenomena there is the oxidation by water.
Zircaloy-4 oxidation in air proceeds in a similar way to that described in the pressurized steam. In this work, we studied the oxidation of the zircaloy-4 in air, and we have chosen various temperatures; 25˚C, 350˚C, 500˚C, 830˚C and 1000˚C.
1.1. Allotropic Phases of the Zr
At atmospheric pressure, and temperature inferior to 866˚C, elemental Zr appears as the hcp α-phase. At temperature between 866˚C and 1850˚C, Zr appears as the bcc β-phase. At room temperature and at pressure of about 2 GPa and more, the hcp α-phase is transformed to the hexagonal ω-phase [2].
1.2. Zr-O System
The phase diagram of the binary system Zr-O presented in Figure 2 [3] shows that a great quantity of oxygen can dissolve in interstitial position (until 30% at 500˚C) on one hand and on the other hand, there exists one stable zirconium oxide with atmospheric pressure, the ZrO2 zirconia.
At atmospheric pressure, and at a temperature that reaches 1170˚C, the crystalline structure of ZrO2 is monoclinic, between 1170˚C and 2370˚C, the monoclinic structure becomes tetragonal structure, and cubic structure from 2370˚C to 2706˚C [4,5].
1.3. Oxidation Mechanism
The oxidation of zirconium and its alloys progresses by a diffusive stage of anionic type, i.e. the growth of the products of the oxidation of metal is carried out by transport of oxygen of the gas environment towards the interface metal/oxide (intern interface) according to the
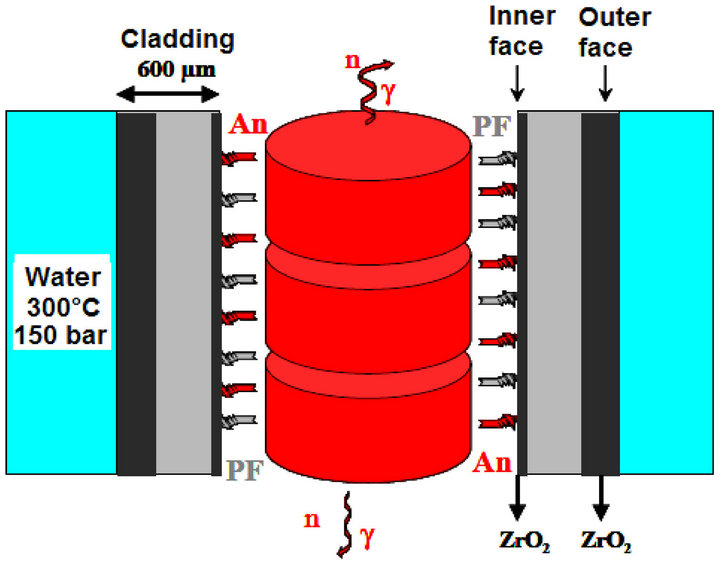
Figure 1. Schematical section of fuel rod in reactor. An and PF represent respectively the actinides and the fission products.
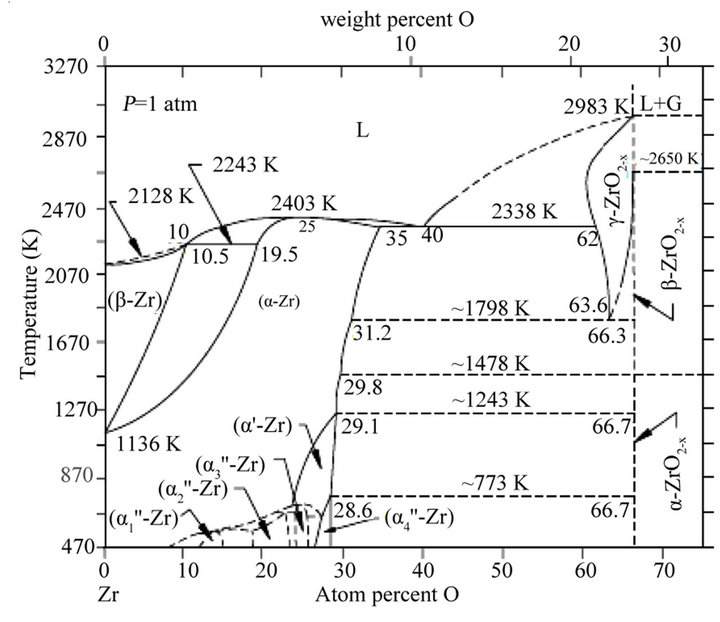
Figure 2. Equilibrium phase diagram for the Zr-O system [2].
following global reaction [6,7]:
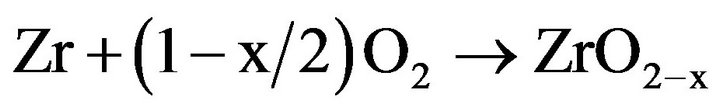
Only, part of the oxygen which arrives at the intern interface enters in this reaction; the other part is dissolved in subjacent metal [8].
Parise gave a relation between the values of oxygen diffusion coefficient in α zirconium between 300˚C and 600˚C [9]:
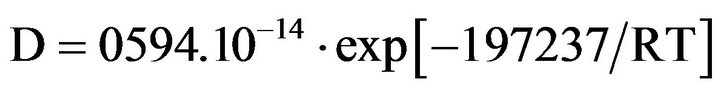
where D is in m2∙s-1, R = 8.314 J∙mol-1∙K-1 and T is in Kelvin.
This same type of relation was obtained by Debuigne for temperatures between 650˚C and 850˚C [8]:
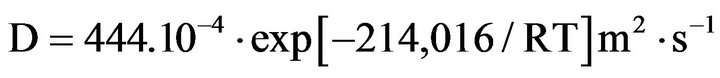
Concerning the oxygen diffusion coefficients in zirconia resulting from the oxidation of metal, Parise gives the following relation between 150˚C and 500˚C:
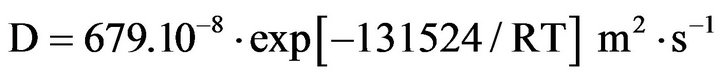
and between 400˚C and 850˚C, Debuigne gives:
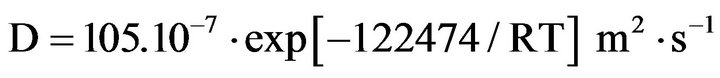
2. Experimental Procedure
We have studied the microstructure of Zry-4 as received (α phase). To follow the oxidation kinetics, we used, in-situ x-ray diffraction for different temperatures (25˚C, 350˚C, 500˚C, 830˚C and 1000˚C), in-situ XRD for temperatures of 350˚C and different times of ageing: 00 min, 10 min, 20 min, 40 min and 60 min.
During a postulated loss-of-coolant-accident (LOCA), the cladding tube is submitted in steam environment to a fast increase in the temperature (up to 800˚C - 1200˚C), which induces an accelerated oxidation phenomenon until quenching [1]. To simulate this case, we have done X-ray diffraction with Zry-4 samples water quenched at 1050˚C with different times at this temperature (05 sec, 10 sec, 30 sec, 01 min, 02 min, 05 min, 10 min and 20 min).
The X-ray diffraction is realized on a diffractometer equipped with a vertical goniometric, provided with x-rays tube with copper anticathode (KaCu: 1,5406 Å), the sample stages are spinner and “HTK-1200” furnace with a temperature that reaches 1200˚C.
The X-rays diffraction analysis parameters are the following:
Scan angle (2q): [15 to 125] and [25 to 65].
Step size (2q): 0,02.
Counting time (s): 1.
The studied material is the zirconium alloy of nuclear usage; the zircaloy-4. The X-Ray Fluorescence technique (XRF) defines the elementary chemical composition of this alloy (Table 1).
3. Results and Discussion
3.1. X-Ray Diffraction
The XRD for Zry-4 sample as received is shown in Figure 3.
Crystallographic structure of this material as received is the hexagonal close-packed structure (hcp-Zrα), this material has a polycrystalline metallographic structure with equiaxes grains and a microhardness Hv(0.5) ≈ 180 (kg/mm²).
3.2. In Situ X-Ray Diffraction (Oxidation Kenitics)
The Zry-4 sample has a diameter of 10 mm and a thick-
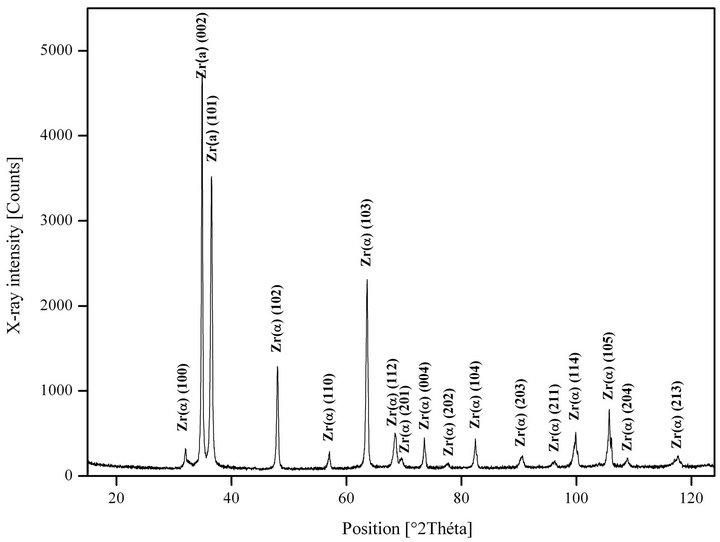
Figure 3. X-ray diffraction pattern of Zry-4 as received.
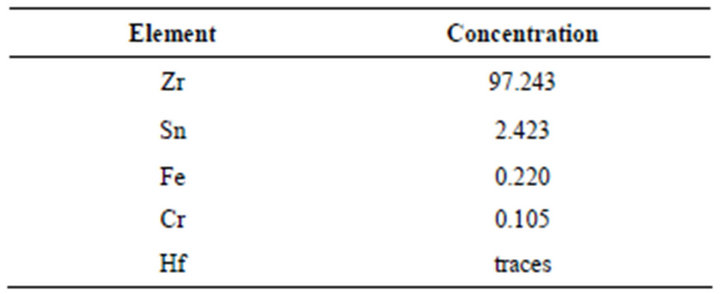
Table 1. Chemical composition of Zircaloy-4 (%).
ness of 0.9 mm, placed in “HTK-1200” furnace of the diffractometer. The different diffraction temperatures in air are: 25˚C, 350˚C, 500˚C, 830˚C and 1000˚C.
When the temperature of furnace is reached, the diffraction is immediately launched and the results are presented in the form of diffractograms.
The results of in-situ XRD diffractograms prove that:
• For 25˚C, there is only Zr-α phase, the principal peak of this phase is in 2θ = 34.83˚.
• The formation of the monoclinic and tetragonal phases start at 350°C, principal peaks of monoclinic and tetragonal phases are respectively at 2θ = 28.24˚ and 30.28˚
• From 350˚C to 1000˚C, there is an important evolution in the two monoclinic and tetragonal zirconias, Figure 4.
• When the temperature is equal or up to 830˚C, there is a formation of Zr-β phase and disappearance of Zr-α phase, the principal peak of Zr-β phase is at 2θ = 35.8˚, Figure 4.
These results give the oxidation kenitic’s of Zry-4, Figure 5.
For the room temperature 25˚C, There is only Zr-α phase, the principal peak intensity is 10,787 counts, this intensity increases to 11,747 counts for 350˚C, and decreases rapidly to 8985 counts for 500˚C until 1197 counts for 800˚C and zero intensity for 1000˚C, which confirms the reduction in the volume fraction of this phase.
The reduction in the volume fraction of Zr-α phase is directly related to the formation of the ZrO2 zirconia on the sample surface.
On the other hand, and for 25˚C, zero intensity for monoclinic and tetragonal ZrO2 zirconias. The X-rays diffracted intensities are increased progressively from 350˚C to 1000˚C.
For monoclinic zirconia: 388 counts for 350˚C, 697 counts for 500˚C, 2957 counts for 830˚C and 2070 counts for 1000˚C.
For tetragonal zirconia: 371 counts for 350˚C, 438 counts for 500˚C, 827 counts for 830˚C and 629 counts for 1000˚C.
Therefore, we have an increase in the volume fraction of zirconias with the increase in the temperature because of the oxygen diffusion.
3.3. Ageing in HTK-1200 Furnace
We have done in-situ XRD at 350˚C and at different times of ageing: 00 min, 10 min, 20 min, 40 min and 60 min (Figure 6).
We put the sample during a fixed time of ageing in 350˚C before starting the diffraction again. The holding time enables us to study the influence of the time on the formed phases.
During a typical Loss-Of-Coolant-Accident (LOCA) transient, the fuel cladding tubes are subject to high temperature oxidation that reaches 1000˚C and finally quenched because of the reflooding of the core. The global oxygen content and its distribution affect strongly the proprieties of the cladding which microstructure can be described in terms of zirconia phase.
We have studied this case with x-ray diffraction technique on Zry-4 samples water quenched at 1050˚C with different ageing times (05 sec, 10 sec, 30 sec, 01 min, 02 min, 05 min, 10 min and 20 min) at this temperature, the results are given in Figure 7.
For The 1050˚C water quenching after 05 seconds, there is no formation of oxide layer, but at 10 seconds to 20 minutes; there is a clearly evolution of monoclinic and tetragonal zirconias on one hand, and on the other hand, there is a disappearance of the Zr-α phase (Figure 7).
3.4. Microhardness
The indentation method allows the hardness of a material to be measured. The Vickers hardness of water quenching Zry-4 with 500 g weight is augmented to 207 (kg/mm2) at 05 seconds of ageing time to 1120 (kg/mm2) at 300 seconds of ageing time (Figure 8).
4. Conclusions
The oxidation of zircaloy-4 changes completely the
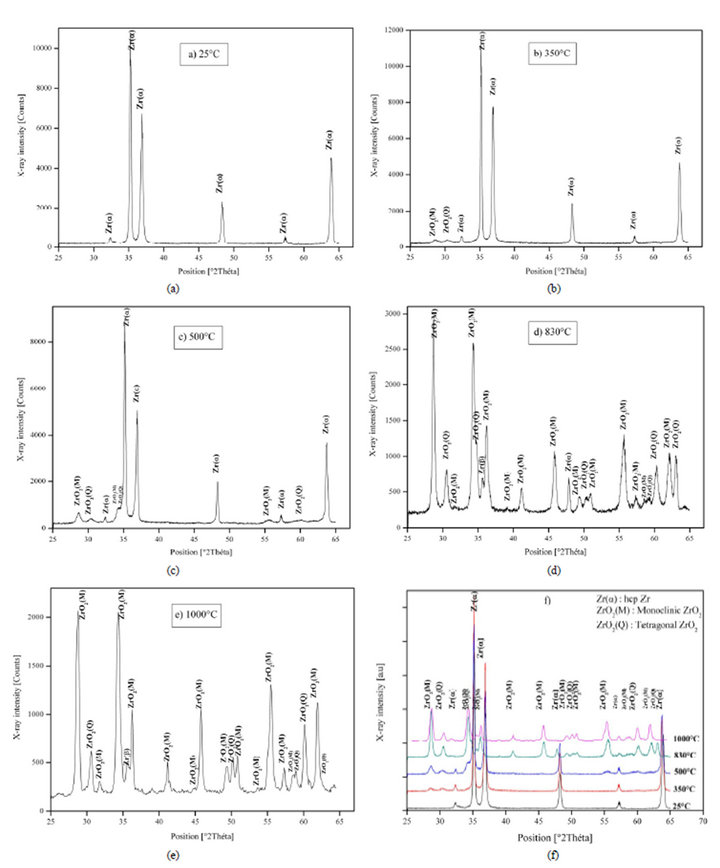
Figure 4. In-situ X-Ray diffraction patterns of Zry-4 for different temperatures: (a) 25˚C; (b) 350˚C; (c) 500˚C; (d) 830˚C; (e) 1000˚C; and (f) Combined graph.
properties of this material. In the presence of oxygen, and in temperature between 25˚C and 350˚C, a great quantity of oxygen can dissolve in interstitial positions, the zircaloy-4 maintains its properties. Between 350˚C and 1050˚C, and with the use of x-ray diffraction technique, we have an evolution of the two phases, monoclinic and
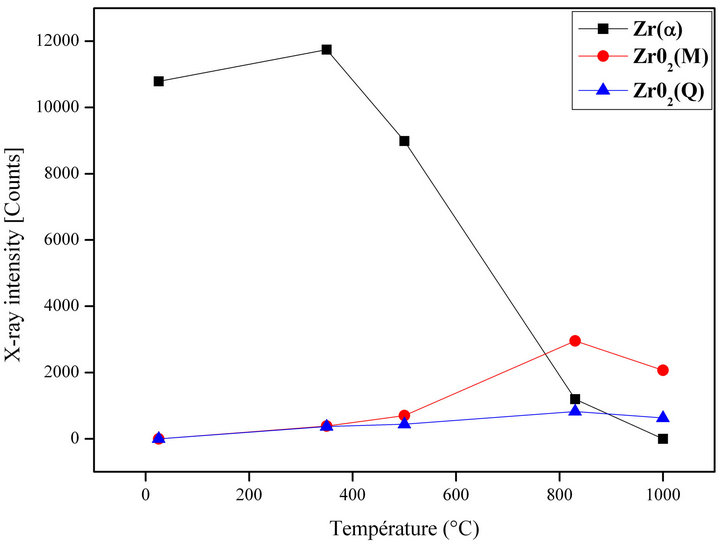
Figure 5. Evolution of the different phases in function of temperature.
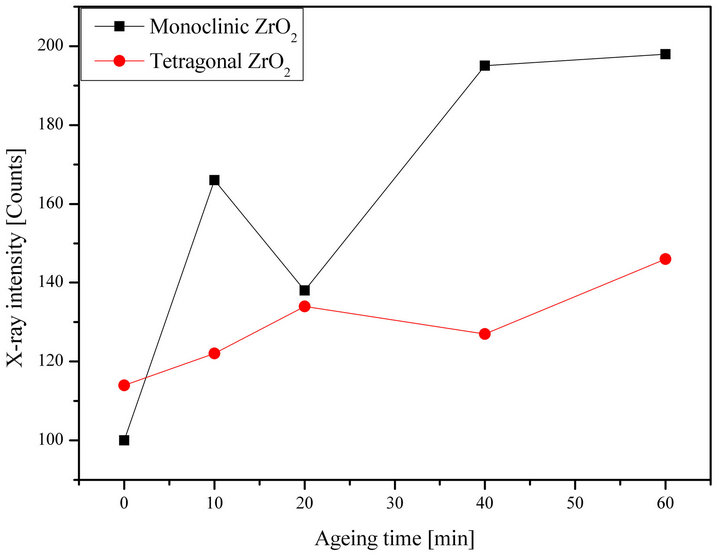
Figure 6. Evolution of the two formed zirconias (monoclinic and tetragonal) in function of ageing time at temperature of 350˚C in the HTK furnace.
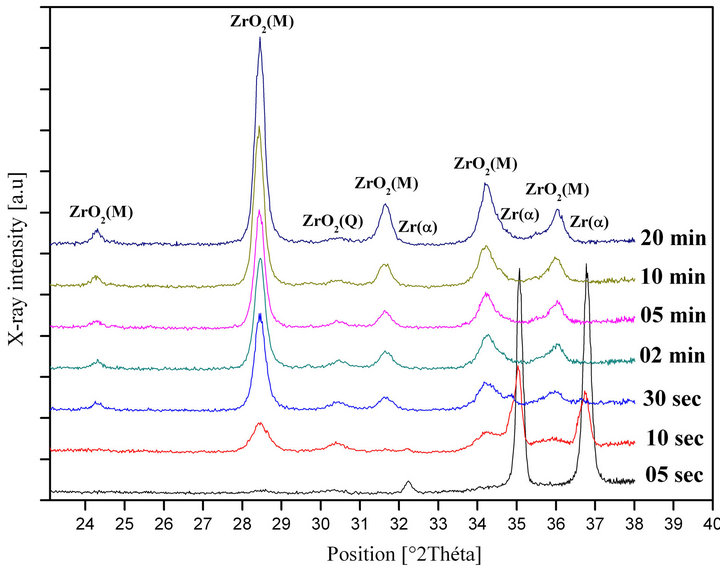
Figure 7. X-ray diffraction pattern of Zry-4 water quenched at 1050˚C with different ageing times at this temperature.
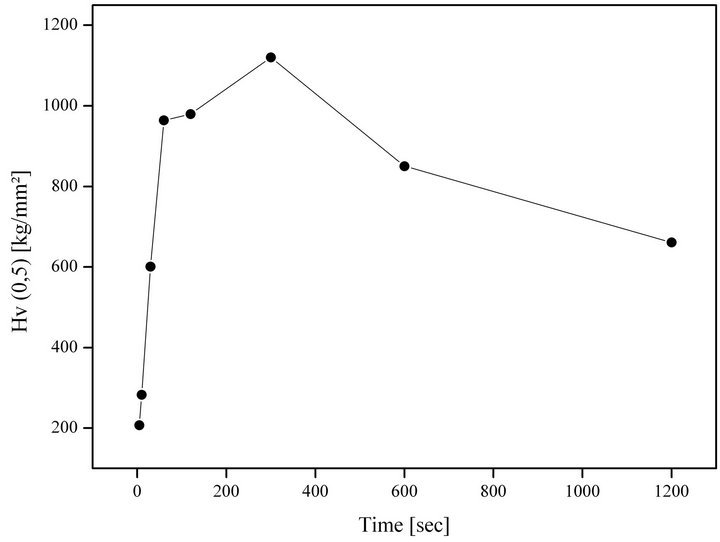
Figure 8. Microhardness (mHv (0.5)) for 1050˚C quenched samples with different ageing times.
tetragonal zirconia where the zircaloys-4 loses its properties.
The formation of monoclinic and tetragonal zirconia leads to the increase in hardness and the reduction in thermal conductivity which leads to the degradation of zircaloy-4 mechanical and thermal properties. The microhardness of these oxides (ZrO2) is very high, which increases the probability of cracking of this material, and consequently, the contamination of the primary circuit.
REFERENCES
- A. Stern, J.-C. Brachet, V. Maillot, D. Hamon, F. Barcelo, S. Poissonnet, A. Pineau, J.-P. Mardon and A. Lesbros, “Investigations of the Microstructure and Mechanical Properties of Prior-Structure as a Function of the Oxygen Content in Two Zirconium Alloys,” Journal of ASTM International, Vol. 5, No. 4, 2008, Article ID: JAI101119.
- S. Banerjee and P. Mukhopadyay, “Phase Transformation: Examples from Titanium and Zirconium Alloys,” Pergamon Press, Oxford, 2007, pp. 6-11, 42.
- D. Yacoub, “Experimental Study of Zirconium Alloys Oxidation at High Water Steam Pressure and Modelling of Mechanisms,” Ph.D. Thesis, Superior National School of Mines, Saint-Etienne, 2007.
- N. Bererd, “Irradiation Effects on the Zirconium Oxidation and the Uranium Diffusion in the Zirconia,” Ph.D. Thesis, Claude Bernard University-Lyon I, Chevarier Alain (Dir.), 2003.
- W. Qin, C. Nam, H. L. Li and J. A. Szpunar, “Tetragonal Phase Stability in ZrO2 Film Formed on Zirconium Alloys and Its Effects on Corrosion Resistance,” Acta Materialia, Vol. 55, No. 5, 2007, pp. 1695-1701. doi:10.1016/j.actamat.2006.10.030
- N. Pétigny, “Comparison of Tow Zirconium Alloys Oxidation by in-Situ and ex-Situ X-Ray Diffraction: Texture, Phase,” Ph.D. Thesis, Bourgogne University, Dijon, 1999.
- P. Putoud, “Alloys Oxidation of the Zircaloy-4 Type with Different Contents of tin at 400°C in the Steam. Analyzes of Phases in Presence and Their Evolution,” Ph.D. Thesis, Compiegne Technology University, Compiègne, 1995.
- J. Debuigne, “Contribution in the Study of the Zirconium Oxidation and the Oxygen Diffusion in the Oxide and the Metal,” Ph.D. Thesis, Paris University, Paris, 1966.
- M. Parise, “Mechanisms of Zirconium Alloys Corrosion: Study of the Oxidation initial Kinetics and the Mechanical Behaviour of the Metal-Oxide System,” Ph.D. Thesis, Superior National School of Mines, Paris, 1996.

