Journal of Biomedical Science and Engineering
Vol.5 No.2(2012), Article ID:17616,4 pages DOI:10.4236/jbise.2012.52006
Synthesis and in vitro antibacterial evaluation of some thiosemicarbazides and thiosemicarbazones
![]()
1Faculty of Pharmacy, Islamic Azad University of Pharmaceutical Sciences, Tehran, Iran
2Agricultural, Medical and Industrial Research School, Nuclear Science and Technology Research Institute (AMIRS-NSTRI), Karaj, Iran
Email: *ajalili@aeoi.org.ir
Received 14 May 2010; revised 25 July 2010; accepted 20 September 2010
Keywords: Synthesis; Antibacterial; Thiosemicarbazides; Thiosemicarbazones
ABSTRACT
In the present study various thiosemicarbazides and thiosemicarbazones were synthesized and screened for their antimicrobial activity. Among the ligands tested, compounds 1, 3, 4 and 5 demonstrated inhibitory effects at 0.4 - 0.5 µM concentrations against E. coli while 1-(4-fluorobenzoyl)-N4-ethyl-thiosemicar-bazide (3) demonstrated the maximum inhibitory effective compound against E. coli, also possessing high water solubility. Instead, compound 3 demonstrated weak antibiotic effects against S. aurous while compounds 4 - 6 demonstrated significant antibacterial effects at 0.4 - 0.42 µM against the latter organism. Chloramphenicol was used as the positive control.
1. INTRODUCTION
The rising incidence of bacterial infections, along with the emergence of resistance to conventionally-utilized antibiotics [1] has added considerable urgency to the pursuit of safe and effective therapies in the past decade. In this respect, the search for new antibacterial agents which are bacteriocidal, have a broad-spectrum of activeity, and have fewer side effects has become critical. Traditionally, small molecules have been a reliable source for discovering novel biologically active compounds. Compared to their natural counterparts with complex structures, these molecules are easily synthesized and their smooth structural optimization would usually lead to a feasible candidate compound. Through utilization of combinatorial chemistry large libraries of small molecules have been generated and screened for specific biological activities.
We have previously reported the synthesis and antimicrobial activity of a series of substituted-thiazolo-1,3,4- thiadiazole, -1,3,4-oxadiazole, and -1,2,4-triazoles [2] and also cytotoxicity and antifungal effects of 1,2,3-thiadiazolo-4H-1,2,4-triazoles [3].
Several small molecule triazole-, thiadiazole-, and oxadiazole-based heterocycles have also been reported to possess potential bioactivity, including antimicrobial, anticonvulsant, antiallergy, etc. [4-7]. In continuation of our current research on the synthesis and biological evaluation of small bioactive heterocyclic molecules series of aryl containing thiosemicarbazides and thiosemicarbazones were synthesized and their antibacterial properties were determined. Figures 1 and 2 demonstrate the structures of the tested compounds in this work.
In order to study the antibacterial activity of these ligands, we performed growth inhibition test on two moicro-organisms. The bioassay was based on the ability to inhibit the liquid culture dilution series in presence of various concentrations of the ligands.
2. MATERIALS AND METHODS
The starting materials were purchased from Aldrich Chemical Company. Proton NMR spectra were obtained on a Brucker FT-80 (80 MHz) or a Varian Unity plus (400 MHz) instrument with tetramethylsilane as internal standard. Mass spectra were recorded by a Finnigan Mat TSQ-70 Spectrometer. All the chemicals were recrystallized repeatedly before use. Infrared spectra were taken on a Perkin-Elmer 781 spectrometer (KBr disks). Thinlayer chromatography (TLC) of products was run on silica gel polymer-backed (F 1500/LS 254, 20 × 20 cm, TLC Ready Foils Schleicher & Schuell) or glass plates (25 × 35 cm, E-Merck). Dimethyl sulfoxide (DMSO) used for labeling experiments were of “Sure-Seal” grade (Aldrich). Analytical HPLC to determine the purity of ligands was performed by a Shimadzu LC-10AT, armed with UV-visible (Shimadzu) using Si Kromasil 100, 5 mm 250 × 4.6 mm (M & W), Inchrom. A mixture of acetonitrile-chloroform (65:35) was used as eluent at a flow rate of 2 mL/min (Rt = 7.5 min). Melting points were
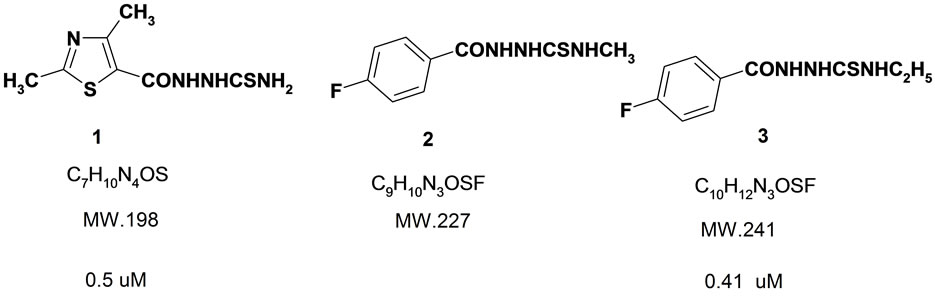
Figure 1. Structural formulas of thiosemicarbazides evaluated for antimicrobial effects.
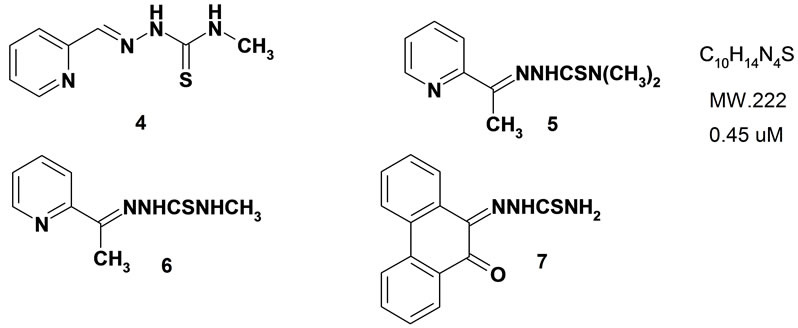
Figure 2. Structural formulas of thiosemicarbazones evaluated for antimicrobial effects.
determined on a Reichert-Jung hot stage microscope and are uncorrected. Elemental microanalyses were within ±0.4% of theoretical values for C, H and N. All stock solutions were passed through a 0.22 micron biological filter, Waters.
2.1. Preparation of Aryl-Carboxylic Acid Hydrazides
Aryl-carboxylic acid hydrazides were prepared according to literature [8]. To a stirred mixture of ethyl aryl-carboxylate (0.05 mole) in ethanol (100 mL), hydrazine hydrate (100 mL) was added dropwise. The reaction mixture was warmed to 50˚C for 2 h with vigorous stirring. The reaction was checked with TLC until completion. The mixture was concentrated at reduced pressure to give a yellow solid which was crystallized in ethanol to give the product [5].
2.2. 1-(Aroyl-5-carboxyl)-4-substitutedthiosemicarbazides (1-3)
Different thiosemicarbazides were prepared according to the method reported previously [9]. A sodium hydroxide solution (10 mL, 1 M) was added dropwise to a stirred mixture of aryl hydrazides (0.01 mole) and ethanol (10 mL). The stirring was continued until a transparent winecolored solution was obtained, the related isothiocyanate (0.01 mole) was then added to above solution in one portion and the mixture was stirred and the progress of the reactions were controlled using TLC. The reaction was cooled to room temperature, filtered, and the filtrate was acidified (pH 5) by the addition of hydrochloric acid solution (1 M). The precipitate was filtered and washed with water and dried to give the related thiosemicarbazides (1- 3), 1-(2,4-dimethylthiazole-5-carboxyl)-N4-ethylthiosemicarbazide (1), 1-(4-fluorobenzoyl)-N4-methylthiosemicarbazide (2), 1-(4-fluorobenzoyl)-N4-ethylthiosemicarbazide (3).
2.2.1. Preparation of Aryl Thiosemicarbazones
These compounds were prepared with slight modifications to the methods of [10]. A mixture of appropriate thiosemicarbazide (2 mmol) in acetic acid solution (5%, prepared with 99% AcOH and MilliQ-H2O) was heated at 50˚C with stirring until a transparent solution was formed. Then freshly distilled carbonyl compound (2 mmol) diluted (1:3) with 5% acetic acid was added drop wise to the mixture during 5 min under a blanket of N2. The mixture was stirred for 3 - 4 h at 50˚C. The hot reaction mixture was filtered off through two layers of Whatman No. 2 filter paper. The filtered mass was washed with MilliQ-H2O (50 mL), rectified ethanol (25 mL) and finally heated in a vacuum oven overnight at 75˚C. The dried powder was refluxed in 80% acetic acid (prepared with MilliQ-H2O) for 2 h. The hot mixture was filtered and the precipitate was washed with MilliQ-H2O (50 mL), rectified ethanol (25 mL) and heated at a vacuum oven overnight at 75˚C. The fine white powder was kept overnight in vacuum. Alternatively the powder can be crystallized from hot ethanol to give a brilliant white powder. 2-Pyridine-aldehyde-4-N-methylthiosemicar-bazone (4), 2-Acetyl pyridine 4-N,N’-dimethylthio-semicarbazone (5): (60%) m.p. 275˚C. 1H NMR (D6-DMSO) d (ppm) 10.28 (bs, 1H, NH-N2), 8.58 - 8.36 (m, 3H, H3&H6 pyridine & NH-N4), 8.1 (bs, 1H, NH-N4), 7.88 - 7.67 (hexlet, 1H, H5 pyridine), 7.43 - 7.29 (m, 1H, H4 pyridine), 2.39 (s, 3H, CH3-C=N). IR (CHCl3) l max 3208, 3132 (N-H), 1470 (C=N), 1160 (C=S). Mass (electrospray) 194.1 (14%), 172(4), 157.1(76), 130(65), Elemental analysis for C8H10N4S, calcd C, 49.46, H, 5.19, N, 28.84; found C, 49.41, H, 5.21, N, 28.91. 2-Acetyl pyridine 4-N-methylthiosemicarbazone (6). 9,10-Phenanthrenequinone thiosemicarbazone (7): IR v max: 3409 (N-H), 3243 (N-H), 3143 (N-H), 1708 (C=O), 1598, 1494 (C=C), 1427 (C=N), 1273 (C=S), 1223, 1169, 1013, 761, 1HNMR (d6-DMSO) 14.37 (s, 1H, NH, H9), 9.36 (s, 1H, NH, H10 or11), 9.05 (s, 1H, NH, H10 or 11), 8.65 (d, 1H, aromatic, H1), 8.63 (d, 1H, aromatic, H8), 8.37 (d, 1H, aromatic, H2), 8.27 (d, 1H, aromatic, H7), 8.22 (d, 1H, aromatic, H4), 8.20 (d, 1H, aromatic, H5), 7.80 (m, 1H, aromatic, H3), 7.54 (m, 2H, aromatic, H6). Mass (EI), 281 (M+).
2.2.2. Preparation of Samples
The chemicals were dried in oven overnight under vacue and 1 mg/mL stock solutions in DMSO were prepared for the preparation of the stock solutions and were filtered through the Millex filters (0.22 micron) and kept in fridge and darkness.
2.2.3. Biological in Vitro Tests-Antibacterial Tests-Agar Dilution
Microorganisms with specific ATCC numbers were purchased from Razi Institute, Karaj, Iran. Staphylococos aureos and Escerichia coli were cultured in their specific media (Tryptic Soy Broth and Lactose Broth respectively) at 37˚C for 15 hours. These cultures were used for innocolation of the test tubes conataining the synthesized compounds. The method used for this test was adopted from Muanza et al. [11] and Mitscher et al. [12] with minor modifications. For each compound, a test tube containing specific media (8 mL) and the test compound stock solution (1 mL) was prepared. A positive control tube containing the media (8 mL) and innoculant (1 mL) and DMSO (1 mL) was also prepared. As the negative control, chloramphenical alcoholic stock solution (200 ul, 1 mg/mL) was added to a test tube containing media (8 mL) and inoculants (1 mL). The mixture of the inoculated media containing compound (9 mL in each tube) were serially diluted in 7 more tubes by the addition of 1 mL of first tube and specific media (8 mL) and finally to each tube, inoculants bacteria (1 mL) was added, while all tubes were shaken for 30 seconds before each dilution and/or inoculants addition. The transparency of all test tubes was checked carefully before incubation. The tube caps were sealed by Parafilm and kept at 37˚C in incubator overnight. After the incubation the tubes were checked for the minimum concentration tube with no M.O. growth. The positive and negative controls were also observed for the accuracy of the tests. The tests were done in triplicates, and the positive antibacterial results were read based on no growth compared to solvent control. Recommendations of NCCLS (1992) were used for broth dilution to measure the minimum inhibitory concentration (MIC) values [13].
3. RESULTS
The synthesis of the titled compounds was performed according to the common chemical processes already. The compounds are synthesized according to the published procedure and their structure are presented in Figures 1 and 2. All the compounds were recrystalized and dehydrated according to the procedures and carefully weighed and the stock solutions were prepared under sterile conditions.
4. DISCUSSION
In order to obtain novel chemotherapeutic heterocyclic cores, we synthesized new thiazolo-, 4-fluorophenylthisemicarbazides and some thiosemicarbazones that showed antibacterial activities on some pathogenic gram positive/ gram negative organisms.
Among thiosemicarbazides tested on E. coli, compounds 1 and 3 demonstrated inhibitory effects at 0.5 and 0.4 uM concentrations. Although compound 2 and 3 vary jut in CH2 moiety their antibacterial effects in E. coli is significant. Compound 3 is the most lipophilic compound in the group and it can influence the penetration through lipophilic LPS gram negative organisms such as E. coli. Compounds 4, 5 demonstrated antibacterial effects on both E. coli and S. auroes samples at 0.l mg/ml. Also compounds 1 and 3 demonstrated specific antibacterial effects on E. coli at 0.1 mg/mL concentrations (Table 1).
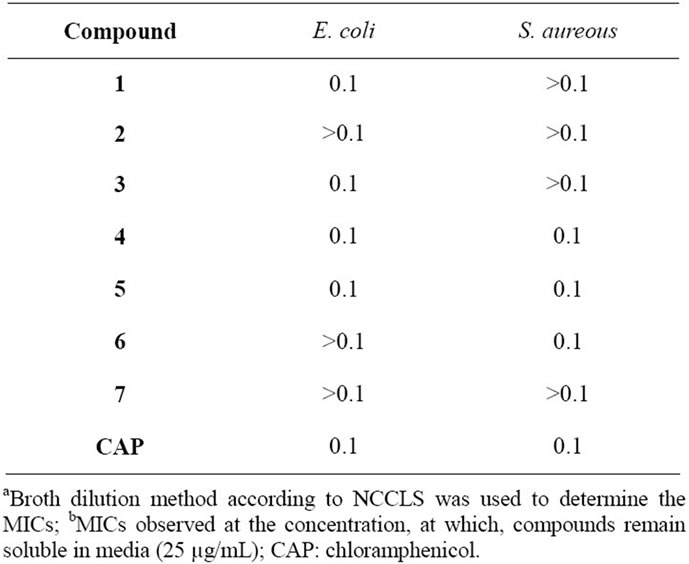
Table 1. Minimum inhibitory concentration (MIC) (mg/mL) of different compounds against selected bacterial species.
Compounds 4 and 5 both contain acetyl pyridine moiety that is mostly incorporated in vitamin B6 cycle and is considered an enzyme inhibitor in pyridoxal cycle. Considering this hypothesis the antibacterial effect in both gram positive and negative strains is explainable. These compounds may also have cytotoxic effects on mammary cells as well. Many antibiotic effects of the thiosemiarbazones have already been explained and there are proposed mechanisms such as DNA synthesis enzymes and ribonuclease reductase inhibition already reported, in complex form with metals and/or in free ligand forms.
![]()
![]()
REFERENCES
- Barchiesi, F., Arzeni, D., Fothergill, A.W., Di Francesco, L.F., Caselli, F., Rinaldi, M.G. and Scalise, G. (1999) In vitro activities of the new antifungal triazole SCH 56592 against common and emerging yeast pathogens. Antimicrob Agents Chemother, 44, 226-229. doi:10.1128/AAC.44.1.226-229.2000
- Shafiee, A., Jalilian, A.R. and Tabatabaiee-Yazdi, M. (1998) Synthesses, antibacterial and antifungal activities of substituted-thiazolo-1,3,4-thiadiazoles, oxadiazoles and 1, 2,4-triazoles. Iranian Journal of Chemistry & Chemical Engineering, 17, 14-20.
- Jalilian, A.R., Sattari, S., Bineshmarvasti, M., Shafiee, A. and Daneshtalab M. (2000) Synthesis and in vitro antifungal and cytotoxicity evaluation of thiazolo-4H-1,2,4- triazoles and 1,2,3-thiadiazolo-4H-1,2,4-triazoles. Arch Pharm (Weinheim), 333, 347-354. doi:10.1002/1521-4184(200010)333:10<347::AID-ARDP347>3.0.CO;2-6
- Jalilian, A.R., Sattari, S., Bineshmarvasti, M. and Daneshtalab, M. (2003) A shafiee, synthesis and in vitro antifungal and cytotoxicity evaluation of substituted 4,5-dihydronaphtho[1,2-d][1,2,3]thia(or selena)diazoles. Il Farmaco, 58, 63-68.
- Shafiee, A.R. and Jalilian, B. (2000) Rezaei, selenium heterocycles XLIV. Syntheses of 8,9-dihydro-1,2,3-thiadiazolo[4,5-a]-4,7-dihydronaphthalene and 1,2,3-selenadiazolo[4,5-a]-4,7-dimethoxynaphthalene. Journal of Heterocyclic Chemistry, 37, 1325-1327. doi:10.1002/jhet.5570370551
- Varvaresou, T., Siatra-Papastaikoudi, A., Tsotinis, D., Tsantili-Kakoulidou, A. and Amvakides, V.A. (1998) Synthesis, lipophilicity and biological evaluation of indole-containing derivatives of 1,3,4-thiadiazole and 1,2,4-triazole. Farmaco, 53, 320-326.
- Bineshmarvasti, M., Sharifzadeh, M., Jalilian, A.R., Soltaninejad, K. and Shafiee, A. (2003) Syntheses and anticonvulsant activity of N4-substituted triazolylthiazoles. Daru, 11, 74-78.
- Boon, W.R. (1945) The action of thionyl chloride on 2: 4-dimethylthiazole-5-carboxylic acid. Journal of the Chemical Society, 162, 601-603. doi:10.1039/jr9450000601
- Kane, J.M., Staeger, M.A., Dalton, C.R., Miller, F.P., Dudley, M.W., Ogden, A.M., Kehne, J.H., Ketteler, H.J., McCloskey, T.C. and Senyah, Y. (1994) 5-Aryl-3-(alkylthio)-4H-1,2,4-triazoles as selective antagonists of strychnine-induced convulsions and potential antispastic agents. Journal of Medicinal Chemistry, 37, 125-132. doi:10.1021/jm00027a015
- Gingras, B.A., Suprunchuk, T. and Bayley, C.H. (1962) The preparation of some thiosemicarbazones and their copper complexes, Part III. Canadian Journal of Chemistry, 40, 1053-1057. doi:10.1139/v62-161
- Muanza, D.N., Kim, B.W., Eluer, K.L. and Williams, L. (1994) Antibacterial and antifungal activities of nine medicinal plants of Zaire. International Journal of Pharmacology, 32, 337-345.
- Mitscher, L., Lue, R., Bathala, M.S., Wu, W. and Beal, J.L. (1972) Antimicrobial agents from higher plants. I. Introduction, rational and methodology. Lloydia, 35, 157- 161.
- National Committee for Clinical Laboratory Standards (1992) Reference method for broth dilution antifungal susceptibility testing for yeasts. Proposed Standards, Document M27-P, Villanova.
NOTES
*Corresponding author.

