Open Journal of Preventive Medicine
Vol.2 No.1(2012), Article ID:17413,5 pages DOI:10.4236/ojpm.2012.21010
Inhibition of calcium oxalate nephrotoxicity with Zamzam water
![]()
Department of Pharmacology and Toxicology, Faculty of Medicine, Umm Al-Qura University, Makkah, Kingdom of Saudi Arabia; dr.s.s.alghamdi@gmail.com
Received 12 October 2011; revised 20 November 2011; accepted 16 December 2011
Keywords: Zamzam Water; Calcium Oxalate; Nephrotoxicity
ABSTRACT
Zamzam water is well known of its high conductivity. For this fact urologist and nephrologists recommend their patients who are suffering from kidney stones not to drink this water because it could worse their health status. This study was conducted to investigate the effect of Zamzam water on calcium oxalate nephrotoxicity in experimentally induced kidney stones in male Wistar albino rats. Calcium oxalate crystals were induced by orally administration of 200 mg of glycolic acid dissolved in the drinking water. The rats were divided into three groups; six rats each. These include positive control group (given glycolic acid), test group (given glycolic acid plus Zamzam water) and negative group (given drinking water only). After two weeks of treatment, blood analysis of blood urea nitrogen (BUN) and creatinine showed significant differences in positive control group compared to the negative control group, whereas no significant differences were noticed in the level of BUN and creatinine between both the negative control and the test group. Moreover, urine analysis showed a high density of calcium oxalate crystals in the positive control group, whereas no crystals were detected in the negative control and the test groups. Histopathological investigations showed damaging in kidneys of the positive control group with no tissue abnormalities in the negative control and the test group. I concluded from this study that Zamzam water prevents the formation calcium oxalate stone, which probably mean that it has no negative effect on patients suffering from kidney disorders due to crystals formation.
1. INTRODUCTION
Zamzam water is located inside the Holy Mosque at about 20 meters east of the Ka’ba in Makkah Al-Mukarramah, Saudi Arabia. The well of Zamzam is hand-excavated and is about 30.5 m deep, with an internal diameter ranging from 1.08 to 2.66 meters.
Zamzam water is different from other water in many ways: first no bacteria can form at its source. Second it doesn’t go mouldy nor does it change colour, taste or smell [1]. Biological growth and vegetation usually take place in most wells. This makes water unpalatable owing to the growth of algae leading to changes in taste and odour. However, in Zamzam water well, there isn’t any sign of biological growth [2].
The Chemical analysis of Zamzam water contains some inorganic elements such as sodium (Na), calcium (Ca), magnesium (Mg), potassiumn (K), bicarbonate (HCO3), chloride (Cl), fluoride (Fl), nitrate (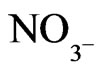 ), sulfate (SO4), and totally dissolved salts (TDS) [3].
), sulfate (SO4), and totally dissolved salts (TDS) [3].
Renal calculi are formed when the urine is supersaturated with salt and minerals such as calcium oxalate, struvite (ammonium magnesium phosphate), uric acid and cystine [4]. In most types of kidney stones, calcium oxalate crystals are found to be the main constituent. The prevalence of calcium oxalate crystals has been constantly increasing during past fifty years in industrialized as well as in developing countries and varied depending on race, sex and geographic locations. Although kidney stones can be traced to the earliest antiquity of human history, the primary causative factors remain obscure. It is suspected that kidney stones have direct relationship to the composition of urine, which depends on the patients’ habits [5,6].
This study was conducted to illustrate the effects of Zamzam water on experimentally induced renal in animal model.
2. MATERIALS AND METHODS
2.1. Urine Analysis
Eighteen male Wistar albino rats weighted between 350 to 410 g were used in the experiments. Rats were divided into three groups: positive control, negative control and test group as 6 rats/group. Each rat placed independently in a metabolic cage.
Rats of the positive control group rats were given 200 mg of glycolic acid orally using gastric gavages for each rat, and supplied with 500 mL bottled drinking water, whereas, the test group was given 200 mg of glycolic acid and supplied with 500 mL Zamzam until the end of experiments. The negative control group rats were supplied with bottled drinking water only. Animal groups were lifted for two weeks. Zamzam water was collected from Zamzam well through the united office of Zamzami, which located in Makkah.
Urine was collected from rats and studied for volume, pH value, and examined microscopically for crystals.
2.2. Blood Analysis
Two ml of blood were collected in plain tube by cutting of two third of the tail of each rat for all groups to measure blood urea nitrogen (BUN) and creatinine.
2.3. Histopathological Study
It has been applied to examine the histopathological changes in the tissues of kidneys for all rats in each groups. Rats were killed using chloroform. The rats were dissected and kidneys were removed and preserved in 10% formaldehyde for fixation of the tissues. Gross from kidney were processed, ambeded and stained using the procedure of H and E methods.
2.4. Statistical Data
Results are presented as mean ± SEM. Means were obtained from six rats for each group. Statistical analysis was performed using student t-test. Statistical significance was assured when P < 0.05.
3. RESULTS
3.1. Urine Excretion
The mean volume of two weeks collected urine shows that the amount of urine excreted by the test group was significantly higher than that excreted by the negative (P = 0.01) and positive (P = 0.007) control groups. Moreover, the amount of urine excreted by the positive control was significantly lower (P = 0.01) than the negative control (Table 1).
3.2. Urine pH
Table 2 shows the pH reading of the positive control group, which was acidic. The acidity was significantly reduced (P < 0.05) by the Zamzam water.
3.3. Microscopic Examination
|

Table 1. The average extracted urine volume of each group for two weeks (Mean ± SEM).

Table 2. The average urine pH of the groups of rats.
both test and negative control group (Figure 1). The pictures were randomly selected due to the similarity of results.
3.4. Blood Analysis
Table 3 shows that both BUN and creatinine levels were significantly higher (P = 0.007) in the positive control group compared to both the negative and the test groups. Moreover, the level of BUN was significantly higher (P = 0.04) in the test group than the negative control group. There was no significant difference in the level of createnine between both the negative and the test group.
3.5. Histopathological Examination
In the cortex of kidney tissues of the negative control group, most of proximal convoluted tubules, which were seen as large, prominent and generally staining a deeper pink than the other tubules. As an artefact of histological preparation, in some sets there were small, white splits in the walls of these tubules. Nuclei were distinct and the brush border was visible on most of the cuboidal cells. Distal tubules were paler in appearance, usually having a smaller diameter, and a low cuboidal epithelium. In the renal corpuscle numerous capillary loops of the glomerulus having a uniform diameter and with flat nuclei belonging to endothelial cells, podocytes and mesangial cells were observed. The collecting tubules were also observed in which the epithelium was simple cuboidal or low columnar, the cell outlines distinct, and the nuclei were prominent and closer together than in proximal or distal tubules. In the medulla the thin portion of the loop of Henle was lined by a simple squamous epithelium with numerous capillaries and sections of collecting ducts.
The proximal tubular epithelium of the Positive control group appeared highly acidophilic and the number of the microvilli (brush border) was reduced. The glomerular capillaries appeared slightly dilated. The distal tubules were
 (a)
(a) (b)
(b) (c)
(c)
Figure 1. Calcium oxalate crystals in the urine of positive control group (a). The test group (b) and negative control group (c) looked the same.
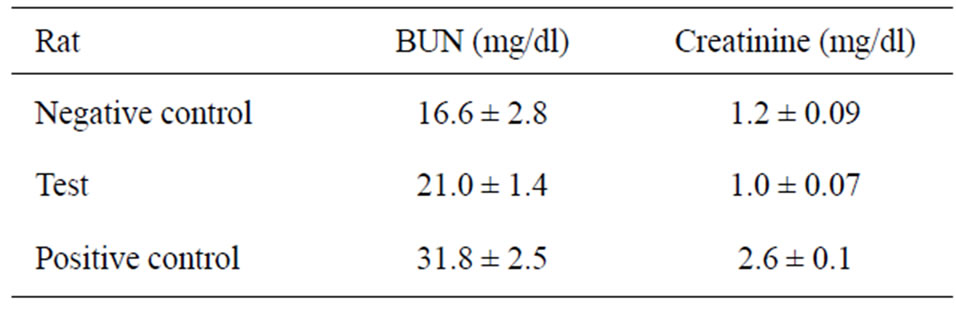
Table 3. The average levels of BUN and creatinine of groups of rats.
normal. The interstitial spaces in the medulla appeared slightly expanded. There were dilated and congested capillaries in the medulla. Small areas of hemorrhage were seen. Some of them were present in the cortex but most of them and more extensive were observed in the medulla.
Glumerular capillaries and mesangial tissue of the test group appeared normal; the capillaries had a regular diameter and did not seem to be dilated. Other components of the renal corpuscle also appeared normal. Proximal tubule had the usual normal cuboidal epithelium; their outline was regular with an intact basal lamina and a prominent brush border. Distal tubules had a regular diameter and slightly paler appearance than the proximal tubules. In the medulla the thin limb of loop of Henle appeared normal. The capillaries were not dilated and there was no congestion in them. The interstitial tissue appeared normal; no areas of hemorrhage were observed (Figure 2). The pictures were randomly selected due to the similarity of results.
4. DISCUSSION
Hyperoxaluria can provoke calcium nephrotoxicity. For the treatment and/or the prevention of calcium oxalate kidney stone formation, different approaches have been tested [7-9]. Oxalate metabolism considered to be almost identical between rats and humans, a rat model of calcium oxalate nephrotoxicity can be used to test the effect of different compounds on oxalate nephrotoxicity and which of them can alter the solubility of oxalate [10].
The chemical analysis of Zamzam water demonstrated highly significant readings in some inorganic elements when compared to the tap water. Laboratory investigations for the chemical composition of Zamzam water demonstrated almost similar readings with no significant differences in inorganic elements and pH but when compared to water collected from different wells in Saudi Arabia. Zamzam water showed highly significant readings in some inorganic elements, including Na, Ca, Mg, K, HCO3, Cl, Fl, NO3, and SO4. The levels of these elements in Zamzam water may play a critical role in its effectives in the inhibition of calcium oxalate formation.
Current evidence suggests that the consumption of diets low in calcium is associated with a higher overall risk for the development of kidney stones [7]. This is perhaps related to the role of calcium in binding ingested oxalate in the gastrointestinal tract. As the amount of calcium intake decreases, the amount of oxalate available for absorption into the bloodstream increases; this oxalate is then excreted in greater amounts into the urine by the kidneys. In the urine, oxalate is a very strong promoter of calcium oxalate precipitation, about 15 times stronger than calcium. In view of the above mentioned levels of ions it can be expected that both magnesium and pH can exert a fine kinetic control on the precipitation and growth of calcium oxalate monohydrate. Magnesium could replace calcium ions affecting

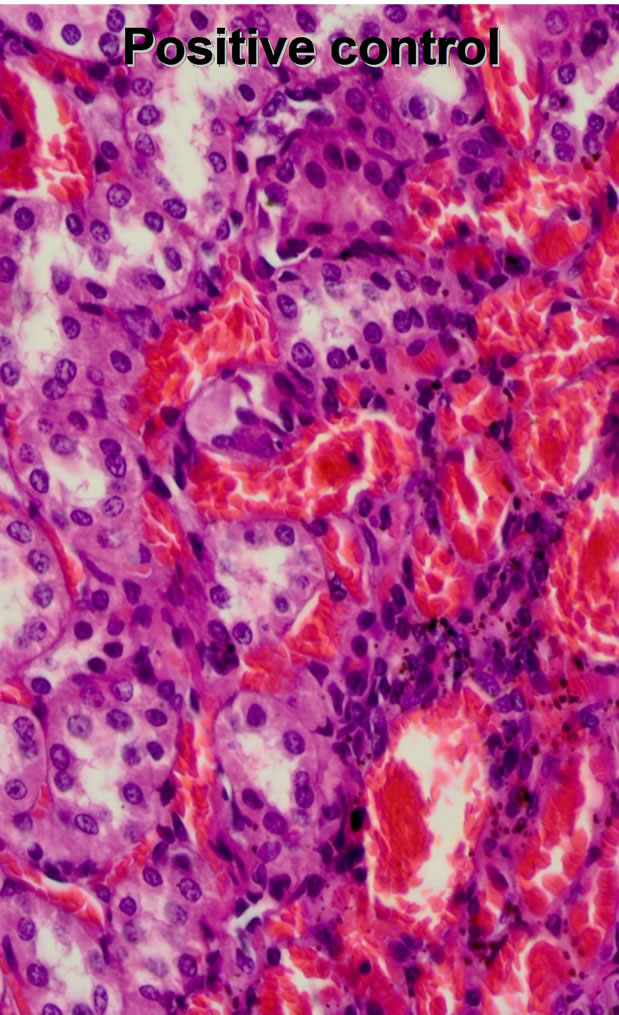
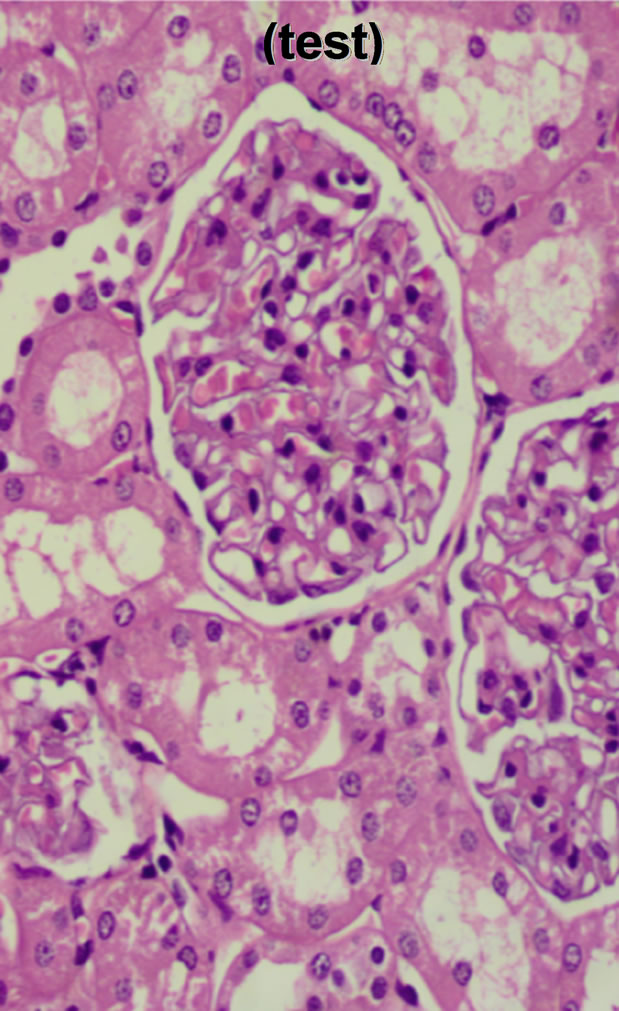
Figure 2. Histopathological changes of the animal groups; negative, positive and test groups (from left to right).
the dissolution of the later with a possible role played by pH. Magnesium decreases the urinary saturation of calcium oxalate by combining with urinary oxalate to form soluble magnesium oxalate so long as it is administered with meals. In addition, the presence of NaCl at a concentration as high as it seems in Zamzam water can affect the dissolution of such stone [10].
In this study, I found that the urine out put of the test group was higher than the other two groups indicating the diuretic effects of the Zamzam water. Moreover, the urine of positive control group was acidic, whereas in the test group, the pH was about 7.3. This could be attributed to the diuretic activity of Zamzam water. This diuretic effect may have an impact on stone precipitation and/or formation within the kidney through increasing the urine flow rates and consequently decreasing the stagnation of crystals within the renal tubules. Moreover, the acidity of urine of the positive control may precipitate stone formation whereas the high pH (7.3) of the test group rats may have a positive lowering effect on kidney stone formation.
Most of the literatures report a positive correlation between high fluoride content of water and stone formation [11,12].
Histopathological investigation obviously reveals the damaging of kidney in positive control, where the function of the kidney is altered. This may explain the high level of blood BUN and creatinine. The damages in the glomerular capillaries in positive control were absent in rats that were drinking Zamzam water because no calcium oxalate crystals were formed as demonstrated by the urine analysis. Damaging of kidneys of the positive control.
In conclusion, the multi-ionic contents of Zamzam water prevent the formation of renal stone that illustrates the special characteristic of this water and make it different from other types of waters.
5. ACKNOWLEDGEMENTS
This work was sponsored by Al-Ghad International Applied Medical Sciences College whom I would like to thank.
REFERENCES
- Koshak, Y.H. (1983) Zamzam. Dar Alelm for Publications, Jeddah.
- Mashat, B.H. (2010) The microbiological quality of sabil free) drinking water in Makkah Al-Mukarramah. JKAU: Met. Env. & Arid Land Agric. Sci., 21, 87-100.
- Al-Zuhair, A., Al-Ghamdi, H. and Noorwali, M. (2005) Analytical report of Zamzam water during the Ramadan and Hajj seasons 1425H. The Institute of the Custodian of the Two Holy Mosques for Al Hajj Research Centre, Om Al Qura University, Makkah.
- Parmar and Malvinder, S. (2004) Kidney stones. British Medical Journal, 328, 1420-1424. doi:10.1136/bmj.328.7453.1420
- Risal, S. (2006) Spectrum of stones composition. 2nd Edition, New York.
- Macaluso, J.N. (1996) Management of stone diseasebearing the burden. The Journal of Urology, 156, 1579- 1580. doi:10.1016/S0022-5347(01)65452-1
- Trinchieri, A., Mandressi, A., Luongo, P., Longo, G. and Pisani, P. (1991) The influence of diet on urinary risk factors for stones in healthy subjects and idiopathic renal calcium stone formers. British Journal of Urology, 67, 230-236. doi:10.1111/j.1464-410X.1991.tb15124.x
- Jaime, U., Oh, M.S. and Carroll, H.J. (1989) Diagnosis and treatment: The first kidney stone. Annals of Internal Medicine, 15, 1006-1009.
- Kumar, S., Sigmon, D., Miller, T., Carpenter, B., Khan, S., Malhotra, R., Scheid, C. and Menon, M. (1991) A new model of nephrolithiasis involving tubular dysfunction/ injury. Journal of Urology, 146, 1384-1389.
- Desmars, J.F. and Tawashi, R. (1973) Dissolution and growth of calcium oxalate monohydrate. I. Effect of magnesium and pH. Biochimica et Biophysica Acta (BBA), 313, 256-267. doi:10.1016/0304-4165(73)90025-1
- Sai Sathish, R., Ranjit, B., Ganesh, K.M., Nageswara Rao, G. and Janardhana, C. (2008) A quantitative study on the chemical composition of renal stones and their fluoride content. Current Sciences, 94, 104-109.
- Singh, P.P., Barjatiya, M.K, Dhing, S., Bhatnagar, R., Kothari, S. and Dhar, V. (2001) Evidence suggesting that high intake of fluoride provokes nephrolithiasis in tribal populations. Urological Research, 29, 238-244. doi:10.1007/s002400100192


