Materials Sciences and Applications
Vol.5 No.9(2014), Article ID:48447,11 pages
DOI:10.4236/msa.2014.59067
Preparation of Porous Silicone Resin Sheet with Phase Inversion in Parallel with Non Solvent Induced Phase Separation and Application to Hollow Particle Formation
Takuya Yokoyama, Yoshinari Taguchi, Masato Tanaka*
Graduate School of Science and Technology, Niigata University, Niigata, Japan
Email: *tanaka@eng.niigata-u.ac.jp
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/
![]()
![]()

Received 6 May 2014; revised 9 June 2014; accepted 28 June 2014
ABSTRACT
We have tried to prepare the porous silicone resin sheet with the phase inversion method in parallel with the non solvent induced phase separation method. In the experiment, ethyl acetate and water were adopted as a good solvent and a poor solvent for silicone resin, respectively and ethyl alcohol as an amphiphilic solvent was used to increase the solubility of ethyl acetate in water and decrease the interfacial tension by mass transfer from water to ethyl acetate. The concentration of silicone resin in ethyl acetate and the oil soluble surfactant species were changed. Increasing the concentration of silicone resin could depress coalescence between the water droplets in the (W/O) dispersion and increase the porosity and pore number density of silicone resin sheet. Span 80 among the oil soluble surfactant species made the porosity and pore number density larger. The effect of physical proparties of liquids concerned on the porosity and pore number density was discussed on the basis of dispersing behavior of liquid droplets in the liquid-liquid dispersion. The hollow silicone resin particles could be prepared by applying the preparation method presented here.
Keywords:Porous Silicone Resin Sheet, Non Solvent Induced Phase Separation, Phase Inversion, Hollow Silicone Resin Particles

1. Introduction
Polymeric porous membranes [1] -[3] and particles [4] [5] have attracted much attention for various applications such as membrane separation and purification, solid supports for catalysts and dyes, scaffolds for biological cells and so on [6] .
The polymeric porous membranes and particles have been prepared by the several methods such as the multiple emulsion method, the phase separation method, the removal method of inorganic salts, carbon dioxide forming method and so on [5] .
The phase separation method among these preparation methods has been of great interest because of the simple and low cost preparation process [7] [8] . Non solvent induced phase separation is on the basis of the removal of the good solvent from the uniform solution composed of polymer, good and poor solvents for polymer. With increasing the removed volume of good solvent, polymer and poor solvent may be separated and the droplets of poor solvent come to be formed in polymer swollen by the good solvent. The polymeric porous membrane can be prepared by removing water from the droplets of poor solvent. Accordingly, in order to prepare the higher porous membrane, it is necessary to stably disperse the smaller droplets of poor solvent in the matrix polymer.
In the case of the system composed of silicone resin, ethyl acetate, water and ethyl alcohol, after complete removing of the good solvent, the silicone resin sheet containing the liquid droplets of poor solvent is prepared. Then, the porous silicone resin sheet can be prepared by removing these liquid droplets due to drying.
It is well known that the diameter and number of dispersed droplets are determined by the dispersing behavior of droplets such as coalescence and breakup which are strongly affected by the turbulent energy level in the continuous phase and the physical properties of liquids concerned [9] -[11] .
Also, the finer liquid droplets are able to be easily prepared by utilizing the phase inversion under mass transfer of amphiphilic solvent [12] .
As the pore size and pore number density in the porous sheet prepared by the phase separation method are inevitably dependent on the diameter and number of liquid droplets of poor solvent formed by removing the good solvent, it is necessary to investigate how the dispersing behavior of dispersed liquid droplets affects the pore size and porosity of sheet in detail.
In this study, it was tried to prepare the porous silicon resin sheet with the phase inversion method in parallel with the non solvent induced phase separation method.
In the experiment, ethyl acetate and water were adopted as the good and the poor solvent for silicone resin, respectively and ethyl alcohol as an amphiphilic solvent was used to increase the solubility of ethyl acetate in water and decrease the interfacial tension between the oil phase and the aqueous solution due to mass transfer.
The purposes of this study are to investigate how the porosity and pore number density of silicone resin sheet are affected by the experimental conditions such as silicone resin concentration and the oil soluble surfactant species, to discuss the effect of dispersing behavior of dispersed liquid droplets of poor solvent on the porosity and pore number density and to apply to the preparation of hollow silicone resin particles.
2. Experimental
2.1. Materials
Silicone resin and hardening agent for silicone resin were silicone KE-106 (Shinetsu Kagaku Kogyo, Tokyo, Japan) and CAT-RG (Shinetsu Kagaku Kogyo, Tokyo, Japan), respectively.
Ethyl acetate (Wako Pure Chemical Industries, Tokyo, Japan) and distilled water were used as a good solvent and a poor solvent for silicone resin, respectively. Ethyl alcohol as an amphiphilic solvent (Wako Pure Chemical Industries, Tokyo, Japan) was used to decrease the interfacial tension between the oil solution and the aqueous solution by rapid mass transfer and to increase the solubility of ethyl acetate in water.
2.2. Preparation of Porous Silicone Resin Sheet
Figure 1 shows the schematic diagram of experimental apparatus together with the flow chart for preparing the porous silicone resin sheet. The reactor was the separable flask with the diameter of 8.5 cm and the effective volume of 300 ml. The aqueous solution dissolving ethyl alcohol was poured into this reactor. Then, the oil solution composed of silicone resin, ethyl acetate, hardening agent and oil soluble surfactant was dropped into the
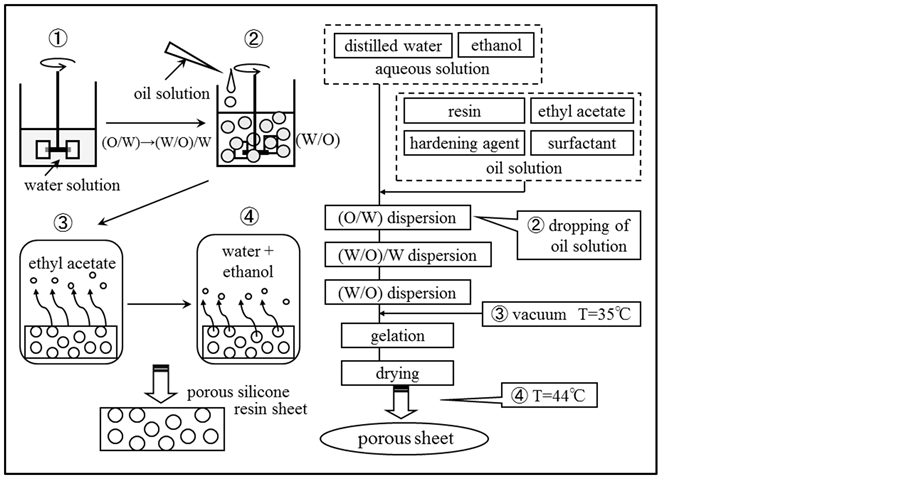
Figure 1. Shematic diagram of experimental apparatus and flow chart for preparing porous silicone resin sheet.
aqueous solution with the syringe pump under stirring with the six-bladed disc turbine impeller (revolution speed: 5 s−1).
First, the (O/W) dispersion was formed at room temperature and then, with increasing the volume of oil solution poured, the (W/O)/W dispersion and the (W/O) dispersion due to phase inversion were formed in turn [12] . The (W/O) dispersion was kept in the thermostatted water bath at temperature of 35˚C. From this point, the hardening reaction and the removal of ethyl acetate and ethyl alcohol were performed for 12 h under reduced pressure. With removing ethyl acetate and ethyl alcohol by the drying-in-liquid operation, the water droplets were formed in the silicone resin sheet swollen by ethyl acetate.
Then, the silicone resin sheet containing the water droplets was dried at temperature of 44˚C for 24 h to remove water perfectly. After this operation, the porous silicone resin sheet has been prepared.
In the fundamental preparation process stated just above, the concentration of silicone resin and the oil soluble surfactant species were changed.
The experimental conditions adopted here are shown in Table1
2.3. Preparation of Hollow Silicone Resin Particle
Figure 2 shows the schematic diagram of experimental apparatus and the flow chart for preparing the hollow silicone resin particles. The reactor was the separable flask with the diameter of 8.5 cm and the effective volume of 400 ml, in which four baffles with 0.85 cm width made from aluminum plate were settled on the reactor wall. This reactor was set in the water bath to keep temperature of the reactor constant. A six bladed disc type impeller with the diameter of 3.5 cm was used to form the (W/O) dispersion and the (W/O)/W dispersion. The inner aqueous solution was prepared by dissolving ethyl alcohol in the water phase. The oil solution was prepared by dissolving ethyl acetate, hardening agent and oil soluble surfactant in silicone resin.
First, the (W/O) dispersion was formed at room temperature by dropping the inner aqueous solution into the oil solution with the syringe pump under stirring with the six bladed disc turbine impeller (revolution speed: 11.7 s−1).
Second, the (W/O)/W dispersion was formed at room temperature by dropping the (W/O) dispersion into the continuous water phase in which sodium dodecyl sulphate was dissolved as a water soluble surfactant.
After formation of the (W/O)/W dispersion, the gelated reaction was performed at temperature of 70˚C with removing ethyl acetate and ethyl alcohol by the drying-in-liquid method. The experimental conditions are shown in Table2
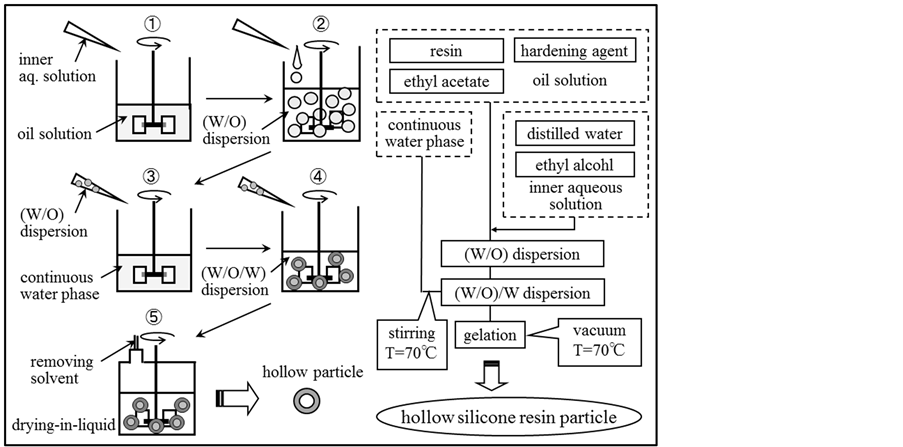
Figure 2. Schematic diagram of experimental apparatus and flow chart for preparing hollow silicone resin particles.
3. Characterization
3.1. Observation of Cross Section of Porous Silicone Resin Sheet
The cross section of porous silicone resin sheet was observed by scanning electron microscopy (JSM-S800; JEOL Ltd., Tokyo, Japan).
From these SEM photographs, the pore size and pore number density were measured directly.
3.2. Measurement of Water Droplet Diameters in the (W/O) Dispersion
The water droplet diameters were measured from the optical microscopic photographs of the water droplets taken by the optical microscope (BH-2; OLYMPUS Grp., Tokyo, Japan) just after formation of the (W/O) dispersion. From these photographs, the mean diameters were measured directly.
3.3. Measurement of Physical Properties of Liquids Concerned
Interfacial tension and viscosity were measured by the Auto Tensiometer (CBVP-A3; Kyowa Kaimen Kogaku Ltd., Tokyo, Japan) and the plate vibrating viscometer (VM-1A-L; Yamaichi Denki Ltd., Tokyo, Japan), respectively.
4. Results and Discussion
4.1. Effect of Concentration of Silicone resin
Figure 3 shows the SEM photographs of the cross section of porous silicone resin sheet prepared at the various concentrations of silicone resin without any oil soluble surfactants.
It is found that the pore sizes drastically decrease with the concentration (Cs) of silicone resin. As the pore sizes are strongly dependent on the diameters of water droplets in the (W/O) dispersion, it is necessary to investigate the dispersion stability of water droplets in the oil solution.
In general, the stability of liquid droplets in the liquid-liquid dispersion is affected by the physical properties of liquids such as the viscosity of continuous phase and dispersed phase, the interfacial tension and the difference in density between the dispersed and the continuous phase under the given conditions of the revolution speed and the concentration of stabilizer [10] . As the viscosity of oil solution and the interfacial tension specially strongly affect the dispersing behavior of droplets, the measured values of the viscosity (μs) of oil solution and the interfacial tension (γ) are shown in Table3 It is found that, with increasing the concentration of silicone re
Table 3. Measured values of viscosity of oil solution and interfacial tension (T = 35˚C).

Figure 3. SEM photographs of silicone resin sheet Cs = (a) 50 (b) 67 (c) 75 (d) 80 wt%.
sin, the interfacial tension slightly increase and the viscosity extremely increase.
Figure 4 shows the dependences of the water droplet
diameters (dp) and the pore sizes (Dp) on the viscosity of oil solution
together with the degree of dispersion of pore sizes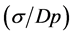 , where σ is the standard deviation of size distribution.
, where σ is the standard deviation of size distribution.
From Figure 4, it is found that the water droplet diameters are almost constant because of almost constant interfacial tension as shown in Table 3 and the pore sizes are very larger than the diameters of the water droplets in the region of the lower concentration of silicone resin, but gradually become close to the diameters of the water droplets with the concentration of silicone resin. Furthermore, the distribution of pore sizes becomes larger over the region of the lower concentration of silicone resin.
In general, the water droplet diameters may inevitably change due to coalescence or breakup of liquid droplets. Also, coalescence between the liquid droplets is depressed by increasing the viscosity of continuous phase. As an example, the dependences of the coalescence velocity (Rc) and the mean liquid droplet diameter (dp) on the physical properties of liquids such as the viscosity of continuous phase (μc) and the interfacial tension (γ) are expressed as follows [10] [13] .
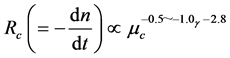 (1)
(1)
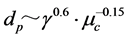 (2)
(2)
where n and t are the number of liquid droplets per unit volume of the dispersion
and time, respectively. Accordingly,
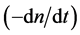 means the decreasing velocity of droplet number due to coalescence.
means the decreasing velocity of droplet number due to coalescence.
From Equation (1), it may be quantitatively understood that the coalescence velocity is able to be decreased with increasing the viscosity of continuous phase and the interfacial tension. On the other hand, the liquid droplet diameters become smaller with increasing the viscosity of continuous phase and decreasing the interfacial tension.
Taking these results into consideration, the following things may be discussed. In the case of the lower concentration of silicone resin, the water droplets frequently coalesce with each other to form the larger water drop-
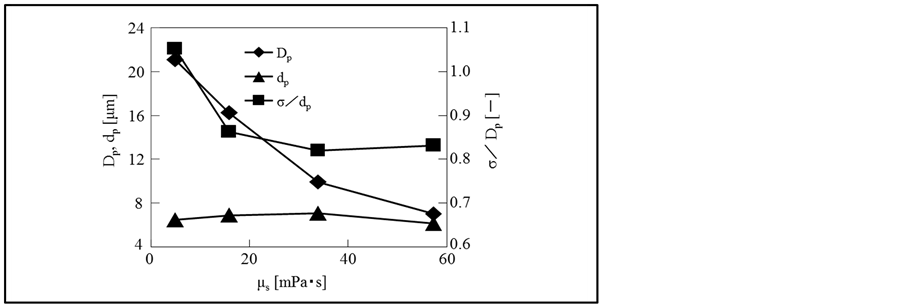
Figure 4. Dependences of mean water droplet diameter, mean pore sizes and degree of dispersion on viscosity of oil solution.
lets during the removal process of good solvent. In the case of the higher concentration
of silicone resin, the water droplets have to be stabilized in the oil solution.
Accordingly, in the range of the lower concentration of silicone resin, the pore
sizes have to become larger due to coalescence of the water droplets. As a result,
the degree of dispersion of pore sizes should become larger. In the range of the
higher concentration of silicone resin, the pore sizes have to become close to the
water droplet diameters due to depression of coalescence. As a result, the degree
of dispersion of pore sizes should become smaller, namely the pore sizes become
more uniform. The degrees of coalescence
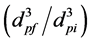 during the drying-in-liquid process at the concentrations of silicone resin of 50
wt% and 80 wt% were found to be ca. 35.4 and 1.6, respectively, where dpi
and dpf are the droplet diameters before and after coalescence, respectively
[14] . Here, the pore sizes (Dp) are used
instead of the droplet diameters after coalescence to estimate the degree of coalescence.
during the drying-in-liquid process at the concentrations of silicone resin of 50
wt% and 80 wt% were found to be ca. 35.4 and 1.6, respectively, where dpi
and dpf are the droplet diameters before and after coalescence, respectively
[14] . Here, the pore sizes (Dp) are used
instead of the droplet diameters after coalescence to estimate the degree of coalescence.
As the degree of coalescence at the concentration of 50 wt% is 35.4, the stability of water droplets in the oil solution is very low in comparison with 1.6 at the concentration of 80 wt%.
Figure 5 shows the dependence of the pore number density on the viscosity of oil solution, where the numerical values in the parentheses are the values at the abscissa and the ordinate, respectively.
On increasing the viscosity of oil solution, the pore number density increases from 28 at 5.0 mPa∙s to 62 at 58.2 mPa∙s.
4.2. Effect of Oil Soluble Surfactant Species
In order to increase the pore number density, it is necessary to decrease the water droplet diameters and to increase the stability of water droplets in the (W/O) dispersion.
According to Equation (2), the water droplet diameters can be decreased by lowering the interfacial tension due to addition of the oil soluble surfactant into the oil solution under the given revolution speed to form the (W/O) dispersion.
In general, it is well known that the effect of surfactant on the stability of the (W/O) dispersion is strongly dependent on the HLB value and molecular structure of oil soluble surfactant, because the amount and configuration of surfactant absorbed on the interface are affected by them. In order to investigate the optimum surfactant species, three kinds of oil soluble surfactants were added.
Figure 6 shows the SEM photographs of the cross section of porous silicone resin sheet prepared by addition of the oil soluble surfactants at the concentration of silicone resin of Cs = 80 wt%.
The pore sizes decrease and the pore number density increases due to addition of the oil soluble surfactants.
In order to quantitatively know the degree of effects of surfactant species, Figure 7 shows the effect of the oil soluble surfactant species on the pore sizes (DP) and the pore number density (N), where the numerical values in the parenthesis are the values at the ordinate. Among of these surfactants, Span80 with the smallest HLB value makes the pore size smaller. Namely, it is found that the pore sizes considerably decrease from 7.5 μm (without surfactant) to 1.8 μm (Span 80) and the pore number density increases from 62 to 148.
As these results are considered to be due to the fact that the interfacial tension and the viscosity of oil solution are changed by addition of the surfactants, these physical properties are measured and shown in Figure 8.
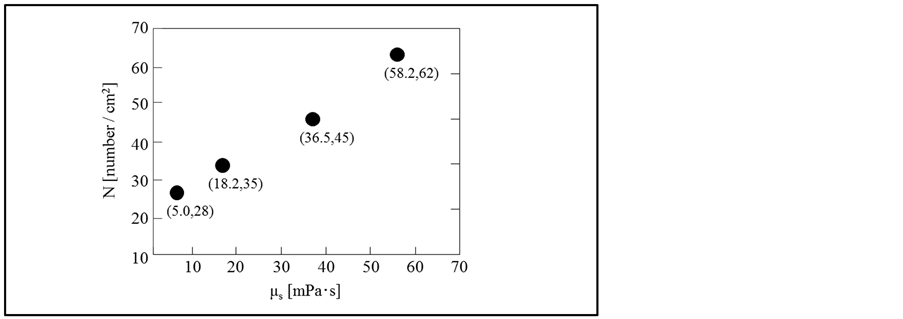
Figure 5. Dependences of mean water droplet diameter, mean pore sizes and degree of dispersion on viscosity of oil solution.

Figure 6. SEM photographs of silicone resin sheet (a) no (b) Span 20 (c) Span 40 (d) Span 80 (effect of surfactant species).
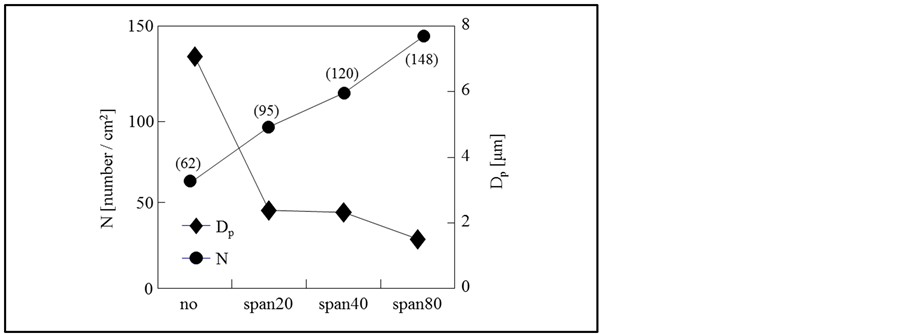
Figure 7. Effect of oil soluble surfactant species on mean pore size and pore number density.
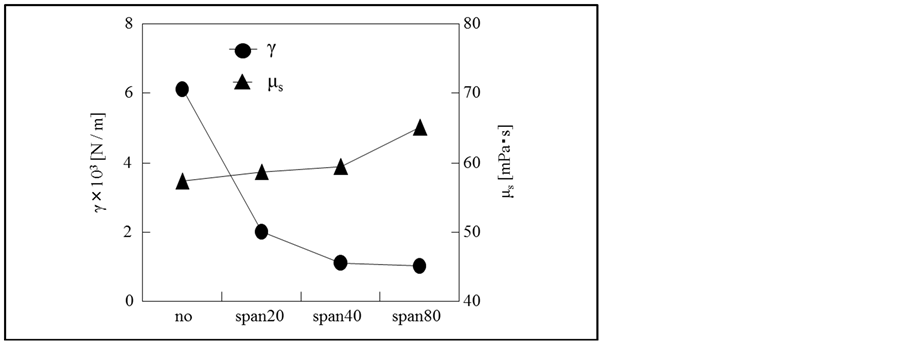
Figure 8. Effect of oil soluble surfactant species on interfacial tension and viscosity of oil solution.
In the case of Span 80, as the interfacial tension considerably decreases and the viscosity slightly increases, the water droplets have to become smaller and been stabilized with both the absorption layer of surfactant and the increase in viscosity of oil solution as stated just above.
4.3. Formation Mechanism of Porous Sheet
Taking the results obtained above into consideration, the formation mechanism of the porous sheet may be discussed as follows.
Figure 9 shows the formation mechanism of the porous silicone resin sheet prepared by the method adopted in this study.
First, the (O/W) dispersion has to be formed in the early stage by pouring the oil solution into the aqueous solution. Ethyl alcohol of amphiphilic solvent may transfer from the aqueous solution to the oil solution and lower the interfacial tension [11] .
Then, the (W/O)/W dispersion has to be formed because of increase in the oil droplet diameter with the volume of oil solution poured and the dispersion of the fine inner water droplets into a larger oil droplet.
Finally, the (W/O) dispersion has to be formed by the phase inversion occurred by addition of the volume of oil solution larger than that of aqueous solution.
The water droplets in the (W/O) dispersion have to be stabilized by the higher viscosity of oil solution and the adsorption layer of surfactant during the removal of solvents such as ethyl acetate and ethyl alcohol.
In the consecutive processes stated above, the fine inner droplets dispersed in the oil droplets can be made smaller by lowering the interfacial tension due to mass transfer of ethyl alcohol [15] .
As a result, it is found that the non solvent induced phase separation method in parallel with phase inversion is extremely effective for preparing the porous silicone resin sheet.
4.4. Application to Preparation of Hollow Silicone Resin Particles
Also, it was tried to apply the method for preparing the porous silicone resin sheet to the formation of the hollow silicone resin particles as follows.
Namely, the (W/O) dispersion before gelation as shown in Figure 2 was poured into the continuous water phase dissolving sodium dodecylbenzen sulphonate (DBS) as a water soluble surfactant to form the (W/O)/W dispersion.
From the formation of the (W/O)/W dispersion, the hardening reaction and the removal of solvents such as ethyl acetate and ethyl alcohol were started under temperature of 70˚C and reduced pressure.
In this preparation method, any oil soluble surfactants were not added to promote coalescence between the fine inner water droplets in the oil droplets and to make the hollow structure.
Figure 10 shows the optical microscopic photograph (a) and the SEM photograph (b) of the inner structure of hollow silicone resin particle. From Figure 10, it is found that the hollow silicone resin particles can be prepared
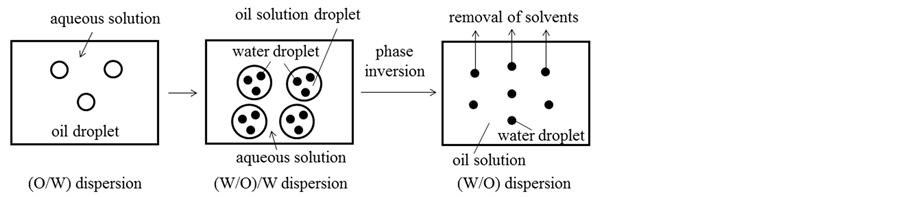
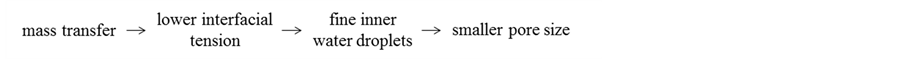
Figure 9. Formation mechanism of porous silicone resin sheet.
 (a) (b)
(a) (b)
Figure 10. Photographs of hollow particle. (a) optical microscopic photograph, (b) SEM photograph.
by the preparation method presented in this study. The formation of the hollow structure is inevitably determined by the coalescence velocity between the fine inner water droplet and the formation velocity of microcapsules shell. Also, these velocities are affected by the concentrations of surfactants and the reaction conditions. Accordingly, it may be necessary to establish the detailed preparation conditions and to develop the simple method for preparing the hollow particles.
5. Conclusions
It was tried to prepare the porous silicone resin sheet with the phase inversion in parallel with the non solvent induced phase separation method. Ethyl acetate and water were adopted as the good solvent and the poor solvent for silicone resin, respectively.
The preparation method presented here was applied to prepare the hollow silicone resin particles. The following results were obtained.
1) With increasing the concentration of silicone resin, coalescence between the water droplets in the (W/O) dispersion could be depressed and the water droplets diameters could be decreased. As a result, the pore sizes could be decreased and the pore number density could be increased.
2) By adding Span 80 (HLB 4.3), the pore sizes could be drastically decreased and the pore number density could be increased.
3) The effects of physical properties of liquids changed by the concentration of silicone resin and Span 80 on the porosity and pore number density was discussed on the basis of dispersing behavior of dispersed droplets in the liquid-liquid dispersion.
4) The hollow silicone resin particles could be prepared by the preparation method prepared in this study.
References
- Matsuyama, H., Teramoto, M. and Uesaka, T. (1997) Membrane Formation and Structure Development by Dry-Cast Process. Journal of Membrane Science, 135, 271-288. http://dx.doi.org/10.1016/S0376-7388(97)00154-3
- Tokuyama, H., Sumida, H., Kanehara, A. and Nii, S. (2009) Effect of Surfactants on the Porous Structure of Poly(NIsopropylacrylamide) Hydrogels Prepared by an Emulsion Templating Method. Colloid and Polymer Science, 287, 115-121. http://dx.doi.org/10.1007/s00396-008-1964-1
- Kim, J.K., Taki, K., Nagamine, S. and Ohshima, M. (2009) Preparation of a Polymeric Membrane with a Fine Porous Structure by Dry Casting. Journal of Applied Polymer Science, 111, 2518-2526. http://dx.doi.org/10.1002/app.29348
- Ji, S., Srivastava, D., Parker, N.J. and Lee, I. (2012) Transitional Behavior of Polymeric Hollow Microsphere Formation in Turbulent Shear flow by Emulsion Diffusion Method. Polymer, 53, 205-212. http://dx.doi.org/10.1016/j.polymer.2011.11.013
- Chen, Y., Chen, Y., Nan, J., Wang, C. and Chu, F. (2012) Hollow Poly (N-Isopropylacrylamide)-Co-Poly(Acrylic Acid) Microgels with High Loading Capacity for Drugs. Journal of Applied Polymer Science, 124, 4678-4685.
- Costa, H.S., Mansur, A.A.P., Barbosa-Stancioli, E.F., Pereira, M.M. and Mansur, H.S. (2008) Morphological, Mechanical, and Biocompatibility Characterization of Macroporous Alumina Scaffolds Coated with Calcium Phosphate/PVA. Journal of Materials Science, 43, 510-524. http://dx.doi.org/10.1007/s10853-007-1849-6
- Graham, P.D., Barton, B.F. and Mchugh, A.J. (1999) Kinetics of Thermally Induced Phase Separation in Ternary Polymer Solutions. II. Comparison of Theory and Experiment. Journal of Polymer Science: Part B: Polymer Physics, 37, 1461-1467. http://dx.doi.org/10.1002/(SICI)1099-0488(19990701)37:13<1461::AID-POLB12>3.0.CO;2-B
- Xin, Y., Fujimoto, T. and Uyama, H. (2012) Facile Fabrication of Polycarbonate Monolith by Non-Solvent Induced Phase Separation method. Polymer, 53, 2847-2853. http://dx.doi.org/10.1016/j.polymer.2012.04.029
- Tanaka, M., Nagada, K. and Oyama, Y. (1970) Coalescence of Dispersed Liquid Droplets in a Batch Type of Stirred Tank Reactor. Kagaku Kogaku, 34, 893-897. http://dx.doi.org/10.1252/kakoronbunshu1953.34.893
- Oshima, E. and Tanaka, M. (1982) Coalescence and Breakup of Droplets in Suspension Polymerization of Styrene. Kagaku Kogaku Ronbunshu, 8, 86-90. http://dx.doi.org/10.1252/kakoronbunshu.8.86
- Tanaka, M. and Morishima, T. (1986) Deposition of Solid Particles on Walls of the Stirred Tank Reactor and Draft Tube. Kagaku Kogaku Ronbunshu, 12, 231-234. http://dx.doi.org/10.1252/kakoronbunshu.12.273
- Ilia Anisa, A.N., Nour, A.H. and Nour, A.H. (2010) Catastrophic and Transitional Phase Inversion of Water-in-Oil Emulsion for Heavy and Light Crude Oil. Journal of Applied Sciences, 10, 3076-3083. http://dx.doi.org/10.3923/jas.2010.3076.3083
- Tanaka, M. and OShima, E. (1982) Effect of Viscosity of Continuous Phase on Stability of Droplets in Suspension Polymerization of Styrene. Kagaku Kogaku Ronbunshu, 8, 734-738.
- Taguchi, Y. and Tanaka, M. (1998) Effect of Solid Powders as Stabilizers on Polymer Particles Sizes in Suspension Polymerization. Kagaku Kogaku Ronbunshu, 24, 633-637. http://dx.doi.org/10.1252/kakoronbunshu.24.633
- Mitomi, R., Taguchi, Y. and Tanaka, M. (2006) Preparation of Polyester Composite Nanoparticles Containing Pigment Using Phase-Inversion Emulsification Followed by Dry-in-Liquid Method. Journal of the Japan Society of Colour Material, 79, 487-491.
NOTES

*Corresponding author.


