World Journal of Condensed Matter Physics
Vol.07 No.02(2017), Article ID:76703,9 pages
10.4236/wjcmp.2017.72005
Influenza of the Cu Ion on the Structural and Optical Properties in Cu + Ce Co-Doped ZnO Compounds
S. López-Romero, M. J. Quiroz Jiménez, M. García-Hipólito, J. Chávez-Ramírez
Departamento de Materia Condensada y Criogenia, Universidad Nacional Autónoma de México, Instituto de Investigaciones en Materiales, Ciudad de México, Mexico

Copyright © 2017 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY 4.0).
http://creativecommons.org/licenses/by/4.0/



Received: April 1, 2017; Accepted: May 28, 2017; Published: May 31, 2017
ABSTRACT
This article showed and explained the effects of the Cu2+ ions on the structural and photoluminescent properties of Ce3+ doped ZnO compounds (ZnO: Ce3+) in Cu2+ + Ce3+ co-doped ZnO (ZnO: Cu2+ + Ce3+) solid solutions powders. The samples were synthesized by a solution combustion method maintaining the Ce3+ ion concentration constant in 3%wt and varying the Cu2+ ion concentration in 0%wt, 1%wt, 2%wt, 3%wt, 10%wt and 20%wt. However, pristine ZnO and Ce3+ doped ZnO were synthesized by the same method for comparison. After the synthesis process all the samples were annealed at 900˚C by 24 h. The pure ZnO, ZnO: Ce3+ and ZnO/Cu2+ + Ce3 powders were structurally characterized using X-ray diffraction (XRD) technique, the XRD patterns showed that for either undoped and doped with the Cu2+ ion both exhibited the hexagonal wurtzite ZnO crystalline structure, also the diffraction peaks of both samples types showed a little change toward lesser angles. The morphology and particle size of the samples were observed by means of a scanner electron microscopy (SEM); from SEM imagen is observed that the crystallites of the samples are agglomerated forming cage-like hollow structures caused by the combustion process. The cage-like structures have approximate size of 800 nm. In addition, the photoluminescence of pure ZnO, ZnO: Ce3+and ZnO: Cu2+ + Ce3+ compounds was measurement as a function of Cu2+ ion concentration under a excitation wavelength of 378 nm in the UV region. As an important result, it is observed that by Auger phenomena of non-radiative recombination, the UV emission of the ZnO is quenching.
Keywords:
Zinc Oxide, Copper-Cerium, Co-Doped, Solution-Combustion

1. Introduction
Actually, there are a lot of scientific literature on the II-VI semiconductor material zinc oxide (ZnO) produced by researchers dedicated to the physical optical study in all the world, because the ZnO has singular and extraordinary physical and chemical properties: presents a wide direct band gap of 3.37 eV at room temperature [1] and has bonding energy of 60 meV that makes ZnO very apt for exciton-based applications [2] . However, such properties make to the ZnOa very versatile material ideal for working in conjunction with other materials. The ZnO has been studied in basic and technological applications: fabrication of the follow devices: electro-optic [3] , solar cells [4] , piezo-electric [5] , magnetic [6] , gas sensors [7] , photoluminescent [8] , etc. The ZnO can be synthesized using various methods, such as: magnetron sputtering [9] , chemical vapor deposition (CVD) [10] , Co-presipitation [11] , metalorganic [12] and solution combustion (SC) technique [13] : this last method is very fast, less expensive, easier variables deposit control, very efficient and highly reproducible. In this work we used SC as synthesis method. In other hand, the Cu atoms incorporation into the crystalline lattice of the ZnO leads to change or improvement in some properties of the ZnO material such as: band gap tailored, electric, magnetic, electronic, thermoluminescent, surface defects control, structural, and photoluminescent [14] [15] [16] [17] . Furthermore, the Cu ion has been used to quenching the Eu3+ PL in glasses and ZnO matrix [18] [19] . In addition, specifically, the ZnO can be doped with various elements of the periodical table (lanthanides, earth rare, metals, phosphorous, etc.) for changing the ZnO intrinsic PL emission spectra as well as its emission intensity [20] [21] [22] [23] . Between the ZnO dopants the most prominent is the cerium atom (Ce3+), the first element of the lanthanide series due to its high PL emission efficiency and wide versatility: according to the studies realized by various researchers [24] - [29] , from your experimental results the authors show that the Ce3+ doped ZnO can presents anomalous PL emission spectra: from the UV radiation until the red color emissions; this multiple color PL emission is attributed to the Ce3+ ion and also depend of the excitation radiation of illumination. In this work, pure ZnO, Ce3+ doped ZnO and Cu2+ + Ce3+ co-doped ZnO solution powders were synthesized by a solution combustion technique. The ZnO/Cu2+ + Ce3+ compound was synthesized as a function of the Cu2+ ion concentration in %wt. maintaining constant the Ce3+ ion concentration at 3% wt. The three type’s samples were annealed at 900˚C by 24 h. Structurally the samples were characterized by means of X-ray diffraction (XRD) technique. Your photoluminescence properties were studied by measuring its PL under an excitation wavelength of 378 nm at room temperature.
2. Experimental Details
The chemical synthesis technique by solution combustion is possible thanks to the development of a chemical reaction of oxidation-reduction (REDOX) between an oxidizer agent and a fuel, the reaction is highly exothermic (1200˚C) [30] producing oxides metallic, water vapor, carbon-dioxide and nitrogen molecular. In our experiment zinc nitrate hexahydrate (Zn(NO3)2∙6H2O) was used as oxidizer agent and urea (5H NCONH)as fuel. In the follow chemical REDOX reaction:
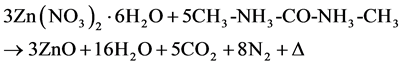 (1)
(1)
The Equation (1) is the stoichiometric form of the REDOX reaction obtained with a equivalence ratio value of unity (i.e. f = O/F = 1) where O and F are the total oxidizing and total reduction valences of the agents and the energy released by the combustion is at maximum [30] . Using the Equation (1), intrinsic ZnO, Ce3+ doped ZnO and Cu2+ + Ce3+ co-doped ZnO solid solution powders were synthesized and after annealed at 900˚C by 24 h. The source of the dopants Cu2+ and Ce3+ were copper chloride (CuCl2) and cerium oxide Ce2O3 respectively. The Ce3+ ion concentration was fixed at 3%wt, while the Cu2+ ion concentration takes the values of 0%wt, 1%wt, 2%wt, 3%wt, 10%wt and 20%wt. The pure ZnO, ZnO/Ce3+, and ZnO/Cu2+ + Ce3+ samples thus obtained were structurally characterized by X-ray diffraction (XRD) technique using a Philips PW 1800 diffractometer with Cu kα radiation (1.5406 Å), the morphology of the powders samples was recorded using a scanning electron microscopy (SEM) JEOL JSM 840. The PL study was realized using a spectrofluorometer Fluoro Max-P that uses a xenon lamp.
3. Results and Discussion
3.1. Structural Study
The Figure 1 shows the XRD patterns of the as prepared samples of ZnO, ZnO: Ce3+ and ZnO: Cu2+ + Ce3+ as a function of the Cu2+ ion concentration in %wt.
Figure 1. XRD pattern of ZnO, Ce3+ doped ZnO and Cu2+ + Ce3+ co-doped ZnO as a function of the Cu2+ ion concentration.
and annealed at 900˚C by 24 h. From the XRD patterns can be observed that all the diffraction peaks can be indexed to the hexagonal wurtzite ZnO structure (JCPDS CARD #89-(102)), no change of the peaks toward lesser angles was observed: the Cu2+ ion incorporation into the ZnO crystalline lattice do not changed the ZnO basic structure. However, the Figure 2 shows the XRD pattern of the ZnO joint to two diffraction peaks at 28.4˚ and 32.5˚: the first is assigned to the residual Ce2O3 phase and the second to the CuO compound. Also is observed that the diffraction peaks are very sharp indicating good crystallization of the products. By using the Scherrer formula the size of the crystallites was estimated in 60 nm.
3.2. Morphology Study
The Figure 3 shows the SEM micrographs imagens of (a) undoped ZnO, and doped ZnO with (b) 3, (c) 10, and (d) 20%wt of Cu ion concentration and 3%wt of Ce ion respectively. For undoped ZnO it is observed that the powders presents particles with amorphous morphology and connected each other forming large network systems with irregular pore sizes and shapes. In Cu + Ce doped ZnO the powders shows cages broken structure with hollows formed by scapin gases during the combustion process. This porous system with lot of voids is typical of solution combustion synthesized powders caused by scaping gases. In higher Cu doping the SEM micrograph also shows the presence of various small crystalites with different sizes and shapes within grains this is due to the non-uniform distribution temperature and mass flow in the combustion flame.
3.3. Photoluminescence Study
The effects of the incorporation of the Cu2+ ion in the lattice crystalline of the Ce doped ZnO matrix on the luminescence of the Cu2+ + Ce3+ co-doped ZnO powders were studied measuring the photoluminescence (PL) of the samples. The PL was obtained at room temperature under the excitation wavelength of 378 nm
Figure 2. XRD patterns of ZnO, Ce2O3 and CuO compounds.


Figure 3. SEM micrographs of the (a) Ce3+ doped ZnO, ZnO: 3%Ce intrinsic; (b) Ce3+ + 2Cu%, ZnO: 3%Ce + 2%Cu; (c) ZnO: 3%Ce3+ + 10%Cu; and (d) Ce3+ + 20%Cu, ZnO: 3%Ce + 20%Cu.
obtained from a He-Cd laser. The Figure 4 exhibits the PL spectra of the ZnO: Cu2+ + Ce3+ compounds as a function of the Cu2+ ion concentration in %wt. In Figure 4 appears four relevant emission peaks for all Cu2+ ion concentration centered about 422, 470, 640 and 665 nm. The first two peaks belong to the blue color region and are attributed to the electric-dipole-allowed transition of the Ce3+ ions [31] . The second two peaks are centered in the redcolor region, and are due to a radiative recombination process that occurs between the donors associated with oxygen vacancies Ov and the acceptors associated with the native defects adjacent to the Cu2+ impurity [32] . It is important to note from Figure 4 that the UV PL was quenching of the PL spectra; this quenching effect is caused by a non-radiative recombination process known as non-radiative Auger recombination phenomena [33] associated to degenerate electrons, in which the energy released by an electron is immediately recombined and absorbed by another electron and the energy involved is dissiped by phonons [21] , and [22] . Auger
Figure 4. PL spectra of the ZnO: Ce3+ + Cu2+ samples as a function of the Cu2+ ion concentration in %wt.
process is considered as the cause major of non-radiative recombination in semiconductor materials. Auger process depends on the doping atoms concentration and defects in the lattice [34] . In our case, when the ZnO: Ce3+ powders are doped with a minimum of Cu2+ atoms (<1%wt) the near band edge emission (NBE) origin of the UV PL in the ZnO semiconductor is quenching in its totality.
4. Conclusion
In this work the effects caused by the Cu2+ ion on the structural and optical properties of co-doped compounds type ZnO: Cu2+ + Ce3+ synthesized by a solution combustion method have been investigated. It is found that the copper ion presence does not change the ZnO crystalline structure. The PL emission spectra consist of four relevant peaks: two peaks centered in the blue color region attributed to an electric-dipole-allowed; and the other two peaks centered in the red color region due to radiative recombination between donoroxygen vacancies defects and the acceptors Cu2+ impurity. However, it is concluded that the Cu2+ ion introduction into the ZnO crystalline lattice quenching the UV Pl of the ZnO semiconductor matrix caused by a non-radiative-recombination Auger effect; this result can be technologically used to fabric devices photoluminescent with ZnO matrix without UV emission.
Acknowledgements
The authors wish to thank to Dr. Ciro Falcony (IPN), Adriana Tejeda (IIM) for the XRD measurements, to Omar Novelo Peralta (IIM) for his SEM study, to M.A. Canseco Martinez (IIM) for their chemical analysis.
Cite this paper
López-Romero, S., Quiroz Jiménez, M.J., García-Hipólito, M. and Chávez-Ramírez, J. (2017) Influenza of the Cu Ion on the Structural and Optical Properties in Cu + Ce Co-Doped ZnO Compounds. World Journal of Condensed Matter Physics, 7, 47-55. https://doi.org/10.4236/wjcmp.2017.72005
References
- 1. Klingshirn, C. (2007) ZnO: From Basics towards Applications. Physic Status Solidi (B), 244, 3027-3073.
https://doi.org/10.1002/pssb.200743072 - 2. Sharma, S.K., Pitale, S.S., Manzar Malik, M., Dubey, R.N., Qureshi, M.S. and Ojha, S. (2010) Influence of Fuel/Oxidizer Ratio on Lattice Parameters and Morphology of Combustion. 307 Synthesized ZnO Powders. Physica B: Condensed Matter, 405, 866-874.
- 3. Sharma, S.K., Pitale, S.K., Manzar Malik, M., Dubey, R.N., Qureshi, M.S. and Lumin, J. (2009) Luminescence Studies on the Blue-Green Emitting Sr4Al14O25:Ce3+ Phosphor Synthesized through Solution Combustion Route. Journal of Luminescence, 129, 140-147.
- 4. Lee, J.-B., Lee, H.-J., Seo, S.-H. and Park, J.-S. (2001) Characterization of Undoped and Cu-Doped ZnO Films for Surface Acoustic Wave Applications. Thin Solid Films, 398-399, 641-646.
- 5. Qi, J., Gao, D.Q., Zhang, L. and Yang, Y.H. (2010) Room-Temperature Ferromagnetism of the Amorphous Cu-Doped ZnO Thin Films. Applied Surface Science, 256, 2507-2508.
- 6. Wei, H.M., Gong, H.B., Wang, Y.Z., Hu, X.L., Chen, L., Xu, H.Y., Liu, P. and Cao, B.Q. (2011) Three Kinds of Cu2O/ZnO Heterostructure Solar Cells Fabricated with Electrochemical Deposition and Their Structure-Related Photovoltaic Properties. CrystEngComm, 13, 6065-6070.
https://doi.org/10.1039/c1ce05540b - 7. Kundu, T.K., Karak, N., Barik, P. and Saha, S. (2011) Optical Properties of ZnO Nanoparticles Prepared by Chemical Method Using Poly(Vinyl Alcohol ) (PVA) as Capping Agent. International Journal of Soft Computing and Engineering (IJSCE), 1, 2231-2307.
- 8. Ge, C.Q. and Xie, C.S. (2007) Preparation and Gas-Sensing Properties of Ce-Doped ZnO Thin-Film Sensors by Dip-Coating. Materials Science and Engineering: B, 137, 53-58.
- 9. Anandana, S. and Miyauchi, S. (2011) Ce-Doped ZnO (CexZn1-xO) Becomes an Efficient Visible-Light-Sensitive Photocatalyst by Co-Catalyst (Cu2+) Grafting. Physical Chemistry Chemical Physics, 13, 14937-14945.
https://doi.org/10.1039/c1cp21514k - 10. Ahn, C.-W., Nahm, S., Ryu, J., Uchino, K., Yoon, S.-J., Jung, S.-J. and Song, J.-S. (2004) Effects of CuO and ZnO Additives on Sintering Temperature and Piezoelectric Properties of 0.41Pb(Ni1/3Nb2/3)O3-0.36PbTiO3-0.23PbZrO3 Ceramics. Japanese Journal of Applied Physics, 43, 205-210.
https://doi.org/10.1143/JJAP.43.205 - 11. Morinaga, Y., Sakuragi, K., Fujimura, K. and Ito, T. (1997) Effect of Ce Doping on the Growth of ZnO Thin Films. Journal of Crystal Growth, 174, 691-695.
- 12. Sinhaa, N., Raya, G. and Bhandaria, S. (2014) Synthesis and Enhanced Properties of Cerium Doped ZnO Nanorods. Ceramics International, 40, 12337-12342.
- 13. Shukla, S., Agorku, E., Mittal, H. and Mishra, A. (2013) Synthesis, Characterization and Photoluminescence Properties of Ce3+ Doped ZnO-Nanophosphors. Chemical Papers, 68, 217-222.
- 14. Sofiania, Z., Derkowskab, B., Dalasińskib, P. and Wojdylab, M. (2006) Optical Properties of ZnO and ZnO:Ce Layers Grown by Spray Pyrolysis. Optics Communications, 267, 433-439.
- 15. Varughese, G., Jithin, P.W. and Usha, K.T. (2015) Determination of Optical Band Gap Energy of Wurtzite ZnO:Ce Nanocrystallites. Physical Science International Journal, 5, 146-154.
https://doi.org/10.9734/PSIJ/2015/14151 - 16. Koaoa, L.F., Dejenea, F.B. and Swartb, H.C. (2013) The Effect of Ce3+ on Structure, Morphology and Optical Properties of Flower-Like ZnO Synthesized Using the Chemical Bath Method. Journal of Luminescence, 143, 463-468.
- 17. Yousefia, M., Amirib, M. and Azimiradc, R. (2011) Enhanced Photoelectrochemical Activity of Ce Doped ZnO Nanocomposite Thin Films under Visible Light. Journal of Electroanalytical Chemistry, 661, 106-112.
- 18. Jimenez, J.A. (2015) Photoluminescence of Eu3+-Doped Glasses with Cu2+ Impurities. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 145, 482-486.
- 19. Lopez-Romero, S., Quiroz-Jiménez, M.J. and Garcia-Hipolito, M. (2016) Quenching Photoluminescence of Eu(III) by Cu(II) in ZnO:Eu3+ + Cu2+ Compounds by Solution Combustion Method. World Journal of Condensed Matter Physics, 6, 269-275.
https://doi.org/10.4236/wjcmp.2016.63025 - 20. Pandey, P., Kurchania, R. and Haque, F.Z. (2015) Structural, Diffused Reflectance and Photoluminescence Study of Cerium Doped ZnO Nanoparticles Synthesized through Simple Sol-Gel Method. Optik-International Journal for Light and Electron Optics, 126, 3310-3315.
- 21. Karunakaran, C., Gomathisankar, P. and Manikandan, G. (2010) Preparation and Characterization of Antimicrobial Ce-Doped ZnO Nanoparticles for Photocatalytic Detoxification of Cyanide Materials. Chemistry and Physics, 123, 585-594.
- 22. Samadia, M., Ziraka, M. and Naserib, A. (2016) Recent Progress on Doped ZnO Nanostructures for Visible-Light Photocatalysis. Thin Solid Films, 605, 2-19.
- 23. Koaoa, L.F., Dejenea, F.B. and Tsegaa, M. (2016) Annealed Ce3+-Doped ZnO Flower-Like Morphology Synthesized by Chemical Bath Deposition Method. Physica B: Condensed Matter, 480, 53-57.
- 24. George, A. and Sharma, S.K. (2011) Detailed of X-Ray Diffraction and Photoluminescence Studies of Ce Dopes ZnO Nanocrystals. Journal of Alloys and Compounds, 509, 5942-5946.
- 25. Ge, C., Xie, C. and Cai, S. (2007) Preparation and Gas-Sensing Properties of Ce-Doped ZnO Thin-Film Sensors by Dip-Coating. Materials Science and Engineering: B, 137, 53-58.
- 26. Li, F., Yan, B., Zhang, J., Jiang, A.X., Shao, C.H., Kong, X.J. and Wang, X. (2007) Study on Desulfurization Efficiency and Products of Ce-Doped Nanosized ZnO Desulfurizer at Ambient Temperature. Journal of Rare Earths, 25, 306-310.
- 27. Yang, J., Gao, M., Yang, L., Zhang, Y., Lang, J., Wang, D., Wang, Y., Liu, H. and Fan, H. (2008) Low-Temperature Growth and Optical Properties of Ce-Doped ZnO Nanorods. Applied Surface Science, 255, 2646-2650.
- 28. Iqbal, J., Liu, X., Zhu, H., Wu, Z.B., Zhang, Y., Yu, D., Yu, R. and Yu, R. (2009) Raman and Highly Ultraviolet Red-Shifted near Band-Edge Properties of LaCe-Co-Doped ZnO Nanoparticles. Acta Materialia, 57, 4790-4796.
- 29. López-Romero, S., Quiroz-Jiménez, M.J. and García-Hipólito, M. (2016) Structural and Optical Properties of Cu2+ + Ce3+ Co-Doped ZnO by Solution Combustion Method. World Journal of Condensed Matter Physics, 6, 300-309.
- 30. Patil, K.C., Hegde, M.S., Tanu, R. and Aruna, S.T. (2008) Chemistry of Nanocrystalline Oxide Materials. Combustion Synthesis, Properties and Applications. World Scientific, Singapore, 52-58.
https://doi.org/10.1142/6754 - 31. Chakraborty, R., Das, U., Mohanta, D. and Chouhury, A. (2009) Anomalous Luminescence Phenomena of Indium Doped ZnO Nano Structures Grown on Silicon Substrates by the Hydro Thermal Method. Indian Journal of Physics, 83, 553-558.
https://doi.org/10.1007/s12648-009-0019-x - 32. Zhang, X.Y., Dai, J.Y, Lam, C.H., Wang, H.T., Webleyle, P.A., Li, Q. and Ong, H. (2007) Zinc/ZnO Core-Shell Hexagonal Nanodisk Dendrites and Their Photoluminescence. Acta Materialia, 55, 5039-5044.
- 33. Vigneshwaran, N., Kumar, S., Kathe, A.A., Varadarajan, P.V. and Prasad, V. (2006) Functional Finishing of Cotton Fabrics Using Zinc Oxide-Soluble Starch Nanocomposites. Nanotechnology, 17, 5087.
https://doi.org/10.1088/0957-4484/17/20/008 - 34. Aliyu, H.S., Abdullah, A.H. and Abbas, Z. (2014) Solid State Characterization of Cu-ZnO Nanocomposite Synthesized via Micro-Wave Irradiation. The International Journal of Engineering and Science (IJES), 3, 47-53.
http://www.theijes.com/Vol,3,Issue,5.html






