Journal of Biomedical Science and Engineering
Vol.08 No.12(2015), Article ID:61960,8 pages
10.4236/jbise.2015.812076
Comparative Analysis of Adaptive Vessel Segmentation―Cerebral Arteriovenous Malformation
Yadalam Kiran Kumar1,2*, Shashi Bhushan Mehta1, Manjunath Ramachandra1
1Manipal University, Manipal, India
2Philips Research, Philips Innovation Campus, Bangalore, India

Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).


Received 21 October 2015; accepted 14 December 2015; published 17 December 2015

ABSTRACT
Aim: Neurovascular abnormalities are extremely complex, due to the multitude of factors acting simultaneously on cerebral hemodynamics. Cerebral Arteriovenous Malformation (CAVM) hemodynamic in one of the vascular abnormality condition results changes in the vessels structures and hemodynamics in blood vessels. The challenge is segmenting accurate vessel region to measure hemodynamics of CAVM patients. The clinical procedure is in-vivo method to measure hemodynamics. The catheter-based procedure is difficult, as it is sometimes difficult to reach vessels substructures. Methods: In this paper, we have proposed adaptive vessel segmentation based on threshold technique for CAVM patients. We have compared different adaptive methods for vessel segmentation of CAVM structures. The sub-structures are modeled using lumped model to measure hemodynamics non-invasively. Results: Twenty-three CAVM patients with 150 different vessel locations of DSA datasets were studied as part of the adaptive segmentation. 30 simulated data has been evaluated for more than 150 vessels locations for sub-segmentation of vessels. The segmentation results are evaluated with accuracy of 93%. The computed p-value is smaller than the significance level 0.05. Conclusion: The adaptive segmentation using threshold based produces accurate vessel segmentation, results in better accuracy of hemodynamic measurements for DSA images for CAVM patients. The proposed adaptive segmentation helps clinicians to measure hemodynamic non-invasively for the segmented sub-structures of vessels.
Keywords:
Adaptive Segmentation, AVM, Lumped Model

1. Introduction
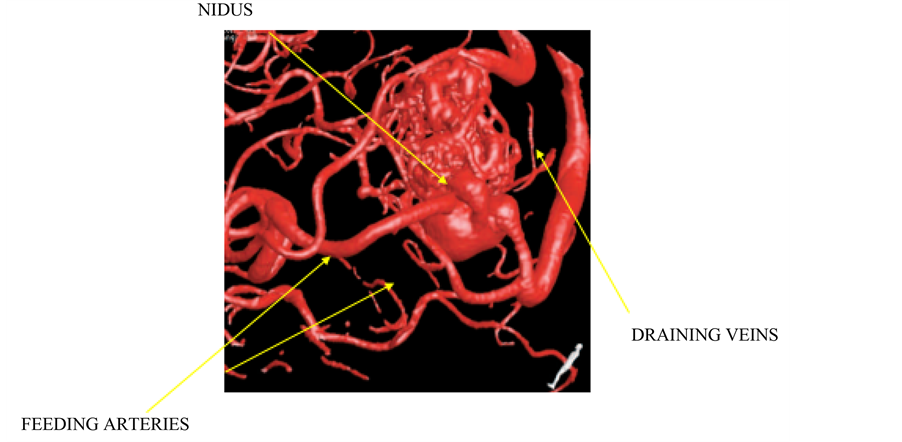
Cerebral Arteriovenous Malformation (CAVM) is one of the neurovascular disease conditions, which changes the cerebral angioarchitecture and hemodynamics changes in the flow and pressure level in blood vessels. CAVM vessels are made of tangled abnormal vessels, which form a complex vessels structure called Nidus. The invasive procedure to measure hemodynamics near Nidus is risky. Figure 1 shows the CAVM complex structure. The studies show that various phase of acquisition of DSA images is used for analysis of vessel segmentation in cerebrovascular patients [1] , but limited to complex structures. The literature shows the recursive tracking techniques to detect the vessel network, which has limitation [2] such as handling of structural variations. The author Nong Sang [3] studied the vessel segmentation of DSA image. However, drawback of his study is thresholding method for non-overlapping sub images is not considered.
In the present study, we have used adaptive threshold based segmentation technique to sub-segment various structures of vessels using DSA images of CAVM. We propose a novel approach for adaptive methodology to segment each variation of vessels. The hemodynamics measurements are modeled for various threshold techniques of segmentation.
2. Methodology
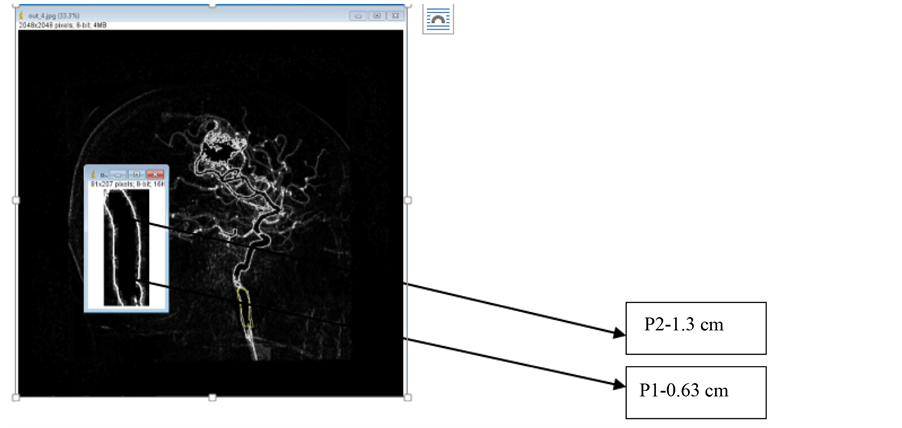
This section describes method for segmenting the Digital Subtraction Angiogram (DSA) images. The data is obtained from KMC Manipal. The input image is preprocessed using Gaussian noise and Hessian matrix based filtering is applied to angiogram images, to detect the tubular structures of vessels using eigenvalues [5] . Figure 2 shows adaptive segmentation technique methodology. The preprocessed image is segmented using OTSU segmentation to create an initial vessel segment as shown in Figure 3. The length and diameter of vessel is calculated using the ROI tools on the vessel segment [6] [7] . The diameter of the initial part of segmented region set as reference node-P1. The diameter is calculated for every small change of the vessel. The sub segment of vessel is created, whenever there is an increase of 10% of diameter of newer segment more than reference diameter, which is shown as segment-P2 as shown in Figure 4. The entire vessel is segmented into smaller sub- structures for variation in diameter with comparison to reference diameter. This is repeated for entire vessel structure. This leads to different combinations of sub-segments of vessels.
The adaptive segmentation technique is modified by changing threshold to 5%. The number of sub segmentation results of vessels is more than threshold by 10%, because diameter variation for angiogram images is more than other modalities. The segmented images are modeled using lumped model to measure hemodynamic parameters such as cerebral pressure, cerebral flow, and cerebral velocity [8] [9] . These parameters are recorded non-invasively, which is used by clinicians for diagnosis.
Figure 1. Cerebral arteriovenous malformation (CAVM). Source: KMC Manipal [4] .
Figure 2. Adaptive segmentation technique.
Figure 3. ROI segmentation image.
Figure 4. Vessel diameter measurement. Source: KMC manipal [10] .
3. Results
Twenty-three CAVM patients obtained from Cathlab, Kasturba Medical College (KMC) Manipal from the study population. The study received ethical clearance from KMC Manipal and written informed consent was obtained from all individuals prior to enrollment in the study. All clinical investigations have been performed according to the principles expressed in the declaration of KMC Manipal.
The adaptive segmentation of threshold based adaptive segmentation for CAVM patients is implemented using MATLAB. Figure 5 shows various segment sub-division based on the threshold factor. The adaptive segmentation using 5% produces more sub-segment and with more accurate in hemodynamics measurements. Twenty-three CAVM patients with 150 different vessel location of DSA datasets were studied as part of the adaptive modeling and 30 simulated data are created with equivalent complexity of DSA, has been evaluated for more than 150 vessels locations for sub-segmentation of vessels.
3.1. Evaluation of Segmentation Results
The number of segments created for the threshold based 10% segmentation is lesser than 5% segmentation method, because vessel structure has variation for every 5% diameter. Table 1 shows comparison analysis between the adaptive segmentation results of DSA image. The result shows that accuracy is improved by nearly 50% with our study. Each sub-segment is modeled independently to measure hemodynamics parameters. The segmented results were evaluated using performance indices such as accuracy, texture-based and shape-based measures [11] . The accuracy measure determines how different the threshold based segmented image from reference image. In our study, we took threshold of 10% segmented output is considered as reference.
The vesselness response function V(x,y;σ) for segmented images is obtained using following Equation (1), where σ is the scalar function, T represents transformation function [12] . The maximum vesselness response is obtained using different ranges for minimum and maximum scales. The ranges used are the weighted scales for our analysis.
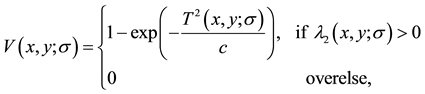 (1)
(1)
3.2. Statistical Analysis
The analysis of segmented results is compared for various statistical parameters such as entropy, standard deviation, mean, orientation, circularity and solidity. Table 2 shows comparative analysis between threshold methods that highlight segmentation accuracy of both proposed adaptive threshold methods. All statistical analyzes were performed using SPSS for Windows (SPSS Inc., Chicago), version 17 [13] . The segmentation results using 5% are evaluated with accuracy of 93%, using hemodynamics measurements and computed p-value is smaller than the significance level 0.05.
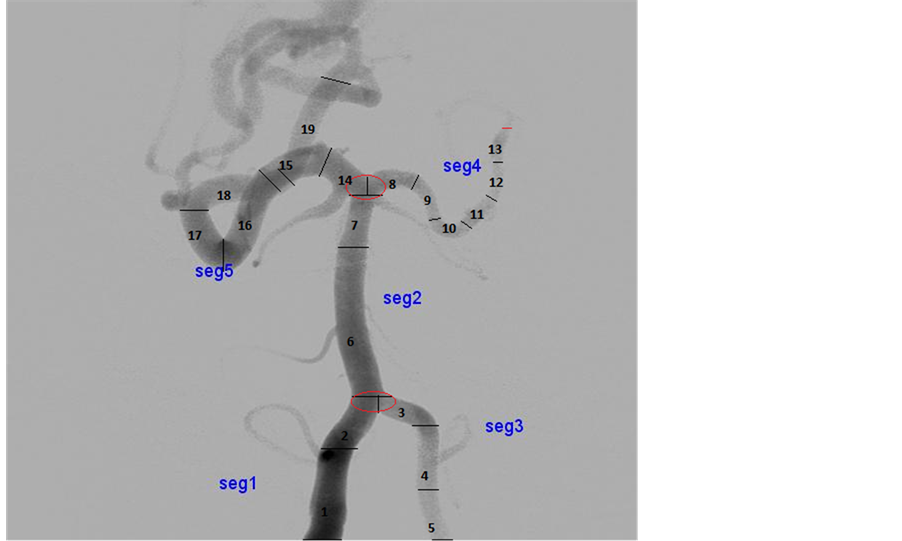
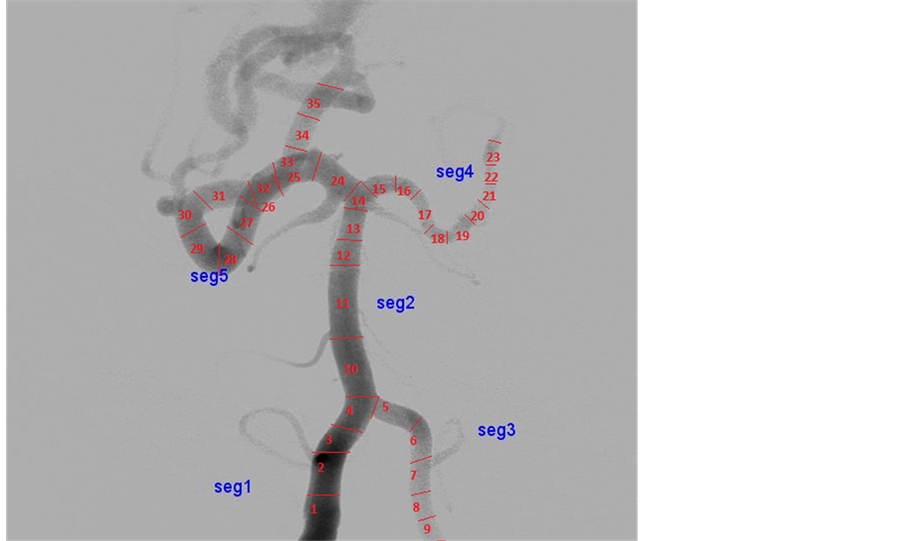
Figure 5. Adaptive threshold―10 and 5 percentage vessels segments. Source: KMC Manipal.
Table 1. Pressure measurement for each segment.
Table 2. Comparative analysis of segmentation techniques.
4. Discussion
The clinical interest in 2D & 3D vascular segmentation creates research interests to biomedical community. In this study, a novel method to sub-segment DSA images of CAVM using adaptive segmentation is presented. The method is based on threshold-based techniques. The use of a priori knowledge on the vessel geometry and structures of the angiogram is important in determining robustness and accuracy of segmentation. The clinical angiographic applications requires combinations of manual and automatic segmentation for analysis, such as our proposed approach of threshold based segmentation, helps clinicians to use segmented image for diagnosis or therapeutic decision. The previous study shows that segmentation of angiograms by Lorenz et al. (2003), analyzed three level of segmentation, but limited to low-level appearance hypotheses [14] . The study by Socher et al., (2008), using a hierarchical marginal space paradigm, trained by vessel positions, widths and lengths is limited by performance [15] . The research studies shows [16] [17] ; achieve very good specificity but not competitive sensitivity and lies far below the ROC curve of the proposed method.
5. Conclusion
The adaptive segmentation using threshold based produces accurate vessel segmentation. This results in better accuracy of hemodynamic measurements of DSA images for CAVM patients. The proposed adaptive segmentation helps clinicians to measure hemodynamic non-invasively for the segmented sub-structures of vessels. The proposed segmentation and non-invasive measurement address the clinical problem, where clinicians find difficult to insert catheter into sub-structures, to measure hemodynamics invasively. Twenty-three CAVM patients with 150 different vessel locations of DSA data sets were studied as part of the adaptive segmentation. 30 simulated data has been evaluated for more than 150 vessels locations for sub-segmentation of vessels. The results are evaluated with accuracy of 93%, and computed p-value is smaller than the significance level 0.05.
Cite this paper
Yadalam KiranKumar,Shashi BhushanMehta,ManjunathRamachandra, (2015) Comparative Analysis of Adaptive Vessel Segmentation—Cerebral Arteriovenous Malformation. Journal of Biomedical Science and Engineering,08,797-804. doi: 10.4236/jbise.2015.812076
References
- 1. Babin, D., et al. (2013) Brain Blood Vessel Segmentation Using Line-Shaped Profiles. Physics in Medicine and Biology, 58, 8041-8061.
http://dx.doi.org/10.1088/0031-9155/58/22/8041 - 2. Liu, I., et al. (1993) Recursive Tracking of Vascular Networks in Angiograms Based on the Detection-Deletion Scheme. IEEE Transactions on Medical Imaging, 12, 334-341.
http://dx.doi.org/10.1109/42.232264 - 3. Sang, N., et al. (2007) Knowledge Based Adaptive Thresholding Segmentaion of Digital Subtraction Angiography Images. Image and Vision Computing, 25, 1263-1270.
http://dx.doi.org/10.1016/j.imavis.2006.07.026 - 4. Kumar, Y.K., Mehta, S.B. and Ramachandra, M. (2014) Vascular Segmentation of Cerebral AVM. AIR, 2, 52-57.
http://dx.doi.org/10.9734/AIR/2014/7044 - 5. Yang, X.L., et al. (2012) An Improved Median-Based Otsu Image Thresholding Algorithm. AASRI Procedia, 3, 468-473.
http://dx.doi.org/10.1016/j.aasri.2012.11.074 - 6. Kumar, Y.K., Mehta, S. and Ramachandra, M. (2013) Review Paper: Cerebral Arteriovenous Malformations Modelling. International Journal of Scientific and Engineering Research, 4, 129-139.
- 7. Tsai, C.-M., et al. (2015) Identifying Regions of Interest in Reading an Image. Displays, 39, 33-41.
http://dx.doi.org/10.1016/j.displa.2015.08.001 - 8. Guglielmi, G. (2006) Electrical Models in the Analysis of Hemodynamic Characteristics of Arteriovenous Malformations. Interventional Neuroradiology, 12, 9-15.
- 9. Kumar, Y.K., Mehta, S. and Ramachandra, M. (2014) Multimodality Vessel Modelling Analysis for Cerebral Arteriovenous Malformation. Journal of Behavioral and Brain Science, 2, 23-26.
http://dx.doi.org/10.4236/jbbs.2014.41003 - 10. Yu, S., et al. (2012) Noncontrast Dynamic MRA in Intracranial Arteriovenous Malformation (AVM): Comparison with Time of Flight (TOF) and Digital Subtraction Angiography (DSA). Magnetic Resonance Imaging, 30, 869-877.
http://dx.doi.org/10.1016/j.mri.2012.02.027 - 11. Betanzosa, A., Varelaa, A. and Martinez, C. (2000) Analysis and Evolution of Hard and Fuzzy Clustering Segmentation Techniques in Burned Patient Images. Image and Vision Computing, 18, 1045.
http://dx.doi.org/10.1016/S0262-8856(00)00045-7 - 12. Tsai, Y.-C., et al. (2015) Automatic Segmentation of Vessels from Angiogram Sequences Using Adaptive Feature Transformation. Computers in Biology and Medicine, 62, 239-253.
http://dx.doi.org/10.1016/j.compbiomed.2015.04.029 - 13. Fic, A.M., Ingham, D.B., Ginalski, M.K., Nowak, A.J. and Wrobel, L.C. (2014) Modelling and Optimisation of the Operation of a Radiant Warmer. Medical Engineering & Physics, 36, 81-87.
http://dx.doi.org/10.1016/j.medengphy.2013.10.003 - 14. Lorenz, C., Carlsen, I., Buzug, T., Fassnacht, C. and Weese, J. (1997) Multi-Scale Line Segmentation with Automatic Estimation of Width, Contrast and Tangential Direction in 2D and 3D Medical Images. Lecture Notes in Computer Science, 1205, 233-242.
http://dx.doi.org/10.1007/BFb0029242 - 15. Socher, R., Barbu, A. and Comaniciu, D. (2008) A Learning Based Hierarchical Model for Vessel Segmentation. 5th IEEE International Symposium on Biomedical Imaging: From Nano to Macro, Paris, 14-17 May 2008, 1055-1058.
http://dx.doi.org/10.1109/isbi.2008.4541181 - 16. Chaudhuri, S., Chatterjee, S., Katz, N., Nelson, M. and Goldbaum, M. (1989) Detection of Blood Vessels in Retinal Images Using Two-Dimensional Matched Filters. IEEE Transactions on Medical Imaging, 8, 263-269.
http://dx.doi.org/10.1109/42.34715 - 17. Martínez-Pérez, M., Hughes, A., Stanton, A., Thom, S., Bharath, A. and Parker, K. (1999) Scale-Space Analysis for the Characterisation of Retinal Blood Vessels. In: Taylor, C. and Colchester, A., Eds., Medical Image Computing and Computer-Assisted Intervention—MICCAI’99, 90-97.
NOTES
*Corresponding author.