Journal of Modern Physics
Vol.5 No.14(2014), Article
ID:49006,11
pages
DOI:10.4236/jmp.2014.514122
Magnetic Hysteresis and Complex Initial Permeability of Cr3+ Substituted Mn-Zn Ferrites
F. Alamwang#_s:sup1*, M. L. Rahman2, M. H. R. Khan3, A. K. M. Akther Hossain4
1Department of Physical Sciences, School of Engineering and Computer Science, Independent University, Dhaka, Bangladesh
2Department of Mathematics and Natural Sciences, BRAC University, Dhaka, Bangladesh
3Department of Arts and Sciences, Ahsanullah University of Science and Technology, Dhaka, Bangladesh
4Departments of Physics, Bangladesh University of Engineering and Technology, Dhaka, Bangladesh
Email: *farhadiub@gmail.com
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 20 May 2014; revised 18 June 2014; accepted 11 July 2014
ABSTRACT
The impact of Cr3+ ion on the magnetic properties of Mn0.50Zn0.50CrxFe2−xO4 (with x = 0.0, 0.1, 0.2, 0.3, 0.4 and 0.5) has been studied. Ferrite samples were synthesized by combustion method and sintered at various temperatures (1250˚C, 1300˚C and 1350˚C). The structural properties were investigated by means of X-ray diffraction patterns and indicated that the samples possess single phase cubic spinel structure. The lattice parameter decreases with the increase in Cr3+ content, as the ionic radius of Cr3+ ion is smaller than that of Fe3+. The average grain size (D), bulk density (ρB) and initial permeability  decreases with increase in Cr3+ content whereas porosity follows its opposite trend. The ρB was found to increase with increase in Cr3+ content as the sintering temperature (Ts) is increased from 1250˚C to 1350˚C. The Ts affects the densification, grain growth and
decreases with increase in Cr3+ content whereas porosity follows its opposite trend. The ρB was found to increase with increase in Cr3+ content as the sintering temperature (Ts) is increased from 1250˚C to 1350˚C. The Ts affects the densification, grain growth and  of the samples. The
of the samples. The 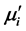 strongly depends on average grain size, density and intragranular porosity. The B-H loops of the compositions were measured at room temperature. The saturation magnetization (Ms), coercivity (Hc) and hysteresis losses were studied as a function of Cr3+ content. The Ms was found to decrease with the increase of Cr3+ content, which is attributed to the dilution of A-B interaction.
strongly depends on average grain size, density and intragranular porosity. The B-H loops of the compositions were measured at room temperature. The saturation magnetization (Ms), coercivity (Hc) and hysteresis losses were studied as a function of Cr3+ content. The Ms was found to decrease with the increase of Cr3+ content, which is attributed to the dilution of A-B interaction.
Keywords:Mn-Zn Ferrites, Initial Permeability, Saturation Magnetization, Hysteresis Loss

1. Introduction
Polycrystalline mixed spinel ferrites are very attractive for their various potential applications. Manganese zinc and substituted manganese zinc ferrites are technologically important materials due to their high magnetic permeability, high saturation magnetization, high resistivity and low power losses. These ferrites have been extensively used in electronic applications such as transformers, choke coils, noise filters, recording heads, multilayer chip inductor, electromagnetic wave absorbers etc. [1] -[5] . Crystallogarphically, spinel ferrites have tetrahedral A-site and octahedral B-site in AB2O4 crystal structure. It shows various magnetic and electrical properties depending on the type of cations and their distribution between the two interstitial sites. Desired electrical and magnetic properties of soft ferrites can be tailored by controlling the different types and amount of substitutes and by choosing suitable synthesize technique [6] -[9] . A large number of mixed ferrites like Mn-In-Zn [10] , Ni-Mn-Zn [11] , Mg-Mn-In and Mg-Mn-Cr [12] , Ni-Zn-Cr [13] , Cd-Mg-Cr [14] have been prepared by various processing techniques and their structural, electrical and magnetic properties have been reported. Properties of ferrites are sensitive to the processing technique which can play a very crucial role in defining the structural and magnetic properties. Many researchers have studied the effect of the substitution of Cr3+ in the spinel structure of ferrites [15] -[17] . However, there are no reports in the literature about Cr3+ substitution for Fe3+ in Mn-Zn ferrites obtained by combustion technique. Compared with the other synthesization methods, the combustion method offers advantages of being fast and simple, short preparation duration and low energy consumption. The present paper therefore reports on a study of Cr3+ substituted Mn-Zn ferrite produced by combustion synthesis, and on its structural and magnetic characterization.
2. Experimental
The nominal chemical compositions of Mn0.50Zn0.50CrxFe2−xO4 (with x = 0.0, 0.1, 0.2, 0.3, 0.4 and 0.5) were synthesized by combustion method. All the raw materials used in this reaction were of analytical grade. The proper amounts of powders of Cr(NO3)3·9H2O, Zn(NO3)2·6H2O, MnCl2·4H2O and Fe(NO3)3·9H2O were dissolved in ethanol (GR grade). Then the solution were placed at a constant temperature bath (~70˚C) followed by an ignition and formed a fluffy loose product of the desired composition. The resultant powders were calcined at 900˚C for five hours in air. The grounded fine powders were mixed with binder, and then pressed into diskand toroid-shaped samples. The samples prepared from each composition were sintered at 1250˚C, 1300˚C and 1350˚C for five hours in air. During sintering, temperature ramps were 10˚C/min for heating and 5˚C/min for cooling. Surfaces of all sintered samples were polished and thermally etched. The crystalline structure and phase of the compositions were identified using X-ray diffraction (XRD) patterns obtained using CuKα radiation (λ = 1.54178Å) for 2θ value. The lattice parameter for each composition was determined using Nelson-Riley function [18] . The ρB, of the samples were determined using the Archimedes principle. The theoretical density (ρth) was calculated using the expression: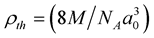 , where NA is Avogadro’s number, M is the molecular weight of the composition and a0 is the lattice parameter. The porosity (P) was calculated from the relation
, where NA is Avogadro’s number, M is the molecular weight of the composition and a0 is the lattice parameter. The porosity (P) was calculated from the relation
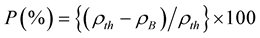 . The samples were visualized under a high-resolution optical microscope (Olympus DP-70) and then photographed. Average grain sizes of the samples were determined from optical micrographs by linear intercept technique. The frequency dependent initial permeability spectra were determined using Agilent Impedance analyzer (Agilent 4294A) on toroid shaped samples. 4 turns of enameled copper wire were wound on the toroid and the inductance (Ls) was measured at room temperature in the frequency range 20 Hz - 120 MHz. The
. The samples were visualized under a high-resolution optical microscope (Olympus DP-70) and then photographed. Average grain sizes of the samples were determined from optical micrographs by linear intercept technique. The frequency dependent initial permeability spectra were determined using Agilent Impedance analyzer (Agilent 4294A) on toroid shaped samples. 4 turns of enameled copper wire were wound on the toroid and the inductance (Ls) was measured at room temperature in the frequency range 20 Hz - 120 MHz. The  and the imaginary part
and the imaginary part 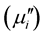 of the complex initial permeability were calculated using the following relations
of the complex initial permeability were calculated using the following relations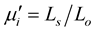 , and
, and , where
, where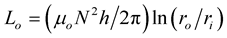 .
.  is the inductance of the winding coil without the sample core, N is the number of turns of the coil , h is the thickness,
is the inductance of the winding coil without the sample core, N is the number of turns of the coil , h is the thickness,  is the outer radius and
is the outer radius and 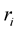 is the inner radius of the toroid-shaped sample. The relative quality factor, Q was calculated from the relation:
is the inner radius of the toroid-shaped sample. The relative quality factor, Q was calculated from the relation: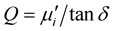 , where tand is the loss factor. The DC magnetization measurements were made using the SQUID magnetometer (MPMS-5S; Quantum design Co. Ltd.). B-H loops measurements were performed at room temperature (20˚C) using an Automatic Magnetic Hysteresis Graph Tracer (Model no. AMH-300, Laboratorio Electrofisico, Italy). The B-H loops were traced from the toroid shaped specimens with primary and secondary windings of 40:10 ratio of enamelled copper wire using a computer assisted hysteresis loop tracer at a constant frequency 1 kHz and sufficient high applied field to get magnetic saturation.
, where tand is the loss factor. The DC magnetization measurements were made using the SQUID magnetometer (MPMS-5S; Quantum design Co. Ltd.). B-H loops measurements were performed at room temperature (20˚C) using an Automatic Magnetic Hysteresis Graph Tracer (Model no. AMH-300, Laboratorio Electrofisico, Italy). The B-H loops were traced from the toroid shaped specimens with primary and secondary windings of 40:10 ratio of enamelled copper wire using a computer assisted hysteresis loop tracer at a constant frequency 1 kHz and sufficient high applied field to get magnetic saturation.
3. Results and Discussion
3.1. Lattice Parameter, Density and Porosity
Figure 1 depicts the X-ray diffraction patterns of various chromium-substituted Mn-Zn ferrites. The patterns exhibit typical reflection of (111), (220), (311), (222), (400), (422), (511) and (440) planes indicating the formation of cubic spinel structure of manganese ferrite. All the diffraction peaks comply with the standard peaks of cubic spinel ferrites. No secondary peaks were detected in XRD patterns of above mentioned samples which ensured the phase purity of each composition. The lattice parameter was determined by using the Nelson-Riley function, which can be expressed as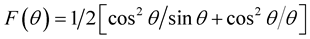 , where
, where 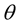 is the Bragg’s angle. The exact values of lattice parameter of each sample was estimated from the extrapolation of all peaks to
is the Bragg’s angle. The exact values of lattice parameter of each sample was estimated from the extrapolation of all peaks to 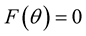 or
or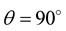 . As the Cr3+ substitution increases, the lattice parameter decreases as shown in Figure 2. It is due to the fact that the ionic radius of substituting Cr3+ (0.62Å) is less than that of Fe3+ (0.64Å) [19] [20] . Taking into consideration the preference of Cr3+ to the octahedral site, the partial replacement of Fe3+ by Cr3+causes a slight effect on the unit cell dimensions [21] . Similar result has been reported in Cr3+ doped Ni-Zn ferrite prepared by combustion synthesis [13] . The lattice parameter, estimated from XRD patterns is tabulated in Table 1 along with density, porosity, average grain size and relaxation frequency for various compositions.
. As the Cr3+ substitution increases, the lattice parameter decreases as shown in Figure 2. It is due to the fact that the ionic radius of substituting Cr3+ (0.62Å) is less than that of Fe3+ (0.64Å) [19] [20] . Taking into consideration the preference of Cr3+ to the octahedral site, the partial replacement of Fe3+ by Cr3+causes a slight effect on the unit cell dimensions [21] . Similar result has been reported in Cr3+ doped Ni-Zn ferrite prepared by combustion synthesis [13] . The lattice parameter, estimated from XRD patterns is tabulated in Table 1 along with density, porosity, average grain size and relaxation frequency for various compositions.
Figure 3 shows the effects of Cr3+ substitutions on bulk density and porosity in Mn0.50Zn0.50CrxFe2−xO4 ferrites. The ρB decreases with increase in Cr3+ content, while porosity increases for Cr3+ substitutions. The decrease in ρB can be attributed to the difference in atomic weight and density of the ferrite components. The atomic weight and density of Cr3+ (51.996 amu and 7.19 g/cm3) is less than that of Fe3+ (55.85 amu and 7.87 g/cm3) [22] . The
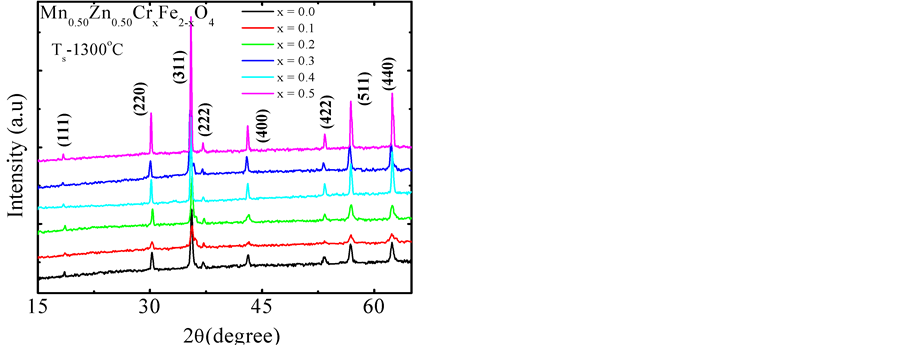
Figure 1. The XRD patterns for Mn0.50Zn0.50CrxFe2−xO4 with x = 0.0, 0.1, 0.2, 0.3, 0.4 and 0.5.
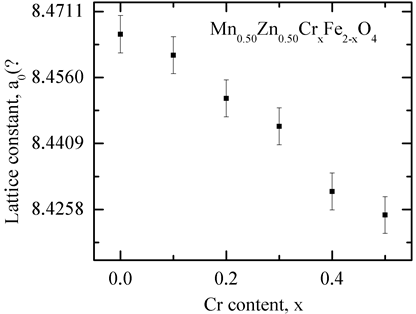
Figure 2. The variation of lattice parameter with Cr3+ content (x) for various Mn0.50Zn0.50CrxFe2−xO4 samples.
Table 1. The lattice parameter, density, porosity, average grain size, real part of initial permeability, coercive field, and room temperature saturation magnetization of various Mn0.50Zn0.50CrxFe2−xO4 sintered at various temperatures in air for 5 h.

Figure 3. The variation of bulk density and porosity as a function of Cr3+ content (x) for various Mn0.50Zn0.50CrxFe2−xO4 sintered at 1300˚C.
increase in porosity with the addition of Cr3+ content may be due to the creation of more cation vacancies with the reduction of oxygen vacancies [23] . It is found that the bulk densities are smaller in magnitude than corresponding theoretical densities. This may be due to the presence of pores in the samples [24] .
Figure 4 shows the variation of ρB and P for Mn0.50Zn0.50Cr0.20Fe1.8O4 sintered at various sintering temperatures. It is noticed that the ρB of Mn0.50Zn0.50CrxFe2−xO4 (x = 0.20) increases with increase in sintering temperature. On the other hand, porosity has the opposite trend. It is also noticed that the ρB for all other compositions increases with increase in sintering temperature, while the porosity of these compositions exhibit opposite behavior which has been tabulated in the Table1 The increases in density with sintering temperature are expected.
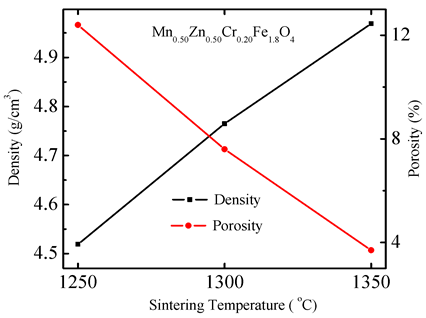
Figure 4. The variation of bulk density and porosity with sintering temperature for Mn0.50Zn0.50Cr0.20Fe1.8O4.
This is because during the sintering process, the thermal energy generates a force that drives the grain boundaries to grow over pores, thereby decreasing the pore volume and densifying the material.
3.2. Microstructure of Mn0.50Zn0.50CrxFe2−xO4
Magnetic properties of ferrites are dependent on their microstructures. Grain size is more important parameter affecting the magnetic properties of ferrites. Grain growth is closely related to the grain boundary mobility. During grain growth there is a competition between the driving force for grain boundary movement and the retarding force exerted by the pores [25] . When the driving force of the grain boundary in each grain is homogeneous, the sintered body attains a uniform grain size distribution. Discontinuous grain growth occurs if this driving force is inhomogeneous. The optical micrographs of Mn0.50Zn0.50CrxFe2−xO4 ferrites are shown in Figure 5. Average grain sizes of the samples determined by the linear intercept technique are presented in Table1 The average grain sizes of the samples vary from 5.7 µm to 3.2 µm. The grain size decreases with increasing Cr3+ substitution. This may be due to the fact that the melting point of Cr (1914˚C) is greater than that of iron (1538˚C). With the substitution of chromium, the appearance of smaller grains has been observed and when the substitution level is higher the population of smaller grains is dominating over the larger grains. The size of the bigger as well as smaller grains is decreased when the Cr3+ concentration is higher. From the microstructures, it is also observed that the porosity is increased when the Cr3+ content is increased [26] .
3.3. Magnetic Properties of Mn0.50Zn0.50CrxFe2−xO4
3.3.1. Frequency Dependence of Complex Permeability
The complex initial permeability spectra for all Mn0.50Zn0.50CrxFe2−xO4 ferrite compositions sintered at various sintering temperatures as a function of frequency are shown in Figure 6. The  decrease with increase in Cr3+ substitution in Mn0.50Zn0.50CrxFe2−xO4 ferrites. Similar trend is followed in Cr3+ substituted Li-Sb ferrites [26] .
decrease with increase in Cr3+ substitution in Mn0.50Zn0.50CrxFe2−xO4 ferrites. Similar trend is followed in Cr3+ substituted Li-Sb ferrites [26] .
The 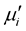 remains steady up to a certain frequency, known as relaxation frequency (fr). It is also noticed that there is a decrease in
remains steady up to a certain frequency, known as relaxation frequency (fr). It is also noticed that there is a decrease in 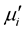 and increase in
and increase in 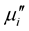 above the fr. The
above the fr. The 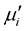 of the samples decrease but the fr shifted to higher values as result of Cr3+ substitutions at higher sintering temperature. The
of the samples decrease but the fr shifted to higher values as result of Cr3+ substitutions at higher sintering temperature. The 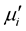 of a ferromagnetic material depends on many factors like reversible displacement of domain walls, bulging of domain walls as well as microstructural parameters like average grain size, intragranular porosity, etc. [27] . Perduijin and Peloschek [28] and Roess et al. [29] found a linear relation between the
of a ferromagnetic material depends on many factors like reversible displacement of domain walls, bulging of domain walls as well as microstructural parameters like average grain size, intragranular porosity, etc. [27] . Perduijin and Peloschek [28] and Roess et al. [29] found a linear relation between the  and grain size in Mn-Zn ferrites. The
and grain size in Mn-Zn ferrites. The  being sensitive to many parameters, it is still difficult to draw a specific conclusion for variation of
being sensitive to many parameters, it is still difficult to draw a specific conclusion for variation of 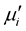 with concentration. However
with concentration. However 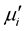 is found to decreases with decreasing of grain size. The
is found to decreases with decreasing of grain size. The 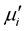 of ferrite follows
of ferrite follows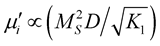 , where Ms is the saturation magnetization is,
, where Ms is the saturation magnetization is,  is the magnetocrystalline anisotropy constant and D is the average grain diameter. The decrease in
is the magnetocrystalline anisotropy constant and D is the average grain diameter. The decrease in  with increase in Cr3+ substitution can be attributed to the decrease in grain size and Ms which has been discussed in DC magnetization section. Decrease in grain size also results in a decrease in the number of domain walls in each grain. The onset of relaxation frequency (due to domain wall oscillations) is drifting towards the higher side with the increase in Cr3+ concentration
with increase in Cr3+ substitution can be attributed to the decrease in grain size and Ms which has been discussed in DC magnetization section. Decrease in grain size also results in a decrease in the number of domain walls in each grain. The onset of relaxation frequency (due to domain wall oscillations) is drifting towards the higher side with the increase in Cr3+ concentration
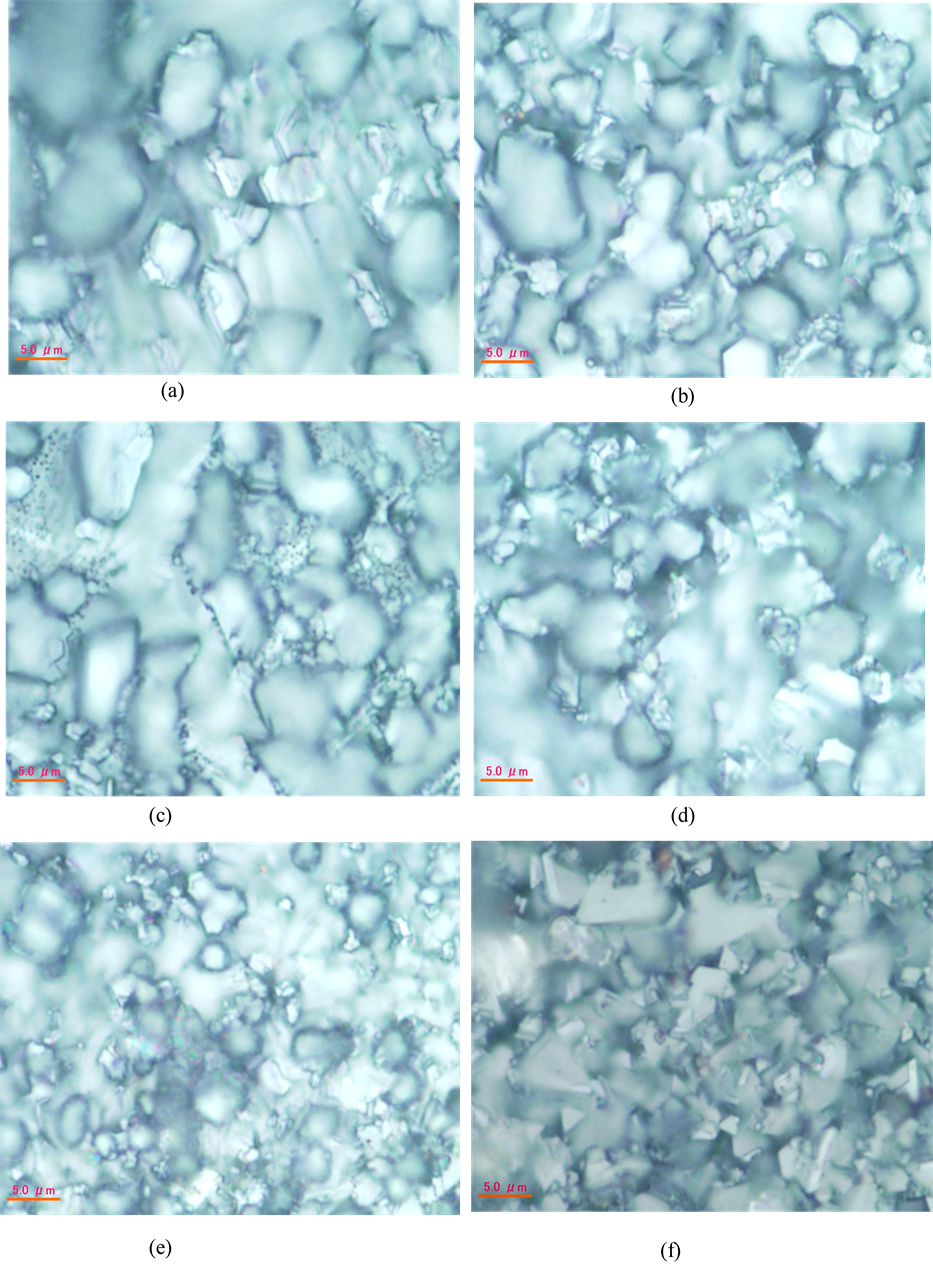
Figure 5. The optical micrographs of Mn0.50Zn0.50CrxFe2−xO4 ferrites: (a) x = 0.0; (b) x = 0.1; (c) x = 0.2; (d) x = 0.3; (e) x = 0.4 and (f) x = 0.5 sintered at 1250˚C.
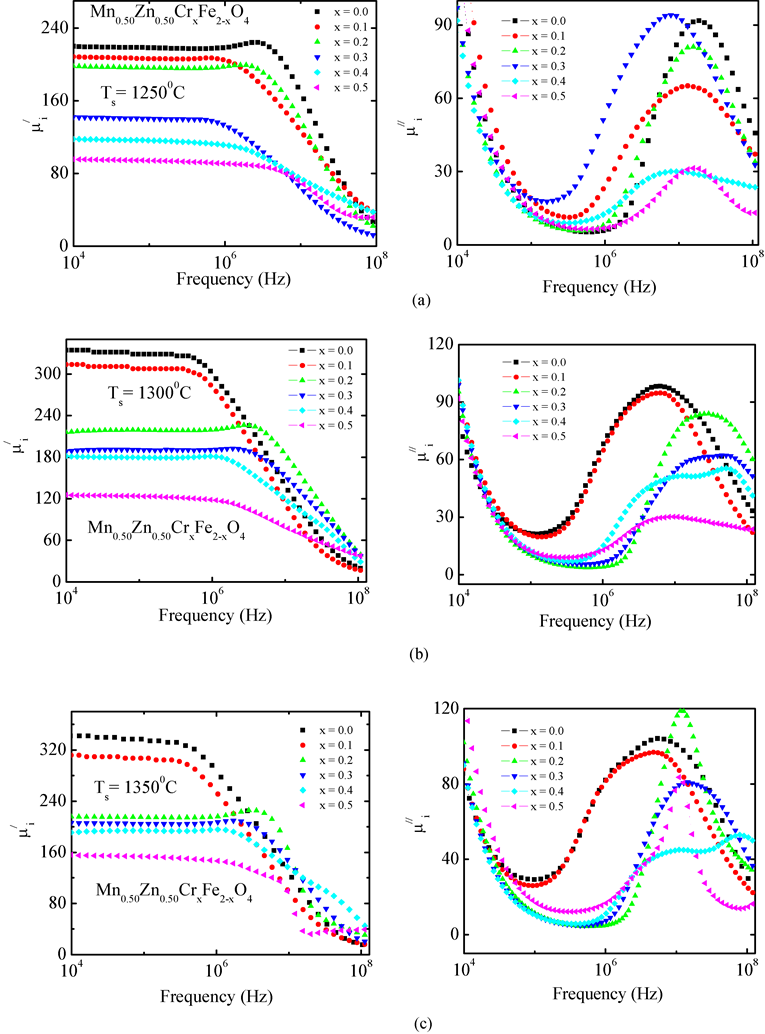
Figure 6. The variation of  and
and 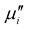 with frequency for Mn0.50Zn0.50CrxFe2−xO4 samples sintered at temperatures (a) 1250 (b) 1300 and (c) 1350˚C in air.
with frequency for Mn0.50Zn0.50CrxFe2−xO4 samples sintered at temperatures (a) 1250 (b) 1300 and (c) 1350˚C in air.
or decrease in . This agrees well with the Globus model [30] .
. This agrees well with the Globus model [30] .
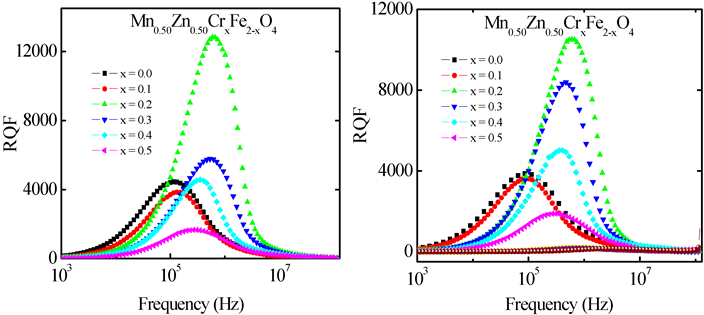
3.3.2. Frequency Dependence of Relative Quality Factor
Figure 7 shows the frequency dependence of relative quality factor (RQF) of the samples sintered at (a) 1300˚C and (b) 1350˚C. The RQF increases with an increase in frequency, showing a peak and then decreases. The variation of RQF with frequency showed a similar trend for all the samples. It is seen that RQF decreases beyond 0.2 MHz i.e. the loss tangent is minimum up to 0.2 MHz and then it rises rapidly. The loss is due to lag of domain wall motion with the applied alternating magnetic field and is attributed to various domain effect, which include non-uniform and non-repetitive domain wall motion, domain wall bowing, localized variation of flux density, nucleation and annihilation of domain wall [31] . The peak corresponding to maxima in quality factor shifts to higher frequency as Cr3+ content increases up to x = 0.20, after that it decreases. RQF has the maximum value for Mn0.50Zn0.50Cr0.20Fe1.8O4 sintered at 1300˚C.
3.3.3. DC Magnetization
The magnetization (M) as a function of applied magnetic field (H) for various Mn0.50Zn0.50CrxFe2−xO4 (with x = 0.0, 0.1, 0.2, 0.3, 0.4 and 0.5) measured at room temperature (300 K) are shown in Figure 8. The magnetization of all samples increases linearly with increasing the applied magnetic field up to 0.1 T. Beyond this applied field magnetization increases slowly and then saturation occurs. The saturation magnetization, Ms, for all sample are determined by the extrapolation of magnetization curve to .
.
L. Néel [32] considered three kinds of exchange interactions between unpaired electrons of two ions lying: (i) both ions at A sites (AA interaction), (ii) both ions at B sites (BB interaction), and (iii) one at a site and the other at B site (AB interaction). AB interaction strongly predominates over AA and BB interactions. The AB interaction aligns all the magnetic spins at A site in one direction and those at B site in opposite direction. The net magnetic moment of the lattice is therefore the difference between the magnetic moments of B and A sublattices, i.e, M = MB − MA. The saturation magnetization is observed to decrease with increasing paramagnetic Cr3+ content throughout the concentration range studied. It is well known that Cr3+ ions preferably occupy B sites [33] . As the number of Fe3+ ions at B sites continuously decreases, the magnetization of B sub lattice decreases which results into the observed decrease in saturation magnetization. A similar decrease was reported in Mn-Zn-Cr [34] and Mg-Mn-Cr ferrites [35] .
 (a) (b)
(a) (b)
Figure 7. The variation of Relative Quality Factor (RQF) with frequency for Mn0.50Zn0.50CrxFe2−xO4 samples sintered at (a) 1300 and (b) 1350ºC.

Figure 8. The magnetization (M) versus the applied magnetic field (H) curves for Mn0.50Zn0.50CrxFe2−xO4 at room temperature.

Figure 9. B-H loops of polycrystalline Mn0.50Zn0.50CrxFe2−xO4 ferrites at room temperature. The inset B-H loops at low field.
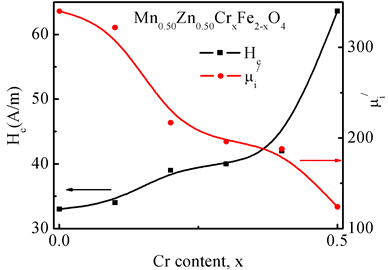
Figure 10. The variation of Hc and 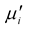 for various Mn0.50Zn0.50CrxFe2−xO4 for various Cr3+ content.
for various Mn0.50Zn0.50CrxFe2−xO4 for various Cr3+ content.
The hysteresis loops of the Mn-Zn-Cr ferrite were recorded by a computer interfaced hysteresis loop tracer at room temperature at constant frequency f = 1 kHz. Figure 9 shows the B-H loops of the samples of the composition Mn0.50Zn0.50CrxFe2−xO4 sintered at 1300˚C. The inclusion of Cr3+ enhances the hysteresis loop, which in turn increased coercive field (Hc), favoring its application in high frequency transformer. The addition of Cr3+ reduced the Ms as expected as it reduces the density of magnetic ions at B site. Hc increases due to the increase of magnetocrystalline anisotropy [36] . It is well known that the shape and size of a B-H loop depends not only on the chemical composition but also on several microstructural properties like grain size, porosity and nature of the pores [37] . Hc is inversely proportional to initial permeability  of the ferrite system as shown in Figure 10. Our present observations of compositional variation of Hc with
of the ferrite system as shown in Figure 10. Our present observations of compositional variation of Hc with  agree with the reported value [38] [39] .
agree with the reported value [38] [39] .
4. Conclusion
Substitution of Cr3+ in Mn-Zn ferrite results in slight shrinkage of the unit cell. D, ρB,  , as well as Ms decrease with Cr3+ content. As
, as well as Ms decrease with Cr3+ content. As  decreases the
decreases the  of the samples shifts towards the higher frequency up to a particular Cr3+ content. The
of the samples shifts towards the higher frequency up to a particular Cr3+ content. The 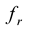 indicates the operational frequency limit of the ferrites in applications, so it is preferable to push this frequency to the higher frequency region. In our present investigation operational frequency range is increased with a particular Cr3+ substitution. Saturation magnetization decreased with Cr3+ content whereas Hc was found to increase at the Cr3+ rich Mn-Zn ferrites.
indicates the operational frequency limit of the ferrites in applications, so it is preferable to push this frequency to the higher frequency region. In our present investigation operational frequency range is increased with a particular Cr3+ substitution. Saturation magnetization decreased with Cr3+ content whereas Hc was found to increase at the Cr3+ rich Mn-Zn ferrites.
Acknowledgements
F. Alam would like to acknowledge CASR of Bangladesh University of Engineering and Technology for financing and Bangladesh Atomic Energy Center, Dhaka for providing B-H loop data. Thanks are also due to M. A. Rahman for valuable discussions for preparation of the manuscript.
References
- Syue, M.-R., Wei, F.-J., Chou, C.-S. and Fu, C.-M. (2011) Journal of Applied Physics, 109, Article ID: 07A324. http://dx.doi.org/10.1063/1.3560880
- Praveena, K., Sadhana, K., Bharadwaj, S. and Murthy, S.R. (2010) Materials Research Innovations, 14, 56-61. http://dx.doi.org/10.1179/143307510X12599329343727
- Praveena, K., Sadhana, K., Bharadwaj, S. and Murthy, S.R. (2009) Journal of Magnetism and Magnetic Materials, 321, 2433-2437. http://dx.doi.org/10.1016/j.jmmm.2009.02.138
- Subramani, A.K., Kondo, K., Tada, M., Abe, M., Yoshimura, M. and Matsushita, N. (2009) Journal of Magnetism and Magnetic Materials, 321, 3979-3983. http://dx.doi.org/10.1016/j.jmmm.2009.07.036
- Latorre-Esteves, M., Cortes, A., Torres-Lugo, M. and Rinaldi, C. (2009) Journal of Magnetism and Magnetic Materials, 321, 3061-3066. http://dx.doi.org/10.1016/j.jmmm.2009.05.023
- Albuquerqe, A.S., Ardisson, J.D., Macedo, W.A.A. and Alves, M.C.M. (2000) Journal of Applied Physics, 87, 4352-4357. http://dx.doi.org/10.1063/1.373077
- Selvan, R.K., Augustin, C.O., Berchmans, L.J. and Saraswathi, R. (2003) Materials Research Bulletin, 38, 41-54. http://dx.doi.org/10.1016/S0025-5408(02)01004-8
- Li, F., Liu, J.J., Evans, D.G. and Duan, X. (2004) Chemistry of Materials, 16, 1597-1602. http://dx.doi.org/10.1021/cm035248c
- Liu, J.H., Wang, L. and Li, F.S. (2005) Journal of Materials Science, 40, 2573-2575. http://dx.doi.org/10.1007/s10853-005-2077-6
- Mathur, P., Thakur, A. and Singh, M. (2008) Journal of Physics and Chemistry of Solids, 69, 187-192. http://dx.doi.org/10.1016/j.jpcs.2007.08.014
- Singh, A.K., Singh, A.K., Goel, T.C. and Mendiratta, R.G. (2004) Journal of Magnetism and Magnetic Materials, 281, 276-280. http://dx.doi.org/10.1016/j.jmmm.2004.04.115
- Laksman, A., Rao, K.H. and Mendiratta, R.G. (2002) Journal of Magnetism and Magnetic Materials, 250, 92-97. http://dx.doi.org/10.1016/S0304-8853(02)00359-1
- Gama, L., Hernandez, E.P., Cornejo, D.R., Costa, A.A., Rezende, S.M., Kiminami, R.H.G.A. and Costa, A.C.F.M. (2007) Journal of Magnetism and Magnetic Materials, 317, 29-33. http://dx.doi.org/10.1016/j.jmmm.2007.04.007
- Masti, S.A., Sharma, A.K., Vasambekar, P.N. and Vainganker, A.S. (2006) Journal of Magnetism and Magnetic Materials, 305, 436-439. http://dx.doi.org/10.1016/j.jmmm.2006.01.229
- El-Sayed, A.M. (2003) Materials Chemistry and Physics, 82, 583-587. http://dx.doi.org/10.1016/S0254-0584(03)00319-5
- Sankpal, A.M., Suryavanshi, S.S., Kakatkar, S.V., Tengshe, G.G., Patil, R.S., Chaaudhari, N.D. and Sawant, S.R. (1998) Journal of Magnetism and Magnetic Materials, 186, 349-356. http://dx.doi.org/10.1016/S0304-8853(97)01156-6
- Rao, K.H., Gaur, N.K., Aggarwal, K. and Mendiratta, R.G. (1982) Journal of Applied Physics, 53, 1122-1126. http://dx.doi.org/10.1063/1.330525
- Nelson, J.B. and Riley, D.P. (1945) Proceedings of the Physical Society, London, 57, 160-177. http://dx.doi.org/10.1088/0959-5309/57/3/302
- Shanon, R.D. (1976) Acta Crystallographica Section A, 32, 751-767. http://dx.doi.org/10.1107/S0567739476001551
- Chae, K.P., Lee, Y.B., Lee, J.G. and Lee, S.H. (2000) Journal of Magnetism and Magnetic Materials, 220, 59-64. http://dx.doi.org/10.1016/S0304-8853(00)00459-5
- Gabal, M.A. and Al Angari, Y.M. (2006) Journal of Magnetism and Magnetic Materials, 322, 3159-3165. http://dx.doi.org/10.1016/j.jmmm.2010.05.054
- Kittel, C. (1976) Introduction to Solid State Physics. 5th Edition, John Wiley & Sons, Inc., New York.
- Abbas, T., Islam, M.U. and Ashraf, Ch.M. (1995) Modern Physics Letters B, 9, 1419-1426.
- Barakat, M.M., Hanaish, M.A., Olofa, S.A. and Tawfik, A. (1991) Journal of Thermal Analysis, 37, 241-248. http://dx.doi.org/10.1007/BF02055926
- Bellad, S.S., Watawe, S.C. and Chougule, B.K. (1999) Journal of Magnetism and Magnetic Materials, 195, 57-64. http://dx.doi.org/10.1016/S0304-8853(98)01073-7
- Laishram, R. and Prakash, C. (2006) Journal of Magnetism and Magnetic Materials, 305, 35-39. http://dx.doi.org/10.1016/j.jmmm.2005.11.037
- Smit, J. and Wijn, H.P.J. (1959) Ferrites. Philips Technical Library, Eindhoven, 150.
- Perduijn, D.J. and Peloschek, H.P. (1968) Proceedings of the British Ceramic Society, 10, 263.
- Roess, E., Hanke, I. and Moser, E. (1964) Zeitschrift für Angewandte Physik, 17, 504.
- Globus, A., Duplex, P. and Guyot, M. (1971) IEEE Transactions on Magnetics, 7, 617-622. http://dx.doi.org/10.1109/TMAG.1971.1067200
- Overshott, K.J. (1981) IEEE Transactions on Magnetics, 17, 2698-2700. http://dx.doi.org/10.1109/TMAG.1981.1061648
- Neel, L. (1948) Annales de Physique, 3, 137-198.
- Rameijin, F.C. (1953) Philips Research Reports, 8, 321.
- Rao, K.H. (1981) Electrical, Magnetic and Mossbauer Studies in Cr, In and Al Doped Zinc-Manganese Ferrites. Ph.D. Thesis, Andhra University, Visakhapatnam.
- Guilland, C. and Creveaux, H. (1950) Comptes Rendus de l'Académie des Sciences, 230, 1256. (France)
- Alone, S.T., Shirsath, S.E., Kadam, R.H. and Jadhav, K.M. (2011) Journal of Alloys and Compounds, 509, 5055-5060. http://dx.doi.org/10.1016/j.jallcom.2011.02.006
- Singh, A.K., Goel, T.C., Mendiratta, R.G., Thakur, O.P. and Prakash, C. (2002) Journal of Applied Physics, 92, 3872. http://dx.doi.org/10.1063/1.1504493
- Singh, A.K., Verma, A., Thakur, O.P., Prakash, C., Goel, T.C. and Mendiratta, R.G. (2003) Materials Letters, 57, 1040.
- Liang, T.J., Nien, H.H. and Chen, J.F. (2007) IEEE Transactions on Magnetics, 43, 3816-3827.
NOTES

*Corresponding author.


