Journal of Biomaterials and Nanobiotechnology
Vol.5 No.1(2014), Article ID:41734,7 pages DOI:10.4236/jbnb.2014.51001
Engineered Hydrophobin for Biomimetic Mineralization of Functional Calcium Carbonate Microparticles
![]()
VTT—Technical Research Centre of Finland, Espoo, Finland.
Email: *hanna.heinonen@vtt.fi
Copyright © 2014 Hanna Heinonen et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. In accordance of the Creative Commons Attribution License all Copyrights © 2014 are reserved for SCIRP and the owner of the intellectual property Hanna Heinonen et al. All Copyright © 2014 are guarded by law and by SCIRP as a guardian.
Received October 16th, 2013; revised November 15th, 2013; accepted December 14th, 2013
KEYWORDS
Biomimetic Mineralization; Engineered Proteins; Hydrophobin; Calcium Carbonate; Functional Microparticles
ABSTRACT
In this study, the modified hydrophobin, engineered for biomimetic mineralization, has been employed as a structure-directing agent for mineralization of calcium carbonate. For the first time amphiphilic calcium carbonate particles have been obtained, using engineered proteins. The mineral microparticles have been characterized by optical microscopy, scanning electron microscopy (SEM) and X-ray diffraction (XRD). While mineralization in the presence of non-modified hydrophobin results in polymorph mineral structures, uniform microspheres with an average particle diameter of one micron are obtained by employing hydrophobin which has been modified with an additional ceramophilic protein sequence. Owing to the tri-functionality of the modified hydrophobin (hydrophilic, hydrophobic and ceramophilic), the obtained mineral microparticles exhibit amphiphilic properties. Potential applications are in the areas of functional fillers and pigments, like biomedical and composite materials. Pickering emulsions have been prepared as a demonstration of the emulsion-stabilizing properties of the obtained amphiphilic mineral microspheres. The structure-directing effects of the studied engineered hydrophobins are compared with those of synthetic polymers (i.e. polycarboxylates) used as crystallization and scaling inhibitors in industrial applications.
1. Introduction
The natural process of biomineralization enables biological materials with complex morphology and functional design. Muscle shells, diatoms, bone and tooth structures are prominent examples and show how small amounts of structure-directing proteins can transform ordinary materials, like inorganic minerals, into high-performance composites [1-8].
Complementary to biomineralization, the term biomemetic mineralization describes the technical process of mineralization in the presence of structure-directing additives, mimicking the natural process of biomineralization. Like other nanoparticle synthesis routes based on self-assembly, it enables morphological control of inorganic and composite particles and hierarchical structures [9]. Earlier approaches of biomimetic mineralization have been focusing on replacing natural proteins with synthetic polymers, mimicking the structural design of the natural proteins controlling the nucleation and growth of biominerals [10-12]. In addition, also different biopolymers have been investigated, such as dextrane, collagen-, and extracted natural proteins [10].
Recent progress in biotechnology enables the engineering of proteins for biomimetic mineralization, not only for their structure-directing properties, but also for inducing additional functionality of the obtained hybrid materials. Such properties can be specific adsorption and drug delivery properties, as well as amphiphilicity.
A highly interesting group of proteins are hydrophobins produced by filamentous fungi [13-15]. Owing to their intrinsic amphiphilic properties they are of high relevance in biological systems, as well as technical applications. Hydrophobins are divided into two classes, class I and class II, according to their sequences, which also relate to their properties. Typically class I hydrophobins exhibit irreversible assembly including conformational changes at interface [16], whereas class II hydrophobins are structurally more stable and such conformational changes have not been observed. Due to their globular and stable conformation and small size, class II hydrophobins have been employed in formation of welldefined layers at various interfaces [17-19]. By genetic engineering, additional functionalities can be brought to the protein layers by fusing the anchoring group with a second functional domain allowing functionalization of such interfacial layer [20,21]. However, only few studies describe the use of this interesting group of self-assembling proteins for the controlled nucleation-and-growth of inorganic materials [22]. For instance, class I hydrophobins have been studied for the growth of titania films [23] and the mineralization of hydroxyapatite at the water-oil interface of emulsions [24]. One reason for their so far very limited use in biomimetic mineralization might be the lack of a higher affinity towards inorganic precursors, like calcium ions.
This study describes the use of engineered hydrophobins, modified with an additional protein sequence for its specific use as a structure-directing additive in biomemetic mineralization. A glutamic acid-containing protein sequence is introduced, resulting in a protein with amphiphilic as well as ceramophilic properties. In this way the engineered protein design follows the very same principles of earlier studied double-hydrophilic block copolymers. The ceramophilic, glutamic acid rich protein sequence (ZE) enables binding of calcium ions and assembly at crystal faces, while the remaining amphiphilic protein sequences lead to self-assembly and stabilization of the mineral suspension in water. Hydrophobin without the additional ZE-protein sequence is known to adsorb also well to surfaces and to self-assemble, however, it is lacking the additional ceramophilic binding capacity. In contrast to the latter this study describes how the mineralization of calcium carbonate in presence of the ceramophilic modified protein results in highly regular, micronsized and amphiphilic vaterite spheres. Pickering emulsions have been prepared as a demonstration of the additional amphiphilic functionality of the obtained mineral particles. Synthetic polycarboxylates, used in industrial applications as crystallization inhibitors and anti-scalants, have been included in this study as reference systems [25].
2. Experiments and Methods
2.1. Reagents and Materials
Hydrophobin (HFB II) has been produced according to the literature [26]. Modification of hydrophobin with a glutamic acid-rich polymer sequence (HFB I-ZE) has been performed by fusing the ZE peptide sequence [27] to the N-terminal of HFBI using established procedures [22]. The polyacrylate copolymers Sokalan CP5 and Sokalan PM10 have been supplied by BASF SE, Ludwigshafen, Germany. As mineral precursors sodium carbonate (Na2CO3, supplied by Sigma-Aldrich) and calcium chloride (CaCl2, supplied by Fluka) have been used as 0.05 M stock solutions.
2.2. Mineralization Experiments
Biomimetic mineralization experiments have been performed using a double-jet-apparatus, consisting of two syringe pumps (for measuring dosage volume and to ensure equal flow) and a 50 ml glass reactor. 30 ml of the additive solution, containing 30 mg of structure-directing additive (protein, respectively polyacrylate copolymer), have been adjusted to pH 8 by addition of 0.1 M sodium hydroxide solution. Subsequently 0.05 M solutions of CaCl2 and Na2CO3 have been simultaneously added at room temperature during magnetic stirring at a rate of 10 ml/h for 10 min.
2.3. Pickering Emulsions
Toluene has been emulsified in water in presence of calcium carbonate microparticles. After suspending the microparticles in water, toluene has been added in a toluene/CaCO3 ratio of 10 with a total oil content of 10 vol%. Emulsification has been performed by using a Vortex mixer.
2.4. Characterization
Optical micrographs have been taken using an Olympus BH-2 microscope. SEM imaging has been performed using a Zeiss Sigma VP microscope operating at 3 kV. The samples have been prepared from aqueous suspensions by air drying at room temperature on a supported, selfadhesive carbon film and sputtered using a platinum target. XRD measurements have been conducted using a Philips PW1830 generator diffractometer with Cu-Kα radiation (l = 1.542 Å). The droplet size distribution of the Pickering emulsion has been measured using light scattering (Beckman Coulter, USA).
3. Result and Discussion
Calcium carbonate is a mineral of high importance in nature and technology, e.g. as a pigment, filler and for biomedical applications [28]. Therefore calcium carbonate has been chosen as a model system for this study. Polymers with specific binding sites for calcium ions have been intensively studied for biomimetic mineralization of calcium minerals [10,29,30]. Especially doublehydrophilic block copolymers have been used very successfully [1]. In addition and owing to their very good cost-performance, polycarboxylate copolymers are often used as nucleation-and-growth controlling agents for technical applications [25]. Therefore as references also two different technical polyacrylates, Sokalan PM10 and Sokalan CP5, have been included in this study. The main focus of this study, however, is the development of engineered proteins, i.e. hydrophobins, for their specific use in biomimetic mineralization. Analogue to the mentioned double-hydrophilic block copolymers and technical polyacrylates, additional calcium binding sites have been introduced to the protein structure of hydrophobin (HFB II), by adding an additional glutamic acid-rich protein sequence. This way the binding capacity of for calcium ions and the affinity to the growing mineral phase is increased. At the same time additional functionality is introduced to the mineral matrix by adding amphiphilicity due to the hydrophilic and hydrophobic protein sequences of the modified hydrophobin. A more detailed description of the studied additives is given in Table 1.
3.1. Biomimetic Mineralization
Biomimetic mineralization of calcium carbonate has been performed using a double-jet device, which is inspired by technical precipitation and pigment synthesis processes [31]. During the mineralization process two precursor solutions of calcium chloride and sodium carbonate are simultaneously dosed to an aqueous solution of the nucleation-and-growth directing additive (engineered protein, respectively polycarboxylate). The precursor solutions are added within 10 min at room temperature in equimolar amounts at concentrations of 0.05 M and a dosage rate of 10 ml/h. The solutions stay clear and transparent throughout the pre-nucleation phase. At the final dosage point all studied systems turn into opaque suspensions of calcium carbonate particles, with the exception of sample HBM 3, which stays clear and transparent throughout the process. This sample contains Sokalan PM10, demonstrating highly efficient crystallization inhibition and complete suppression of macroscopic crystal growth within the investigated parameter range.
The obtained mineral particles of all samples have been investigated by optical microscopy directly after mineralization. In case of the reference sample with no additive (HBM 1), characteristic calcite rhomboeders can be observed besides some irregular particles already at low magnifications (see Figure 1). In contrast, the sample containing Sokalan PM10 (HBM 3) does not show any larger mineral particles, indicating sufficient nucleation inhibition. All other mineral suspensions show microscopic calcium carbonate particles, though of quite

Table 1. Overview of additives and their physicochemical properties.
 (a)
(a) (b)
(b) (c)
(c) (d)
(d) (e)
(e) (f)
(f)
Figure 1. Optical micrographs of mineral suspensions: (a) reference sample with no additive (HBM1), (b) mineral particles obtained in the presence of Sokalan CP5 (HBM2), (c) full crystallization inhibition in presence of Sokalan PM10 (HBM3), (d) polymorphous mineral phase in the presence of HFB-II (HBM4), (e) and (f) uniform microparticles in presence of HFB I-ZE (HBM5).
different sizes and shapes. The most striking difference has been observed for the sample mineralized in the presence of the modified hydrophobin, HFB I-ZE, with additional ceramophilic binding sites (HBM 5): the monodisperse mineral particles of this sample show under the microscope Brownian motion, indicating very small particle sizes.
3.2. Microparticle Morphology—Uniform Mineral Microspheres
For further characterization all mineral suspensions have been centrifuged and washed three times to remove residual dissolved additives and to isolate the mineral phase. Samples for scanning electron microscopy (SEM) have been prepared from the purified suspensions (see Figure 2). Subsequently the suspensions have been air-dried at room temperature and the remaining mineral crystals have been studied by X-ray diffraction (XRD). The results of the characterization of the obtained mineral particles are summarized in Table 2.
SEM micrographs of the purified and dried samples are well in accordance with optical micrographs, obtained in suspension directly after mineralization. In addition they show very interesting additional features. As described in earlier studies, the reference sample with no additive (HBM 1) has a polymorphous appearance [10]: Besides rhombohedral calcite also spherical particles are obtained, indicating thermodynamically instable vaterite or amorphous calcium carbonate (Figure 2(a)). X-ray diffraction of the purified and air-dried sample indicates a transformation into calcite only (see Figure 3(a)), as expected for additive-free, non-stabilized calcium carbonate. As already indicated by optical microscopy, no macroscopic mineral particles are observed for sample HBM 3, confirming the very efficient crystal inhibition by Sokalan PM10 (Figure 2(c)). To investigate the influence of the polymer on the mineral crystal modification the clear solution has been air dried. The obtained solid, white powder has been washed with water and centrifuged to remove any access polymer. X-ray diffraction of this air-dried sample shows that the mineral phase consists mainly of calcite and some sodium chloride crystals, which remained by inclusion within the loose sediment during centrifugation. In contrast to Sokalan PM10, the copolymer Sokalan CP5 did not fully suppress the formation of calcium carbonate crystals under the studied reaction conditions. The observed mineral particles, however, show a very interesting morphology: a roughly spherical superstructure of clustered, distorted calcite rhomboeders, showing the strong interaction between the mineral phase and the structure-directing polymer (Figure 2(d)).
Biomimetic mineralization, in presence of non-modified hydrophobin (HBM 4), results in a polymorphous mineral phase, similar to the reference sample with no additive (Figure 2(d)). However, in contrast to the de-
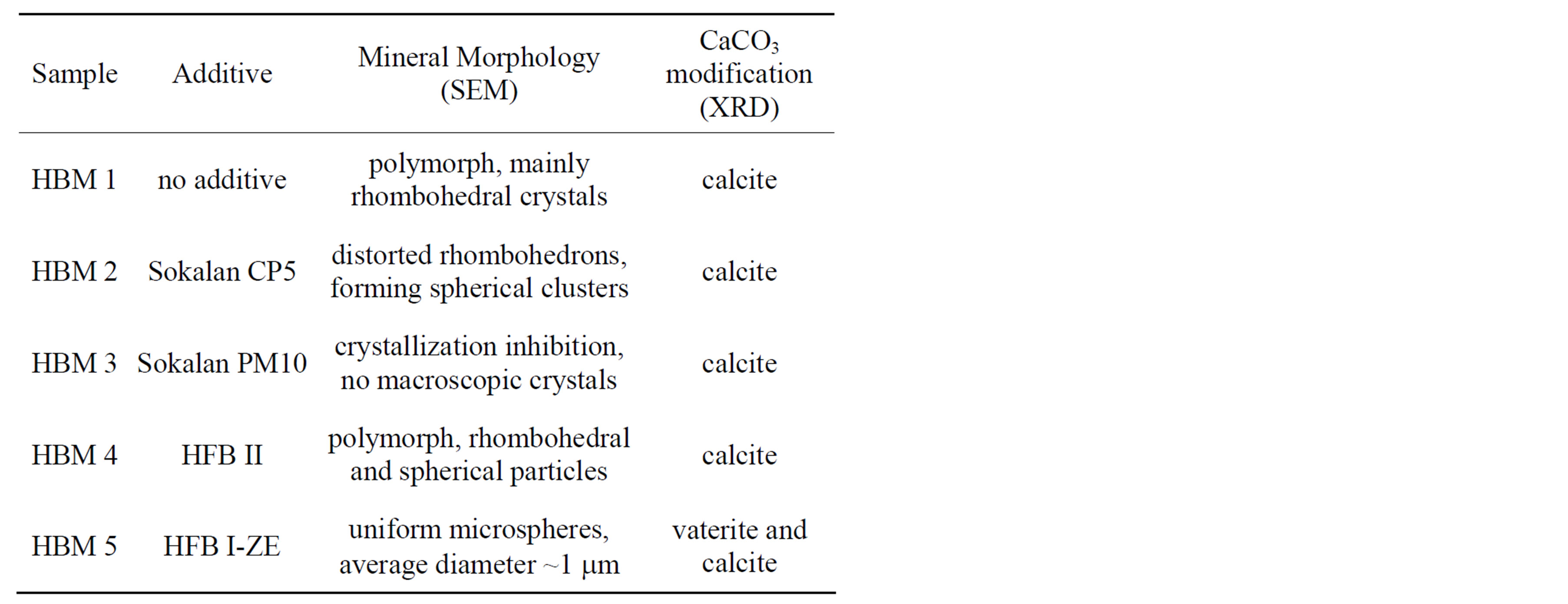
Table 2. Characterization of mineral modification.
 (a)
(a) (b)
(b) (c)
(c) (d)
(d) (e)
(e) (f)
(f)
Figure 2. Scanning electron micrographs: (a) reference sample with no additive (HBM1), mineral particles obtained in the presence of (b) Sokalan CP5 (HBM2) and (c) Sokalan PM10 (HBM3), (d) polymorphous mineral phase obtained in the presence of HFB-II (HBM4), (e) and (f) uniform microspheres obtained in presence of HFB I-ZE (HBM5).
fault sample (HBM 1), a much stronger tendency towards the formation of spherical particles is observed. The diffractogram of the dried mineral particles shows no indication of vaterite formation (Figure 3(b)), and the obtained particles are about double the size compared to the sample containing ceramophilic modified hydrophobin (HBM 5).
Already during mineralization the uniqueness of mineralization in presence of the ceramophilic modified hydrophobin with the additional acidic amino acid sequence (HFB I-ZE) becomes obvious, as obtained mineral particles have a much lower tendency for sedimentation. Scanning electron microscopy reveals the very homogeneous and spherical structure of the microparticles (Figures 2(e), (f)). In contrast to all other samples, XRD of the air dried sample of HBM 5 indicates, besides some calcite, under normal conditions thermodynamically instable vaterite as the predominant crystal modification (Figure 3(c)). If the two crystalline phases coexist as separate particles or as a mixed phase requires further investigation. The primary particle size of the obtained vaterite crystals, estimated on basis of the Scherrer equation from the diffraction peak broadening, is about 6 nm, indicating a non-classical, nanoparticle mediated crystallization mechanism.
3.3. Formation of Pickering Emulsions
Even after air-drying the aggregated minerals are fully redispersible as discrete microparticles. In contrast to non-modified calcite microparticles (HBM 1), they are suitable stabilizers for Pickering emulsions [32,33]. For this purpose oil-in-water emulsions have been prepared at a weight ratio of mineral particles (HBM 5) to oil of 10. Optical micrographs of the emulsion droplets show a golf-ball-like morphology, indicating the adsorption of the amphiphilic calcium carbonate spheres at the oilwater interface and distortion of the droplet surface (see Figure 4). The default experiment, using non-modified calcium carbonate particles (HBM 1), results in immediate separation of oil and water phases.
As the biotechnological production of the engineered proteins on lab scale is limited to the milligram scale, no detailed variation of process parameters could be conducted in this study. However, the presented proof-ofconcept of employing proteins specifically engineered for biomimetic mineralization opens up manifold opportunities for future research, including mechanistic studies. In addition to the demonstrated amphiphilicity of the resulting mineral particles other fusion protein functionnalities could be considered, such as specific binding sides, biocompatibility and biological activity. The demonstrated use of engineered, ceramophilic fusion proteins for the production of functional fillers and pigments for future studies might be especially interesting at the very
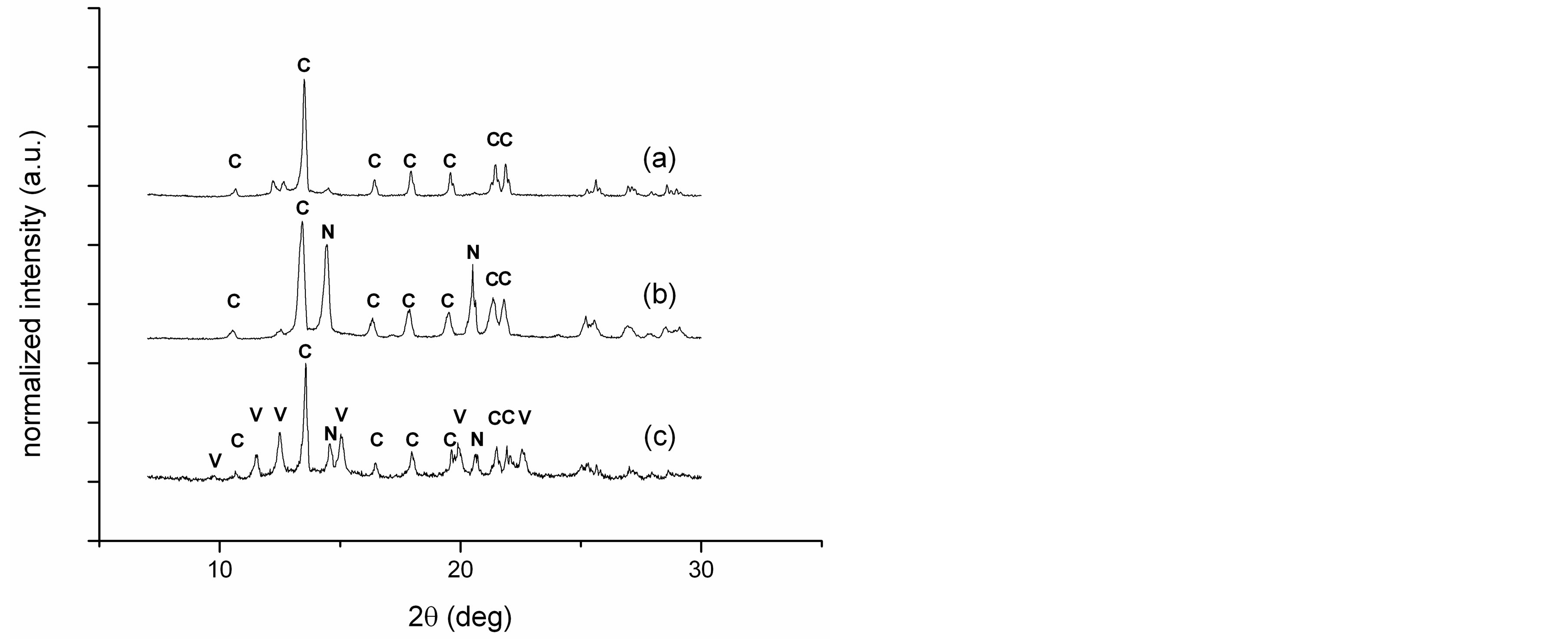
Figure 3. X-ray diffractograms of calcium carbonate samples obtained with no additive, HBM1 (a), with non-modified hydrophobin, HBM 4 (b) and with ceramophilic modified hydrophobin, HBM5 (c). Main peaks of the calcium carbonate crystal modifications calcite (C) and vaterite (V) are indicated, as well as inclusions of sodium chloride (N).

Figure 4. Oil droplet of an oil-in-water Pickering emulsion, showing the amphiphilic calcium carbonate microspheres absorbed at the oil-water interface.
exciting and fascinating interface between biological systems, nanoand micromaterials.
4. Conclusion
The study demonstrates for the first time the synthesis of amphiphilic calcium carbonate particles by using engineered proteins. Mimicking the natural process of biomineralization enables the synthesis of mineral particles with potential application as novel functional fillers and pigments [34]. Earlier studies of natural biomineralization processes have revealed the crucial importance of structure-directing agents, enabling binding of metal ions and adsorption at crystal faces [35,36]. Synthetic polymers, such as double-hydrophilic block copolymers and polycarboxylates, have been successfully employed for the morphological control of biomimetic minerals and as crystallization inhibitors. In his study the concept of biomimetic mineralization has been extended from synthetic polymers and natural proteins, to novel engineered fusion proteins with additional ceramophilic binding sites, i.e. hydrophobin containing an additional glutamic acidrich protein sequence (HFB I-ZE). In contrast to nonmodified hydrophobin, the calcium carbonate particles obtained from biomimetic mineralization, are uniform in size and morphology. The dried mineral phase shows under normal conditions thermodynamically instable vaterite as the predominant crystal modification. In addition to the observed size control, the engineered protein (HFB I-ZE) induces additional functionality to the resulting mineral particles. Pickering emulsions have been prepared as a demonstration of their amphiphilicity and emulsion stabilizing properties.
Acknowledgements
Funding is gratefully acknowledged by VTT’s Frontier Project Biomimetic Hybrid Materials. The authors thank Dr. Jens Rieger (BASF SE) for the kind supply of polyacrylate copolymers and (Sokalan PM10 and Sokalan CP5) and Ulla Kanerva and Jari Keskinen for conducting XRD measurements. This article is dedicated to Heimo Schnablegger (1963-2012).
REFERENCES
- H. Cölfen and S. Mann, “Higher-Order Organization by Mesoscale Self-Assembly and Transformation of Hybrid Nanostructures,” Angewandte Chemie International Edition, Vol. 42, No. 21, 2003, pp. 2350-2365. http://dx.doi.org/10.1002/anie.200200562
- S. Mann, “Biomineralization—Principles and Concepts in Bioinorganic Materials Chemistry,” Oxford University Press, New York, 2001.
- F. C. Meldrum, “Calcium Carbonate in Biomineralisation and Biomimetic Chemistry,” International Materials Reviews, Vol. 48, No. 3, 2003, pp. 187-224. http://dx.doi.org/10.1179/095066003225005836
- M. Breulmann, H. Cölfen, H.-P. Hentze, M. Antonietti, D. Walsh and S. Mann, “Elastic Magnets: Template-Controlled Mineralization of Iron Oxide Colloids in a SpongeLike Gel Matrix,” Advanced Materials, Vol. 10, No. 3, 1998, pp. 237-241. http://dx.doi.org/10.1002/(SICI)1521-4095(199802)10:3<237::AID-ADMA237>3.0.CO;2-6
- P. Laaksonen, G. R. Szilvay and M. B. Linder, “Genetic Engineering in Biomimetic Composites,” Trends in Biotechnology, Vol. 30, No. 4, 2012, pp. 191-197. http://dx.doi.org/10.1016/j.tibtech.2012.01.001
- A. Walther, I. Bjurhager, J.-M. Malho, J. Peere, J. Ruokolainen, L. A. Berglund and O. Ikkala, “Large-Area, Lightweight and Thick Biomimetic Composites with Superior Material Properties via Fast, Economic, and Green Pathways,” Nano Letters, Vol. 10, No. 8, 2010, pp. 2742- 2748. http://dx.doi.org/10.1021/nl1003224
- N. Yin, S. Y. Chen, Y. Ouyang, L. Tang, J. X. Jang and H.P. Wang, “Biomimetic Mineralization Synthesis of Hydroxyapatite Bacterial Cellulose Nanocomposites,” Progress in Natural Science: Materials International, Vol. 21, No. 6, 2011, pp. 472-477. http://dx.doi.org/10.1016/S1002-0071(12)60085-9
- P. Fratzl, “Biomimetic Materials Research: What Can We Really Learn from Nature’s Structural Materials?” Journal of the Royal Society Interface, Vol. 4, No. 15, 2007, 637-642. http://dx.doi.org/10.1098/rsif.2007.0218
- M. Li, H. Schnablegger and S. Mann, “Coupled Synthesis and Self-Assembly of Nanoparticles to Give Structures with Controlled Organization,” Nature, Vol. 402, 1999, pp. 393-395. http://dx.doi.org/10.1038/46509
- H. Cölfen, “Precipitation of Carbonates: Recent Progress in Controlled Production of Complex Shapes,” Current Opinion in Colloid & Interface Science, Vol. 8, No. 1, 2003, pp. 23-31. http://dx.doi.org/10.1016/S1359-0294(03)00012-8
- A. Peytcheva, H. Cölfen, H. Schnablegger and M. Antonietti, “Calcium Phosphate Colloids with Hierarchical Structure Controlled by Polyaspartates,” Colloid and Polymer Science, Vol. 280, No. 3, 2002, pp. 218-227. http://dx.doi.org/10.1007/s00396-001-0600-0
- D. Gebauer and H. Cölfen, “Prenucleation Clusters and Non-Classical Nucleation,” Nano Today, Vol. 6, No. 6, 2011, pp. 564-584. http://dx.doi.org/10.1016/j.nantod.2011.10.005
- H. A. B. Wosten, “Hydrophobins: Multipurpose Proteins,” Annual Review of Microbiology, Vol. 55, No. 1, 2001, pp. 625-646. http://dx.doi.org/10.1146/annurev.micro.55.1.625
- M. B. Linder, G. R. Szilvay, T. Nakari-Setälä and M. E. Penttilä, “Hydrophobins: The Protein-Amphiphiles of Filamentous Fungi,” FEMS Microbiology Reviews, Vol. 29, No. 5, 2005, pp. 877-896. http://dx.doi.org/10.1016/j.femsre.2005.01.004
- M. B. Linder, “Hydrophobins: Proteins That Self Assemble at Interfaces,” Current Opinion in Colloid & Interface Science, Vol. 14, No. 5, 2009, pp. 356-363. http://dx.doi.org/10.1016/j.cocis.2009.04.001
- M. L. De Vocht, I. Reviakine, W. P. Ulrich, W. BergsmaSchutter, H. A. B. Wosten, H. Vogel, A. Brisson, J. G. H. Wessels and G. T. Robillard, “Self-Assembly of the Hydrophobin SC3 Proceeds via Two Structural Intermediates,” Protein Science, Vol. 11, No. 5, 2002, pp. 1199- 1205. http://dx.doi.org/10.1110/ps.4540102
- G. R. Szilvay, A. Paananen, K. Laurikainen, E. Vuorimaa, H. Lemmetyinen, J. Peltonen and M. B. Linder, “SelfAssembled Hydrophobin Protein Films at the Air-Water Interface: Structural Analysis and Molecular Engineering,” Biochemistry, Vol. 46, No. 9, 2007, pp. 2345-2354. http://dx.doi.org/10.1021/bi602358h
- P. Laaksonen, J. Kivioja, A. Paananen, M. Kainlauri, K. Kontturi, J. Ahopelto and M. B. Linder, “Selective Nanopatterning Using Citrate-Stabilized Au Nanoparticles and Cystein-Modified Amphiphilic Protein,” Langmuir, Vol. 25, No. 9, 2009, pp. 5185-5192. http://dx.doi.org/10.1021/la804001e
- P. Laaksonen, M. Kainlauri, T. Laaksonen, A. Shchepetov, H. Jiang, J. Ahopelto and M. B. Linder, “Interfacial Engineering by Proteins: Exfoliation and Functionalization of Graphene by Hydrophobins,” Angewandte Chemie International Edition, Vol. 49, No. 29, 2010, pp. 4946- 4949. http://dx.doi.org/10.1002/anie.201001806
- M. B. Linder, M. Qiao, F. Laumen, K. Selber, T. Hyytia, T. Nakari-Setala, M. E. Penttila, “Efficient Purification of Recombinant Proteins Using Hydrophobins as Tags in Surfactant-Based Two-Phase Systems,” Biochemistry, Vol. 43, No. 37, 2004, pp. 11873-11882. http://dx.doi.org/10.1021/bi0488202
- S. Varjonen, P. Laaksonen, A. Paananen, H. Valo, H. Hähl, T. Laaksonen and M. B. Linder, “Self-Assembly of Cellulose Nanofibrils by Genetically Engineered Fusion Proteins,” Soft Matter, Vol. 7, No. 6, 2011, pp. 2402-2411. http://dx.doi.org/10.1039/c0sm01114b
- S. S. Behrens, “Synthesis of Inorganic Nanomaterials Mediated by Protein Assemblies,” Journal of Materials Chemistry, Vol. 18, 2008, pp. 3788-3798. http://dx.doi.org/10.1039/b806551a
- D. Santhiya, Z. Burghard, C. Greiner, L. P. H. Jeurgens, T. Subkowski and J. Bill, “Bioinspired Deposition of TiO2 Thin Films Induced by Hydrophobins,” Langmuir, Vol. 26, No. 9, 2010, pp. 6494-6502. http://dx.doi.org/10.1021/la9039557
- A. Schulz, B. M. Liebeck, D. John, A. Heiss, T. Subkowski and A. Boker, “Protein-Mineral Hybrid Capsules from Emulsions Stabilized with an Amphiphilic Protein,” Journal of Materials Chemistry, Vol. 21, 2011, pp. 9731- 9736. http://dx.doi.org/10.1039/c1jm10662g
- J. Rieger, E. Hädicke, I. U. Rau and D. Boeckh, “A Rational Approach to the Mechanism of Incrustation Inhibition by Polymeric Additives,” Tenside, Surfactants, Detergents, Vol. 34, 1997, pp. 430-435.
- M. B. Linder, K. Selber, T. Nakari-Setälä, M. Qiao, M.-R. Kula and M. Penttilä, “The Hydrophobins HFBI and HFBII from Trichoderma reesei Showing Efficient Interactions with Nonionic Surfactants in Aqueous TwoPhase Systems,” Biomacromolecules, Vol. 2, No. 2, 2001, pp. 511-517. http://dx.doi.org/10.1021/bm0001493
- K. Zhang, M. R. Diehl and D. A. Tirrell, “Artificial Polypeptide Scaffold for Protein Immobilization,” Journal of the American Chemical Society, Vol. 127, No. 29, 2005, pp. 10136-10137. http://dx.doi.org/10.1021/ja051457h
- D. V. Volodkin, R. von Klitzing and H. Moehwald, “Pure Protein Microspheres by Calcium Carbonate Templating,” Angewandte Chemie International Edition, Vol. 49, No. 48, 2010, pp. 9258-9261. http://dx.doi.org/10.1002/anie.201005089
- H. Imai, Y. Oaki and A. Kotachi, “A Biomimetic Approach for Hierarchically Structured Inorganic Crystals through Self-Organization,” Bulletin of the Chemical Society of Japan, Vol. 79, No. 12, 2006, pp. 1834-1851. http://dx.doi.org/10.1246/bcsj.79.1834
- N. A. J. M. Sommerdijk and G. D. With, “Biomimetic CaCO3 Mineralization Using Designer Molecules and Interfaces,” Chemical Reviews, Vol. 108, No. 11, 2008, pp. 4499-4550. http://dx.doi.org/10.1021/cr078259o
- M. Sedlák, M. Antonietti and H. Cölfen, “Synthesis of a New Class of Double-Hydrophilic Block Copolymers with Calcium Binding Capacity as Builders and for Biomimetic Structure Control of Minerals,” Macromolecular Chemistry and Physics, Vol. 199, No. 2, 1998, pp. 247- 254. http://dx.doi.org/10.1002/(SICI)1521-3935(19980201)199:2<247::AID-MACP247>3.0.CO;2-9
- Z.-G. Cui, C.-F. Cui, Y. Zhu and B. P. Binks, “Multiple Phase Inversion of Emulsions Stabilized by in Situ Surface Activation of CaCO3 Nanoparticles via Adsorption of Fatty Acids,” Langmuir, Vol. 28, No. 1, 2012, pp. 314- 320. http://dx.doi.org/10.1021/la204021v
- R. Aveyard, B. P. Binks and J. H. Clint, “Emulsions Stabilised Solely by Colloidal Particles,” Advances in Colloid and Interface Science, Vol. 100-102, 2003, pp. 503- 546. http://dx.doi.org/10.1016/S0001-8686(02)00069-6
- G. Jutz and A. Böker, “Bionanoparticles as Functional Macromolecular Building Blocks—A New Class of Nanomaterials,” Polymer, Vol. 52, No. 2, 2011, pp. 211-232. http://dx.doi.org/10.1016/j.polymer.2010.11.047
- G. Falini, S. Albeck, S. Weiner and L. Addadi, “Control of Aragonite or Calcite Polymorphism by Mollusk Shell Macromolecules,” Science, Vol. 271, No. 5245, 1996, pp. 67-69. http://dx.doi.org/10.1126/science.271.5245.67
- S. Weiner and L. Addadi, “Crystallization Pathways in Biomineralization,” Annual Review of Materials Research, Vol. 21, 2011, pp. 21-40. http://dx.doi.org/10.1146/annurev-matsci-062910-095803
NOTES
*Corresponding author.

