Open Journal of Obstetrics and Gynecology
Vol.3 No.1(2013), Article ID:26503,6 pages DOI:10.4236/ojog.2013.31010
Does robotic surgical staging of endometrial cancer compromise outcomes?
![]()
1Women’s Cancer Care of Seattle, Northwest Hospital & Medical Center, University of Washington Medicine, Seattle, USA
2Department of Obstetrics & Gynecology, Division of Gynecologic Oncology, University of Rochester School of Medicine, Rochester, USA
Email: *holly.r.sato@gmail.com
Received 26 October 2012; revised 28 November 2012; accepted 7 December 2012
Keywords: Endometrial Cancer; Surgical Staging; Robotic-Assisted Hysterectomy; Outcomes; Recurrence
ABSTRACT
Objective: To compare intra-operative and post-operative complications as well as recurrence in women with endometrial cancer undergoing surgical staging with robotic-assisted laparoscopic hysterectomy (RALH) versus total abdominal hysterectomy (TAH). Methods: A retrospective review was performed of 416 patients who underwent surgical staging for endometrial adenocarcinoma from January 1, 2008 to December 31, 2010. Demographics, intraand post-operative complications, surgical-pathologic data, subsequent treatments, and recurrence were compared in women treated with TAH versus RALH. A Student’s t-test, Mann Whitney U analysis, or chi-squared analysis were used for statistical analysis. Results: One hundred thirty seven and 279 patients underwent TAH and RALH, respectively. Post-operative complications were lower in the RALH group (16.8% vs 8.2%, p = 0.009) and mean hospital stay for the TAH group versus the RALH group was 3 days versus 1 day (p < 0.001). Demographics and intra-operative complications were similar (p = 0.94). Uterine weight (108 g vs 103 g, p = 0.36), pelvic lymph nodes retrieved (9 vs 9, p = 0.18), and presence of lymph-vascular involvement (11.7% vs 10.8%, p = 0.38) were similar between groups, as were aggressive histologic subtypes (p = 0.52) and grade (p = 0.15). Recurrence occurred in 4.4% of the TAH group and 4.3% of the RALH group (p = 0.97) with death from disease occurring in 1.5% of the TAH group and 2.2% of the RALH group (p = 0.64). Conclusions: RALH for endometrial adenocarcinoma is associated with fewer post-operative complications and a shorter hospital stay than TAH. Recurrence and death due to disease are not compromised by this minimally invasive approach.
1. INTRODUCTION
Endometrial cancer is the most common gynecologic malignancy in the United States, with an estimated 47,130 new cases and 8010 related deaths in 2012 [1]. The International Federation of Gynecology and Obstetrics recommended surgical staging for endometrial cancer in 1988 based on findings that 20% of women with clinical stage I disease have evidence of extra-uterine disease at the time of surgery [2,3]. Surgical staging entails total hysterectomy and bilateral salpingo-oophorectomy (BSO). Pelvic and para-aortic lymphadenectomy is routinely performed in some centers while others reserve these procedures for patients who are at intermediate to high risk for lymph node spread (grade 3 tumors, grade 2 tumors > 2 cm in diameter, clear cell or papillary serous histology, >50% myometrial invasion, or cervical extension) [4]. Although surgical staging has allowed for more tailored adjuvant treatment, there is no proof of enhanced survival and increased morbidity is a concern.
Minimally invasive surgery (MIS) has become more prominent within the field of gynecologic oncology although standard laparoscopic surgery has not been widely adopted for management of patients with endometrial cancer despite the decreased operative morbidity with this technique [5-7]. A drawback of MIS is decreased tactile sense which is more pronounced in robotic surgery, and there were concerns that metastatic disease, normally able to be palpated, would be missed with laparoscopic surgery. Other concerns with laparoscopic surgery were changes in recurrence patterns due to insufflation and the associated high intra-abdominal pressure, port site metastases, and tumor spillage related to the uterine manipulator. The recent GOG LAP2 study [8] addressed several of these concerns finding no statistical difference in recurrence rates in patients with clinical stage I to IIA endometrial cancer who underwent minimally invasive (standard laparoscopy and robotic-assisted laparoscopy) versus laparotomy staging. While this study is reassuring, it did not address robotic surgery as its own entity nor did it address long term outcomes for more advanced stage disease.
The recent introduction of the da Vinci Surgical System™ (Intuitive Surgical, Inc., Sunnyvale, CA) has advanced the field of gynecologic oncology by allowing more widespread use of a minimally invasive approach for the surgical management of endometrial cancer. The da Vinci Surgical System™ offers advantages over traditional laparoscopy such as high definition, 3-dimensional images of the operative field, instruments with a wrist-like range of motion for improved surgical dexterity, the lack of a fulcrum effect, a faster learning curve, and improved ergonomics to reduce surgeon fatigue [9]. There are currently 1615 da Vinci Surgical Systems™ installed in the United States [10].
Few studies have compared the long term outcomes of robotic-assisted laparoscopic hysterectomy (RALH) versus total abdominal hysterectomy (TAH) for endometrial cancer staging [11]. The purpose of this study is to compare surgical morbidity, recurrence, and mortality in subjects with endometrial cancer undergoing TAH versus RALH.
2. METHODS
Institutional IRB approval was obtained for this study to ensure patient privacy. Patient permission was not needed given that the study was a retrospective chart review not impacting patient privacy or treatment. Billing and procedure codes were used to identify 488 patients from a gynecologic oncology office who underwent surgery for endometrial cancer from January 1, 2008 to December 31, 2010. Of the 488 subjects identified, 32 were excluded for having insufficient data and 40 were excluded for having a prior hysterectomy, a gynecologic malignancy other than endometrial adenocarcinoma, complex endometrial hyperplasia, or no gynecologic malignancy.
A retrospective study was performed of the remaining 416 patients using clinic and hospital charts to collect demographics including age, gravidity, parity, body mass index (BMI), prior abdominal surgery, and medical comorbidities. The surgery performed, intraand postoperative complications within 6 weeks of follow-up, and length of hospital stay were recorded. Surgical-pathologic data including uterine weight, tumor histology, grade, myometrial invasion, stage, lymph-vascular involvement, and number of pelvic and para-aortic lymph nodes collected as well as number of positive nodes were collected. Subsequent treatment with radiation and/or chemotherapy, recurrence, and death due to disease were noted.
The surgical approach was at the discretion of the attending surgeon, and the series reflects the learning curve for robotics by members of the division. An open approach was more likely if there was a history of multiple laparotomies, prior pelvic radiation, or suspected metastatic or upper abdominal disease. All patients underwent pelvic lymphadenectomy. Para-aortic lymph node dissection was performed, if feasible, for those with aggressive histologies, high-grade lesions, deep myometrial invasion, or large tumors noted on preoperative evaluation or frozen section.
A Student’s t-test or Mann Whitney U analysis was used for continuous variables that were normally distributed and not normally distributed, respectively. Chisquared analysis was used to examine discrete variables. A p-value of <0.05 was used to determine statistical significance.
3. RESULTS
Of the 416 study patients with endometrial adenocarcinoma, 137 (33%) underwent TAH-BSO and lymphadenectomy while 279 (67%) underwent RALH-BSO and lymphadenectomy reflecting the introduction of robotic-assisted surgery into our practice. Seven of the RALH (2.5%) were converted to TAH but were kept in the RALH cohort for analysis. Four were converted due to adhesions, one due to major vessel injury, one due to inability to place a trocar due to the patient’s morbid obesity, and one due to a combination of adhesions and inability to adequately ventilate the patient in deep Trendelenberg.
Baseline demographics including age, gravidity, parity, BMI, prior abdominal surgery, and medical co-morbidities for the TAH and RALH groups are shown in Table 1. The average age (63.5 versus 63.4), prior abdominal surgery (61% versus 52%), medical comorbidities, body mass index (34.0 kg/m2 versus 34.8 kg/m2), and distribution of body mass index classification were similar between the TAH and RALH groups.
Both groups had similar rates of intra-operative complications but there were significantly fewer post-operative complications in the RALH group (16.8% vs 8.2%, p = 0.009) (Table 2). Wound complications occurred in only 1.4% of the RALH group versus 7.3% of the TAH group (p = 0.002). The mean length of stay was a shorter for the RALH group (3 days vs 1 day, p < 0.001). Acute renal injury occurred more frequently in the TAH group (2.9% vs 0.4%, p = 0.02).
Histology, grade distribution, uterine weight, lymph-vascular involvement, and number of pelvic lymph nodes retrieved were not different between the TAH and RALH groups (Table 3). There were more para-aortic lymph nodes retrieved in the TAH group (2 vs 1, p = 0.003) with the TAH group also having more pelvic and para-aortic lymph nodes positive for carcinoma (pel-
Table 1. Demographics and medical co-morbidities of subjects undergoing surgical staging for endometrial cancer.

Table 2. Intraoperative and postoperative complications in subjects undergoing TAH versus RALH for endometrial cancer staging.
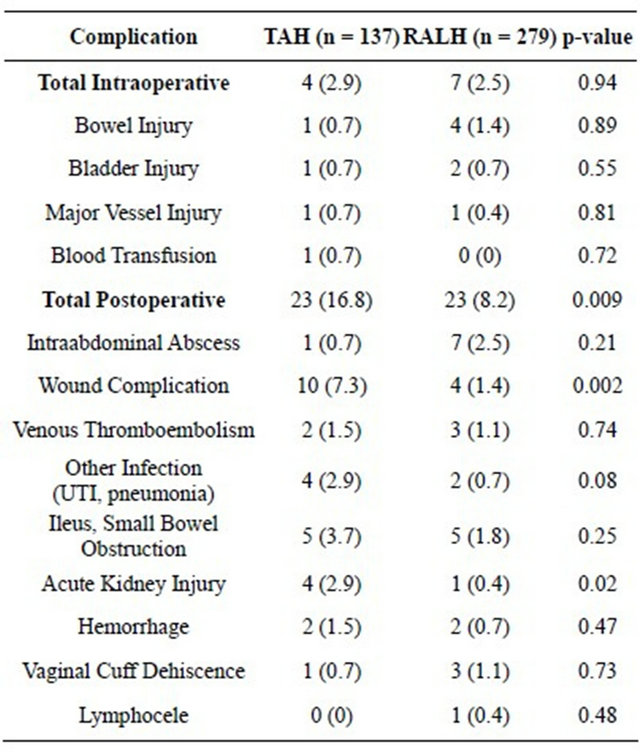
Table 3. Surgical-pathologic data and stage of subjects undergoing TAH versus RALH.

vic: 11.8% vs 5.5 %, p = 0.02, para-aortic: 8.4% vs 1.3%, p = 0.03). Deep myometrial invasion occurred more frequently among patients in the TAH group (35.8% vs 23.7%, p = 0.009).
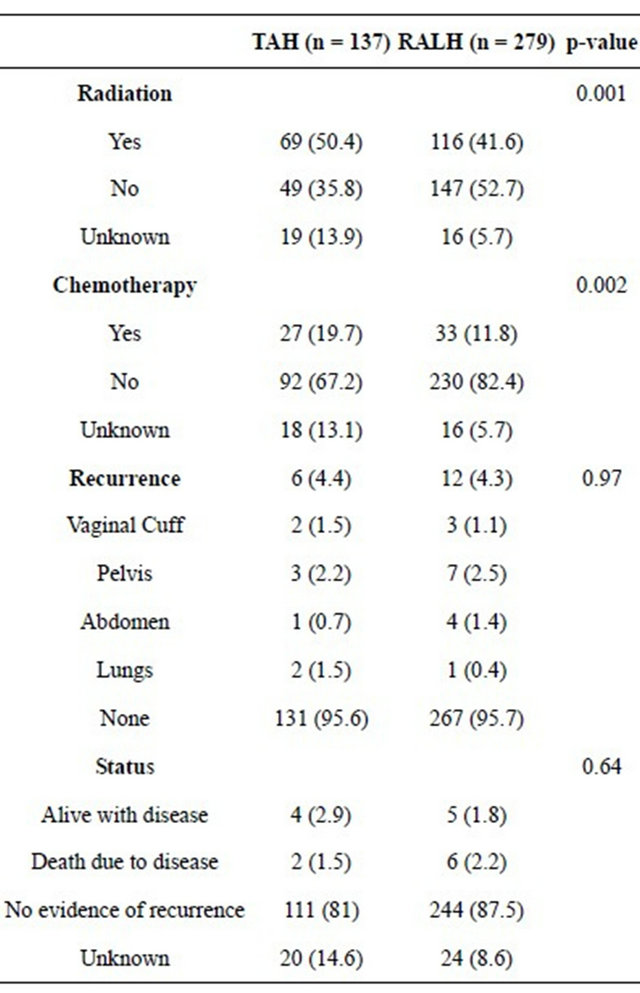
Based on the surgical-pathologic data, more advanced stages of endometrial cancer were seen in subjects who underwent TAH with a statistically higher rate of Stage III disease in the TAH subjects (16.1% vs 7.9%, p = 0.02) (Table 3). The TAH group received more adjuvant radiation (50.4% vs 41.6%, p = 0.001) and chemotherapy (19.7% vs 11.8%, p = 0.002) (Table 4).
The median length of follow-up was 835 days (range 264 to 1276 days) for the TAH group and 518 days (range 18 to 1237 days) for the RALH group (p < 0.001).
Overall, there was no difference in recurrence (4.4% vs 4.3%, p = 0.97) between patients undergoing TAH versus RALH (Table 4). Recurrences occurred in the vaginal cuff (1.5% vs 1.1%), pelvis (2.2% vs 2.5%), abdomen (0.7% vs 1.4%), and lung (1.5% vs 0.4%) with some subjects recurring in multiple sites. Two subjects died of their disease in the TAH group versus six in the RALH group (1.5% vs 2.2%, p = 0.36).
4. DISCUSSION
Minimally invasive surgery has decreased morbidity for women undergoing surgical staging for endometrial cancer [12]. However, the use of laparoscopic surgical staging has been limited due to the longer operative times and prolonged learning curve associated with these complex surgical procedures [7,12,13]. The introduction of the da Vinci Surgical System™ has expanded the use of minimally invasive techniques for endometrial cancer surgical staging.
Table 4. Adjuvant treatment, recurrence, and status of subjects undergoing TAH versus RALH for endometrial cancer.
This study found significantly fewer post-operative complications in patients undergoing treatment with RALH compared to TAH (8.2% v 16.8%), a finding noted by several prior investigators [5,7,13,14]. Patients who were staged with TAH underwent a large vertical midline abdominal incision with wound complications (cellulitis, superficial dehiscence, fascial dehiscence) occurring in 7.3% compared to 1.4% of patients in the RALH group. In addition, the average BMI of our patients was 34 kg/m2. Obesity is a well-known risk factor for endometrial cancer with nearly half of patients with endometrial cancer having a BMI greater than 30 kg/m2. We found that robotic surgery, with its significantly smaller incisions, decreased the risk of post-operative wound complications in a population at high risk for these problems, which confirms findings noted in a number of previous studies [5,7,14-16].
The overall recurrence rate was 4.4% in the TAH group versus 4.3% in the RALH group. In addition, we did not find a significant difference in the risk of cuff recurrence with 1.5% in the TAH group versus 1.1% in the RALH group. Thus, despite the theoretical risk of increased manipulation of the uterus during robotic hysterectomy and the difficulty of performing para-aortic node dissections with the robotic approach, we did not find any impact on recurrence risk. Although the survival value of lymphadenectomy is controversial, it is prognostic and useful in selecting appropriate adjuvant therapy [17].
In our study, the mean duration of follow-up was 835 days for the TAH group and 518 days for the RALH group, reflecting the recent introduction of robotics at our institution. There have been few long-term studies comparing the recurrence of endometrial cancer in subjects undergoing staging with RALH versus TAH. Lau et al. found a lower endometrial cancer recurrence rate in women who underwent robotic staging compared to women who underwent staging with laparotomy or traditional laparoscopy (7.7% vs 11.9%, p < 0.001), when followed for two years [18], and the GOG LAP2 study [8] found no significant difference in recurrence rate over 59 months in patients with clinical stage I to IIA endometrial cancer undergoing minimally invasive (traditional laparoscopy and robotic-assisted laparoscopy) versus laparotomy staging. Similarly, Fader et al. examined the use of MIS compared to laparotomy in high grade endometrial cancers [11]. Of the minimally invasive subjects, 65% were stage robotically and 35% were staged using traditional laparoscopy. Similar to our study, they found fewer complications without compromise of survival outcomes in those undergoing minimally invasive versus laparotomy staging. Although our median follow-up for the robotic group was shorter, 60% of recurrences of endometrial cancer occur within 2 years, so we feel we have captured a majority of those destined to recur [19]. Based on our study, the studies by Lau et al. and Fader et al., and the GOG LAP2 study, it is increasingly apparent that the initial concerns regarding an increased risk of recurrent cancer for patients undergoing minimally invasive procedures were unfounded, and that there are clear benefits with MIS in terms of operative morbidity and length of stay.
The main weakness of this study is that it is retrospective and the two groups are not comparable. The TAH staged group had higher rates of significant myometrial invasion and more positive lymph nodes with a corresponding higher proportion of stage III and stage IV cases compared to the RALH group. Although there was no recall bias given that the study was retrospective, there may have been selection bias when choosing laparotomy versus robotic staging for patients. If preoperative testing had findings concerning for more advanced disease such as lesions concerning for metastases or lymphadenopathy, the surgeon may have been more likely to choose laparotomy. The TAH group had a higher proportion of both positive pelvic and para-aortic lymph nodes. Both the TAH and RALH cohorts had similar numbers of total pelvic lymph nodes retrieved so the difference in positive pelvic lymph nodes was likely not due to a difference in surgical technique. However, fewer para-aortic lymph nodes were retrieved in the RALH group, likely due to the learning curve associated with introducing a new modality. The RALH cohort also had fewer positive para-aortic lymph nodes, which is in accord with the lower percentage of positive pelvic nodes in this group. While it is certainly possible that some in the RALH were under-staged, particularly with respect to the para-aortic node dissection, this does not appear to have an impact on the risk of recurrence [17].
The introduction of robotic surgery has improved accessibility to MIS for endometrial cancer staging. Although widely accepted as a modality for endometrial cancer staging, it is still a relatively new technique and few studies with long-term outcomes have been reported. We found fewer post-operative complications and a shorter hospital stay in those undergoing robotic treatment of endometrial cancer, confirming previous studies. We also found no difference in recurrence risk with robotic surgery. We believe that minimally invasive approaches to the treatment of patients with endometrial cancer have clear benefits and that this approach will dominate in the future.
5. CONCLUSION
RALH for endometrial adenocarcinoma is associated with fewer post-operative complications and a shorter hospital stay than TAH. Recurrence and death due to disease are not compromised by this minimally invasive approach.
6. ACKNOWLEDGEMENTS
Thank you to J. Christopher Glantz, MD for help with statistical analysis.
![]()
![]()
REFERENCES
- Siegel, R., Naishadham, J. and Jemal, A. (2012) Cancer statistics 2012. CA Cancer Journal for Clinicians, 62, 10- 29. doi:10.3322/caac.20138
- Creasman, W.T., Morrow, C.P., Bundy, B.N., Homesley, H.D., Graham, J.E. and Heller, P.B. (1987) Surgical pathologic spread patterns of endometrial cancer: A gyncecology oncology group study. Cancer, 60, 2035-2041. doi:10.1002/1097-0142(19901015)60:8+<2035::AID-CNCR2820601515>3.0.CO;2-8
- Mikuta, J.J. (1993) International federation of gynecology and obstetrics staging of endometrial cancer 1988. Cancer, 71, 1460-1463. doi:10.1002/cncr.2820710409
- Neubauer, N.L. and Lurain, J.R. (2011) The role of lymphadenectomy in surgical staging of endometrial cancer. International Journal of Surgical Oncology, 2011, Article ID: 814649.
- Boggess, J.F., Gehrig, P.A., Cantrell, L., et al. (2008) A comparative study of 3 surgical methods for hysterictomy with staging for endometrial cancer: Robotic assistance, laparoscopy, laparotomy. American Journal of Obstetrics & Gynecology, 199, 360e1-360e9.
- Eletabbakh, G.H. (2002) Analysis of survival after laparoscopy in women with endometrial carcinoma. Cancer, 95, 1894-901. doi:10.1002/cncr.10928
- Zullo, F., Falbo, A. and Palomba, S. (2012) Safety of laparoscopy vs laparotomy in the surgical staging of endometrial cancer: A systematic review and metaanalysis of randomized controlled trials. American Journal of Obstetrics & Gynecology, 26, 94-100. doi:10.1016/j.ajog.2012.01.010
- Walker, J.K., Piedmonte, M.R., Spirtos, N.M., et al. (2012) Recurrence and survival after random assignment to laparoscopy versus laparotomy for comprehensive surgical staging of uterine cancer: Gynecologic oncology group LAP2 study. Journal of Clinical Oncology, 20, 695-700. doi:10.1200/JCO.2011.38.8645
- Ahlering, T.E., Skarecky, D., Lee, D. and Clayman, R.V. (2003) Successful transfer of open surgical skills to a laparoscopic environment using a robotic interface: Initial experience with laparoscopic radical prostatectomy. The Journal of Urology, 170, 1738-1741. doi:10.1097/01.ju.0000092881.24608.5e
- Intuitive Surgical (2012) Second quarter earnings 2012. http://phx.corporate-ir.net/phoenix.zhtml?c=122359&p=irol-IRHome
- Fader, A.N., Seamon, L.G., Escobar, P.F., et al. (2012) Minimally invasive surgery versus laparotomy in women with high grade endometrial cancer: A multi-site study performed at high volume cancer centers. Gynecologic Oncology, 126, 180-185. doi:10.1016/j.ygyno.2012.04.028
- Ghezzi, F., Cromi, A., Uccella, S., et al. (2010) Laparoscopic versus open surgery for endometrial cancer: A minimum 3-year follow-up study. Annals of Surgical Oncology, 17, 271-278. doi:10.1245/s10434-009-0720-1
- Wattiez, A., Soriano, D., Cohen, S.B., et al. (2002) The learning curve of total laparoscopic hysterectomy: Comparative analysis of 1647 cases. The Journal of the American Association of Gynecologic Laparoscopists, 9, 339- 345. doi:10.1016/S1074-3804(05)60414-8
- DeNardis, S.A., Holloway, R.W., Bigsby IV, G.E., Pikaart, D.P., Ahmad, S. and Finkler, N.J. (2008) Robotically assisted laparoscopic hysterectomy versus total abdominal hysterectomy and lyphadenectomy for endometrial cancer. Gynecologic Oncology, 111, 412-417. doi:10.1016/j.ygyno.2008.08.025
- Lim, P.C., Kang, E. and Park, D.H. (2011) A comparative detail analysis of the learning curve and surgical outcome for robotic hysterectomy with lymphadenectomy versus laparoscopic hysterectomy with lymphadenectomy in treatment of endometrial cancer: A case-matched controlled study of the first one hundred twenty two patients. Gynecologic Oncology, 120, 413-418. doi:10.1016/j.ygyno.2010.11.034
- Backes, F.J., Brudie, L.A., Farrell, M.R., et al. (2012) Shortand long-term morbidity and outcomes after robotic surgery for comprehensive endometrial cancer staging. Gynecologic Oncology, 125, 546-551. doi:10.1016/j.ygyno.2012.02.023
- Sorosky, J.I. (2012) Endometrial cancer. Gynecologic Oncology, 120, 383-397. doi:10.1097/AOG.0b013e3182605bf1
- Lau, S., Vaknin, Z., Ramana-Kumar, A.V., Halliday, D., Franco, E.L. and Gotlieb, W.H. (2012) Outcomes and cost comparisons after introducing a robotics program for endometrial cancer surgery. Gynecologic Oncology, 119, 717-724. doi:10.1097/AOG.0b013e31824c0956
- Creutzberg, C.L., van Putten, W.L., Koper, P.C., et al. (2003) Survival after relapse in patients with endometrial cancer: Results from a randomized trial. Gynecologic Oncology, 89, 201-209. doi:10.1016/S0090-8258(03)00126-4
NOTES
*Corresponding author.

