Materials Sciences and Applications
Vol.06 No.07(2015), Article ID:57791,12 pages
10.4236/msa.2015.67063
Inclusion-Interaction Assembly Strategy for Constructing pH/Redox Responsive Micelles for Controlled Release of 6-Mercaptopurine
Mingyuan Tan*, Hua Zheng, Xueqiong Zhang*, Zhiyan Yu, Zhao Ye, Liyan Shi, Kaikai Ding
Department of Pharmaceutical Engineering, School of Chemistry, Chemical Engineering and Life Sciences, Wuhan University of Technology, Wuhan, China
Email: *tanmingyuan@qq.com, *365464583@qq.com
Copyright © 2015 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 15 May 2015; accepted 5 July 2015; published 8 July 2015
ABSTRACT
An inclusion-interaction assembly strategy was used to construct novel pH/redox responsive core-shell micelles with hydrophobic drug as the core and hydrophilic polymer as the shell. At first, a dimer of hydrophobic drug 6-mercaptopurine and a hydrophilic β-CD grafted carboxymethyl chitosan were synthesized. Then, a novel amphiphilic inclusion complex was prepared with the dimer being partially embedded into the cavity of β-CD moiety. It self-assembled into pH/redox responsive core-shell micelles in distilled water. TEM confirmed that the micelles possessed a spherical core-shell configuration with a mean size of about 160 nm. DLS showed that the micelles were stable in aqueous solution. Their particle diameters altered with pH values as well as glutathione (GSH) concentrations and respectively attained a maximum value at pH 6.0 and
Keywords:
Chitosan, Cyclodextrin, 6-Mercaptopurine, pH-Sensitive, Redox-Responsive, Controlled Release

1. Introduction
β-Cyclodextrin (β-CD) is a natural cyclic oligosaccharide composed of 7 glucose units linked by α-1, 4 glycosidic bonds, with a hydrophilic outer surface and a hydrophobic inner cavity [1] - [3] . Due to inclusion interactions, a wide range of hydrophobic drugs can be embedded into the hydrophobic cavities to form inclusion complexes, thus significantly increasing the solubility and permeability of the drugs [4] . Moreover, grafting β-CD onto polymers can result in materials with unique features and excellent biocompatibility [5] . A lot of β-CD containing polymers have been prepared and utilized to build supramolecular systems [6] .
In recent years, our group has prepared a variety of polymeric micelles, in which cores and shells are linked by covalent bonds [7] - [10] . Recently, we developed a novel hydrogen-bonding strategy to build pH-sensitive micelles, using hydrophilic polymer and hydrophobic drug as building blocks [11] . It resulted in a micelle, in which only hydrogen bonds rather than covalent bonds existed between core and shell. Further, it achieved the integration of drug-loading and self-assembly in preparing drug-loaded micelles, avoiding the multiple steps used in the covalent-bond method. This result aroused our interest to construct a novel non-covalently connected micelle, with inclusion interaction serving as driving force.
We assumed that, if a hydrophilic β-CD containing polymer and a dimer of hydrophobic drug could form an amphiphilic inclusion complex, with the dimer being partially embedded into the cavity of β-CD moiety, the complex should be able to self-assemble into a micelle, with dimer as core and the polymer as shell. Meanwhile, it was mentioned that the GSH concentration in human cell (millimolar level) was much higher than that in bloodstream (micromolar level), and that the GSH concentration in tumor cells was at least 4-fold higher than that in normal cells [12] . Therefore, if we chose the dimer that two monomer units were joined by a disulfide bond, it would be able to remain stable in bloodstream, and become easy to cleavage once the micelle had entered into tumor cells, and the controlled release of drug might be achieved.
Based on the above assumptions, dimer of 6-mercaptopurine (DMP) was determined. First, from the chemical structure, DMP was a homodimer consisting of two 6-mercaptopurine (6-MP) monomers joined by a disulfide bond. Second, DMP had good hydrophobility and could form an inclusion complex with β-CD with its molecule being partially embedded into the cavity of β-CD. Third, 6-MP was an anti-cancer drug with low bioavailability (about 16%) as well as short plasma half-time (0.5 - 1.5 h), due to its poor solubility and a free sulfhydryl group easily reacting with the plasma proteins [13] . With the micellization of DMP, these two defects could be avoided.
Carboxymethyl chitosan (CMCS), obtained from carboxymethylation of chitosan, was also determined. On one hand, CMCS was biopolymer with good aqueous solubility, low toxicity and good biocompatibility [14] . On the other hand, it was reported that polymeric micelles with CMCS as shells usually could swell in slightly acid aqueous solution, due to a lot of reversibly ionized groups existing in their structures [10] . Taking account of the slightly acid intracellular environments of tumor cells (lysosome pH 4.5 - 5.0 and endosome pH 5.0 - 6.5) [15] , this result might contribute to exposing the disulfide bonds in the core of the micelle to the GSH in tumor cells.
The aim of this study was to employ an inclusion-interaction assembly strategy to construct a novel pH/redox responsive micelle system for controlled release of 6-MP. At first, a hydrophilic β-CD grafted carboxymethyl chitosan (CMCS-g-β-CD) and a dimer of 6-MP were synthesized. Then, an amphiphilic inclusion complex (CMCS-g-β-CD・DMP) was prepared with the dimer being partially embedded into the cavity of β-CD moiety. It self-assembled into micelles in distilled water. Their structure and morphology were observed by transmission electron microscopy (TEM). The stability, pH-sensitivity and reduction-response were investigated by dynamic light scattering (DLS). Their stimuli-responsive release properties and anti-tumor activity were also studied.
2. Materials and Methods
2.1. Materials
Chitosan (CS, MW: 560 kDa, degree of decetylation: 91%) was purchased from Yuhuan Ocean Biochemical Co., Ltd. (
2.2. Synthesis of CMCS
Chitosan (
2.3. Synthesis of CM β-CD
β-CD (
2.4. Synthesis of DMP
DMP was synthesized following the method mentioned in the literature [16] . 6-Mercaptopurine (
2.5. Synthesis of CMCS-g-β-CD
CM β-CD (
2.6. Preparation of CMCS-g-β-CD・DMP
CMCS-g-β-CD (
2.7. Characterization
The FT-IR spectra of CS, CMCS, β-CD, CM β-CD, DMP, CMCS-g-β-CD, the physical mixture of CMCS- g-β-CD and DMP and CMCS-g-β-CD・DMP were determined by a FT-IR spectrometer (Avator 360, Nicolet, MA, USA) using KBr pellets. The 1H-NMR spectra of CMCS, CMCS-g-β-CD, CMCS-g-β-CD・DMP were determined on a Varian 600 spectrometer (Varian,
2.8. Fabrication and Characterization of CMCS-g-β-CD・DMP Micelles
CMCS-g-β-CD・DMP (
Table 1. Characteristic data of CMCS-g-β-CD・DMP with different DMP contents.
Then, this dispersion was sonicated by a probe type sonifer (JY92-2D, made by Ningbo Xinzhi Bio-tech Co., Ltd) at 60 W for 3 min and a CMCS-g-β-CD・DMP micelle solution was obtained. The micelles prepared from CMCS-g-β-CD・DMP1, CMCS-g-β-CD・DMP2 and CMCS-g-β-CD・DMP3 were named M-1, M-2 and M-3, respectively. Their mean diameters and distributions were determined by DLS using a Nano-ZS3600 (
2.9. Stability Studies of CMCS-g-β-CD・DMP Micelles
The stability study of CMCS-g-β-CD・DMP micelles was performed by DLS. The micelle dry powders (
2.10. pH-Sensitivity and Reduction-Response of CMCS-g-β-CD・DMP Micelles
The pH-sensitivity of the micelles was studied in media at various pH values. The micelle dry powders (
The reduction-response of the micelles was investigated in media containing different GSH concentrations. The micelle dry powders (
2.11. Stimuli-Responsive Release of 6-MP from the Micelles
The stimuli-responsive release of 6-MP from the micelles was investigated in three different release media: 1) pH 7.4 PBS with 10 μM GSH; 2) pH 7.4 PBS with
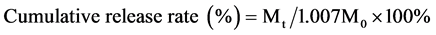 (1)
(1)
where Mt refers to the amount of 6-MP released at time t and M0 refers to the total amount of DMP in micelles.
2.12. Anti-Tumor Activity of CMCS-g-β-CD・DMP Micelles
MTT assay was employed to study the anti-tumor activity of CMCS-g-β-CD・DMP micelles. HeLa cells were seeded onto a 96-well plate in DMEM containing 10% FBS with a density of 1 × 104 cells/well. The cells were incubated at 37˚C for 1 day and exposed to 20 μL dispersions of micelles, and 20 μL dispersions of 6-MP and CMCS-g-β-CD with equivalent concentration. A 20 μL buffer was also added and used as a control. The cells were incubated for another 48 h. Then, 10 μL MTT solutions (5 mg/mL) were added into each well. After the cells were incubated for 4 h, 10 μL DMSO was added to each well to dissolve the formazan crystals. Finally, the optical densities (OD) were observed by a microplate reader at 570 nm. The inhibition ratios were calculated using following equation:
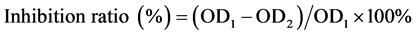 (2)
(2)
where OD1 refers to optical density of the control and OD2 refers to the optical densities of the cells exposed to the micelles, 6-MP and CMCS-g-β-CD.
3. Results and Discussion
3.1. Preparation and Structural Analysis of CMCS-g-β-CD・DMP
The synthetic route of CMCS-g-β-CD・DMP was shown in Figure 1. Firstly, CS and β-CD were modified with
Figure 1. Synthetic route and assembly process of CMCS-g-β-CD・DMP.
chloroacetic acid to obtain CMCS and CM β-CD. Then, CMCS-g-β-CD was obtained via the formation of amide bonds between the carboxyl groups of CM β-CD and the amino groups of CMCS. At last, CMCS-g- β-CD DMP was synthesized with DMP being partially embedding into the cavity of β-CD moiety.
The FT-IR spectra of CS, CMCS, β-CD, CM β-CD, DMP, CMCS-g-β-CD, the physical mixture of DMP and CMCS-g-β-CD (DMP content 9.37 wt%), 6-MP and CMCS-g-β-CD・DMP2 were shown in Figure 2. Compared with the spectrum of CS, the new characteristic bands at 1596 cm−1 and
The DSC data shown in Figure 3 further supported the result. In the DSC profile of CMCS-g-β-CD, a broad endothermic peak could be observed, which was related to water evaporation. However, in the profile of the inclusion complex, the peak that represented water loses was vastly reduced in intensity and shifted to a lower temperature. This result suggested that a structure of lower water capacity was formed, because of the extrusion of water molecules from β-CD cavities, resulting from DMP being embedded into β-CD cavities [17] .
The 1H-NMR spectra of CMCS, CMCS-g-β-CD and CMCS-g-β-CD・DMP in D2O were shown in Figure 4. Compared with curve a, the new signals in the region of 5.0 - 5.2 ppm in curve b could be ascribed to the existence of D-glucopyranosyl residues of β-CD, suggesting that β-CD was successfully grafted onto CMCS. The signals at 3.86 - 3.90 ppm and 3.61 - 3.64 ppm in curve b were respectively assigned to two protons positioned
Figure 2. (A) FT-IR spectra of (a) CS, (b) CMCS, (c) β-CD and (d) CM β-CD; (B) FT-IR spectra of (e) CMCS-g-β-CD, (f) 6-MP, (g) DMP, (h) physical mixture of CMCS-g-β-CD and DMP (DMP content 9.37 wt%) and (i) CMCS-g-β-CD・DMP2.
Figure 3. DSC curves of (A) CMCS-g-β-CD・DMP2 and (B) CMCS-g-β-CD.
Figure 4. (A) 1H-NMR spectra in D2O of (a) CMCS, (b) CMCS-g-β-CD and (c) CMCS-g-β-CD・DMP; (B) Enlarge figures of the regions of (d) and (e).
inside the β-CD cavity, namely H
3.2. Fabrication and Characterization of CMCS-g-β-CD・DMP Micelles
The fabrication procedures of the core-shell micelles were illustrated in Figure 1. Amphiphilic CMCS-g-β- CD・DMP was firstly synthesized and then underwent self-assembly to form a core-shell micelle. The TEM image of M
With DMP being partially embedded into the cavity of β-CD moiety, the non-covalently connected complex CMCS-g-β-CD・DMP obtained an amphiphilic macromolecule composed of a hydrophilic polymer backbone and hydrophobic DMP moieties that were located outside β-CD. After it dissolved in water, driven by the strong hydrophobic interactions, these hydrophobic drug moieties rapidly self-aggregated to form a hydrophobic core. And then, because of a high affinity with water, those hydrophilic polymer chains extended in the water and then frizzled to form a hydrophilic shell on the surface of the hydrophobic core. It should be noted that these inter- and/or intra-molecular hydrogen bonds in the hydrophilic shell also promoted the assembly process by increasing the stability of the shell [19] [20] .
Figure 5. TEM image of M
Table 2. The mean diameters and distributions of the micelles in pH 7.4 PBS for their stability.
3.3. Stability of CMCS-g-β-CD・DMP Micelles
The stability study of the micelles was performed using DLS by placing the micelles in pH 7.4 PBS for 30 days. Before and after storage, their mean diameters and distributions were both determined and summarized in Table 2. It could be easily found that the micelles only had little diameter and polydispersity index (PDI) value change which indicated the micelles had good stability. This result might be ascribed to the formation of intra- and/or inter-molecular hydrogen bonds in the hydrophilic shell which made the micelles a non-covalently cross-linked hydrogel configuration.
3.4. pH-Sensitivity of CMCS-g-β-CD・DMP Micelles
The changes of the micelle diameters with pH alteration determined by DLS were shown in Figure 6. As the pH value increased from 3.0 to 6.0, the mean diameters of the three samples increased significantly and reached a maximum value at pH 6.0. This might be attributed to increasing of protonation of the carboxyl groups in the shells. After being protonated, the polysaccharide chains would become more hydrophilic and tend to repulse each other which finally led to a swell of the hydrophilic shell. As the pH value further increased to 8.0, however, a sudden decrease of particle diameters was observed, which could be attributed to the weaker electrostatic repulsion weakened by the charge screening effect of counter ions.
3.5. Reduction-Response of CMCS-g-β-CD・DMP Micelles
The reduction-response of the micelles was investigated by DLS in media containing various concentrations of GSH (0, 10 μM,
Figure 6. pH-sensitivity of the micelles.
Table 3. The mean diameters and distributions of the micelles in pH 7.4 media with various concentrations of GSH for 24 h.
diameters and PDI values of the micelles in the buffer with 10 μM GSH changed little after incubated for 24 h. As the GSH concentration increased to
3.6. Stimuli-Responsive Release of 6-MP from the Micelles
The stimuli-responsive release of 6-MP from the micelles was investigated with M-2. As shown in Figure 7, 6-MP cumulative release from the micelles was about 27.3 wt% in pH 7.4 PBS with 10 μM GSH (mimicking the blood circulation) after 48 h. When the GSH concentration increased to
The release mechanism of hydrophobic 6-MP was highly dependent on diffusion process and degradation of the micelles. 6-MP could be released from DMP through two procedures. The first was that via an exchange reaction between DMP and GSH, one 6-MP was released in free form and the other was released in form of 6-MP-GSH. After that, 6-MP-GSH could again release free 6-MP by an exchange reaction with another GSH [7] . In pH7.4 PBS with 10 μM GSH, drug release was likely to obey the diffusion mechanism and was controlled by the swelling degree of the micelles. In pH 7.4 PBS with
The effect of drug content on the release behavior of the micelles was investigated in pH 7.4 PBS with
Figure 7. Drug release profiles from M
50.1 wt% after 48 h. The release rate from M-2 reached about 28 wt% in initial 3 h and over 59 wt% after 48 h, whereas M-3 reached about 29 wt% in initial 3 h and over 69 wt% after 48 h. The data showed the release rate increased with the increase of drug content in CMCS-g-β-CD・DMP micelles.
3.7. Anti-Tumor Activity of CMCS-g-β-CD・DMP Micelles
The cytotoxicity of the CMCS-g-β-CD・DMP micelles was evaluated in HeLa cells using MTT assay. As shown in Figure 9, free 6-MP was employed as a positive control and blank CMCS-g-β-CD was employed as a negative control. The negative control experiment revealed that CMCS-g-β-CD was nearly non-toxicity to HeLa cells, indicating the polymer had good biocompatibility. The M-2, however, exhibited a dose-dependent cytotoxicity
Figure 8. Drug release profiles from the micelles of various DMP contents in pH 7.4 PBS with
Figure 9. Cell inhibition ratio of in vitro cytotoxicity of CMCS-g-β-CD, CMCS-g-β-CD・DMP micelles and free DMP at various concentrations against HeLa cells.
for HeLa cells which was comparable to that of free 6-MP after incubation for 48 h. This result should be attributed to the fast release of 6-MP from M
4. Conclusion
In summary, we had shown that: 1) an inclusion-interaction assembly strategy was used to construct novel pH/ redox responsive micelles, with the drug as the core and the polymer as the shell; this strategy could achieve integration of drug-loading and self-assembly, avoiding the multiple steps used in covalent-bond method; 2) TEM photograph confirmed that the micelles had a spherical core-shell structure; 3) investigations by DLS showed that the micelles were stable in water and had a narrow size distribution as well as a good pH/redox sensitivity; 4) in vitro drug release showed a selective release in pH 5.0 medium containing
Acknowledgements
This work was financially supported by Natural National Science Foundation of China (Nos. 51373130, 51273156 and 31300791).
Cite this paper
Mingyuan Tan,Hua Zheng,Xueqiong Zhang,Zhiyan Yu,Zhao Ye,Liyan Shi,Kaikai Ding, (2015) Inclusion-Interaction Assembly Strategy for Constructing pH/Redox Responsive Micelles for Controlled Release of 6-Mercaptopurine. Materials Sciences and Applications,06,605-616. doi: 10.4236/msa.2015.67063
References
- 1. Zhang, J., et al. (2005) Temperature Sensitive Poly[N-isopropylacrylamide-co-(acryloylβ-cyclodextrin)] for Improved Drug Release. Macromolecular Bioscience, 5, 192-196.
http://dx.doi.org/10.1002/mabi.200400167 - 2. Thorsteinn, L. and Dominique, D. (2007) Cyclodextrin and Their Pharmaceutical Applications. International Journal of Pharmaceutics, 329, 1-11.
http://dx.doi.org/10.1016/j.ijpharm.2006.10.044 - 3. He, H., Chen, S., Zhou, J., Dou, Y., Song, L., Che, L., Zhou, X., Chen, X., Jia, Y., Zhang, J., Li, S. and Li, X. (2013) Cyclodextrin-Derived pH-Responsive Nanoparticles for Delivery of Paclitaxel. Biomaterials, 34, 5344-5358.
http://dx.doi.org/10.1016/j.biomaterials.2013.03.068 - 4. Yuan, Z., Ye, Y., Gao, F., Yuan, H., Lan, M., Lou, K. and Wang, W. (2013) Chitosan-Graft-β-Cyclodextrin Nanoparticles as a Carrier for Controlled Drug Release. International Journal of Pharmaceutics, 446, 191-198.
http://dx.doi.org/10.1016/j.ijpharm.2013.02.024 - 5. Zhang, J. and Ma, P. (2013) Cyclodextrin-Based Supramolecular Systems for Drug Delivery: Recent Progress and Future Perspective. Advanced Drug Delivery Reviews, 65, 1215-1233.
http://dx.doi.org/10.1016/j.addr.2013.05.001 - 6. Wang, J. and Jiang, M. (2006) Polymeric Self-Assembly into Micelles and Hollow Spheres with Multiscale Cavities Driven by Inclusion Complexation. Journal of American Chemical Society, 128, 3703-3708.
http://dx.doi.org/10.1021/ja056775v - 7. Zheng, H., Rao, Y., Yin, Y., Xiong, X., Xu, P. and Lu, B. (2011) Preparation, Characterization, and in Vitro Drug Release Behavior of 6-Mercaptopurine-carboxymethyl Chitosan. Carbohydrate Polymer, 83, 1952-1958.
http://dx.doi.org/10.1016/j.carbpol.2010.10.069 - 8. Gong, X., Yin, Y., Huang, Z., Lu, B., Xu, P., Zheng, H., Xiong, F., Xu, H., Xiong, X. and Gu, X. (2012) Preparation, Characterization and in Vitro Release Study of a Glutathione-Dependent Polymeric Prodrug Cis-3-(9H-purin-6-ylthio)- acrylic acid-graft-carboxymethyl chitosan. International Journal of Pharmaceutics, 436, 240-247.
http://dx.doi.org/10.1016/j.ijpharm.2012.06.043 - 9. Chang, D., Lei, J., Cui, H., Lu, N., Sun, Y., Zhang, X., Gao, C., Zheng, H. and Yin, Y. (2012) Disulfide Cross-Linked Nanospheres from Sodium Alginate Derivative for Inflammatory Bowel Disease: Preparation, Characterization, and in Vitro Drug Release Behavior. Carbohydrate Polymer, 88, 663-669.
http://dx.doi.org/10.1016/j.carbpol.2012.01.020 - 10. Gao, C., Liu, T., Dang, Y., Yu, Z., Wang, W., Guo, J., Zhang, X., He, G., Zheng, H., Yin, Y. and Kong, X. (2014) pH/Redox Responsive Core Cross-Linked Nanoparticles from Thiolated Carboxymethyl Chitosan for in Vitro Release Study of Methotrexate. Carbohydrate Polymer, 111, 964-970.
http://dx.doi.org/10.1016/j.carbpol.2014.05.012 - 11. Wang, W., Yang, H., Kong, X., Ye, Z., Yin, Y., Zhang, X., He, G., Xu, P. and Zheng, H. (2014) Hydrogen-Bonding Strategy for Constructing pH-Sensitive Core-Shell Micelles with Hydrophilic Polymer as the Shell and Hydrophobic Drug as the Core. RSC Advances, 4, 28499-28503.
http://dx.doi.org/10.1039/c4ra03716b - 12. Schafer, F.Q. and Buettner, G.R. (2001) Redox Environment of the Cell as Viewed through the Redox State of the Glutathione Disulfide/Glutathione Couple. Free Radical Biology Medicine, 30, 1191-1212.
http://dx.doi.org/10.1039/c4ra03716b - 13. Zacchigna, M., Cateni, F., Di-Luca, G. and Drioli, S. (2007) A Simple Method for the Preparation of PEG-6-mercap- topurine for Oral Administration. Bioorganic & Medicinal Chemistry, 17, 6607-6609.
http://dx.doi.org/10.1016/j.bmcl.2007.09.064 - 14. Chen, Y., Peng, C. and Lu, Y. (2015) Responsiveness and Release Characteristic of Semi-IPN Hydrogels Consisting of Nano-Sized Clay Crosslinked Poly(Dimethylaminoethyl Methacrylate) and Linear Carboxymethyl Chitosan. Journal of Nanoscience and Nanotechnology, 15, 164-171.
http://dx.doi.org/10.1166/jnn.2015.8768 - 15. Gerweck, L.E. and Seetharaman, K. (1996) Cellular pH Gradient in Tumor versus Normal Tissue: Potential Exploitation for Treatment of Cancer. Cancer Research, 56, 1194-1198.
- 16. Doerr, I.L., Wempen, I., Clarke, D.A. and Fox, J.J. (1961) Thiation of Nucleosides. III. Oxidation of 6-Mercaptopu- rines. Journal of Organic Chemistry, 26, 3401-3409.
http://dx.doi.org/10.1021/jo01067a093 - 17. Rajendiran, N., Mohandoss, T. and Sankaranarayanan, R.K. (2014) Nanostructure Formed by Cyclodextrin Covered Procainamide through Supramolecular Self-Assembly—Spectral and Molecular Modeling Study. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 136, 875-883.
http://dx.doi.org/10.1016/j.saa.2014.09.108 - 18. Arti, M., Manisha, S. and Deepak, S. (2011) Investigation of the Binding of Roxatidine Acetate Hydrochloride with Cyclomaltoheptaose (β-Cyclodextrin) Using IR and NMR Spectroscopy. Carbohydrate Research, 346, 1809-1813.
http://dx.doi.org/10.1016/j.carres.2011.07.003 - 19. Warayuth, S., Onanong, N., Pattarapond, G., Somsak, S., Issara, S., Apinan, S., Satit, P. and Uracha, R. (2012) Water-Soluble β-Cyclodextrin Grafted with Chitosan and Its Inclusion Complex as a Mucoadhesive Eugenol Carrier. Carbohydrate Polymer, 89, 623-631.
http://dx.doi.org/10.1016/j.carbpol.2012.03.060 - 20. Yuan, P., Chengde, L., Zhongxing, Z., Kerh, L., Jianhai, C. and Jun, L. (2011) Chitosan-Graft-(PEI-β-Cyclodextrin) Copolymers and Their Supramolecular PEGylation for DNA and siRNA Delivery. Biomaterials, 32, 8328-8341.
http://dx.doi.org/10.1016/j.biomaterials.2011.07.038
NOTES
*Corresponding authors.













