Materials Sciences and Applications
Vol.4 No.6A(2013), Article ID:32987,4 pages DOI:10.4236/msa.2013.46A001
Heterostructure Solar Cells Based on Sol-Gel Deposited SnO2 and Electrochemically Deposited Cu2O
![]()
Department of Engineering Physics, Electronics and Mechanics, Nagoya Institute of Technology, Nagoya, Japan.
Email: ichimura.masaya@nitech.ac.jp
Copyright © 2013 Akito Fukuda, Masaya Ichimura. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received April 8th, 2013; revised May 11th, 2013; accepted May 20th, 2013
Keywords: sol-gel deposition; electrochemical deposition; Cu2O; SnO2; solar cell
ABSTRACT
To fabricate a heterostructure solar cell using environmentally friendly materials and low cost techniques, tin oxide (SnO2) and cuprous oxide (Cu2O) were deposited by the sol-gel method and the electrochemical deposition, respectively. The SnO2 films were deposited from a SnCl2 solution containing ethanol and acetic acid. The Cu2O films were deposited using a galvanostatic method from an aqueous bath containing CuSO4 and lactic acid at a temperature of 40˚C. The Cu2O/SnO2 heterostructure solar cells showed rectification and photovoltaic properties, and the best cell showed a conversion efficiency of 6.6 × 10−2 % with an open-circuit voltage of 0.29 V, a short-circuit current of 0.58 mA/cm2, and a fill factor of 0.39.
1. Introduction
The production of silicon solar cells is expanding, but not sufficiently fast, because of high cost and large energy consumption for the material purification and cell manufacturing. Thus extensive researches are going on, aiming the development of solar cells using cost-effective and environmentally benign materials prepared by simple low-cost techniques. In this work, we attempt to use tin oxide (SnO2) deposited by a sol-gel technique for the n-type semiconductor of a solar cell. SnO2 thin films have been attracting interest since they have many important applications such as gas sensors [1,2] and transparent electrodes [3,4]. SnO2 films have been fabricated by a number of techniques, including spray pyrolysis [5-6], sputtering [7-9], chemical vapor deposition (CVD) [10-12], and sol-gel deposition [3,13,14]. The sol-gel technique has several advantages, such as easy control of film thickness, ability to coat large area, and cost-effectiveness. In this work, we fabricate heterostructure solar cell based on sol-gel deposited SnO2. It should be noted here that there are very few reports of application of SnO2 for n-type layer of a heterostructure solar cell [15- 18] although SnO2 is often used as an electrode material.
On the other hand, cuprous oxide (Cu2O) has gathered much attention as a p-type absorption layer of solar cells owing to its suitable band gap around 2 eV, material abundance, and non-toxicity. The solar cells based on electrochemically deposited Cu2O have been fabricated with pn heterojunction with ZnO [19-21], TiO2 [22,23], and indium-tin-oxide (ITO) [24]. The highest solar conversion efficiency of 1.28% was obtained from ZnO/ Cu2O solar cell [19]. In this work, we attempt fabrication of heterostructure solar cells based on electrochemically deposited Cu2O and sol-gel deposited SnO2. One paper has been published on Cu2O/SnO2 heterostructure solar cells based on sputtered SnO2 [16], but to our best knowledge, there are no reports on fabrication of heterostructure cells by a combination of sol-gel deposition of SnO2 and electrochemical deposition (ECD) of Cu2O.
2. Experimentals
2.1. SnO2 Deposition and Characterization
SnO2 films were deposited by the sol-gel method on ITO coated glass substrates. (For the XRD measurement, we used a glass substrate.) 0.7 M SnCl2∙2H2O, 10 mL ethanol and 0.25 mL acetic acid were used as a starting source material, solvent and catalyst, respectively. The coating solution was stirred at 70˚C for 30 min and aged at 40˚C for 3 hours, and then dropped on the ITO substrate, which was rotated at 3000 rpm for 20 sec. Then, the films were dried at 150˚C for 5 min. The procedures from the spin coating to the drying were repeated 2 - 5 times, and then the film was annealed in air at 300˚C for 1 hour.
The compositional analysis was carried out by Auger electron spectroscopy (AES) using the model JEOL JAMP 7800. Profile meter Accretech Surfcom-1400D was used to measure the thickness of the film. The X-ray diffraction (XRD) measurement was carried out by the Rigaku SmartLab X-ray diffractometer using CuKa1 radiation. The optical transmission measurement was performed using a JASCO-570 spectrometer with the ITO substrate as the reference. In addition, the conduction type and photosensitivity of the films were examined by means of the photoelectrochemical (PEC) measurements. The PEC measurement was carried out using the threeelectrode cell with saturated calomel electrode (SCE) as the reference electrode. The deposited film was used as the working electrode, and 100 mM of Na2SO3 was used for the electrolyte. The backside of the sample was illuminated by pulsed light coming from a Xe lamp (about 100 mW/cm2). The incident light was turned off and on mechanically every five seconds. A ramp voltage was applied between the working and the reference electrodes, and the released current was monitored.
2.2. Cu2O/SnO2 Heterostructure Fabrication and Characterization
The heterostructure solar cells were fabricated by depositing Cu2O by ECD on the SnO2 film. We used a solution containing 0.2 M CuSO4 and 1.6 M lactic acid. pH of the solution was adjusted to 12.5 by adding KOH. Cu2O was deposited galvanostatically at 40˚C with a current density of −1.0 mA/cm2. All samples were deposited for 10 min. Indium metal electrodes were deposited by thermal evaporation on the Cu2O layer. The electrode size is 1 mm2. Photovoltaic properties were characterized using an AM 1.5 solar simulator. The radiation power of light was about 100 mW/cm2. The light was illuminated on the ITO side (SnO2 side) of the sample.
3. Results and Discussion
3.1. SnO2 and Cu2O Films
The thickness of the SnO2 film was about 0.6 μm when the coating-drying cycle was repeated 3 times. The film is transparent in the visible range, and the band gap obtained from the optical transmission spectrum is 3.9 eV. The differential AES spectrum of the prepared film is shown in Figure 1. The O/Sn composition ratio obtained from it is about 2, i.e., the film is almost stoichiometric. A weak chlorine signal was observed near 190 eV. The XRD pattern of the film deposited on the glass substrate is shown in Figure 2. The pattern shows three characteristic XRD peaks of SnO2 with a broad background signal due to glass. The SnO2 peaks are broad, which indicates that the SnO2 film consists of nano crystallites.
The PEC results for the SnO2 film are shown in Figure 3. The current was not changed by illumination for the cathodic scan, while during the anodic scan, the current was changed due to the light chopping. When the junction of the semiconductor-electrolyte is illuminated, photogenerated electrons/holes are separated in the space charge region. The photogenerated minority carriers
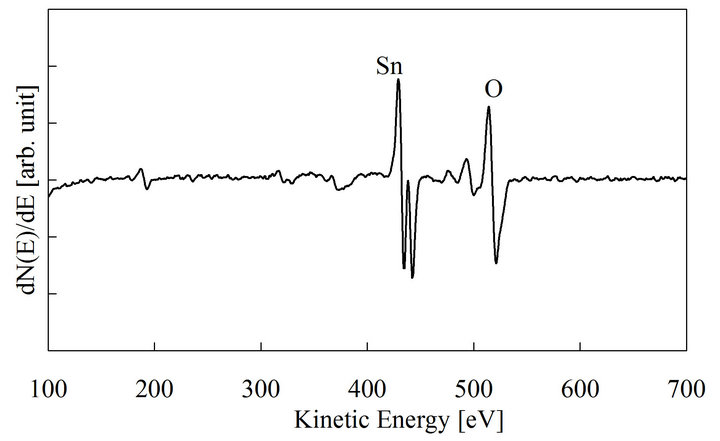
Figure 1. AES spectrum for sol-gel deposited SnO2.
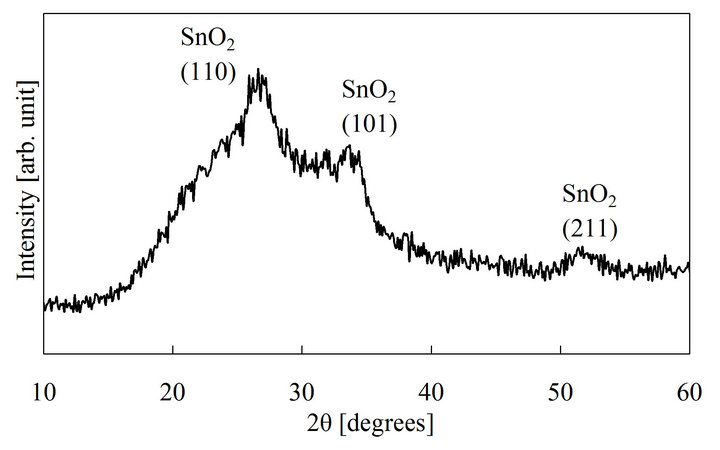
Figure 2. X-ray diffraction pattern of sol-gel deposited SnO2.
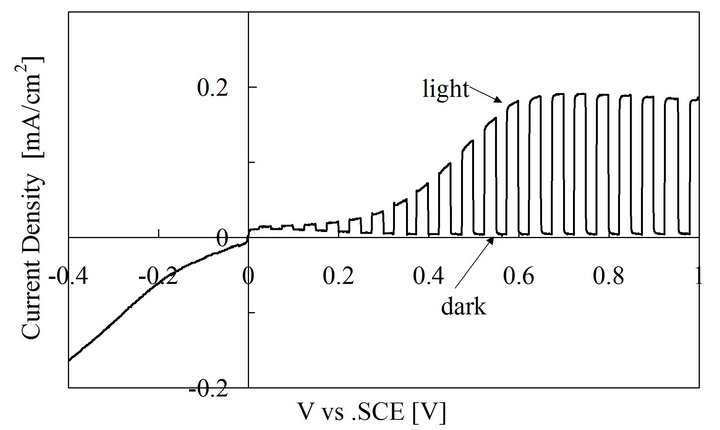
Figure 3. Photoelectrochemical measurement results for SnO2.
arrive at the interface of the semiconductor-electrolyte to participate in the electrochemical reaction at the film/ electrolyte interface. The current becomes more positive under the light illumination during the anodic scan. This implies that the minority carriers generated here are holes. Thus, the prepared film is n-type semiconductor.
The film thickness of ECD-Cu2O was about 0.7 μm and the direct band gap obtained from the optical transmission was about 2.5 eV. The PEC results for the Cu2O film are shown in Figure 4. The current was not changed by illumination for the anodic scan, and thus only the results for the cathodic scan are shown there. Negative photo current was observed, which implies that the minority carriers generated here are electrons. Thus, the prepared film is a p-type semiconductor with a good photosensitivity.
3.2. Cu2O/SnO2 Heterostructures
The heterostructures were fabricated with SnO2 as a window layer and Cu2O as an absorption layer. Figure 5 shows I-V characteristic of the Cu2O/SnO2 heterostructure cell fabricated by repeating the coating-drying cycle 3 times for the SnO2 deposition. The rectification and photovoltaic behaviors appeared. The heterostructure cells were fabricated with different repetition time of the coating-drying cycle, and the photovoltaic properties of all the samples are shown in table 1. The properties strongly depends on the repetition number of the coat-
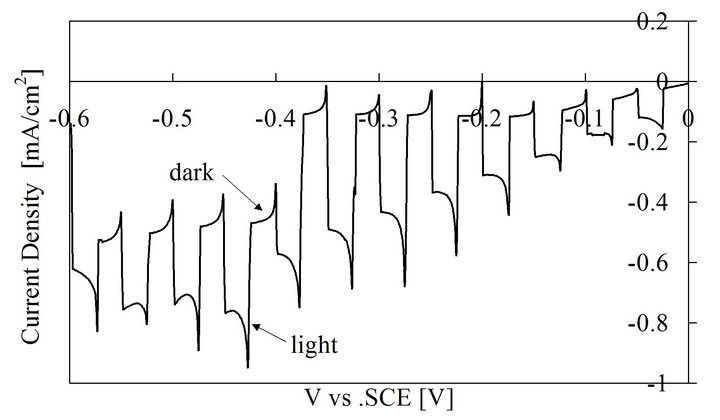
Figure 4. Photoelectrochemical measurement results for ECD-Cu2O.
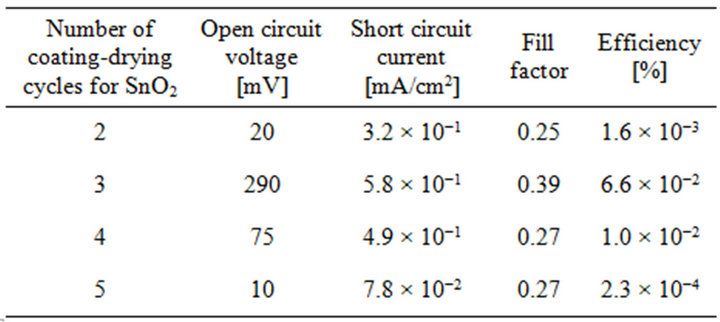
Table 1. Photovoltaic properties for the Cu2O/SnO2 heterostructure cells.
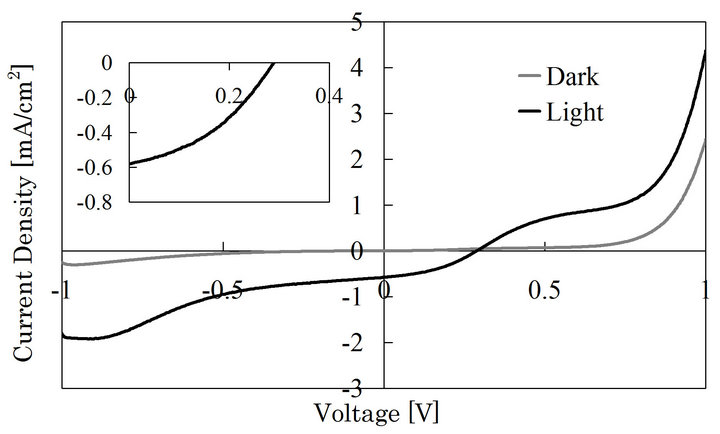
Figure 5. I-V characteristic of the heterostructure cell consisting of sol-gel deposited SnO2 and ECD-Cu2O. The coating-drying cycle was repeated 3 times in the sol-gel process. The inset is an expanded figure of photovoltaic properties.
ing-drying cycle, and the highest efficiency of 6.6 × 10−2 % was obtained with three times repetitions. The reason for dependence on the repetition number is not understood. The film thickness increases in proportion to the repetition number, and too large thickness may results in generation of defects such as crack because of strain.
Since each of the layers in the heterostructure showed fairly high photosensitivity as shown in Figures 3 and 4, the poor photovoltaic characteristics will be due to some disorder at the interface. Apparently, the SnO2 layer did not dissolve in the Cu2O deposition solution, but we cannot exclude possibility that the SnO2 surface reacted with the Cu2O deposition solution, to form a thin defective layer. Another possible reason of the poor performance is the Cu2O/electrode interface. The forward characteristics under the illumination is not a simple diode curve but has a plateau around 0.7 V. This may be due to non-ohmic characteristics of the interface with the electrode.
4. Conclusion
SnO2 films have been deposited by the sol-gel method. The films showed clear n-type conduction and photosensitivity. We have fabricated Cu2O/SnO2 heterostructure cells by depositing Cu2O by ECD on the sol-gel deposited SnO2 layer. Rectification property was observed, and the best cell showed an efficiency of 6.6 × 10−2 % under AM1.5 illumination.
5. Acknowledgements
We would like to thank Dr. M. Kato for the useful discussion.
REFERENCES
- C. Cobianu, C. Savaniu, P. Siciliano, S. Capone, M. Utriainen and L. Niinisto, “SnO2 Sol-Gel Derived Thin Films for Integrated Gas Sensors,” Sensors and Actuators B: Chemical, Vol. 77, No. 1, 2001, pp. 496-502.
- W. Schmid, N. Barsan and U. Weimar, “Sensing of Hydrocarbons with Tin Oxide Sensors: Possible Reaction Path as Revealed by Consumption Measurements,” Sensors and Actuators B: Chemical, Vol. 89, No. 3, 2003, pp. 232-236. doi:10.1016/S0925-4005(02)00470-7
- S.-C. Leen, J.-H. Lee, T.-S. Oh and Y.-H. Kim, “Fabrication of Tin Oxide Film by Sol-Gel Method for Photovoltaic Solar Cell System,” Solar Energy Materials and Solar Cells, Vol. 75, No. 3, 2003, pp. 481-487.
- Y. Wang, I. Ramos and J. J. Santiago-Aviles, “Optical Bandgap and Photoconductance of Electrospun Tin Oxide Nanofibers,” Journal of Applied Physics, Vol. 102, No. 9, 2007, Article ID: 093517.
- S. Shanthi, C. Subramanian and P. Ramasamy, “Growth and Characterization of Antimony Doped Tin Oxide Thin Films,” Journal of Crystal Growth, Vol. 197, No. 4, 1999, pp. 858-864.
- E. Shanthi, “Electrical and Optical Properties of Undoped and Antimony-Doped Tin Oxide Films,” Journal of Applied Physics, Vol. 51, No. 12, 1980, pp. 6243-6251. doi:10.1063/1.327610
- H. W. Lehmann and R. Widmer, “Preparation and Properties of Reactively Co-Sputtered Transparent Conducting Films,” Thin Solid Films, Vol. 27, No. 2, 1975, pp. 359- 368. doi:10.1016/0040-6090(75)90041-3
- K. Suzuki and M. Mizuhashi, “Structural, Electrical and Optical Properties of r.f.-Magnetron-Sputtered SnO2:Sb Film,” Thin Solid Films, Vol. 97, No. 2, 1982, pp. 119- 127. doi:10.1016/0040-6090(82)90221-8
- H. Ahna, J. H. Noha, S.-B. Kima, R. A. Overfelta, Y. S. Yoonb and D.-J. Kim, “Effect of Annealing and Argonto-Oxygen Ratio on Sputtered SnO2 Thin Film Sensor for Ethylene Gas Detection,” Materials Chemistry and Physics, Vol. 124, No. 1, 2010, pp. 563-568. doi:10.1016/j.matchemphys.2010.07.012
- D. Davazoglou, “Optical Properties of SnO2 Thin Films Grown by Atmospheric Pressure Chemical Vapour Deposition Oxiding SnCl4,” Thin Solid Films, Vol. 302, No. 1, 1997, pp. 204-213. doi:10.1016/S0040-6090(96)09601-0
- G. Sanon, A. Banerjee and A. Mansingh, “Growth and Characterization of Tin Oxide Films Prepared by Chemical Vapour Deposition,” Thin Solid Films, Vol. 190, No. 2, 1989, pp. 287-301. doi:10.1016/0040-6090(89)90918-8
- M. Maleki and S. M. Rozati, “Structural, Electrical and Optical Properties of Transparent Conducting SnO2 Films: Effect of the Oxygen Flow Rate,” Physica Scripta, Vol. 86, No. 1, 2012, Article ID: 015801. doi:10.1088/0031-8949/86/01/015801
- T. M. Racheva and G. W. Critchlow, “SnO2 Thin Films Prepared by the Sol-Gel Process,” Thin Solid Films, Vol. 292, 1997, pp. 299-302. doi:10.1016/S0040-6090(96)08956-0
- M. A. Dal Santos, A. C. Antunes, C. Ribeiro, C. P. F. Borges, S. R. M. Antunes, A. J. Zara and S. A. Pianaro, “Electric and Morphologic Properties of SnO2 Films Prepared by Modified Sol-Gel Process,” Materials Letters, Vol. 57, 2003, pp. 4378-4381. doi:10.1016/S0167-577X(03)00328-8
- J. Calderer, J. Esta, H. Luquet and M. Savelli, “Preparation and Characterization of SnO2 (Spray)/CdTe (n or p) Photovoltaic Cells,” Solar Energy Materials, Vol. 5, 1981, pp. 337-347. doi:10.1016/0165-1633(81)90003-4
- L. Panadimitrious, N. A. Economou and D. Trivich, “Heterojunction Solar Cells on Cuprous Oxide,” Solar Cells, Vol. 3, 1981, pp. 73-80. doi:10.1016/0379-6787(81)90084-3
- M. Ristov, G. Sinadinovski, M. Mitreski and M. Ristova, “Photovoltaic Cells Based on Chemically Deposited pType SnS,” Solar Energy Materials and Solar Cells, Vol. 69, 2001, pp. 17-24. doi:10.1016/S0927-0248(00)00355-X
- J. J. M. Vequizo and M. Ichimura, “Fabrication of Electrodeposited SnS/SnO2 Heterojunction Solar Cells,” Japanese Journal of Applied Physics, Vol. 51, 2012, Article ID: 10NC38-1-4. doi:10.1143/JJAP.51.10NC38
- M. Izaki, T. Shinagawa, K. Mizuno, Y. Ida, M. Inaba and A. Tasaka, “Electrochemically Constructed p-Cu2O/n-ZnO Heterojunction Diode for Photovoltaic Device,” Journal of Physics D: Applied Physics, Vol. 40, 2007, pp. 3326- 3330. doi:10.1088/0022-3727/40/11/010
- K. Akimoto, S. Ishizuka, M. Yanagita, Y. Nawa, G. K. Paul and T. Sakurai, “Thin Film Deposition of Cu2O and Application for Solar Cells,” Solar Energy, Vol. 80, No. 6, 2006, pp. 715-722. doi:10.1016/j.solener.2005.10.012
- J. Katayama, K. Ito, M. Matsuoka and J. Tamaki, “Performance of Cu2O/ZnO Solar Cell Prepared by Two-Step Electrodeposition,” Journal of Applied Electrochemistry, Vol. 34, No. 7, 2004, pp. 687-692. doi:10.1023/B:JACH.0000031166.73660.c1
- Y. Luo, L. Wang, Y. Zou, X. Sheng, L. Chang and D. Yang, “Electrochemically Deposited Cu2O on TiO2 Nanorod Arrays for Photovoltaic Application,” Electrochemical and Solid-State Letters, Vol. 15, No. 2, 2012, pp. H34-H36. doi:10.1149/2.016202esl
- A. R. Zainun, T. Sakamoto, U. M. Noor, M. Rusop and M. Ichimura, “New Approach for Generating Cu2O/TiO2 Composite Films for Solar Cell Applications,” Materials Letters, Vol. 66, 2012, pp. 254-256. doi:10.1016/j.matlet.2011.08.032
- V. Georgieva and M. Ristov, “Electrodeposited Cuprous Oxide on Indium Tin Oxide for Solar Applications,” Solar Energy Materials and Solar Cells, Vol. 73, 2002, pp. 67-73. doi:10.1016/S0927-0248(01)00112-X

