World Journal of Nano Science and Engineering
Vol.04 No.04(2014), Article ID:51563,14 pages
10.4236/wjnse.2014.44015
Research Advances in Photocatalysis of Inorganic Hollow Spheres
Ting Tian1, Jing Hu1,2*, Zuobing Xiao1,2
1School of Perfume and Aroma Technology, Shanghai Institute of Technology, Shanghai, China
2Shanghai Research Institute of Fragrance & Flavor Industry, Shanghai, China
Email: *hujing@sit.edu.cn
Copyright © 2014 by authors and Scientific Research Publishing Inc.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/



Received 29 August 2014; revised 30 September 2014; accepted 30 October 2014
ABSTRACT
Inorganic hollow spheres have shown their superiority in photocatalytic area due to the large specific surface area, controllable structure and their own special optical, electrical, magnetic properties. According to the classification of inorganic hollow spheres as photocatalysts, recent research progress and application status have been summarized in this paper. At last, the future developments of inorganic hollow spheres in photocatalytic field have been discussed.
Keywords:
Inorganic Hollow Spheres, Photocatalysis, Application, Progress

1. Introduction
Hollow spheres, also known as 0D nanomaterial, were spheroidal aggregates with hollow structure assembled by their 1D nanomaterial. Due to their characteristics of controlled morphology, uniform size, large specific surface area and low density, extensive attention has been paid so far in material area [1] -[5] . Particularly, inorganic hollow spheres with special optical, electrical, magnetic, mechanical and catalytic properties have been widely used in bio-pharmaceutical, catalyst, carrier, controlled release, photonic crystal, electrochemistry and environmental protection [6] -[11] . Under the urgent situation of environmental pollution, inorganic hollow spheres as photocatalyst turned into a way to solve environmental problems. Inorganic hollow spheres have peculiar hierarchical porous structure, which makes reactant molecules easily transfer to the active sites of porous wall to improve their photocatalytic efficiency. Moreover, the hollow structure allows multiple reflections of ultraviolet and visible light in inner hole to enhance utilization of light [12] . In this paper, according to the classification of photocatalytic inorganic hollow spheres, recent research progress has been introduced and application status has been concluded. The future developments of inorganic hollow spheres in photocatalytic field have been discussed.
2. Metal Oxide Hollow Spheres
2.1. Titanium Dioxide (TiO2)
The valence and conduction band of TiO2 is composed of the filled 2p orbitals of oxygen and 3d, 4s and 4p orbitals of titanium, respectively. Its energy gap is about 3.2 eV, which makes TiO2 being the most promising photocatalyst. The specific band potential gives its strong oxidizing property, high chemical durability and photoelectric conversion efficiency, as well as price superiority. Furthermore, the large specific surface area and low density of hollow structure can effectively enhance photocatalytic activity of TiO2 [13] . The TiO2 hollow spheres obtained by Li and co-workers can completely remove azo-dye Rhodamine B (Rh B) under visible light irradiation in 240 min, which is only 50% for DeGussa P25 under the same experimental conditions [14] . However, TiO2 can be excited only under ultraviolet light (less than 5% of the full solar spectrum). Besides, easily recombination of photogenerated electrons and holes and low interfacial charge transfer rate into TiO2 lead to reduction of photocatalytic efficiency. Therefore, three principal methods have been used to enhance photocatalytic activity of TiO2: ameliorate crystal composition, enlarge specific surface area and modify surface of TiO2 hollow sphere. In the process of crystal growth, the more exposed of {001} and {110} facets which have high interface energy means higher photocatalytic activity [15] -[17] . Wang et al. [18] prepared TiO2 hollow sphere with sixty percent exposure of {001} facets, which has higher photocatalytic activity than that of commercial photocatalyst P25. Figure 1 shows XRD pattern , SEM images and TEM images of anatase TiO2 hollow microspheres with exposed {001} facets. Jiao et al. [19] discovered that the interface energy of {116} facets was similar to that of {110} facets through X-ray diffraction analysis. The photocatalytic property of TiO2 hollow sphere consisting of highly active {116} plane-oriented crystallites exceed that of common TiO2 hollow sphere, because curved {116} facets can multi-reflect incident light to enhance capture rate. The TiO2 hollow spheres with special morphology were obtained to add specific surface area thus improve photocatalytic performance such as multi-shell structure [20] [21] . Tao et al. [22] prepared flower-like TiO2 hollow spheres assembled by nanosheet, which has a higher surface area (65 m2/g) than that value of commercial TiO2 (7 m2/g). Therefore, Degradation performance of methyl orange (MO) by the flower-like TiO2 hollow sphere is better than that of commercial TiO2.
Figure 1. XRD pattern (a), SEM images (b) (c) and TEM images of anatase TiO2 hollow microspheres with exposed {001} facets (d) [18] .
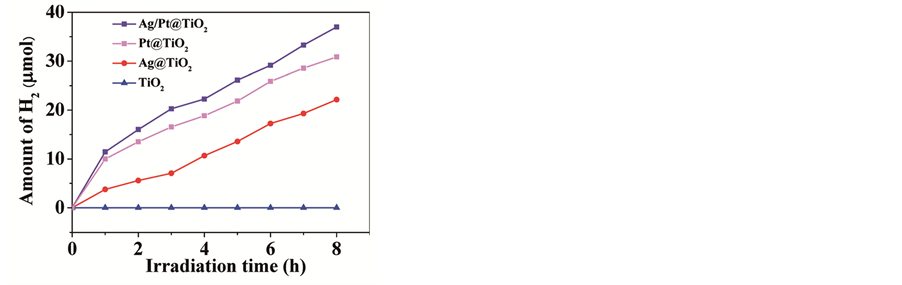
In recent years, surface modification of TiO2 hollow spheres has been widely researched to improve their photocatalytic efficiency. 1) Doping metal ion into TiO2 lattice to create defect position can capture electron from conduction band, reduce recombination of photogenerated electrons and holes in photocatalytic process and accelerate interface charge-transfer to raise photocatalytic activity. Transition metal, noble metal and rare- earth metal can be incorporate elements, such as vanadium, platinum, cerium, neodymium, etc. Vanadium- doped TiO2 hollow spheres prepared by Liu et al. [23] can quickly degrade methylene blue under visible-light irradiation to show their excellent photocatalytic activity and renewability. Feng and coworkers [24] prepared platinum doped TiO2 hollow spheres using carbon spheres as templates through hydrothermal precipitation method and adding hydrazine hydrate to reduce chlomplatinie acid. When platinum-doped content is 2.0%, the decolorization rate of obtained TiO2 hollow spheres can achieve 100% under 2 hour ultraviolet light irradiation. Wang and coworkers [25] [26] use carbon spheres to prepare cerium and neodymium doped TiO2 hollow spheres with dope concentration of 4% and 3.9% and apply them to dye decomposition. Results show that the apparent rate constant of doped TiO2 hollow spheres in degrading dye is 31 and 9 times as that of P25. 2) Non-metal elements also can be doped, such as boron [27] , nitrogen [28] [29] , carbon [30] [31] , fluorine [32] , etc. Non-metal ion possess relatively high energy orbit. Once doped it can replace some part of oxygen in the TiO2 lattice, thus bring in new level to lower energy gap and extend absorption wavelength. Yu et al. [33] prepared trifluoroacetic acid modified TiO2 hollow spheres by one-pot hydrothermal treatment using titanium sulfate as titanium source, the photocatalytic activity of which manifested 2 times higher than that of P25. In order to further improve photocatalytic activity of TiO2 hollow spheres, two or more kinds of ion can be doped to simultaneously provide electrons and holes trap to effectively suppress the recombination. Wang et al. [34] prepared cerium and nitrogen co-doped TiO2 hollow spheres owning enhanced visible light photocatalytic performance in dye X-3B decolorization because nitrogen decreased the energy gap and photogenerated electron of TiO2 valence band can transfer to the 4f orbit of cerium to inhibiting recombination. 3) Noble metal particles deposited onto the surface of TiO2 hollow spheres can take advantage of their surface plasmon resonance to extend absorption wavelength to visible light region, therefore enhance photocatalytic performance [35] [36] . Xiang et al. [37] combine microwave and hydrothermal treatment to prepare TiO2 hollow spheres with silver (Ag) nanoparticles deposited onto the surface, which have obvious absorption in the wavelength region from 400 to 600 nm and show better photocatalytic activity than pure TiO2 and commercial Degussa P25 powders. The single-crystalline anatase TiO2 hollow nano-hemispheres with bimetallic Ag/Pt nanoparticles uniformly loaded on both interior and exterior of the nano-hemispheres prepared by Jiang and co-workers exhibited excellent photocatalytic ability in the degradation of Rh B/ciprofloxacin (RhB/CIP) and hydrogen generation [38] . Figure 2 shows H2 production rates of TiO2, Ag@TiO2, Pt@TiO2 and Ag/Pt@TiO2 photocatalysts with CH3OH as the sacrificial reagent.
2.2. Other Metal Oxide
Oxide of d region elements also own good photocatalytic activity, such as vanadium pentoxide [39] , manganese dioxide [40] , nickel oxide [41] , tantalum oxide [42] , ferric oxide (Fe2O3) [43] , copper (I) oxide (Cu2O) [44] , tungsten oxide (WO3) [45] [46] , zinc oxide (ZnO) [47] [48] , etc. Patrinoiu et al. [49] impregnated carbonaceous
Figure 2. H2 production rates of TiO2, Ag@TiO2, of and Ag/Pt@TiO2 photocatalysts with CH3OH as sacrificial reagent [38] .
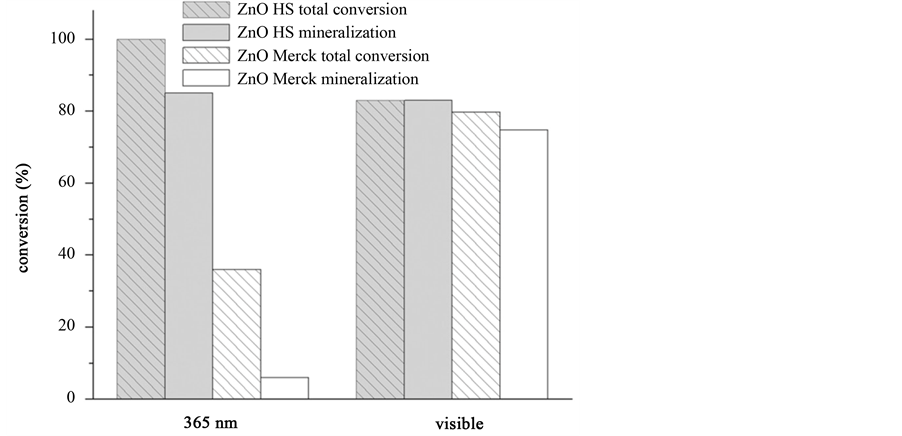
spheres templates with hydrated zinc acetate and then removed templates by thermal treatment to obtain zinc oxide hollow spheres. Under ultraviolet light irradiation, the phenol mineralization rate by zinc oxide hollow spheres can exceed 80%, which notably superior to commercial zinc oxide (Merck). Figure 3 shows effects of ZnO hollow sphere (HS) and commercial product (Merck) nature on degradation of phenol under same irradiation conditions. Cao and co-workers [50] prepared hexagonal α-Fe2O3 hollow spheres assembled by nanosheets through a microwave-assisted solvothermal route using ferric trichloride hexahydrate, sodium hydroxide and sodium dodecyl benzene sulfonate as raw materials and ethylene glycol as solvent. Degradation percentage of salicylic acid by the obtained hollow spheres under ultraviolet light in one hour can reach 60% and overtop 40% after using 2 times. Li et al. [51] prepared Nb2O5 hollow nanospheres with high surface energy (001) planes via ostwald ripening process, which owned high thermal stability, strong intensity of blue emission and efficiently split water under visible light irradiation.
Moreover, oxide of some p region elements also have photocatalytic activity, such as tin oxide (SnO2), indium oxide (In2O3), etc. Manjula et al. [52] utilize glucose as structure-directing agent to prepare porous tin oxide hollow spheres, which not only can photodegrade dye but also can be reused. The indium oxide hollow spheres gained by Li et al. [53] using emulsion vesicles as templates can photodecompose Rh B under ultraviolet light irradiation.
Surface modification still can be employed to enhance photocatalytic property of the aforementioned metal oxide hollow spheres. Ma et al. [54] doped tungsten oxide hollow spheres with silver-silver chloride to further improve their photocatalytic activity. Rahimi et al. [55] successfully prepared Ba-Cd-Sr-Ti doped ferroferric oxide (Fe3O4) nanohollow spheres via a simple solvothermal method without any templates. The photocatalytic degradation rate of congo red solution by this nanohollow spheres under visible light irradiation can reach to 99.5% when pH is 6.
2.3. Composite Metal Oxide
When two or more kinds of semiconductor form a compound system with fixed microstructure, it can restrain the recombination of photogenerated electrons and holes to significantly improve the photocatalytic performance. Hence, composite metal oxide hollow spheres owned a more excellent photocatalytic activity than that of single metal oxide spheres. Composite metal oxide can be divided into metal oxide modification and non-metal oxide modification. 1) Two kinds of metal oxide can be mutually compounded, such as ZnO [56] , Fe3O4 [57] , zirconium oxide (ZrO2) [58] composited with TiO2; In2O3 [59] , SnO2 [60] , CuO [61] , GeO2 [62] composited with ZnO; Fe2O3 composited with MnO [63] ; bismuth oxide (Bi2O3) composited with WO3 [64] , V2O5 [65] [66] , etc. When WO3 which has a low band gap combined with TiO2, it suppresses recombina-
Figure 3. Effects of ZnO hollow sphere (HS) and commercial product (Merck) nature on degradation of phenol under same irradiation conditions [49] .
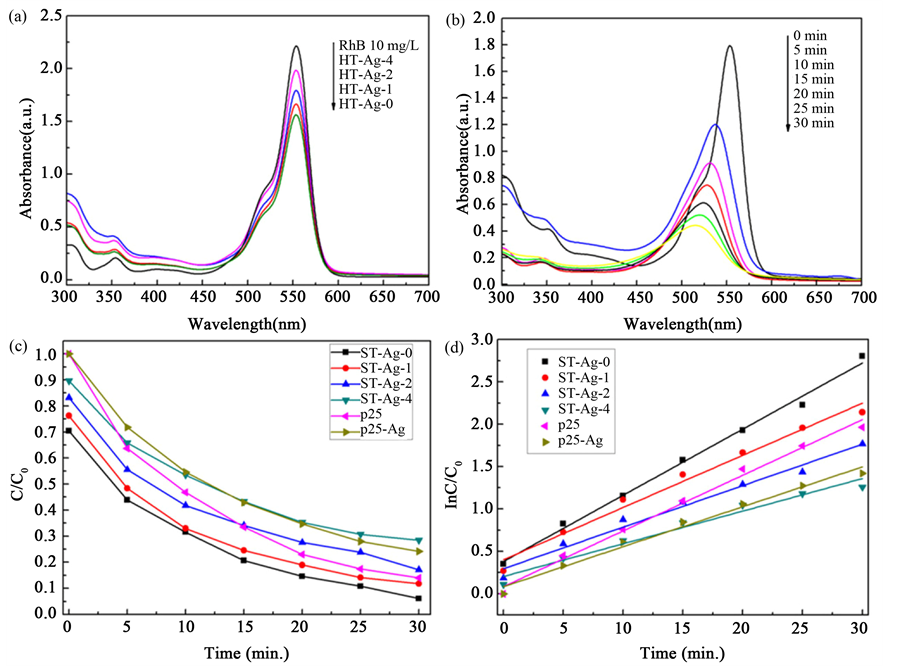
tion of photogenerated electrons and holes on hollow spheres surface as well as broadens photo-responsive scope to enhance utilization rate for optical energy. Tian et al. [67] prepared hierarchical flower-like Bi2MoO6 hollow spheres via a solvothermal process in the presence of ethylene glycol, which can remove over 95% Rh B within 2 h under visible-light irradiation. Li et al. [68] prepared bismuth tungstate (Bi2WO6) hollow spheres using polystyrene particles as the template, with Bi2O3 deposited on their surface and sequentially calcination to obtain double-shell Bi2O3-Bi2WO6 hollow spheres. The internal electric field of p-n junction formed by close contact between p-type Bi2O3 and n-type Bi2WO6 can speed up separation of photogenerated charge to boost photocatalytic performance, which makes this composite hollow spheres can completely decompose Rh B in 3 hours under visible light irradiation. An Ag2ZnGeO4 photocatalyst was obtained by Zhang and co-workers [69] via an ion-exchange reaction between amorphous Zn2GeO4 suspension and Ag ions solutions. The Ag2ZnGeO4 hollow sphere obtained through ostwald ripening process shows superior photocatalytic activity. 2) Few research has been focused on non-metal oxide modification. The composite between TiO2 and silicon oxide (SiO2) is the relatively familiar one. Li et al. [70] prepared composite SiO2-TiO2 hollow spheres by coating colloid carbon microspheres template with Si-doped TiO2 layer in a one-pot hydrothermal approach and sequentially calcination. The photodegradation rate of methylene blue solution under ultraviolet irradiation in 2 hours by the composite SiO2-TiO2 hollow spheres (80%) exceeds that of TiO2 hollow spheres (54%). In order to further improve photocatalytic activity of the above composite hollow spheres, measures such as surface noble metal deposition and element doping can be taken. Zhao et al. [71] prepared Ag modified hollow SiO2/TiO2 hybrid spheres through successively coating polystyrene spheres (PS) with SiO2 and TiO2 layer, evenly loading Ag nanoparticles by reducing silver nitrate onto surface of TiO2 layer, and then carrying out a calcination process to eliminate the template. The modified hollow spheres can efficiently degrade Rh B under both ultraviolet and visible light irradiation. Figure 4 shows UV-vis spectra of Rh B with Ag modified hollow SiO2/TiO2 hybrid
Figure 4. (a) UV-vis spectra of Rh B with Ag modified hollow SiO2/TiO2 hybrid spheres after 1 h dark absorption, photocatalytic degradation of Rh B under UV irradiation; (b) UV-vis spectra of the aqueous solutions of Rh B dye; (c) Photodegradation of the Rh B dye; (d) The corresponding pseudo-first-order kinetic rate plot [71] .
spheres after 1 h dark absorption, photocatalytic degradation of Rh B under UV irradiation, UV-vis spectra of the aqueous solutions of Rh B dye, photodegradation plot of the Rh B dye and the corresponding pseudo- first-order kinetic rate plot. Zhang et al. [72] prepared hollow cobalt, nitrogen co-doped TiO2/SiO2 microspheres using PS microspheres as templates, tetraethylorthosilicate and tetrabutyltitanate as precursors. The compound hollow spheres have a wide absorption wavelength to 600 nm and their photodegradation rate for Rh B under visible light irradiation in 40 minute can reach to 98%.
3. Metal Sulfide Hollow Spheres
3.1. Single Metal Sulfide
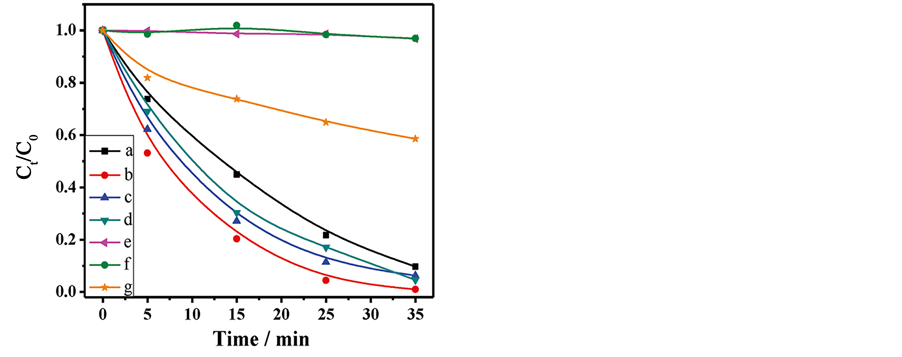
Metal sulfide as photocatalysts have attracted researchers’ general attention in recent years. Cadmium sulfide (CdS), a typical semiconductor of II-VI group with the band gap at 2.42 eV, owns outstanding photocatalytic property. Li et al. [73] prepared hollow CdS nanospheres with a diameter of about 130 nm and controllable shell thickness through 1-butyl-3-methylimidazolium-bis(trifluoro-methylsulfonyl)-imide ionic liquids as the templates, polyvinylpyrrolidone adjusting the formation of spheres and hexamethylenetetramine regulating the size and shell thickness. The photodegradation rate for methylene blue by the hollow CdS spheres can achieve 81% under ultraviolet irradiation in 80 minutes. Elements doping can effectively overcome its instability for light. Luo and co-workers [74] prepared nickel ion (Ni2+) doped CdS hollow spheres via a template-free one-pot method. The obtained 1.2 mol% Ni-doped CdS hollow spheres can completely decompose Rh B under visible light (λ > 420 nm) in 35 minutes and the degradation rate keeps over 98% after using 4 times. Figure 5 shows the photogagradation plot for Rh B under visible light of Ni-doped CdS hollow spheres.
Besides, zinc sulfide (ZnS), indium sulfide (In2S3) and copper sulfide (CuS) also have a certain degree of photocatalytic property. Yu and co-workers [75] prepared hexagonal wurtzite ZnS hollow spheres via one-pot template-free hydrothermal route using zinc acetate dehydrate and thiourea as raw materials. The ZnS hollow sphere shows strong absorption less than 365 nm. Zhang et al. [76] successfully synthesized Bi-doped ZnS hollow spheres (BZ) with enhanced ultraviolet and visible-light photocatalytic activity because of the formation of an isolated state originating from Bi 6s above the top of the valence band of ZnS and the electron excitation from Bi 6s state to the conduction band occurred under visible light irradiation. Under the optimal content of Bi dopant (R = 0.3, which is defined as the nominal atomic ratio of Bi to Zn), that is 0.3 at%, the hydrogen production rate is 1030 and 134 μmol∙h−1∙g−1 under UV and visible-light irradiation, respectively.
Rengaraj et al. [77] obtained tetragonal porous In2S3 hollow spheres composed of two-dimensional nanosheets and nanorods via a one-step solvothermal method using thiosemic arbazide as both sulfur source and capping ligand. The above-mentioned hollow spheres can photodegrade 30% percentage of methyl blue solution
Figure 5. Ct/C0 versus time curves of Rh B photodegradation under visible-light (λ > 420 nm) irradiation over (a) CdS; (b) 1.2 mol% Ni-doped CdS; (c) 3 mol% Ni-doped CdS; (d) 5 mol% Ni-doped CdS; (e) Catalyst free; (f) Light off; and (g) Degussa P25 [74] .
under visible light irradiation in 3 hours. Meng et al. [78] prepared hierarchical flower-like CuS hollow nanos- pheres via a solvothermal approach. The CuS hollow nanospheres obtained can photodegrade Rh B and 2,4-dichlorophenol aqueous solution because of the synergistic effect of surface hierarchical structure with large surface area, porous hollow sphere structure and high visible light utilization.
3.2. Composite Metal Sulfide
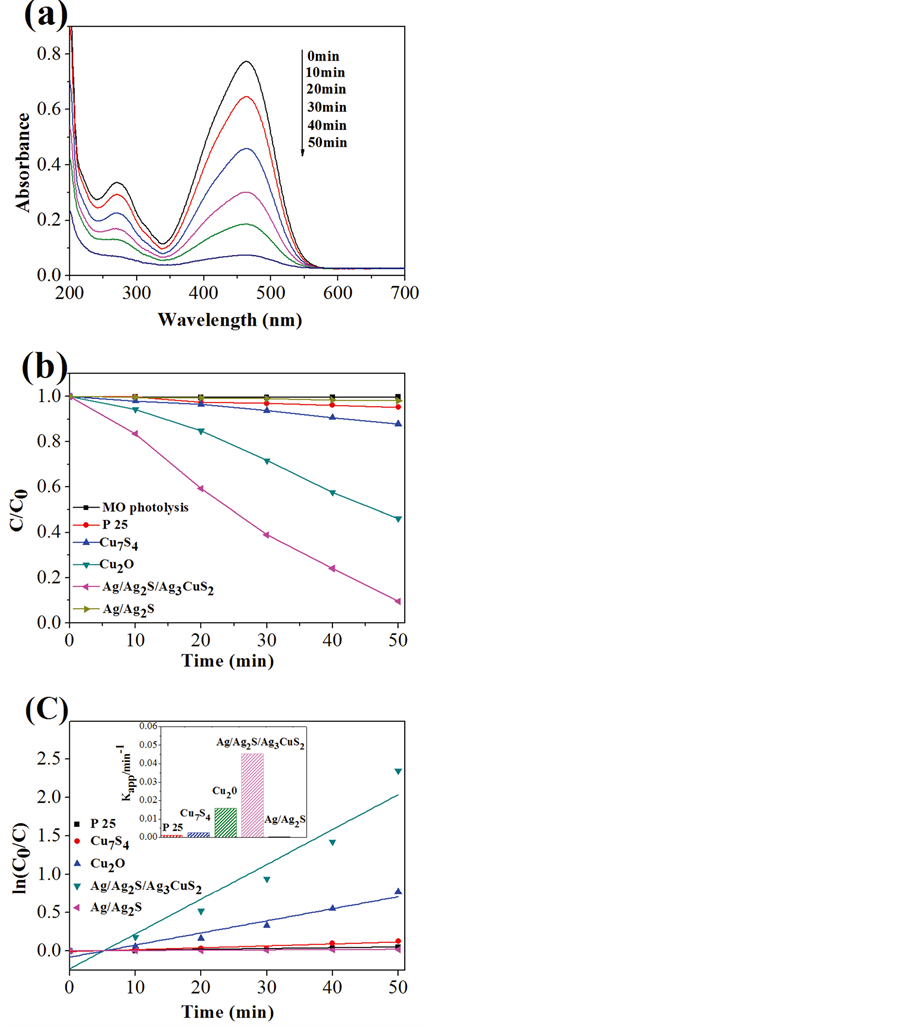
Compared to single metal sulfide, composite metal sulfide has more superior photocatalytic activity and broaden light responsive scope. However, until now few studies of composite metal sulfide hollow spheres concentrated on phohocatalytic realm. The way to get composite metal sulfide hollow spheres is mainly by simple coupling and doping. Zhu et al. [79] prepared composite (Ag, Cu)2S hollow spheres with cation exchange method using spherical aggregates of CuS nanoparticles as templates. The composite (Ag, Cu)2S hollow spheres have strong absorption at ultraviolet light scope from 200 nm to 500 nm and infrared light scope from 1000 nm to 2500 nm, but weak absorption at visible light scope from 500 nm to 800 nm, which makes selective light absorption come true. Yu et al. [80] prepared composite CuS-ZnS hollow nanospheres by an ion-exchange method using ZnS solid spheres as a precursor and copper nitrate as raw materials. The photodegradation rate for Rh B by the composite hollow nanospheres is obviously higher than that of CuS and ZnS solid spheres. Bhirud et al. [81] prepared CdIn2S4 hollow spheres assembled by nanoparticles with a flower like morphology using cadmium nitrate tetrahydrate, indium nitrate trihydrate, thiourea and cetyl trimethyl ammonium bromide as raw materials. The rate of hydrogen production from H2S photodecomposition for CdIn2S4 hollow spheres is 3171 μmol∙h−1, which is almost three fold enhancement than the highest rate of hydrogen production from normal bulk CdS (847 μmol∙h−1). Xing and co-workers [82] prepared a type of ternary Ag/Ag2S/Ag3CuS2 hollow microspheres with Cu7S4 hollow submicrospheres as the template, the photocatalytic activity of which was higher than those of Ag/Ag2S, Cu2O, Cu7S4 and P25 for the photodegradation of MO under visible light irradiation. Superoxide radicals and holes were confirmed to be the main reactive species for MO degradation through radical scavenger experiments. Figure 6 shows photogagradation plot for MO under visible light of the as-obtained Ag/Ag2S/ Ag3CuS2 hollow microspheres.
4. Application
4.1. Sewage Treatment
Organic pollutants durably presented in sewage often have reducibility. Consequently, inorganic hollow sphere photocatalysts can randomly oxidize organic pollutants by photo-generated holes and hydroxyl to carbon dioxide (CO2), water and other nontoxic inorganic substances to purify water, as well as avoiding secondary water pollution. At present, photocatalytic technology can effectively degrade halogenated aliphatic hydrocarbon, dye, nitroaromatic compounds, polycyclic aromatic hydrocarbon, heterocyclic compounds, hydrocarbon, phenols, surfactants, pesticides and so on. For example, nitrogen doped TiO2 hollow spheres can decompose bisphenol A [83] , carbon, nitrogen co-doped TiO2 hollow spheres can remove dye X-3B [84] , copper ion doped TiO2 hollow spheres can degrade chlorotetracycline [85] , zirconium oxide doped TiO2 hollow spheres can decompose Rh B [58] , bismuth molybdate hollow spheres can decompose phenol [86] and Zn2GeO4 hollow spheres can degrade antibiotic metronidazole [87] . While inorganic hollow spheres as water purification material have achieved great progress, it also exists limitations such as unable to efficiently resolve high concentration wastewater and difficult to recycle for powder catalyst.
4.2. Air Purification
Due to the vast application of organic materials and continuous off-gas emissions from automobile, there is a large amount of poisonous organic gas and nitrogen oxides suspending in our breathing air. Air purification by photocatalysts can operate at normal temperature and pressure, do no harm to environmental and human body, costs little, completely decompose organics. Dong et al. [88] prepared hierarchical (BiO)2CO3 hollow microspheres which can be reused as NO removal material in indoor air. The indium vanadate hollow spheres prepared by Ai et al. [89] can oxidize NO to nitric acid under visible light and maintain their photocatalytic ratio after reused. Ikeda et al. [90] encapsulates TiO2 into a hollow SiO2 shell to attain a type of composite material, which can decompose gas-phase acetone and isopropanol into CO2. Nevertheless, absence of applied research on
Figure 6. (a) UV-visible absorption spectra of degradation of MO by Ag/Ag2S/Ag3CuS2 under visible light (λ > 420 nm); (b) photocatalytic degradation of MO over different photocatalysts; (c) Linear transformation 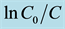 = Kappt of the kinetic curves of MO degradation over different photocatalysts. The inset shows the Kapp for MO degradation over different photocatalysts [82] .
= Kappt of the kinetic curves of MO degradation over different photocatalysts. The inset shows the Kapp for MO degradation over different photocatalysts [82] .
air-handling equipment gives rise to the unrealizable industrialization of air purification by nanosize photocatalysts.
4.3. Hydrogen Production
Hydrogen gas is a kind of clean energy sources with a high combustion value, superior efficiency and environmental friendly. The hydrogen production chiefly depends on coal and natural gas so far, which could aggravate consumption of nonrenewable resources and environmental pollution. The most ideal way to settle down the problem is to transfer solar energy into hydrogen energy with photocatalyst using renewable substance as raw materials such as water and biomass. The hierarchical Sn2Nb2O7 hollow spheres synthesized by Zhou et al. shows a superior visible-light-driven photocatalytic H2 production activity(3 μmol∙h−1), which is about 4 times higher than that of the bulk Sn2Nb2O7 sample prepared by a conventional high temperature solid state reaction method [91] . Zhang [92] and co-workers prepared composite CdS-TiO2 hollow sphere by coupling TiO2 to CdS which had a conduction band potential more negative than that of H+/H2 via a hydrothermal process, second step impregnation method and sol-gel method in sequence. The hydrogen production rate by the composite hollow spheres from water under ultraviolet light irradiation in one hour is 0.81 ml/g. After that, they prepared NiO- CdS hollow spheres with p-n junction, the hydrogen production rate by which under ultraviolet light irradiation in one hour (1.81 ml/g) precede the common composite CdS-TiO2 hollow spheres [93] . The reason is that interior electric field of p-n junction boosting transport rate of photo-production electronics. While great progresses have been achieved in hydrogen production by photocatalysts until now, it fails to meet practical applications for the low efficiency of directly water splitting.
4.4. CO2 Reduction
Greenhouse gas CO2 is a major cause to global warming. It is also a kind of potential carbon resources. Hence, effective control and utilization of CO2 becomes research focus. Present emission reduction technology requires numerous energy and exists potential safety problems in application. In the process of constant exploring emission reduction technology, it has been found that photogenerated electronics can change CO2 to organic compounds with high application value such as methane and methanol. Tu et al. [94] prepared hollow spheres consisting of alternating Ti0.91O2 nanosheets and graphene nanosheets with polymer beads as sacrificial templates and via a microwave irradiation technique. The ultrathin Ti0.91O2 nanosheets allow charge carriers to move rapidly onto the surface to take part in the photoreduction reaction and the alternating compact stacking structure allow the photogenerated electron to transfer fast from Ti0.91O2 nanosheets to graphene nanosheets to extend lifetime of the charge carriers. The photocatalytic activity of the hollow spheres is 9 times higher than P25. It can reduce CO2 to carbonic oxide and methane. Photocatalytic reduction for CO2 operates at normal temperature and pressure, directly ultilizes solar light with no energy consumption, does no harm to environment, is the most promising conversion method.
4.5. Other Application
Photogenerated holes and electronics can directly react with compounds which deactive cytoderm, cytomembrane and cell leading to the death of bacterium. So, photocatalysts can be used as a new type of antibacterial materials. Feng et al. [95] prepared Ag-doped TiO2 hollow spheres by hydrothermal precipitation method using carbon spheres as the templates. TiO2 hollow sphere doping 9.4 mol% Ag has excellent antibacterial activities against scherichia coli, staphylococcus aurels and candida albicans at room temperature. Furthermore, hollow spheres with photocatalytic properties can be photoelectric detector [96] .
5. Outlook
Inorganic hollow spheres show their excellent performance in photocatalytic area, which belongs to their large specific surface area, controllable structure and their own special optical, electrical, magnetic properties. They can be widely applied in sewage treatment, hydrogen production, air purification and other field. Metal oxide, metal sulfide and composite hollow spheres formed by metal oxide or sulfide have successfully prepared and primarily improve their photocatalytic property by controlling surface morphology and doping various other semiconductors. However, owing to the limitations of synthetic method, there exist some drawbacks including poor structure controllability, uneven shell thickness and broad particle size distribution, which are adverse to photocatalytic activity. Therefore, inorganic hollow spheres with well structure and property controllability remain to be one of the principal development trends in photocatalytic field. On the other hand, various types of inorganic hollow spheres own photocatalytic property, but the photodegradation efficiency is low. Rare earth elements possess unique f electronic configuration and abundant storage. So, hollow spheres composed of rare earth elements will be one of the research focuses in photocatalytic area.
Acknowledgements
The support from National Natural Science Foundation of China (No. 21106084), Shanghai Science and Research Innovation Foundation (No. 14zz164) and Shanghai Rising-Star Program (14QA143300) was appreciated.
References
- Hu, J., Chen, M., Fang, X. and Wu, L. (2011) Fabrication and Application of Inorganic Hollow Spheres. Chemical Society Reviews, 40, 5472-5491. http://dx.doi.org/10.1039/c1cs15103g
- Gao, R., Chen, M., Li, W., Zhou, S. and Wu, L. (2013) Facile Fabrication and Some Specific Properties of Polymeric/ Inorganic Bilayer Hybrid Hollow Spheres. Journal of Materials Chemistry A, 1, 2183-2191. http://dx.doi.org/10.1039/c2ta00837h
- Zhu, B.T., Wang, Z., Ding, S., Chen, J.S. and Lou, X.W. (2011) Hierarchical Nickel Sulfide Hollow Spheres for High Performance Supercapacitors. RSC Advances, 1, 397-400. http://dx.doi.org/10.1039/c1ra00240f
- Guan, J., Mou, F., Sun, Z. and Shi, W. (2010) Preparation of Hollow Spheres with Controllable Interior Structures by Heterogeneous Contraction. Chemical Communications, 46, 6605-6607. http://dx.doi.org/10.1039/c0cc01044h
- Li, P., Fan, H., Cai, Y. and Xu, M. (2014) Zn-Doped In2O3 Hollow Spheres: Mild Solution Reaction Synthesis and Enhanced Cl2 Sensing Performance. CrystEngComm, 16, 2715-2722. http://dx.doi.org/10.1039/c3ce42325e
- Xu, S., Hessel, C.M., Ren, H., Yu, R.B., Jin, Q., Yang, M., et al. (2014) α-Fe2O3 Multi-Shelled Hollow Microspheres for Lithium Ion Battery Anodes with Superior Capacity and Charge Retention. Energy & Environmental Science, 7, 632-637. http://dx.doi.org/10.1039/c3ee43319f
- Tian, W., Zhang, C., Zhai, T.Y., Li, S.L., Wang, X., Liao, M.Y., et al. (2013) Flexible SnO2 Hollow Nanosphere Film Based High-Performance Ultraviolet Photodetector. Chemical Communications, 49, 3739-3741. http://dx.doi.org/10.1039/c3cc39273b
- Cong, H., Wang, Y., Yu, B., Wang, J. and Jiao, M. (2014) Synthesis of Anisotropic TiO2 Hollow Microspheres Using Cave Particles as Templates and Application in Water Treatment. New Journal of Chemistry, 38. 2564-2568. http://dx.doi.org/10.1039/c3nj01302b
- Xue, C., Wang, T., Yang, G., Yang, B. and Ding, S. (2014) A Facile Strategy for the Synthesis of Hierarchical TiO2/ CdS Hollow Sphere Heterostructures with Excellent Visible Light Activity. Journal of Materials Chemistry A, 2, 7674- 7679. http://dx.doi.org/10.1039/c4ta01190b
- Wu, L., Feng, H., Liu, M., Zhang, K. and Li, J. (2013) Graphene-Based Hollow Spheres as Efficient Electrocatalysts for Oxygen Reduction. Nanoscale, 5, 10839-10843. http://dx.doi.org/10.1039/c3nr03794k
- Li, R., Li, L., Han, Y., Gai, S., He, F. and Yang, P. (2014) Core-Shell Structured Gd2O3:Ln@mSiO2 Hollow Nanospheres: Synthesis, Photoluminescence and Drug Release Properties. Journal of Materials Chemistry B, 2, 2127-2135. http://dx.doi.org/10.1039/c3tb21718c
- Sun, Z., Liao, T., Kim, J.G., Liu, K., Jiang, L., Kim, J.H., et al. (2013) Architecture Designed ZnO Hollow Microspheres with Wide-Range Visible-Light Photoresponses. Journal of Materials Chemistry C, 1, 6924-6929. http://dx.doi.org/10.1039/c3tc31649a
- Ye, M., Chen, Z., Wang, W., Shen, J. and Ma, J. (2010) Hydrothermal Synthesis of TiO2 Hollow Microspheres for the Photocatalytic Degradation of 4-Chloronitrobenzene. Journal of Hazardous Materials, 184, 612-619. http://dx.doi.org/10.1016/j.jhazmat.2010.08.080
- Li, G., Zhang, H., Lan, J., Li, J., Chen, Q., Liu, J., et al. (2013) Hierarchical Hollow TiO2 Spheres: Facile Synthesis and Improved Visible-Light Photocatalytic Activity. Dalton Transactions, 42, 8541-8544. http://dx.doi.org/10.1039/c3dt50503k
- Gong, X.Q. and Selloni, A. (2005) Reactivity of Anatase TiO2 Nanoparticles: The Role of the Minority (001) Surface. Journal of Physical Chemistry B, 109, 19560-19562. http://dx.doi.org/10.1021/jp055311g
- Nakamura, R., Ohashi, N., Imanishi, A., Osawa, T., Matsumoto, Y., Koinuma, H., et al. (2005) Crystal-Face Dependences of Surface Band Edges and Hole Reactivity, Revealed by Preparation of Essentially Atomically Smooth and Stable (110) and (100) n-TiO2 (Rutile) Surfaces. Journal of Physical Chemistry B, 109, 1648-1651. http://dx.doi.org/10.1021/jp044710t
- Ong, W.J., Tan, L.L., Chai, S.P., Yong, S.T. and Mohamed, A.R. (2014) Highly Reactive {001} Facets of TiO2-Based Composites: Synthesis, Formation Mechanism and Characterization. Nanoscale, 6, 1946-2008. http://dx.doi.org/10.1039/c3nr04655a
- Wang, X., He, H., Chen, Y., Zhao, J. and Zhang, X. (2012) Anatase TiO2 Hollow Microspheres with Exposed {001} Facets: Facile Synthesis and Enhanced Photocatalysis. Applied Surface Science, 258, 5863-5868. http://dx.doi.org/10.1016/j.apsusc.2012.02.117
- Jiao, Y., Peng, C., Guo, F., Bao, Z., Yang, J., Schmidt-Mende, L., et al. (2011) Facile Synthesis and Photocatalysis of Size-Distributed TiO2 Hollow Spheres Consisting of {116} Plane-Oriented Nanocrystallites. The Journal of Physical Chemistry C, 115, 6405-6409. http://dx.doi.org/10.1021/jp200491u
- Li, S., Chen, J., Zheng, F., Li, Y. and Huang, F. (2013) Synthesis of the Double-Shell Anatase-Rutile TiO2 Hollow Spheres with Enhanced Photocatalytic Activity. Nanoscale, 5, 12150-12155. http://dx.doi.org/10.1039/c3nr04043g
- Zeng, Y., Wang, X., Wang, H., Dong, Y., Ma, Y. and Yao, J. (2010) Multi-Shelled Titania Hollow Spheres Fabricated by a Hard Template Strategy: Enhanced Photocatalytic Activity. Chemical Communications, 46, 4312-4314. http://dx.doi.org/10.1039/c0cc00706d
- Tao, Y.G., Xu, Y., Pan, J., Gu, H., Qin, C. and Zhou, P. (2012) Glycine Assisted Synthesis of Flower-Like TiO2 Hierarchical Spheres and Its Application in Photocatalysis. Materials Science and Engineering: B, 177, 1664-1671. http://dx.doi.org/10.1016/j.mseb.2012.08.010
- Liu, J., Chang, L., Wang, J., Zhu, M. and Zhang, W. (2010) A Facile One-Step Approach to Visible-Light-Sensitive Vanadium-Doped TiO2 Hollow Microspheres. Materials Science and Engineering: B, 172, 142-145. http://dx.doi.org/10.1016/j.mseb.2010.04.037
- Feng, X., Yang, O., Yang, L., Guang, Q. and Liu, Y.L. (2010) Preparation of Pt-Doped TiO2 Hollow Spheres by Hydrothermal Precipitation and Their Photocatalytic Activities in Rh B Degradation. Journal of Jinan University, 31, 495- 499.
- Wang, C., Ao, Y., Wang, P., Hou, J., Qian, J. and Zhang, S. (2010) Preparation, Characterization, Photocatalytic Properties of Titania Hollow Sphere Doped with Cerium. Journal of Hazardous Materials, 178, 517-521. http://dx.doi.org/10.1016/j.jhazmat.2010.01.111
- Wang, C., Ao, Y., Wang, P., Hou, J. and Qian, J. (2010) Preparation, Characterization and Photocatalytic Activity of the Neodymium-Doped TiO2 Hollow Spheres. Applied Surface Science, 257, 227-231. http://dx.doi.org/10.1016/j.apsusc.2010.06.071
- Xu, J., Ao, Y. and Chen, M. (2009) Preparation of B-Doped Titania Hollow Sphere and Its Photocatalytic Activity under Visible Light. Materials Letters, 63, 2442-2444. http://dx.doi.org/10.1016/j.matlet.2009.08.031
- Ao, Y., Xu, J., Fu, D. and Yuan, C. (2009) A Simple Method to Prepare N-Doped Titania Hollow Spheres with High Photocatalytic Activity under Visible Light. Journal of Hazardous Materials, 167, 413-417. http://dx.doi.org/10.1016/j.jhazmat.2008.12.139
- Ao, Y., Xu, J., Zhang, S. and Fu, D. (2010) A One-Pot Method to Prepare N-Doped Titania Hollow Spheres with High Photocatalytic Activity under Visible Light. Applied Surface Science, 256, 2754-2758. http://dx.doi.org/10.1016/j.apsusc.2009.11.023
- Zhuang, J., Tian, Q., Zhou, H., Liu, Q., Liu, P. and Zhong, H. (2012) Hierarchical Porous TiO2@C Hollow Microspheres: One-Pot Synthesis and Enhanced Visible-Light Photocatalysis. Journal of Materials Chemistry, 22, 7036- 7042. http://dx.doi.org/10.1039/c2jm16924j
- Shi, J.W., Chen, J.W., Cui, H.J., Fu, M.L., Luo, H.Y., Xu, B., et al. (2012) One Template Approach to Synthesize C- Doped Titania Hollow Spheres with High Visible-Light Photocatalytic Activity. Chemical Engineering Journal, 195- 196, 226-232. http://dx.doi.org/10.1016/j.cej.2012.04.095
- Zhou, J.K., Lv, L., Yu, J., Li, H.L., Guo, P.Z., Sun, H., et al. (2008) Synthesis of Self-Organized Polycrystalline F- Doped TiO2 Hollow Microspheres and Their Photocatalytic Activity under Visible Light. The Journal of Physical Che- mistry C, 112, 5316-5321. http://dx.doi.org/10.1021/jp709615x
- Yu, J. and Shi, L. (2010) One-Pot Hydrothermal Synthesis and Enhanced Photocatalytic Activity of Trifluoroacetic Acid Modified TiO2 Hollow Microspheres. Journal of Molecular Catalysis A: Chemical, 326, 8-14. http://dx.doi.org/10.1016/j.molcata.2010.04.016
- Wang, C., Ao, Y., Wang, P., Hou, J. and Qian, J. (2011) Preparation of Cerium and Nitrogen Co-Doped Titania Hollow Spheres with Enhanced Visible Light Photocatalytic Performance. Powder Technology, 210, 203-207. http://dx.doi.org/10.1016/j.powtec.2011.03.015
- Lu, J., Su, F., Huang, Z., Zhang, C., Liu, Y., Ma, X., et al. (2013) N-Doped Ag/TiO2 Hollow Spheres for Highly Efficient Photocatalysis under Visible-Light Irradiation. RSC Advances, 3, 720-724. http://dx.doi.org/10.1039/c2ra22713d
- Wang, S., Qian, H., Hu, Y., Dai, W., Zhong, Y., Chen, J., et al. (2013) Facile One-Pot Synthesis of Uniform TiO2-Ag Hybrid Hollow Spheres with Enhanced Photocatalytic Activity. Dalton Transactions, 42, 1122-1128. http://dx.doi.org/10.1039/c2dt32040a
- Xiang, Q., Yu, J., Cheng, B. and Ong, H.C. (2010) Microwave-Hydrothermal Preparation and Visible-Light Photoacti- vity of Plasmonic Photocatalyst Ag-TiO2 Nanocomposite Hollow Spheres. Chemistry: An Asian Journal, 5, 1466-1474. http://dx.doi.org/10.1002/asia.200900695
- Jiang, Z., Zhu, J., Liu, D., Wei, W., Xie, J. and Chen, M. (2014) In Situ Synthesis of Bimetallic Ag/Pt Loaded Single- Crystalline Anatase TiO2 Hollow Nano-Hemispheres and Their Improved Photocatalytic Properties. CrystEngComm, 16, 2384-2394. http://dx.doi.org/10.1039/c3ce41949e
- Fei, H.L., Zhou, H.J., Wang, J.G., Sun, P.C., Ding, D.T. and Chen, T.H. (2008) Synthesis of Hollow V2O5 Microspheres and Application to Photocatalysis. Solid State Sciences, 10, 1276-1284. http://dx.doi.org/10.1016/j.solidstatesciences.2007.12.026
- Duan, Z.Y. and Chen, Y.C. (2010) Hydrothermal Preparation for Self-Assembled γ-MnO2 Hollow Spheres and Their Photocatalytic Activity. Journal of Anqing Teachers College (Natural Science), 16, 88-91. http://dx.doi.org/10.3969/j.issn.1007-4260.2010.02.026
- Song, X.F. and Gao, L. (2008) Facile Synthesis and Hierarchical Assembly of Hollow Nickel Oxide Architectures Bea- ring Enhanced Photocatalytic Properties. Journal of Physical Chemistry C, 112, 15299-15305. http://dx.doi.org/10.1021/jp804921g
- Guo, G.L. and Huang, J.H. (2011) Ta2O5 Hollow Sphere: Fabrication and Photocatalytic Activity. Chinese Journal of Inorganic Chemistry, 27, 214-218. http://d.wanfangdata.com.cn/Periodical_wjhxxb201102002.aspx
- Xu, J.S. and Zhu, Y.J. (2011) α-Fe2O3 Hierarchically Hollow Microspheres Self-Assembled with Nanosheets: Surfactant-Free Solvothermal Synthesis, Magnetic and Photocatalytic Properties. CrystEngComm, 13, 5162-5169. http://dx.doi.org/10.1039/c1ce05252g
- Yu, Y., Zhang, L., Wang, J., Yang, Z., Long, M., Hu, N., et al. (2012) Preparation of Hollow Porous Cu2O Microspheres and Photocatalytic Activity under Visible Light Irradiation. Nanoscale Research Letters, 7, 347-353.
- Huang, J., Xu, X., Gu, C., Fu, G., Wang, W. and Liu, J. (2012) Flower-Like and Hollow Sphere-Like WO3 Porous Na- nostructures: Selective Synthesis and Their Photocatalysis Property. Materials Research Bulletin, 47, 3224-3232. http://dx.doi.org/10.1016/j.materresbull.2012.08.009
- Yu, J., Qi, L., Cheng, B. and Zhao, X. (2008) Effect of Calcination Temperatures on Microstructures and Photocatalytic Activity of Tungsten Trioxide Hollow Microspheres. Journal of Hazardous Materials, 160, 621-628. http://dx.doi.org/10.1016/j.jhazmat.2008.03.047
- Yu, J.G. and Yu, X.X. (2008) Hydrothermal Synthesis and Photocatalytic Activity of Zinc Oxide Hollow Spheres. Environmental Science &Technology, 42, 4902-4907. http://dx.doi.org/10.1021/es800036n
- Khoa, N.T., Kim, S.W., Thuan, D.V., Yoo, D.H., Kim, E.J. and Hahn, S.H. (2014) Hydrothermally Controlled ZnO Nanosheet Self-Assembled Hollow Spheres/Hierarchical Aggregates and Their Photocatalytic Activities. CrystEngComm, 16, 1344-1350. http://dx.doi.org/10.1039/c3ce41763h
- Patrinoiu, G., Tudose, M., Calderón-Moreno, J.M., Birjega, R., Budrugeac, P., Ene, R., et al. (2012) A Green Chemical Approach to the Synthesis of Photoluminescent ZnO Hollow Spheres with Enhanced Photocatalytic Properties. Journal of Solid State Chemistry, 186, 17-22. http://dx.doi.org/10.1016/j.jssc.2011.11.024
- Cao, S.W. and Zhu, Y.J. (2008) Hierarchically Nanostructured α-Fe2O3 Hollow Spheres: Preparation, Growth Mecha- nism, Photocatalytic Property and Application in Water Treatment. Journal of Physical Chemistry C, 112, 6253-6257. http://dx.doi.org/10.1021/jp8000465
- Li, L., Deng, J., Yu, R., Chen, J., Wang, Z. and Xing, X. (2013) Niobium Pentoxide Hollow Nanospheres with Enhanced Visible Light Photocatalytic Activity. Journal of Materials Chemistry A, 1, 11894-11900. http://dx.doi.org/10.1039/c3ta12599h
- Manjula, P., Boppella, R. and Manorama, S.V. (2012) A Facile and Green Approach for the Controlled Synthesis of Porous SnO2 Nanospheres: Application as an Efficient Photocatalyst and an Excellent Gas Sensing Material. ACS Applied Materials & Interfaces, 4, 6252-6260. http://dx.doi.org/10.1021/am301840s
- Li, B.X., Xie, Y., Jing, M., Rong, G., Tang, Y. and Zhang, G. (2006) In2O3 Hollow Microspheres: Sythesis from Designed In(OH)3 Precursors and Applications in Gas Sensors and Photocatalysis. Langmuir, 22, 9380-9385. http://dx.doi.org/10.1021/la061844k
- Ma, B., Guo, J., Dai, W.L. and Fan, K. (2012) Ag-AgCl/WO3 Hollow Sphere with Flower-Like Structure and Superior Visible Photocatalytic Activity. Applied Catalysis B: Environmental, 123-124, 193-199. http://dx.doi.org/10.1016/j.apcatb.2012.04.029
- Rahimi, R., Tadjarodi, A., Rabbani, M., Kerdari, H. and Imani, M. (2012) Preparation, Characterization and Photocatalytic Properties of Ba-Cd-Sr-Ti Doped Fe3O4 Nanohollow Spheres on Removal of Congo Red under Visible-Light Ir- radiation. Journal of Superconductivity and Novel Magnetism, 26, 219-228. http://dx.doi.org/10.1007/s10948-012-1716-9
- Agrawal, M., Gupta, S., Pich, A., Zafeiropoulos, N.E. and Stamm, M. (2009) A Facile Approach to Fabrication of ZnO-TiO2 Hollow Spheres. Chemistry of Materials, 21, 5343-5348. http://dx.doi.org/10.1021/cm9028098
- Xuan, S.H., Jiang, W.Q., Gong, X.L., Hu, Y. and Chen, Z.Y. (2009) Magnetically Separable Fe3O4/TiO2Hollow Spheres: Fabrication and Photocatalytic Activity. Journal of Physical Chemistry C, 113, 553-558. http://dx.doi.org/10.1021/jp8073859
- Sun, C., Liu, L., Qi, L., Li, H., Zhang, H., Li, C., et al. (2011) Efficient Fabrication of ZrO2-Doped TiO2 Hollow Nanospheres with Enhanced Photocatalytic Activity of Rhodamine B Degradation. Journal of Colloid and Interface Sci- ence, 364, 288-297. http://dx.doi.org/10.1016/j.jcis.2011.07.055
- Li, W.B., Bu, Y.Y. and Yu, J.Q. (2012) Preparation of ZnO/In2O3 Composite Hollow Spheres and Their Photoelectrocatalytic Properties to Glucose Degradation. Acta Physical-Chimica Sinica, 28, 2676-2682. http://dx.doi.org/10.3866/PKU.WHXB201207101
- Zhang, C., Yin, L., Zhang, L., Qi, Y. and Lun, N. (2012) Preparation and Photocatalytic Activity of Hollow ZnO and ZnO-CuO Composite Spheres. Materials Letters, 67, 303-307. http://dx.doi.org/10.1016/j.matlet.2011.09.073
- Wang, W.W., Zhu, Y.J. and Yang, L.X. (2007) ZnO-SnO2 Hollow Spheres and Hierarchical Nanosheets: Hydrothermal Preparation, Formation Mechanism, and Photocatalytic Properties. Advanced Functional Materials, 17, 59-64. http://dx.doi.org/10.1002/adfm.200600431
- Liang, J., Xu, J., Long, J., Zhang, Z. and Wang, X. (2013) Self-Assembled Micro/Nano-Structured Zn2GeO4 Hollow Spheres: Direct Synthesis and Enhanced Photocatalytic Activity. Journal of Materials Chemistry A, 1, 10622-10625. http://dx.doi.org/10.1039/c3ta12183f
- Chen, A.X. (2007) Preparation of ZrTiO4 Composite Hollow Spheres and Their Photocatalytic Properties (Jinan). M.E. Thesis, Shangdong University, Jinan.
- Dai, X.J., Luo, Y.S., Zhang, W.D. and Fu, S.Y. (2010) Facile Hydrothermal Synthesis and Photocatalytic Activity of Bismuth Tungstate Hierarchical Hollow Spheres with an Ultrahigh Surface Area. Dalton Transactions, 39, 3426-3432. http://dx.doi.org/10.1039/b923443h
- Lu, Y., Luo, Y., Xiao, H.M. and Fu, S.Y. (2014) Novel Core-Shell Structured BiVO4 Hollow Spheres with an Ultra- High Surface Area as Visible-Light-Driven Catalyst. CrystEngComm, 16, 5059-6065. http://dx.doi.org/10.1039/c4ce00379a
- Chen, X., Liu, J., Wang, H., Ding, Y., Sun, Y. and Yan, H. (2013) One-Step Approach to Novel Bi4V2O11 Hierarchical Hollow Microspheres with High Visible-Light-Driven Photocatalytic Activities. Journal of Materials Chemistry A, 1, 877-883. http://dx.doi.org/10.1039/c2ta00312k
- Tian, G., Chen, Y., Zhou, W., Pan, K., Dong, Y., Tian, C., et al. (2011) Facile Solvothermal Synthesis of Hierarchical Flower-Like Bi2MoO6 Hollow Spheres as High Performance Visible-Light Driven Photocatalysts. Journal of Materials Chemistry, 21, 887-892. http://dx.doi.org/10.1039/c0jm03040f
- Li, X., Huang, R., Hu, Y., Chen, Y., Liu, W., Yuan, R., et al. (2012) A Templated Method to Bi2WO6 Hollow Microspheres and Their Conversion to Double-Shell Bi2O3/Bi2WO6 Hollow Microspheres with Improved Photocatalytic Per- formance. Inorganic Chemistry, 51, 6245-6250. http://dx.doi.org/10.1021/ic300454q
- Zhang, N., Ouyang, S., Kako, T. and Ye, J. (2012) Synthesis of Hierarchical Ag2ZnGeO4 Hollow Spheres for Enhanced Photocatalytic Property. Chemical Communications, 48, 9894-9896. http://dx.doi.org/10.1039/c2cc34738e
- Li, G., Liu, F. and Zhang, Z. (2010) Enhanced Photocatalytic Activity of Silica-Embedded TiO2 Hollow Microspheres Prepared by One-Pot Approach. Journal of Alloys and Compounds, 493, L1-L7. http://dx.doi.org/10.1016/j.jallcom.2009.12.046
- Zhao, W., Feng, L., Yang, R., Zheng, J. and Li, X. (2011) Synthesis, Characterization, and Photocatalytic Properties of Ag Modified Hollow SiO2/TiO2 Hybrid Microspheres. Applied Catalysis B: Environmental, 103, 181-189. http://dx.doi.org/10.1016/j.apcatb.2011.01.025
- Zhang, L., Li, X., Chang, Z. and Li, D. (2011) Preparation, Characterization and Photoactivity of Hollow N, Co Co- Doped TiO2/SiO2 Microspheres. Materials Science in Semiconductor Processing, 14, 52-57. http://dx.doi.org/10.1016/j.mssp.2011.01.004
- Li, X., Gao, Y., Yu, L. and Zheng, L. (2010) Template-Free Synthesis of CdS Hollow Nanospheres Based on an Ionic Liquid Assisted Hydrothermal Process and Their Application in Photocatalysis. Journal of Solid State Chemistry, 183, 1423-1432. http://dx.doi.org/10.1016/j.jssc.2010.04.001
- Luo, M., Liu, Y., Hu, J., Liu, H. and Li, J. (2012) One-Pot Synthesis of CdS and Ni-Doped CdS Hollow Spheres with Enhanced Photocatalytic Activity and Durability. ACS Applied Materials & Interfaces, 4, 1813-1821. http://dx.doi.org/10.1021/am3000903
- Yu, X., Yu, J., Cheng, B. and Huang, B. (2009) One-Pot Template-Free Synthesis of Monodisperse Zinc Sulfide Hollow Spheres and Their Photocatalytic Properties. Chemistry: A European Journal, 15, 6731-6739. http://dx.doi.org/10.1002/chem.200900204
- Zhang, J., Liu, S., Yu, J. and Jaroniec, M. (2011) A Simple Cation Exchange Approach to Bi-Doped ZnS Hollow Spheres with Enhanced UV and Visible-Light Photocatalytic H2-Production Activity. Journal of Materials Chemistry, 21, 14655-14662. http://dx.doi.org/10.1039/c1jm12596f
- Rengaraj, S., Venkataraj, S., Tai, C., Kim, Y., Repo, E. and Sillanpää, M. (2011) Self-Assembled Mesoporous Hierarchical-Like In2S3 Hollow Microspheres Composed of Nanofibers and Nanosheets and Their Photocatalytic Activity. Langmuir, 27, 5534-5541. http://dx.doi.org/10.1021/la104780d
- Meng, X., Tian, G., Chen, Y., Zhai, R., Zhou, J., Shi, Y., et al. (2013) Hierarchical CuS Hollow Nanospheres and Their Structure-Enhanced Visible Light Photocatalytic Properties. CrystEngComm, 15, 5144-5149. http://dx.doi.org/10.1039/c3ce40195b
- Zhu, H., Lian, C. and Wu, D. (2011) Room-Temperature Synthesis of (Ag, Cu)2S Hollow Spheres by Cation Exchange and Their Optical Properties. Materials Chemistry and Physics, 127, 24-27. http://dx.doi.org/10.1016/j.matchemphys.2011.02.023
- Yu, J.G., Zhang, J. and Liu, S.W. (2010) Ion-Exchange Synthesis and Enhanced Visible-Light Photoactivity of CuS/ ZnS Nanocomposite Hollow Spheres. Journal of Physical Chemistry C, 114, 13642-13649. http://dx.doi.org/10.1021/jp101816c
- Bhirud, A., Chaudhari, N., Nikam, L., Sonawane, R., Patil, K., Baeg, J.O., et al. (2011) Surfactant Tunable Hierarchical Nanostructures of CdIn2S4 and Their Photohydrogen Production under Solar Light. International Journal of Hydrogen Energy, 36, 11628-11639. http://dx.doi.org/10.1016/j.ijhydene.2011.06.061
- Xing, C., Zhang, Y., Wu, Z., Jiang, D. and Chen, M. (2014) Ion-Exchange Synthesis of Ag/Ag2S/Ag3CuS2 Ternary Ho- llow Microspheres with Efficient Visible-Light Photocatalytic Activity. Dalton Transactions, 43, 2772-2780. http://dx.doi.org/10.1039/c3dt52875h
- Subagio, D.P., Srinivasan, M., Lim, M. and Lim, T.T. (2010) Photocatalytic Degradation of Bisphenol-A by Nitrogen- Doped TiO2 Hollow Sphere in a Vis-LED Photoreactor. Applied Catalysis B: Environmental, 95, 414-422. http://dx.doi.org/10.1016/j.apcatb.2010.01.021
- Ao, Y., Xu, J., Fu, D. and Yuan, C. (2009) Visible-Light Responsive C,N-Codoped Titania Hollow Spheres for X-3B Dye Photodegradation. Microporous and Mesoporous Materials, 118, 382-386. http://dx.doi.org/10.1016/j.micromeso.2008.09.010
- Bu, D. and Zhuang, H. (2013) Biotemplated Synthesis of High Specific Surface Area Copper-Doped Hollow Spherical Titania and Its Photocatalytic Research for Degradating Chlorotetracycline. Applied Surface Science, 265, 677-685. http://dx.doi.org/10.1016/j.apsusc.2012.11.080
- Yin, W., Wang, W. and Sun, S. (2010) Photocatalytic Degradation of Phenol over Cage-Like Bi2MoO6 Hollow Spheres under Visible-Light Irradiation. Catalysis Communications, 11, 647-650. http://dx.doi.org/10.1016/j.catcom.2010.01.014
- Liu, J., Zhang, G., Yu, J.C. and Guo, Y. (2013) In Situ Synthesis of Zn2GeO4 Hollow Spheres and Their Enhanced Photocatalytic Activity for the Degradation of Antibiotic Metronidazole. Dalton Transactions, 42, 5092-5099. http://dx.doi.org/10.1039/c2dt32623j
- Dong, F., Lee, S.C., Wu, Z., Huang, Y., Fu, M., Ho, W.K., et al. (2011) Rose-Like Monodisperse Bismuth Subcarbonate Hierarchical Hollow Microspheres: One-Pot Template-Free Fabrication and Excellent Visible Light Photocatalytic Activity and Photochemical Stability for NO Removal in Indoor Air. Journal of Hazardous Materials, 195, 346-354. http://dx.doi.org/10.1016/j.jhazmat.2011.08.050
- Ai, Z.H., Zhang, L.Z. and Lee, S.C. (2010) Efficient Visible Light Photocatalytic Oxidation of NO on Aerosol Flow- Synthesized Nano-Crystalline InVO4 Hollow Microspheres. Journal of Physical Chemistry C, 114, 18594-18600. http://dx.doi.org/10.1021/jp106906s
- Ikeda, S., Kobayashi, H., Ikoma, Y., Harada, T., Yamazaki, S. and Matsumura, M. (2009) Structural Effects of Titanium(IV) Oxide Encapsulated in a Hollow Silica Shell on Photocatalytic Activity for Gas-Phase Decomposition of Organics. Applied Catalysis A: General, 369, 113-118. http://dx.doi.org/10.1016/j.apcata.2009.09.008
- Zhou, C., Zhao, Y., Bian, T., Shang, L., Yu, H., Wu, L.Z., et al. (2013) Bubble Template Synthesis of Sn2Nb2O7 Hollow Spheres for Enhanced Visible-Light-Driven Photocatalytic Hydrogen Production. Chemical Communications, 49, 9872-9874. http://dx.doi.org/10.1039/c3cc45683h
- Zhang, Y.J., Li, X.J., Min, C., Wang, Y.C. and Li, S. (2010) Preparation of NiO-CdS Composite Hollow Spheres and Their Application for H2-Production to Water Photodegradation. China Patent No. CN101623644.
- Zhang, Y.J., Li, X.J., Min, C., Wang, Y.C. and Li, S. (2010) Preparation of Composite Hollow Spheres with p-n Junction and Their Application for H2-Production to Water Photodegradation. China Patent No. CN101623645.
- Tu, W.G., Zhou, Y., Liu, Q., Tian, Z., Gao, J., Chen, X., et al. (2012) Robust Hollow Spheres Consisting of Alternating Titania Nanosheets and Graphene Nanosheets with High Photocatalytic Activity for CO2 Conversion into Renewable Fuels. Advanced Functional Materials, 22, 1215-1221. http://dx.doi.org/10.1002/adfm.201102566
- Feng, X., Yang, L., Guan, Q. and Liu, Y.L. (2010) Preparation of Ag-Doped TiO2 Hollow Spheres and Their Antibacterial Activity. Ecological Science, 29, 251-255.
- Hu, L., Chen, M., Shan, W., Zhan, T., Liao, M., Fang, X., et al. (2012) Stacking-Order-Dependent Optoelectronic Pro- perties of Bilayer Nanofilm Photodetectors Made from Hollow ZnS and ZnO Microspheres. Advanced Materials, 24, 5872-5877. http://dx.doi.org/10.1002/adma.201202749
NOTES
*Corresponding author.